Dayang Chem (Hangzhou) Co.,Ltd.
Dayangchem's R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. DayangChem can provide different quantities
Cas:16052-42-9
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquiryChemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Cas:16052-42-9
Min.Order:5 Kiloliter
FOB Price: $1.2 / 5.0
Type:Manufacturers
inquiryHenan Tianfu Chemical Co., Ltd.
Now we would like to update our product list for you, kindly check the below information: 1. Catalyst series ( such as Noble Metal Catalyst, Phosphorous ligand, etc ) 2. Pharmaceutical Intermediate ( such as diabtes series, Anti
Cas:16052-42-9
Min.Order:1 Kilogram
FOB Price: $1.0
Type:Lab/Research institutions
inquiryZhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:16052-42-9
Min.Order:1 Kilogram
FOB Price: $139.0 / 210.0
Type:Trading Company
inquiryHebei Nengqian Chemical Import and Export Co., LTD
Our advantages: 1. All inquiries will be replied within 12 hours. 2. Dedication to quality, supply & service. 3. Strictly on selecting raw materials. 4. Reasonable & competitive price, fast lead time. 5. Sample is available for your eva
Cas:16052-42-9
Min.Order:1 Kilogram
FOB Price: $9.0 / 99.0
Type:Trading Company
inquiryShandong Hanjiang Chemical Co., Ltd.
Hello, dear friend! I'm Hansen and Allen from China. Welcome to my lookchem mall! The following is a brief introduction of our company's products and services. If you are interested in our products, please contact us by emai
Cas:16052-42-9
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHenan Wentao Chemical Product Co., Ltd.
Henan Wentao Chemical Product Co.,Ltd is Located in Zhengzhou High-tech Development Zone with import and export license, We passed ISO 9001:2008 as well, Henan Wentao has developed more than 1000 compounds, which are widely used in the fields of prod
Hangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
TAIZHOU ZHENYU BIOTECHNOLOGY CO., LTD
Zhenyu biotech exported this product to many countries and regions at best price. if you are looking for the material's manufacturer or supplier in china, zhenyu biotech is your best choice. pls contact with us freely for getting detailed
Cas:16052-42-9
Min.Order:1 Kilogram
FOB Price: $2.0
Type:Lab/Research institutions
inquiryKAISA GROUP INC
1.Applied in food field.it can improve the immune system and prolong life. 2.Appliedin cosmetic field.it can improve the skin care. 3.Applied in pharmaceutical field.it can treat various dieases. 4.Our product quality assurance will make our customer
Cas:16052-42-9
Min.Order:1 Metric Ton
FOB Price: $7.0 / 8.0
Type:Trading Company
inquiryHenan Allgreen Chemical Co.,Ltd
high quality Storage:Sealed, dry, microtherm , avoid light and smell. Package:According to the demand of customer Application:Organic synthesis Transportation:by air or by sea
Xi`an Eastling Biotech Co., Ltd.
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:Foil bag; Drum; Plastic bottle Application:Pharma;Industry;Agricultural Transportation:by sea or air Port:Beijing or Guangzhou
Antimex Chemical Limied
(-)-Menthyl chlorideAppearance:detailed see specifications Storage:Keep away of light, cool place Package:25kg/drum;200kg/drum Application:It is an important raw material and intermediate used in Organic Synthesis, Pharmaceuticals, Agrochemicals and
Changchun Artel lmport and Export trade company
Supply top quality products with a reasonable price Application:api
Hangzhou Fandachem Co.,Ltd
(-)-Menthyl chloride cas 16052-42-9Appearance:white crystalline powder Storage:Store in dry, dark and ventilated place Package:25KG drum Application:intermediate Transportation:by air, by sea, by express
ZHEJIANG JIUZHOU CHEM CO.,LTD
factory?direct?saleAppearance:White powder Storage:Sealed and preserved Package:200/Kilograms Application:healing drugs Transportation:By sea Port:Shanghai/tianjin
Jilin haofei import and export trade Co.,Ltd
Price, service, company and transport advantage: 1. Best service, place of origin China, high quality, and reasonable price. 2. It's customers' right to choose the package (EMS, DHL, FEDEX, UPS). 3. It's customers' right
Watson International Ltd
Watson International Ltd' has a very strong R&D and technical capacity supported by FCAD's platform. The subsidiaries under FCAD Group have accumulated much know-how of different fine chemical branches. For example, Apnoke Scientific L
Cas:16052-42-9
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryShanghai united Scientific Co.,Ltd.
United Scientific Company Located in Shanghai of China , is a competitive player in the global specialty and fine chemical market. Fenghua has both the expertise and flexibility to produce a wide range of chemicals. Focusing on developing the innovat
Finetech Industry Limited
FINETECH INDUSTRY LIMITED is a LONDON based CRO company providing drug discovery & development services to worldwide clients. FINETECH INDUSTRY LIMITED supplies the (-)-MENTHYL CHLORIDE, CAS:16052-42-9 with the most competitive price and the best qua
Changzhou Foreign Trade Corp
CFTC?PharmaChem?is?a?service?based?company?in?China,?a?pharmachem?division?of?Changzhou?Foreign?Trade?Corp.,supplying?raw?materials,?intermediates,?APIs?and?fine?chemicals?for?the?pharmaceutical?and?specialty?chemical?industries?worldwide. Applicatio
Cas:16052-42-9
Min.Order:0
Negotiable
Type:Trading Company
inquiryChemical Co.Ltd
Manufacturer,strong R&D,professional team Storage:Store in a cool, dry place. Store in a tightly closed container. Package:according to your requirement Application:ZhiShang Chemical is owned by ZhiShang Group, is a professional new-type chemicals en
NovaChemistry
high purity Application:Drug intermediates Materials intermediates and active molecules
Synthetic route
-
-
2216-51-5
(-)-menthol

