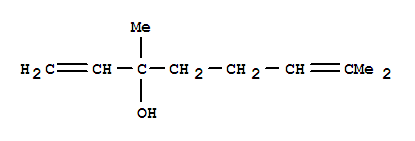Dayang Chem (Hangzhou) Co.,Ltd.
As a leading manufacturer and supplier of chemicals in China, DayangChem not only supply popular chemicals, but also DayangChem's R&D center offer custom synthesis services. DayangChem can provide different quantities of custom synthesis ch
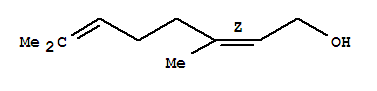
Cas:3779-61-1
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquiryShanghai Upbio Tech Co.,Ltd
1.In No Less 10 years exporting experience. you can 100% received goods 2.Lower Price with higher quality 3,Free sample 4,We are sincerely responsible for the "product quality" and "After Service" Upbio is Specializ
Cas:3779-61-1
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryQingdao Beluga Import and Export Co., LTD
(E)-3,7-dimethylocta-1,3,6-triene CAS:3779-61-1 Qingdao Belugas Import and Export Co., Ltd. is a scientific and technological company integrating research and development, production and trade of chemical intermediates, specializing in high quality
Cas:3779-61-1
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryShandong Hanjiang Chemical Co., Ltd.
Hello, dear friend! I'm Hansen and Allen from China. Welcome to my lookchem mall! The following is a brief introduction of our company's products and services. If you are interested in our products, please contact us by emai
Cas:3779-61-1
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHenan Wentao Chemical Product Co., Ltd.
Henan Wentao Chemical Product Co.,Ltd is Located in Zhengzhou High-tech Development Zone with import and export license, We passed ISO 9001:2008 as well, Henan Wentao has developed more than 1000 compounds, which are widely used in the fields of prod
Cas:3779-61-1
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryZibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:3779-61-1
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquiryHangzhou Huarong Pharm Co., Ltd.
Hangzhou Huarong Pharm Co., Ltd.established since 2006 , has been actively developing specialty products for Finished Dosages, APIs, Intermediates, and Fine chemicals markets in North America, Europe, Korea, Japan, Mid-East and all over the World. Hu
Hangzhou J&H Chemical Co., Ltd.
J&H CHEM is one of China's leading providers of integrated fine chemical services including offering, research and development, Custom manufacturing business, as well as other Value-added customer services, for diversified range products of chemicals
Cas:3779-61-1
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryHenan Tianfu Chemical Co., Ltd.
1.Our services:A.Supply sampleB.The packing also can be according the customers` requirmentC.Any inquiries will be replied within 24 hoursD.we provide Commerical Invoice, Packing List, Bill of loading, COA , Health certificate and Origin certificate.
Cas:3779-61-1
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryAntimex Chemical Limied
Ansciep Chemical is a professional enterprise manufacturing and distributing fine chemicals and speciality chemicals. We have been dedicated to heterocycle compounds and phenyl rings for tens of years. This is our mature product for export. Our quali
Cas:3779-61-1
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryHenan Kanbei Chemical Co.,LTD
factory?direct?saleAppearance:White powder Storage:Sealed and preserved Package:200/Kilograms Application:healing drugs Transportation:By sea Port:Shanghai/tianjin
Cas:3779-61-1
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryShanghai Chinqesen Biotechnology Co., Ltd.
Good Quality Package:1kg/bag Application:Medical or chemical Transportation:Air/Train/Sea Port:Shenzhen
Shanghai Yuanye Bio-Technology Co., Ltd.
good quality, competitive price, thoughtful after sale serviceAppearance:white powder Storage:Keep it in dry,shady and cool place Package:10mg Application:Pharma;Industry;Agricultural;chemical reaserch Transportation:by express or by sea Port:Any por
ZHEJIANG JIUZHOU CHEM CO.,LTD
factory?direct?saleAppearance:White powder Storage:Sealed and preserved Package:200/Kilograms Application:healing drugs Transportation:By sea Port:Shanghai/tianjin
Nanjing Raymon Biotech Co., Ltd.
1,3,6-Octatriene,3,7-dimethyl-, (3E)-Appearance:Off white to slight yellow solid Storage:Store in dry and cool condition Package:25kg or according to cutomer's demand Application:Chemical research/Pharmaceutical intermediates Transportation:By Sea,by
Cas:3779-61-1
Min.Order:0
Negotiable
Type:Trading Company
inquiryHebei Ruishun Trade Co.,Ltd
Supply top quality products with a reasonable price Application:api
Chemical Co.Ltd
1,3,6-Octatriene,3,7-dimethyl-, (3E)-Appearance:Off white to slight yellow solid Storage:Stored in shaded, cool and dry places Package:1L 5L 10L 25L bottle Application:pharma intermediate Transportation:Handle with cares to avoid damaging the package
Cas:3779-61-1
Min.Order:0
Negotiable
Type:Trading Company
inquirySynthetic route
-
-
94987-59-4
2-methyl-4-(3-methyl-3-sulfolen-2-yl)-2-butene

