Hangzhou Dingyan Chem Co., Ltd
Items Standard Result Appearance Colorless,transparent liquid Complies Content% ≥99.90
Cas:106-25-2
Min.Order:1 Kilogram
FOB Price: $3.0 / 10.0
Type:Manufacturers
inquiryAlity Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Xi'an Xszo Chem Co., Ltd.
1. Factory price and high quality must be guaranteed, base on 8 years of production and R&D experience2. Free samples will be provided,ensure specifications and quality are right for customer3. Customers will receive the most professional technical s
Cas:106-25-2
Min.Order:1 Gram
FOB Price: $0.1
Type:Manufacturers
inquiryChengdu Lemeitian Pharmaceutical Technology Co,. Ltd
ADVANTAGE: 1. More than 3,000 Chinese medicine reference substances / standard products available from stock, issued on the same day, multiple cities can be delivered the next day 2. The products are provided with COA, HPLC, NMR, quality assurance,
Dayang Chem (Hangzhou) Co.,Ltd.
As a leading manufacturer and supplier of chemicals in China, DayangChem not only supply popular chemicals, but also DayangChem’s R&D center offer custom synthesis services. DayangChem can provide different quantities of custom synthesis
Chemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Henan Tianfu Chemical Co., Ltd.
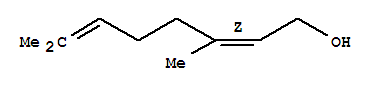
Product Name: NEROL Synonyms: NEROL BRI (98+%) FCC;Nerol solution;cis-3,7-DiMethyl-2,6-octadien-1-ol 97%;(2Z)-3,7-Dimethyl-2,6-octadien-1-ol;2, 6-DIMETHYL-CIS-2, 6-OCTADIEN-8-OL;2,6-OCTADIEN-1-OL, 3,7-DIMETHYL;2,6-DIMETHYL-2,6-OCTADIEN-8-OL;3, 7-D
Cas:106-25-2
Min.Order:1 Kilogram
FOB Price: $230.0
Type:Lab/Research institutions
inquiryHebei Nengqian Chemical Import and Export Co., LTD
Our Advantage Rich Experience Our products are sold all over Europe,North&South America, Sino-East, Asia and pacific area as well as Africa,we establish long term. Quality service Company cooperates with research institutes. We strictly con
Cas:106-25-2
Min.Order:1 Kilogram
FOB Price: $1.0 / 10.0
Type:Trading Company
inquiryZhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:106-25-2
Min.Order:1 Kilogram
FOB Price: $139.0 / 210.0
Type:Trading Company
inquiryHebei yanxi chemical co.,LTD.
hebei yanxi chemical co., LTD who registered capital of 10 million yuan, nearly to $2 million, we have a pharmaceutical raw materials factory production of pharmaceutical raw materials, and a reagent r&d center, and we do research and developmen
Cas:106-25-2
Min.Order:1 Kilogram
FOB Price: $2.0 / 6.0
Type:Trading Company
inquiryLeader Biochemical Group
About Product Details
Cas:106-25-2
Min.Order:1 Kilogram
FOB Price: $1.0 / 2.0
Type:Lab/Research institutions
inquiryShanghai Upbio Tech Co.,Ltd
1.No Less 8 years exporting experience. Clients can 100% received goods 2.Lower Price with higher quality 3,Free sample 4,We are sincerely responsible for the "product quality" and "After Service" Upbio is Specialized
Cas:106-25-2
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryShandong Hanjiang Chemical Co., Ltd.
Hello, dear friend! I'm Hansen and Allen from China. Welcome to my lookchem mall! The following is a brief introduction of our company's products and services. If you are interested in our products, please contact us by emai
Cas:106-25-2
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHangzhou Sartort Biopharma Co., Ltd
Appearance:Colorless liquid Storage:R.T Package:25kg/Barrel Application:Edible spices Transportation:Express/Sea/Air Port:Any port in China
Cas:106-25-2
Min.Order:25 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHenan Wentao Chemical Product Co., Ltd.
Henan Wentao Chemical Product Co.,Ltd is Located in Zhengzhou High-tech Development Zone with import and export license, We passed ISO 9001:2008 as well, Henan Wentao has developed more than 1000 compounds, which are widely used in the fields of prod
Cas:106-25-2
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryHubei Jiutian Bio-medical Technology Co., Ltd