-
-
16052-42-9
(1R,2S,5R)-menthyl chloride

| Conditions | Yield |
|---|---|
| With hydrogenchloride; zinc(II) chloride In water at 0 - 20℃; for 48h; Schlenk technique; | 99% |
| With hydrogenchloride; zinc(II) chloride Substitution; | 92% |
| With hydrogenchloride; zinc(II) chloride at 60℃; for 12h; | 91% |
-
-
61548-81-0
(1R,2S,5R)-2-isopropyl-5-methylcyclohexyl methanesulfonate

-
-
16052-42-9
(1R,2S,5R)-menthyl chloride

| Conditions | Yield |
|---|---|
| With chloro-trimethyl-silane; iron(III)-acetylacetonate In dichloromethane at 20℃; for 3h; Inert atmosphere; | 99% |
-
-
15356-60-2
(1S,2R,5S)-(+)-menthol

-
-
16052-42-9
(1R,2S,5R)-menthyl chloride

| Conditions | Yield |
|---|---|
| With Lucas' reagent | 91% |
-
-
1082742-74-2
C22H34O7S

-
-
16052-42-9
(1R,2S,5R)-menthyl chloride

| Conditions | Yield |
|---|---|
| With titanium tetrachloride In dichloromethane at -78℃; Inert atmosphere; | 90% |
-
-
2230-82-2
menthyl tosylate

-
-
16052-42-9
(1R,2S,5R)-menthyl chloride

| Conditions | Yield |
|---|---|
| With titanium tetrachloride In dichloromethane at -78℃; Inert atmosphere; | 90% |
| With chloro-trimethyl-silane; iron(III)-acetylacetonate In dichloromethane at 20℃; for 20h; Inert atmosphere; | 75% |
-
-
117800-93-8
l-menthyl 8-quinolinesulfonate

-
-
16052-42-9
(1R,2S,5R)-menthyl chloride

| Conditions | Yield |
|---|---|
| With titanium tetrachloride In dichloromethane at -78℃; Inert atmosphere; | 90% |