-
-
3779-61-1
trans ocimene

| Conditions | Yield |
|---|---|
| In pyridine at 125℃; for 7h; | 85% |
-
-
53776-92-4
(E)-3-methyl-5-bromopenta-1,3-diene

-
-
38614-36-7
2-methylpropen-1-ylmagnesium bromide

-
-
3779-61-1
trans ocimene

| Conditions | Yield |
|---|---|
| With dilithium tetrachlorocuprate In tetrahydrofuran; 1,2-dimethoxyethane at 0 - 20℃; for 18.75h; Grignard reaction; optical yield given as %de; | 68% |
-
-
106-24-1
Geraniol

-
A
-
78-70-6
3,7-dimethylocta-1,6-dien-3-ol

-
B
-
3338-55-4
(Z)-ocimene

-
C
-
3779-61-1
trans ocimene

| Conditions | Yield |
|---|---|
| With germacrene A synthase from Nostoc sp. PCC7120 (NS1) In terpene synthase buffer at 25℃; for 18h; Enzymatic reaction; | A 66% B 25% C 9% |


| Conditions | Yield |
|---|---|
| With sesquiterpene synthases Cop4 from Coprinus cinereus In terpene synthase buffer at 25℃; for 18h; Enzymatic reaction; | A 61% B 9.1% C 23.7% |


-
A
-
123-35-3
7-methyl-3-methene-1,6-octadiene

-
B
-
3338-55-4
(Z)-ocimene

-
C
-
3779-61-1
trans ocimene

-
D
-
2885-58-7
3,5,7-trimethyltropolone

| Conditions | Yield |
|---|---|
| at 180℃; pyrolysis; | A 30% B 8% C 49% D n/a |
| at 140℃; pyrolysis; | A 23% B 12% C 36% D n/a |

-
-
106-24-1
Geraniol

-
A
-
78-70-6
3,7-dimethylocta-1,6-dien-3-ol

-
B
-
3338-55-4
(Z)-ocimene

-
C
-
3779-61-1
trans ocimene

-
D
-
138-86-3
limonene.

| Conditions | Yield |
|---|---|
| With sesquiterpene synthases Cop6 from Coprinus cinereus In terpene synthase buffer at 25℃; for 18h; Enzymatic reaction; | A 34.6% B 9.2% C 7.11% D 45% |


| Conditions | Yield |
|---|---|
| at 140℃; | A 29% B 9% C 27% |
| at 140℃; pyrolysis; | A 29% B 9% C 27% |

-
-
106-24-1
Geraniol

-
A
-
123-35-3
7-methyl-3-methene-1,6-octadiene

-
B
-
3338-55-4
(Z)-ocimene

-
C
-
3779-61-1
trans ocimene

-
D
-
586-62-9
Terpinolene

-
E
-
138-86-3
limonene.

| Conditions | Yield |
|---|---|
| With tin(ll) chloride In dimethyl sulfoxide at 139.84℃; for 2h; | A 27% B 24% C 17% D 10% E 22% |


| Conditions | Yield |
|---|---|
| at 180℃; for 15h; Further byproducts given; | A 15% B 6% C 19% D n/a |
| at 180℃; for 0.25h; pyrolysis; Further byproducts given; | A 15% B 6% C 19% D n/a |
| at 180℃; for 15h; pyrolysis; Further byproducts given; | A 15 % Chromat. B 6 % Chromat. C 19 % Chromat. D n/a |