1,we produce and sell good chemicals around the world. 2,our success rate is about 95%. this means, if customer order is accepted, the probability that the customer will obtain the ordered substances, is 95%. 3,our staff consists of highly qualifie
Cas:106-25-2
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHangzhou Keyingchem Co.,Ltd
Hangzhou KeyingChem Co., Ltd. exported this product to many countries and regions at best price. If you are looking for the material’s manufacturer or supplier in China, KeyingChem is your best choice. Pls contact with us freely for getting det
Hangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Cas:106-25-2
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryHANGZHOU YUNUO CHEMICAL CO.,LTD
superior quality Appearance:Clear colorless to almost colorless liquid Storage:Stored in cool, dry and ventilation place; Away from fire and heat Package:1kg/bag, 1kg/drum or 25kg/drum or as per your request. Application:Nerol is an isomer of Geran
Cas:106-25-2
Min.Order:1 Kilogram
Negotiable
Type:Trading Company
inquiryXiamen Jenny Chemical Technology Co., Ltd.
GMP standard, high purity, competitive price, in stock 1. Quick Response: within 6 hours after receiving your email. 2. Quality Guarantee: All products are strictly tested by our QC, confirmed by QA, and approved by a third-party lab in China, USA,
Hubei CuiRan Biotechnology Co., Ltd
Hubei CuiRan Biotechnology Co., Ltd is a leading company in the research, development, manufacture and marketing of High Quality Phytochemicals and Extracts(especially Active Ingredients from Traditional Chinese Medicine,Traditional Chinese Medicine)
Hangzhou Huarong Pharm Co., Ltd.
Hangzhou Huarong Pharm Co., Ltd.established since 2006 , has been actively developing specialty products for Finished Dosages, APIs, Intermediates, and Fine chemicals markets in North America, Europe, Korea, Japan, Mid-East and all over the World. Hu
KAISA GROUP INC
1.Applied in food field.it can improve the immune system and prolong life. 2.Appliedin cosmetic field.it can improve the skin care. 3.Applied in pharmaceutical field.it can treat various dieases. 4.Our product quality assurance will make our customer
Cas:106-25-2
Min.Order:1 Metric Ton
FOB Price: $7.0 / 8.0
Type:Trading Company
inquiryHunan chemfish Pharmaceutical co.,Ltd
Appearance:95%+ Package:R&D,Pilot run Transportation:per client require Port:Express ,Air, Sea
Hefei Zhaobo Technology Co., Ltd.
Nerol is a valuable spice. Used to prepare floral essences such as rose and orange blossom. Category: Found in neroli oil, rose oil, etc. It is a valuable spice. Nerolidol is a flavor raw material with a rose scent and is widely used in the preparat
Cas:106-25-2
Min.Order:1 Kiloliter
FOB Price: $1.0 / 50.0
Type:Trading Company
inquiryHubei Vanz Pharm Co.,Ltd
ISO/factory/goodqualityAppearance:off white Storage:Dry,cool place Package:drum Application:active pharmaceutical ingredients Transportation:by air/sea/express Port:shenzhen/shanghai
Kono Chem Co.,Ltd
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:drum and bag Application:for pharma use Transportation:by sea or air Port:Beijing or Guangzhou
GIHI CHEMICALS CO.,LIMITED
high purity,in stock Package:25kg/drum,or as per customers'demand Application:API,or Intermediates,fine chemicals Transportation:air,sea,courier
Shanghai Acmec Biochemical Technology Co., Ltd.
Acmec is a leading manufacturer and supplier of biochemical reagents and life science products. We have over 40,000 items in stock (real-time inventory) and offer discounted prices to registered members of the online store ( www.acmec.com.cn ) Appea
Cas:106-25-2
Min.Order:1 bottle
Negotiable
Type:Lab/Research institutions
inquiryHenan Allgreen Chemical Co.,Ltd
high quality Storage:Sealed, dry, microtherm , avoid light and smell. Package:According to the demand of customer Application:Organic synthesis Transportation:by air or by sea
Synthetic route