| Conditions | Yield |
|---|---|
| With phosphorus pentachloride In pentane at -20℃; for 2.5h; Product distribution; | A 50% B 30% |
| With phosphorus pentachloride In pentane at -20℃; for 2.5h; | A 50% B 30% |
| With phosphorus pentachloride; iron(III) chloride In hexane at 0℃; for 1.5h; Title compound not separated from byproducts.; | |
| With N-chloro-succinimide; para-thiocresol In dichloromethane Overall yield = 80 %Spectr.; | A n/a B n/a |
| Multi-step reaction with 3 steps 1.1: diethyl ether / 1.5 h / 0 - 23 °C / Inert atmosphere 1.2: 0 - 23 °C / Inert atmosphere 2.1: caesium carbonate / tetrahydrofuran; water / 0.17 h / 23 °C / Inert atmosphere 3.1: [4,4’-bis(1,1-dimethylethyl)-2,2’-bipyridine-N1,N1‘]bis [3,5-difluoro-2-[5-(trifluoromethyl)-2-pyridinyl-N]phenyl-C]iridium(III) hexafluorophosphate; Ethyl trichloroacetate / dichloromethane / 24 h / 27 °C / Inert atmosphere; Sealed tube; Irradiation View Scheme |
-
-
72020-06-5, 80924-03-4, 97526-56-2, 123121-06-2, 128946-28-1, 128946-29-2, 128946-30-5, 128946-31-6, 134052-28-1
2-((1R,2S,5R)-2-isopropyl-5-methylcyclohexyloxy)tetrahydro-2H-pyran

-
A
-
16052-42-9
(1R,2S,5R)-menthyl chloride

-
B
-
13371-12-5
neomenthyl chloride

-
C
-
5113-93-9
(1R)-trans-p-menth-2-ene

| Conditions | Yield |
|---|---|
| With dichloromethylenedimethyliminium chloride In acetonitrile at 0℃; | A 50 % Spectr. B 45 % Spectr. C 5 % Spectr. |
| With dichloromethylenedimethyliminium chloride In dichloromethane at 0℃; | A 18 % Spectr. B 42 % Spectr. C 40 % Spectr. |

-
-
87161-57-7
(1S,2S,4R)-2-bromo-1-isopropyl-4-methylcyclohexane

-
A
-
16052-42-9
(1R,2S,5R)-menthyl chloride

-
B
-
13371-12-5
neomenthyl chloride

| Conditions | Yield |
|---|---|
| With bismuth(III) chloride In 1,2-dichloro-ethane for 2h; Heating; |

-
-
16052-42-9
(1R,2S,5R)-menthyl chloride

| Conditions | Yield |
|---|---|
| With phosphorus pentachloride; iron(III) chloride; Petroleum ether Kuehlung; |
-
-
2216-51-5
(-)-menthol

-
A
-
16052-42-9
(1R,2S,5R)-menthyl chloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: triethylamine / dichloromethane / 20 °C / Inert atmosphere 2: ferric(III) bromide; chloro-trimethyl-silane / dichloromethane / 0.17 h / 20 °C / Inert atmosphere View Scheme |
-
-
61548-81-0
(1R,2S,5R)-2-isopropyl-5-methylcyclohexyl methanesulfonate

-
A
-
16052-42-9
(1R,2S,5R)-menthyl chloride

| Conditions | Yield |
|---|---|
| With ferric(III) bromide; chloro-trimethyl-silane In dichloromethane at 20℃; for 0.166667h; Inert atmosphere; |
-
-
1170718-78-1
L-menthol chlorosulfite ester

-
-
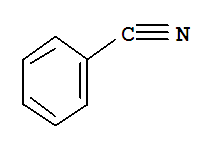
100-47-0
benzonitrile

-
A
-
16052-42-9
(1R,2S,5R)-menthyl chloride

-
B
-
1147723-25-8
(-)-N-((1R,2S,5R)-5-methyl-2-(1-methylethyl)cyclohexyl)benzamide

| Conditions | Yield |
|---|---|
| With titanium(IV) fluoride In dichloromethane at 0℃; for 2h; Inert atmosphere; |

-
-
2216-51-5
(-)-menthol

-
A
-
16052-42-9
(1R,2S,5R)-menthyl chloride

-
B
-
1147723-25-8
(-)-N-((1R,2S,5R)-5-methyl-2-(1-methylethyl)cyclohexyl)benzamide

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: thionyl chloride / dichloromethane / 1 h / Inert atmosphere; Cooling with ice 2: titanium(IV) fluoride / dichloromethane / 2 h / 0 °C / Inert atmosphere View Scheme |
-
-
1170718-78-1
L-menthol chlorosulfite ester

-
-
16052-42-9
(1R,2S,5R)-menthyl chloride

| Conditions | Yield |
|---|---|
| With titanium tetrachloride In dichloromethane at 0℃; for 0.25h; Inert atmosphere; | |
| With titanium tetrachloride at -78 - 0℃; for 1.25h; Catalytic behavior; Reagent/catalyst; |
-
-
2216-51-5
(-)-menthol