-
-
105-87-3
3,7-dimethyl-2E,6-octadien-1-yl acetate

-
A
-
123-35-3
7-methyl-3-methene-1,6-octadiene

-
B
-
3338-55-4
(Z)-ocimene

-
C
-
3779-61-1
trans ocimene

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); triethylamine In tetrahydrofuran for 48h; Product distribution; Ambient temperature; other dienylacetate, var. basic reagents; | A 41 % Chromat. B n/a C n/a |
| With tetrakis(triphenylphosphine) palladium(0) In tetrahydrofuran for 16h; Inert atmosphere; |


| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); 2-Propynylzinc bromide In tetrahydrofuran for 3h; Ambient temperature; Yield given. Yields of byproduct given. Title compound not separated from byproducts; |
-
-
562-74-3
TERPINEN-4-OL

-
A
-
3338-55-4
(Z)-ocimene

-
B
-
3779-61-1
trans ocimene

-
C
-
99-87-6
4-methylisopropylbenzene

-
D
-
586-82-3
(+/-)-4-isopropyl-1-methyl-3-cyclohexen-1-ol

-
E
-
470-67-7
4-methyl-1-propan-2-yl-7-oxabicyclo[2.2.1]heptane

| Conditions | Yield |
|---|---|
| With abietic acid In water at 100℃; Product distribution; sealed tube; | A n/a B n/a C 25 % Chromat. D 24 % Chromat. E 11 % Chromat. F 17 % Chromat. |

-
-
82859-38-9
(2E)-1-iodo-3,7-dimethylocta-2,6-diene

-
A
-
123-35-3
7-methyl-3-methene-1,6-octadiene

-
B
-
3338-55-4
(Z)-ocimene

-
C
-
3779-61-1
trans ocimene

-
D
-
138-86-3
limonene.

| Conditions | Yield |
|---|---|
| With triethylamine In hexane at 25℃; for 6h; Irradiation; Yield given. Yields of byproduct given; | |
| With triethylamine In hexane at 25℃; for 6h; Product distribution; Irradiation; |

-
-
89111-64-8
neryl iodide

-
A
-
123-35-3
7-methyl-3-methene-1,6-octadiene

-
B
-
3338-55-4
(Z)-ocimene

-
C
-
3779-61-1
trans ocimene

-
D
-
586-62-9
Terpinolene

-
E
-
138-86-3
limonene.

| Conditions | Yield |
|---|---|
| With triethylamine In hexane at 25℃; for 6h; Product distribution; Irradiation; | |
| With triethylamine In tetrahydrofuran at 50℃; for 2h; Product distribution; Irradiation; also in the presence of CuCl; similar product distribution by photoirradiation of geranyl iodide; |

-
-
89111-64-8
neryl iodide

-
A
-
123-35-3
7-methyl-3-methene-1,6-octadiene

-
B
-
3779-61-1
trans ocimene

-
C
-
586-62-9
Terpinolene

-
D
-
138-86-3
limonene.

| Conditions | Yield |
|---|---|
| With triethylamine In hexane at 25℃; for 6h; Irradiation; Yield given. Yields of byproduct given; |


| Conditions | Yield |
|---|---|
| Pd(0)-bis(diphenylphosphino)ethane In N,N-dimethyl-formamide at 120℃; for 5h; Yield given. Yields of byproduct given; |


| Conditions | Yield |
|---|---|
| Pd(0)-bis(diphenylphosphino)ethane In N,N-dimethyl-formamide at 120℃; for 5h; Yield given. Yields of byproduct given; |

-
-
115-95-7
linalool acetate

-
A
-
123-35-3
7-methyl-3-methene-1,6-octadiene

-
B
-
3338-55-4
(Z)-ocimene

-
C
-
3779-61-1
trans ocimene

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0) In tetrahydrofuran Product distribution; Mechanism; other allylic acetates; var. solv.; also in presence var. bases and organometallic reagents; kinetic isotope effect; |

-
-
106-24-1
Geraniol

-
A
-
78-70-6
3,7-dimethylocta-1,6-dien-3-ol

-
B
-
123-35-3
7-methyl-3-methene-1,6-octadiene

-
C
-
3779-61-1
trans ocimene

-
D
-
98-55-5
terpineol

| Conditions | Yield |
|---|---|
| With sulfuric acid; potassium tartrate In ethanol at 50℃; for 4h; pH=3; Product distribution; Further Variations:; pH-values; Reaction partners; Reagents; Temperatures; Hydrolysis; |


| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 82 percent 2: NaI / acetone / 1 h / 0 - 2 °C 3: triethylamine / hexane / 6 h / 25 °C / Irradiation View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 86 percent 2: NaI / acetone / 1 h / 0 - 2 °C 3: triethylamine / hexane / 6 h / 25 °C / Irradiation View Scheme |
-
-
20536-36-1
neryl chloride