-
-
106-25-2
Nerol

| Conditions | Yield |
|---|---|
| With palladium diacetate; sodium azide; trisodium tris(3-sulfophenyl)phosphine In water; acetonitrile at 25℃; for 1h; | 98% |

| Conditions | Yield |
|---|---|
| With isopropyl alcohol; zirconium(IV) oxide for 6h; Rate constant; Heating; | 97% |
| With isopropyl alcohol; zirconium(IV) oxide for 6h; Heating; | 97% |
| With sodium formate; RuCl2(m-SPPh2)2 In water at 80℃; for 7h; | 95% |
-
-
80873-76-3
1-(tert-butyldimethylsiloxy)-3,7-dimethyl-2,6-octadiene

-
-
106-25-2
Nerol

| Conditions | Yield |
|---|---|
| With acetonyltriphenylphosphonium bromide In methanol; dichloromethane at 20℃; for 0.0833333h; | 95% |
| In methanol; dichloromethane for 3.66667h; Irradiation; | |
| In methanol; dichloromethane for 3.66667h; Product distribution; Irradiation; other silyl ethers; phenanthrene, var. time; |

-
-
106-25-2
Nerol

| Conditions | Yield |
|---|---|
| With lithium; ethylenediamine In tetrahydrofuran at -10℃; for 2h; | 93% |
-
-
52537-25-4
(Z)-1-(allyloxy)-3,7-dimethylocta-2,6-diene

-
-
106-25-2
Nerol

| Conditions | Yield |
|---|---|
| With samarium diiodide; water; isopropylamine In tetrahydrofuran at 20℃; for 0.0333333h; | 88% |

| Conditions | Yield |
|---|---|
| With indium tri-isopropoxide supported on mesoporous SBA-15 In isopropyl alcohol at 80℃; for 7h; Reagent/catalyst; Meerwein-Ponndorf-Verley Reduction; Inert atmosphere; Schlenk technique; chemoselective reaction; | 87.6% |
| With potassium hydroxide; isopropyl alcohol; dichlorotetrakis(dimethylsulfoxide)ruthenium at 20℃; for 3h; | 86.8% |
| With Triisopropyl borate; isopropyl alcohol at 27℃; for 20h; Kinetics; Reagent/catalyst; Meerwein-Ponndorf-Verley reduction; Inert atmosphere; chemoselective reaction; | 83.1% |
-
-
70473-31-3
neryl 2-tetrahydropyranyl ether

-
-
106-25-2
Nerol

| Conditions | Yield |
|---|---|
| ammonium cerium(IV) nitrate In alkaline aq. solution; acetonitrile at 70℃; for 2h; pH=8; Decomposition; | 86% |
| With tin(ll) chloride In methanol Substitution; | 80% |
| With boron trifluoride diethyl etherate; dimethylthio-dimethyl-tin In toluene at -20 - 0℃; for 12h; | 82 % Chromat. |
-
-
13058-12-3
ethyl 3,7-dimethyl-2,6-octadienoate

-
-
106-25-2
Nerol

| Conditions | Yield |
|---|---|
| With lithium aluminium tetrahydride | 81% |
| With diisobutylaluminium hydride In diethyl ether; hexane at -78 - 20℃; for 12.5h; | 0.9 g |
-
-
74323-42-5
morpholide of 1-hydroxy-3,7-dimethylocta-2Z,6-diene-4-sulfonic acid