-
A
-
16052-42-9
(1R,2S,5R)-menthyl chloride

-
B
-
13371-12-5
neomenthyl chloride

-
E
-
26510-92-9
dimenthyl sulfite

| Conditions | Yield |
|---|---|
| With thionyl chloride In dichloromethane at 45℃; for 18h; |

-
-
70894-19-8
2-(((1R,2S,5R)-2-isopropyl-5-methylcyclohexyl)oxy)-2-oxoacetic acid

-
A
-
16052-42-9
(1R,2S,5R)-menthyl chloride

-
B
-
13371-12-5
neomenthyl chloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: caesium carbonate / tetrahydrofuran; water / 0.17 h / 23 °C / Inert atmosphere 2: [4,4’-bis(1,1-dimethylethyl)-2,2’-bipyridine-N1,N1‘]bis [3,5-difluoro-2-[5-(trifluoromethyl)-2-pyridinyl-N]phenyl-C]iridium(III) hexafluorophosphate; Ethyl trichloroacetate / dichloromethane / 24 h / 27 °C / Inert atmosphere; Sealed tube; Irradiation View Scheme |

-
A
-
16052-42-9
(1R,2S,5R)-menthyl chloride

-
B
-
13371-12-5
neomenthyl chloride

| Conditions | Yield |
|---|---|
| With Ethyl trichloroacetate; [4,4’-bis(1,1-dimethylethyl)-2,2’-bipyridine-N1,N1‘]bis [3,5-difluoro-2-[5-(trifluoromethyl)-2-pyridinyl-N]phenyl-C]iridium(III) hexafluorophosphate In dichloromethane at 27℃; for 24h; Inert atmosphere; Sealed tube; Irradiation; Overall yield = 50 %; Overall yield = 25.3 mg; stereoselective reaction; | A n/a B n/a |
-
-
16052-42-9
(1R,2S,5R)-menthyl chloride

-
-
63474-24-8
(1R,2S,5R)-(-)menthylmagnesium chloride

| Conditions | Yield |
|---|---|
| With ethyl bromide; magnesium In tetrahydrofuran 1.) 4 h, 50 degC, 2.) 1 h, 70 deg C; | 95% |
-
-
124-38-9
carbon dioxide

-
-
16052-42-9
(1R,2S,5R)-menthyl chloride

-
-
16052-40-7
(-)-(1R,3R,4S)-4-isopropyl-1-methylcyclohexane-3-carboxylic acid

| Conditions | Yield |
|---|---|
| Stage #1: (1R,2S,5R)-menthyl chloride With iodine; magnesium; ethylene dibromide at 70 - 75℃; for 4h; Inert atmosphere; Autoclave; Stage #2: carbon dioxide at 85 - 90℃; for 7.5h; Stage #3: With hydrogenchloride In water Temperature; | 93% |
| With manganese; 2,9-dibutyl-3,4,7,8-tetramethyl-1,10-phenanthroline; NiBr2(monoglyme)2; lithium chloride In N,N-dimethyl-formamide under 760.051 Torr; Schlenk technique; Heating; | 57% |
| Stage #1: carbon dioxide; (1R,2S,5R)-menthyl chloride With magnesium; iodine In tetrahydrofuran Stage #2: With hydrogenchloride In tetrahydrofuran; water Ice; Stage #3: With hydrogenchloride; sodium hydroxide more than 3 stages; | 40% |
-
-
16052-42-9
(1R,2S,5R)-menthyl chloride

-
-
73183-34-3
bis(pinacol)diborane

-
-
1357000-40-8
2-((1R,2R,5R)-2-isopropyl-5-methylcyclohexyl)-4,4,5,5-tetramethyl-1,3,2-dioxaborolane

| Conditions | Yield |
|---|---|
| With potassium tert-butylate; 4,5-bis(diphenylphosphino)-9,9-dimethylxanthene In tetrahydrofuran at 20℃; for 92h; Inert atmosphere; Schlenk technique; | 93% |
| With potassium tert-butylate; 4,5-bis(diphenylphosphino)-9,9-dimethylxanthene; copper(l) chloride In tetrahydrofuran at 20℃; for 48h; Inert atmosphere; | 91% |
| With potassium tert-butylate; 4,5-bis(diphenylphosphino)-9,9-dimethylxanthene; copper(l) chloride In tetrahydrofuran at 20℃; for 31h; Inert atmosphere; optical yield given as %de; diastereoselective reaction; | 85% |
-
-
83095-83-4, 14647-25-7
[platinum(II)dichloride(1,2-bis(diphenylphosphino)ethane)]