-
-
3779-61-1
trans ocimene

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: NaI / acetone / 1 h / 0 - 2 °C 2: triethylamine / hexane / 6 h / 25 °C / Irradiation View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: NaI / acetone / 1 h / 0 - 2 °C 2: triethylamine / hexane / 6 h / 25 °C / Irradiation View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 2: 82 percent 3: NaI / acetone / 1 h / 0 - 2 °C 4: triethylamine / hexane / 6 h / 25 °C / Irradiation View Scheme | |
| Multi-step reaction with 4 steps 2: 86 percent 3: NaI / acetone / 1 h / 0 - 2 °C 4: triethylamine / hexane / 6 h / 25 °C / Irradiation View Scheme |

| Conditions | Yield |
|---|---|
| at 336.84℃; Kinetics; Mechanism; Temperature; |

-
-
62054-49-3
ethyl (E)-3-formyl-2-butenoate

-
-
3779-61-1
trans ocimene

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1.1: n-butyllithium / tetrahydrofuran; hexane / -78 - 0 °C 1.2: -78 - 20 °C 2.1: lithium aluminium tetrahydride / diethyl ether / 1 h / Inert atmosphere; Reflux 3.1: phosphorus tribromide / diethyl ether / 1 h / 0 °C / Inert atmosphere 4.1: dilithium tetrachlorocuprate / tetrahydrofuran; 1,2-dimethoxyethane / 18.75 h / 0 - 20 °C View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: phosphorus tribromide / diethyl ether / 1 h / 0 °C / Inert atmosphere 2: dilithium tetrachlorocuprate / tetrahydrofuran; 1,2-dimethoxyethane / 18.75 h / 0 - 20 °C View Scheme |
-
-
37850-26-3
methyl-3 pentadiene-2,4 oate d'ethyle E

-
-
3779-61-1
trans ocimene

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: lithium aluminium tetrahydride / diethyl ether / 1 h / Inert atmosphere; Reflux 2: phosphorus tribromide / diethyl ether / 1 h / 0 °C / Inert atmosphere 3: dilithium tetrachlorocuprate / tetrahydrofuran; 1,2-dimethoxyethane / 18.75 h / 0 - 20 °C View Scheme |
-
-
763-10-0
geranyl diphosphate

-
A
-
99-83-2
p-mentha-1,5-diene

-
B
-
123-35-3
7-methyl-3-methene-1,6-octadiene

-
C
-
3338-55-4
(Z)-ocimene

-
D
-
3779-61-1
trans ocimene

-
E
-
586-62-9
Terpinolene

-
F
-
138-86-3
limonene.

| Conditions | Yield |
|---|---|
| With recombinant (+)-cadinene synthase from Gossypium arboreum; magnesium chloride; D,L-dithiothreitol In pentane at 25℃; for 24h; pH=7.5; HEPES buffer; Enzymatic reaction; |

-
-
3779-61-1
trans ocimene

-
-
20426-48-6
(E)-1-(3-chlorophenyl)-3-phenylprop-2-en-1-one

| Conditions | Yield |
|---|---|
| With aluminum tri-bromide In toluene at 28℃; for 3h; Diels-Alder Cycloaddition; Inert atmosphere; diastereoselective reaction; | 74% |

| Conditions | Yield |
|---|---|
| With aluminum tri-bromide In toluene at 28℃; for 24h; Reagent/catalyst; Temperature; Diels-Alder Cycloaddition; Inert atmosphere; diastereoselective reaction; | 72% |
-
-
3779-61-1
trans ocimene

-
-
156723-02-3
(+/-)-12-hydroxy-9(10->20)-5aH-abeo-abieta-1(10),8(9),12(13)-triene-11,14-dione

| Conditions | Yield |
|---|---|
| With boron trifluoride diethyl etherate In toluene at 0 - 50℃; for 11.5h; Inert atmosphere; | 71% |

| Conditions | Yield |
|---|---|
| With aluminum tri-bromide In dichloromethane at 28℃; for 3h; Diels-Alder Cycloaddition; Inert atmosphere; diastereoselective reaction; | 71% |
-
-
3779-61-1
trans ocimene