-
A
-
106-25-2
Nerol

-
B
-
16750-94-0
2,6-dimethyl-2,5-octadien-8-ol

| Conditions | Yield |
|---|---|
| With dibenzo-18-crown-6; sodium In tetrahydrofuran; benzene-d6 at -70℃; for 0.0333333h; | A 75% B 4% |
| With dibenzo-18-crown-6; sodium In ammonia at -70℃; for 2h; | A 75% B 4% |
| With sodium; tert-butyl alcohol In benzene-d6 at -70℃; Yield given. Yields of byproduct given; | |
| With lithium; tert-butyl alcohol In ammonia at -70℃; Product distribution; also Na, (DB18C6) or Na, tBuOH in NH3; | |
| With lithium In diethyl ether; ammonia at -65℃; Product distribution; other reagent, solvent, temperature; |

| Conditions | Yield |
|---|---|
| With tris(2,4,6-trimethoxyphenyl)phosphine In methanol at 50℃; for 24h; | 73% |

| Conditions | Yield |
|---|---|
| With bis-trimethylsilanyl peroxide; bis(acetylacetonate)oxovanadium In dichloromethane at 25℃; for 7h; | A 68% B 8% C 2% |
| bis(acetylacetonate)oxovanadium; bis-trimethylsilanyl peroxide In dichloromethane at 25℃; for 7h; | A 68 % Chromat. B 8 % Chromat. C 2 % Chromat. |
| bis(acetylacetonate)oxovanadium; bis-trimethylsilanyl peroxide In dichloromethane at 25℃; for 7h; Product distribution; | A 68 % Chromat. B 8 % Chromat. C 2 % Chromat. |

-
-
50727-95-2
(2R*,3R*)-2,3-epoxy-3,7-dimethyl-6-octenyl acetate

-
A
-
78-70-6
3,7-dimethylocta-1,6-dien-3-ol

-
B
-
106-25-2
Nerol

| Conditions | Yield |
|---|---|
| With tellurium; lithium triethylborohydride In tetrahydrofuran for 0.25h; Ambient temperature; | A 62% B 5.5% |
| With tellurium; lithium triethylborohydride In tetrahydrofuran for 0.25h; Product distribution; Mechanism; Ambient temperature; other reducing Te agent; various time; also in the presence of fluoride ion; | A 62% B 5.5% |
-
-
115-18-4
2-methyl-3-buten-2-ol

-
A
-
106-25-2
Nerol

-
B
-
106-24-1
Geraniol

-
C
-
58461-27-1
(+/-)-lavandulol

-
D
-
78-79-5
isoprene

| Conditions | Yield |
|---|---|
| With oxalic acid In water for 5h; Heating; Further byproducts given. Yields of byproduct given; | A n/a B n/a C n/a D 46% |

-
-
106-24-1
Geraniol

-
A
-
106-22-9
Citronellol

-
B
-
106-25-2
Nerol

-
C
-
23062-07-9
(E)-2,6-dimethyl-oct-2-ene-1,8-diol

-
D
-
26488-98-2
(2E,6Z)-2,6-dimethylocta-2,6-diene-1,8-diol

-
E
-
26488-97-1
8-hydroxygeraniol

| Conditions | Yield |
|---|---|
| With 2,4-Dichlorophenoxyacetic acid; sucrose; cultured cells of Catharanthus roseus In water at 25℃; for 48h; Product distribution; other reaction time; | A n/a B n/a C 32.9% D 25.3% E 41.8% |


| Conditions | Yield |
|---|---|
| With hydrogen bromide Behandeln des erhaltenen Bromids mit Kaliumacetat in Dimethylformamid und Erwaermen des Reaktionsprodukts mit wss.Natronlauge; | |
| pyridine; tungsten oxomethoxide at 200℃; for 3h; | |
| pyridine; tungsten oxomethoxide at 200℃; for 3h; Product distribution; other catalysts and time; |
-
-
555-31-7
aluminum isopropoxide