-
-
16052-42-9
(1R,2S,5R)-menthyl chloride

-
-
7439-95-4
magnesium

-
-
106-93-4
ethylene dibromide

| Conditions | Yield |
|---|---|
| In tetrahydrofuran (N2), Schlenk techniques; addn. of 1,2-dibromoethane to Mg in THF at 50°C, after 10 min addn. of menthyl chloride, heating under reflux for 5 h at 65°C, addn. to soln. of Au complex in THF, stirring for1 h at 50°C; cooling to room temp., addn. of satd. aq. NH4Cl, opening to air, extn. (CH2Cl2), drying (MgSO4), filtration (Celite), evapn. in vacuo, washing with petroleum ether, drying in vacuo, elem. anal.; | 82% |


| Conditions | Yield |
|---|---|
| With HCl; PPh3; tert-butylhydroperoxide In tetrahydrofuran byproducts: Ph3PO; stirring (room temp., 1.5 h), refluxing (30 min), HCl addn., extg. (CH2Cl2), drying (MgSO4), evapn. (vac.), hexane and CH2Cl2 addn., fitering Ph3P off, tBuOOH addn., stirring (15 min), H2O addn.; chromy. (SiO2, hexane / CH2Cl2 = 80 : 20); elem. anal.; | 73% |


| Conditions | Yield |
|---|---|
| Stage #1: (1R,2S,5R)-menthyl chloride With magnesium In tetrahydrofuran; ethylene dibromide at 60℃; for 1h; Schlenk technique; Inert atmosphere; Reflux; Stage #2: With zinc(II) chloride In tetrahydrofuran at 20℃; Negishi Coupling; Schlenk technique; Inert atmosphere; Stage #3: 2-iodo-4-methylaniline diastereoselective reaction; | 73% |
-
-
16052-42-9
(1R,2S,5R)-menthyl chloride

-
-
64662-57-3
2,6-diiodo-4-methylaniline

| Conditions | Yield |
|---|---|
| Stage #1: (1R,2S,5R)-menthyl chloride With magnesium In tetrahydrofuran at 60℃; for 1h; Negishi Coupling; Schlenk technique; Inert atmosphere; Reflux; Stage #2: With zinc(II) chloride In tetrahydrofuran at 20℃; Negishi Coupling; Inert atmosphere; Schlenk technique; Stage #3: 2,6-diiodo-4-methylaniline With dicyclohexyl-(2',6'-dimethoxybiphenyl-2-yl)-phosphane; bis(dibenzylideneacetone)-palladium(0) In tetrahydrofuran at 20℃; for 16h; Inert atmosphere; Schlenk technique; | 73% |
-
-
14243-64-2
(triphenylphosphine)gold(I) chloride

-
-
16052-42-9
(1R,2S,5R)-menthyl chloride

-
-
7439-95-4
magnesium

| Conditions | Yield |
|---|---|
| With 1,2-dibromoethane In tetrahydrofuran (N2), Schlenk techniques; addn. of 1,2-dibromoethane to Mg in THF at 50°C, after 20 min addn. of menthyl chloride, heating under reflux for 5 h at 65°C, addn. to soln. of Au complex in THF, stirring for2 h; addn. of dilute H2SO4, opening to air, extn. (CH2Cl2), drying (MgSO4), treatment with charcoal, filtration (Celite), evapn. in vacuo, washing with petroleum ether, drying in vacuo, elem. anal.; | 67% |

-
-
16052-42-9
(1R,2S,5R)-menthyl chloride

-
-
4376-01-6
sodium diphenylphosphide

-
A
-
43077-29-8
((1S,2S,5R)-2-Isopropyl-5-methyl-cyclohexyl)-diphenyl-phosphane

-
B
-
43077-30-1
neomenthyldiphenylphosphine oxide

-
C
-
829-85-6
diphenylphosphane

| Conditions | Yield |
|---|---|
| With N,N,N,N,N,N-hexamethylphosphoric triamide In tetrahydrofuran at 25 - 67℃; for 16h; | A 66% B 24% C 5% |