-
-
5392-40-5
(E/Z)-3,7-dimethyl-2,6-octadienal

-
A
-
106-25-2
Nerol

-
B
-
106-24-1
Geraniol

-
C
-
67-64-1
acetone

| Conditions | Yield |
|---|---|
| analoge Reaktionen mit Aldehyden und Ketonen bei hoeherer Temperatur; |


| Conditions | Yield |
|---|---|
| With sodium amalgam; ethanol | |
| With aluminum isopropoxide; isopropyl alcohol | |
| With hydrotalcite; isopropyl alcohol at 82℃; for 5h; Title compound not separated from byproducts; |

| Conditions | Yield |
|---|---|
| beim Kochen der Natriumverbindung; | |
| With hydrogen iodide; benzene und Erwaermen des Reaktionsproduktes mit alkoh.Natronlauge; | |
| With hydrogen iodide; acetic acid und Erwaermen des Reaktionsproduktes mit alkoh.Natronlauge; |

| Conditions | Yield |
|---|---|
| With hydrogen bromide; acetic acid und Abspaltung von HBr aus dem Reaktionsprodukts; isolinalool; |

| Conditions | Yield |
|---|---|
| With lithium aluminium tetrahydride |
-
-
2414-98-4
magnesium ethylate

-
-
67-63-0
isopropyl alcohol

-
-
5392-40-5
(E/Z)-3,7-dimethyl-2,6-octadienal

-
A
-
106-25-2
Nerol

-
B
-
106-24-1
Geraniol

-
-
555-31-7
aluminum isopropoxide

-
-
67-63-0
isopropyl alcohol

-
-
5392-40-5
(E/Z)-3,7-dimethyl-2,6-octadienal

-
A
-
106-25-2
Nerol

-
B
-
106-24-1
Geraniol

-
-
67-63-0
isopropyl alcohol

-
-
5392-40-5
(E/Z)-3,7-dimethyl-2,6-octadienal

-
A
-
106-25-2
Nerol

-
B
-
106-24-1
Geraniol

| Conditions | Yield |
|---|---|
| With magnesium ethylate | |
| With magnesium ethylate |


| Conditions | Yield |
|---|---|
| beim Erhitzen; |

| Conditions | Yield |
|---|---|
| bis(acetylacetonate)oxovanadium; bis-trimethylsilanyl peroxide In dichloromethane at 25℃; for 12h; Product distribution; | A 5 % Chromat. B 8 % Chromat. C 2 % Chromat. |
| With dodecansulfonic acid; water at 25℃; Rate constant; other acids, other solvent and temperature; |

-
-
70473-50-6
2-(phenylthio)-3-methyl-3-buten-1-ol

-
-
870-63-3
prenyl bromide

-
A
-
106-25-2
Nerol

-
B
-
106-24-1
Geraniol

-
C
-
58461-27-1
(+/-)-lavandulol

| Conditions | Yield |
|---|---|
| With hexamethyldistannane In benzene at 25℃; for 5h; Irradiation; Yield given. Yields of byproduct given. Title compound not separated from byproducts; |


| Conditions | Yield |
|---|---|
| With indium; oxygen 1.) DMF, 1 h, RT, 2.) room temperature, 3 h; Yield given. Multistep reaction. Yields of byproduct given; |
-
-
5392-40-5
(E/Z)-3,7-dimethyl-2,6-octadienal

-
A
-
106-22-9
Citronellol

-
B
-
106-23-0, 26489-02-1
3,7-dimethyl-oct-6-enal

-
C
-
106-25-2
Nerol

-
D
-
106-24-1
Geraniol

| Conditions | Yield |
|---|---|
| With methanol; samarium diiodide In tetrahydrofuran for 24h; Ambient temperature; Yields of byproduct given. Title compound not separated from byproducts; | A 18 % Chromat. B 8 % Chromat. C n/a D n/a |
| With methanol; samarium diiodide In tetrahydrofuran for 24h; Ambient temperature; Yields of byproduct given. Title compound not separated from byproducts; | |
| With hydrogen; MgCo6Ge6 In hexane at 174.84℃; under 56255.6 Torr; for 2.5h; Product distribution; Further Variations:; Catalysts; Pressures; time; |


| Conditions | Yield |
|---|---|
| With aluminum oxide; borane pyridine complex In cyclohexane for 10h; Ambient temperature; Yield given. Yields of byproduct given. Title compound not separated from byproducts; | |
| With carbon monoxide; water; hexarhodium hexadecacarbonyl; N,N,N'N'-tetramethyl-1,3-propanediamine In 2-ethoxy-ethanol at 30℃; under 7600 Torr; for 48h; | A 6 % Chromat. B 94 % Chromat. |
| With formic acid In water; N,N-dimethyl-formamide Product distribution; the effect of the nature and concentration of the acid on the yield and ratio of the products of electrochemical hydrogenation, other reagent; | |
| With sodium tetrahydroborate; ErCpCl2(THF)3 In methanol for 6h; Ambient temperature; Yield given. Yields of byproduct given. Title compound not separated from byproducts; |
-
-
5392-40-5
(E/Z)-3,7-dimethyl-2,6-octadienal

-
A
-
106-22-9
Citronellol

-
B
-
106-25-2
Nerol

-
C
-
106-24-1
Geraniol


| Conditions | Yield |
|---|---|
| With oxygen; Pd561phen60(OAc)180 In acetic acid; benzene at 60℃; for 24h; | 100% |
| With 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical; [bis(acetoxy)iodo]benzene In acetonitrile at 0℃; for 3h; pH=7; Inert atmosphere; | 100% |
| With 4-acetylamino-2,2,6,6-tetramethylpiperidine-1-oxoammonium perchlorate In dichloromethane | 99% |

| Conditions | Yield |
|---|---|
| With pyridine; dmap | 100% |
| With pyridine; dmap for 18h; | 100% |
| With dmap; triethylamine In dichloromethane at 0℃; for 0.333333h; Inert atmosphere; | 98% |

| Conditions | Yield |
|---|---|
| With pyridine In dichloromethane for 0.666667h; | 100% |
| With pyridine In dichloromethane at 0℃; Inert atmosphere; | 100% |
| With dmap; triethylamine at 0℃; for 3h; | 95% |
| With N,N-dimethyl-aniline In diethyl ether for 2h; Heating; | 90% |
-
-
106-25-2
Nerol

-
-
58479-61-1
tert-butylchlorodiphenylsilane

-
-
139109-03-8
(Z)-1-[(tert-butyldiphenylsilyl)oxy]-3,7-dimethyl-2,6-octadiene

| Conditions | Yield |
|---|---|
| With 1H-imidazole In N,N-dimethyl-formamide for 0.25h; Ambient temperature; | 100% |
| With 1H-imidazole In N,N-dimethyl-formamide | 99% |
| With 1H-imidazole In N,N-dimethyl-formamide at 20℃; for 16h; | 98% |
| With 1H-imidazole In N,N-dimethyl-formamide at 20℃; for 16h; | 98% |
| With 1H-imidazole In dichloromethane; N,N-dimethyl-formamide for 3h; Ambient temperature; | 95% |
-
-
106-25-2
Nerol

-
-
50727-94-1
[3-methyl-3-(4-methylpent-3-enyl)oxiran-2-yl]methanol

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; bis(acetylacetonate)oxovanadium In chlorobenzene at 80℃; for 5h; | 100% |
| With dihydrogen peroxide; Sucrose; molybdic acid In phosphate buffer at 2℃; for 48h; pH=7; | 92% |
| With tert.-butylhydroperoxide; bis(acetylacetonate)oxovanadium In decane; dichloromethane at 20℃; for 1.5h; Inert atmosphere; | 75% |

| Conditions | Yield |
|---|---|
| With tetra-(n-butyl)ammonium iodide; potassium hydroxide at 20℃; for 18h; | 100% |
| With sodium hydride In tetrahydrofuran |

| Conditions | Yield |
|---|---|
| With hydrogen; aluminum oxide; copper In isopropyl alcohol at 90℃; for 12h; | 100% |
| With dichloro(η3:η2:η3-dodeca-2,6,10-triene-1,12-diyl)ruthenium(IV); caesium carbonate; isopropyl alcohol at 82℃; for 23h; Inert atmosphere; chemoselective reaction; | 95 %Chromat. |