-
-
16052-42-9
(1R,2S,5R)-menthyl chloride

-
-
4102-54-9
2-iodo-4,6-dimethylaniline

| Conditions | Yield |
|---|---|
| Stage #1: (1R,2S,5R)-menthyl chloride With magnesium In tetrahydrofuran at 60℃; for 1h; Schlenk technique; Inert atmosphere; Reflux; Stage #2: With zinc(II) chloride In tetrahydrofuran at 20℃; Negishi Coupling; Inert atmosphere; Schlenk technique; Stage #3: 2-iodo-4,6-dimethylaniline With dicyclohexyl-(2',6'-dimethoxybiphenyl-2-yl)-phosphane; bis(dibenzylideneacetone)-palladium(0) In tetrahydrofuran at 20℃; for 18h; Inert atmosphere; Schlenk technique; diastereoselective reaction; | 64% |
-
-
124-38-9
carbon dioxide

-
-
16052-42-9
(1R,2S,5R)-menthyl chloride

-
A
-
70985-58-9
(1S,2S,5R)-2-isopropyl-5-methylcyclohexane-1-carboxylic acid

-
B
-
16052-40-7
(-)-(1R,3R,4S)-4-isopropyl-1-methylcyclohexane-3-carboxylic acid

| Conditions | Yield |
|---|---|
| Stage #1: (1R,2S,5R)-menthyl chloride With magnesium In tetrahydrofuran at 50℃; for 2h; Stage #2: carbon dioxide In tetrahydrofuran at 20℃; Inert atmosphere; Schlenk technique; | A 7.6% B 60.2% |

-
-
16052-42-9
(1R,2S,5R)-menthyl chloride

-
-
829-85-6
diphenylphosphane

-
-
43077-29-8
((1S,2S,5R)-2-Isopropyl-5-methyl-cyclohexyl)-diphenyl-phosphane

| Conditions | Yield |
|---|---|
| With cesium hydroxide; 4 Angstroem MS In N,N-dimethyl-formamide at 23℃; for 63h; | 54% |
-
-
16052-42-9
(1R,2S,5R)-menthyl chloride

-
-
4376-01-6
sodium diphenylphosphide

-
A
-
43077-29-8
((1S,2S,5R)-2-Isopropyl-5-methyl-cyclohexyl)-diphenyl-phosphane

-
B
-
829-85-6
diphenylphosphane

| Conditions | Yield |
|---|---|
| With 15-crown-5 In tetrahydrofuran at 25 - 67℃; for 16h; | A 15% B 53% |


| Conditions | Yield |
|---|---|
| With HCl In tetrahydrofuran; benzene refluxing (5 h), stirring (room temp., 16 h), HCl and H2O addn., extg. (ether), drying (MgSO4), evapn. (vac.); recrystn. (EtOH); elem. anal.; | 50% |

-
-
16052-42-9
(1R,2S,5R)-menthyl chloride

-
-
1135-99-5
diphenyltin(IV) dichloride

-
-
182160-38-9, 916576-67-5, 250636-43-2
di(-)menthyldiphenyltin

| Conditions | Yield |
|---|---|
| With magnesium In tetrahydrofuran Ar-atmosphere; treatment of menthyl chloride with Mg, dropwise addn. of Sn-compd. (0°C, over 1 h), stirring at room temp. for 48 h; dropwise addn. of water at 0°C, stirring for 30 min at room temp., ether addn., washing of org. layer (water), drying (MgSO4), solvent removal, distn. (150-180°C/0.01 Torr); elem. anal.; | 49% |

| Conditions | Yield |
|---|---|
| With magnesium In tetrahydrofuran Ar-atmosphere; treatment of menthyl chloride with Mg, dropwise addn. of Sn-compd. (0°C, over 1 h), stirring at room temp. for 48 h; dropwise addn. of water at 0°C, stirring for 30 min at room temp., drying (MgSO4), solvent removal, distn. (90°C/40 mbar); elem. anal.; | 48.2% |
-
-
124-38-9
carbon dioxide

-
-
16052-42-9
(1R,2S,5R)-menthyl chloride

-
-
70-11-1
α-bromoacetophenone

-
A
-
1537179-13-7
2-oxo-2-phenylethyl (1R,2S,5R)-2-isopropyl-5-methylcyclohexanecarboxylate