| Conditions | Yield |
|---|---|
| With di-isopropyl azodicarboxylate; triphenylphosphine In tetrahydrofuran; toluene at 0℃; for 1h; Inert atmosphere; | 100% |

| Conditions | Yield |
|---|---|
| With Candida antarctica lipase B at 50℃; for 4h; Molecular sieve; Ionic liquid; Green chemistry; Enzymatic reaction; | 99.9% |
| With N,N′,N′′,N′′′-hexadecyltrimethylammonium bis(trifluoromethylsulfonyl)imide; Novozym 435 (immobilized Candida antarctica lipase B) at 50℃; for 4h; Green chemistry; Enzymatic reaction; | |
| at 59.84℃; for 3h; | |
| With dmap; dicyclohexyl-carbodiimide In dichloromethane Steglich Esterification; |

| Conditions | Yield |
|---|---|
| With Candida antarctica lipase B at 50℃; for 4h; Molecular sieve; Ionic liquid; Green chemistry; Enzymatic reaction; | 99.9% |
| With dmap; dicyclohexyl-carbodiimide In dichloromethane Steglich Esterification; |
-
-
106-25-2
Nerol

-
-
20536-36-1
neryl chloride

| Conditions | Yield |
|---|---|
| With N-chloro-succinimide; dimethylsulfide In dichloromethane at -40 - 0℃; for 2h; | 99% |
| With chloro-trimethyl-silane; potassium carbonate In Petroleum ether at 0℃; for 2h; | 93% |
| With dimethylsulfide; methanesulfonyl chloride; lithium chloride In N,N-dimethyl-formamide at 0 - 20℃; for 0.75h; Inert atmosphere; | 90% |
-
-
106-25-2
Nerol

-
-
25996-10-5
(Z)-neryl bromide

| Conditions | Yield |
|---|---|
| With carbon tetrabromide; triphenylphosphine In benzene at 0℃; for 2h; | 99% |
| With carbon tetrabromide; triphenylphosphine In benzene at 0℃; for 2h; | 99% |
| With 1H-imidazole; iodine In dichloromethane at 20℃; for 2h; | 91% |
-
-
75-11-6
diiodomethane

-
-
106-25-2
Nerol

-
-
97231-34-0, 98678-70-7, 109801-02-7, 109801-03-8
<(1RS,2RS)-2-methyl-2-(4'-methyl-3'-pentenyl)cyclopropyl>methanol

| Conditions | Yield |
|---|---|
| With samarium In tetrahydrofuran at -78 - 20℃; | 99% |
-
-
106-25-2
Nerol

-
-
4648-54-8
trimethylsilylazide

-
-
72237-31-1
O-(cis-3,7-dimethylocta-2,6-dien-1-yl)trimethylsilane

| Conditions | Yield |
|---|---|
| tetrabutylammomium bromide at 30℃; for 0.00333333h; | 99% |

| Conditions | Yield |
|---|---|
| With Candida antarctica lipase B at 50℃; for 4h; Molecular sieve; Ionic liquid; Green chemistry; Enzymatic reaction; | 98.7% |
| With dmap; dicyclohexyl-carbodiimide In dichloromethane Steglich Esterification; |

| Conditions | Yield |
|---|---|
| With bis(pentafluorophenyl)borinic acid; magnesium sulfate; pivalaldehyde In toluene for 3h; Ambient temperature; | 98% |
| With Ba In dichloromethane for 48h; | 95% |
| With 4-acetylamino-2,2,6,6-tetramethylpiperidine-N-oxyl; toluene-4-sulfonic acid In dichloromethane 1) 0 deg C, 1 h, 2) r.t., 2 h; | 91% |
-
-
106-25-2
Nerol