-
B
-
1537179-15-9
2-oxo-2-phenylethyl (1S,2S,5R)-2-isopropyl-5-methylcyclohexanecarboxylate

-
C
-
1537179-16-0
2-((1R,2S,5R)-2-isopropyl-5-methylcyclohexyl)-2-oxoethyl benzoate

| Conditions | Yield |
|---|---|
| Stage #1: (1R,2S,5R)-menthyl chloride With chloro-trimethyl-silane; magnesium; ethylene dibromide In tetrahydrofuran at 70℃; for 3h; Inert atmosphere; Stage #2: carbon dioxide In tetrahydrofuran; 1-methyl-pyrrolidin-2-one at 70 - 80℃; for 2h; Inert atmosphere; Stage #3: α-bromoacetophenone In tetrahydrofuran; 1-methyl-pyrrolidin-2-one at 80℃; Inert atmosphere; | A 44% B 15% C 1% |
| Stage #1: (1R,2S,5R)-menthyl chloride With chloro-trimethyl-silane; magnesium; ethylene dibromide In tetrahydrofuran at 70℃; for 3h; Inert atmosphere; Stage #2: carbon dioxide In tetrahydrofuran; 1-methyl-pyrrolidin-2-one at 70 - 80℃; for 2h; Stage #3: α-bromoacetophenone In tetrahydrofuran; 1-methyl-pyrrolidin-2-one at 80℃; | A 44% B 15% C 1% |

-
-
16052-42-9
(1R,2S,5R)-menthyl chloride

-
-
567-19-1
3-chloro-1,2-benzisothiazole 1,1-dioxide

-
A
-
18715-95-2
3-[(1'R)-menthoxy]-1,2-benzisothiazole 1,1-dioxide

| Conditions | Yield |
|---|---|
| Stage #1: (1R,2S,5R)-menthyl chloride With ethyl bromide; magnesium In tetrahydrofuran for 0.5h; Metallation; Heating; Stage #2: 3-chloro-1,2-benzisothiazole 1,1-dioxide In tetrahydrofuran at 0℃; Grignard reaction; Further stages.; | A 15% B 38% C 13% |

-
-
16052-42-9
(1R,2S,5R)-menthyl chloride

-
A
-
619-52-3
(1R)-menth-3-ene

-
B
-
135637-19-3
(+)-Menthyllithium

-
C
-
3294-50-6
(2R,5S,2'S,5'R)-2,2'-Diisopropyl-5,5'-dimethyl-bicyclohexyl

| Conditions | Yield |
|---|---|
| With lithium In pentane 1) 2h, reflux, 2) 48h; | A 15.6% B 37% C n/a |

-
-
16052-42-9
(1R,2S,5R)-menthyl chloride

-
-
201733-56-4
2-(5,5-dimethyl-1,3,2-dioxaborinan-2-yl)-5,5-dimethyl-1,3,2-dioxaborinane

| Conditions | Yield |
|---|---|
| With potassium tert-butylate; 4,5-bis(diphenylphosphino)-9,9-dimethylxanthene; copper(l) chloride In tetrahydrofuran for 24h; Inert atmosphere; | 28% |
-
-
16052-42-9
(1R,2S,5R)-menthyl chloride

-
-
65567-06-8, 4541-02-0
lithium diphenylphosphide

-
-
43077-29-8
((1S,2S,5R)-2-Isopropyl-5-methyl-cyclohexyl)-diphenyl-phosphane

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 40℃; for 40h; | 24% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran stirring (room temp., 44 h), H2O addn., extg. (ether), drying (MgSO4), evapn. (vac.); chromy. (SiO2, hexane / CH2Cl2 = 97 : 3), evapn.; elem. anal.; | 22% |

-
-
16052-42-9
(1R,2S,5R)-menthyl chloride

-
-
4376-01-6
sodium diphenylphosphide

-
-
43077-29-8
((1S,2S,5R)-2-Isopropyl-5-methyl-cyclohexyl)-diphenyl-phosphane

| Conditions | Yield |
|---|---|
| With 15-crown-5 In tetrahydrofuran at 25 - 67℃; for 16h; | 15% |
Related products
Raw Materials
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View