-
-
102699-81-0, 103583-58-0
(E)-3-Methyl-oct-2-en-6-yn-1-ol

| Conditions | Yield |
|---|---|
| With sodium nitrite In water; acetic acid 1.) 0 deg C 2.) 60 deg C, 0.5 h; | 98% |

| Conditions | Yield |
|---|---|
| With pyridine In dichloromethane at -78 - 20℃; for 1.08333h; Inert atmosphere; Schlenk technique; Cooling with acetone-dry ice; | 98% |
| With pyridine In dichloromethane at -78℃; Inert atmosphere; |

| Conditions | Yield |
|---|---|
| With Candida antarctica lipase B at 50℃; for 4h; Molecular sieve; Ionic liquid; Green chemistry; Enzymatic reaction; | 97.8% |
-
-
106-25-2
Nerol

-
-
882-33-7
diphenyldisulfane

-
-
31162-76-2, 35162-74-4, 35162-79-9
(cis-3,7-Dimethyl-2,6-octadienyl) phenyl sulfide

| Conditions | Yield |
|---|---|
| With tributylphosphine In tetrahydrofuran for 2h; Ambient temperature; | 97.2% |
-
-
106-25-2
Nerol

-
-
593-71-5
Chloroiodomethane

-
-
499155-10-1
cis-1-(hydroxymethyl)-2-methyl-2-(4-methyl-3-pentenyl)cyclopropane

| Conditions | Yield |
|---|---|
| With samarium; mercury dichloride In tetrahydrofuran at -78 - 20℃; | 97% |

| Conditions | Yield |
|---|---|
| With tetra-(n-butyl)ammonium iodide; sodium hydride In N,N-dimethyl-formamide at 0 - 20℃; for 7h; | 97% |
| Stage #1: Nerol With sodium hydride In N,N-dimethyl-formamide at 20℃; for 1h; Stage #2: p-methoxybenzyl chloride With tetra-(n-butyl)ammonium iodide In N,N-dimethyl-formamide at 0 - 20℃; for 7h; |

| Conditions | Yield |
|---|---|
| With dmap; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In dichloromethane at 22℃; for 1h; Inert atmosphere; | 97% |
-
-
106-25-2
Nerol

-
-
106-96-7
propargyl bromide

-
-
33470-50-7
(Z)-3,7-dimethyl-1-(prop-2-yn-1-yloxy)octa-2,6-diene

| Conditions | Yield |
|---|---|
| Stage #1: Nerol With sodium hydride In tetrahydrofuran at 40℃; for 15h; Inert atmosphere; Stage #2: propargyl bromide In tetrahydrofuran at 40℃; for 7h; Inert atmosphere; | 97% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide; dipotassium peroxodisulfate; nickel(II) sulphate In dichloromethane; water for 24h; Ambient temperature; Yields of byproduct given; | A 94% B n/a |
| With 6C16H36N(1+)*2Zn(2+)*4Na(1+)*[Bi2Zn2(ZnW9O34)2](14-); urea hydrogen peroxide adduct In acetonitrile at 70℃; for 1h; Ene Reaction; | A 89% B 11% |
| With bis(cyclopentadienyl)dihydrozirconium; benzaldehyde In toluene at 110℃; for 8h; | A 83% B 12% |
-
-
106-25-2
Nerol

-
-
18162-48-6
tert-butyldimethylsilyl chloride

-
-
80873-76-3
1-(tert-butyldimethylsiloxy)-3,7-dimethyl-2,6-octadiene

| Conditions | Yield |
|---|---|
| With 1H-imidazole In N,N-dimethyl-formamide for 1h; Ambient temperature; | 94% |
| With diisopropylamine In dichloromethane at 20℃; for 5h; Inert atmosphere; | 64% |
-
-
106-25-2
Nerol

-
-
62960-04-7
(+/-)-2,3-epoxygeraniol

| Conditions | Yield |
|---|---|
| With sodium hydroxide; sodium tungstate; aluminium trichloride; dihydrogen peroxide; toluene-4-sulfonic acid; magnesium chloride In methanol Ambient temperature; | 94% |
Related products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View
 Xi
Xi