-
-
80783-99-9, 124931-15-3, 113474-86-5
N-methoxy-N-methylcinnamamide

| Conditions | Yield |
|---|---|
| In neat (no solvent) at 140℃; for 72h; Diels-Alder Cycloaddition; Inert atmosphere; Sealed tube; regioselective reaction; | 70% |
-
-
3779-61-1
trans ocimene

-
-
22966-22-9
(E)-1-(4-chlorophenyl)-3-phenyl-2-propen-1-one

| Conditions | Yield |
|---|---|
| With aluminum tri-bromide In toluene at 28℃; for 3h; Diels-Alder Cycloaddition; Inert atmosphere; diastereoselective reaction; | 70% |
-
-
3779-61-1
trans ocimene

-
-
24721-26-4, 956-04-7, 22252-16-0
4-chlorochalcone

| Conditions | Yield |
|---|---|
| With aluminum tri-bromide In toluene at 28℃; for 3h; Diels-Alder Cycloaddition; Inert atmosphere; diastereoselective reaction; | 69% |
-
-
3779-61-1
trans ocimene

-
-
75927-49-0
pinacol vinylboronate

| Conditions | Yield |
|---|---|
| With (η6-naphthalene)(η4-1,5-cyclooctadiene)ruthenium(0) In benzene at 30℃; for 1h; Inert atmosphere; Schlenk technique; | 67% |
-
-
3779-61-1
trans ocimene

| Conditions | Yield |
|---|---|
| With aluminum tri-bromide In toluene at 28℃; for 3h; Diels-Alder Cycloaddition; Inert atmosphere; diastereoselective reaction; | 64% |
-
-
3779-61-1
trans ocimene

-
-
14802-30-3
(E)-1-(4-methylphenyl)-3-phenyl-2-propen-1-one

| Conditions | Yield |
|---|---|
| With aluminum tri-bromide In toluene at 28℃; for 3h; Diels-Alder Cycloaddition; Inert atmosphere; diastereoselective reaction; | 62% |
-
-
3779-61-1
trans ocimene

-
-
22966-09-2
(2E)-3-(4-bromophenyl)-1-phenylprop-2-en-1-one

| Conditions | Yield |
|---|---|
| With aluminum tri-bromide In toluene at 28℃; for 3h; Diels-Alder Cycloaddition; Inert atmosphere; diastereoselective reaction; | 62% |
-
-
3779-61-1
trans ocimene

-
-
142132-39-6
(+)-perovskone

| Conditions | Yield |
|---|---|
| With boron trifluoride diethyl etherate In dichloromethane at 0 - 50℃; for 11.5h; Inert atmosphere; | 57% |
-
-
3779-61-1
trans ocimene

-
-
72758-69-1, 2403-27-2, 22966-23-0
(E)-1-(4-bromophenyl)-3-phenylprop-2-en-1-one

| Conditions | Yield |
|---|---|
| With aluminum tri-bromide In toluene at 28℃; for 3h; Diels-Alder Cycloaddition; Inert atmosphere; diastereoselective reaction; | 56% |

| Conditions | Yield |
|---|---|
| With 1,8-diazabicyclo[5.4.0]undec-7-ene In toluene at 150℃; for 5h; Inert atmosphere; Sealed tube; | 55% |
| In toluene at 150℃; for 5h; Inert atmosphere; Sealed tube; | 55% |
-
-
21264-30-2, 90243-59-7, 14592-56-4
dichloro bis(acetonitrile) palladium(II)

-
-
3779-61-1
trans ocimene

| Conditions | Yield |
|---|---|
| With lithium acetate; acetic acid In N,N,N,N,N,N-hexamethylphosphoric triamide a soln. of (MeCN)2PdCl2 in HMPA followed by trans-ocimene was added under N2 to a soln. of LiOAc*2H2O in AcOH and HMPA (total HMPA:water=10/1 v/v), mixt. was stirred at room temp. for 8 h; reaction mixt. was extd. with benzene, ext. washed with water, dried over CaCl2, solvent was removed under reduced pressure, residue purified by column chromy. on SiO2 with 10/3 benzene-EtOAc; elem. anal.; | 54% |

| Conditions | Yield |
|---|---|
| In toluene at 150℃; for 7h; Diels-Alder Cycloaddition; Inert atmosphere; Sealed tube; stereoselective reaction; | 54% |

| Conditions | Yield |
|---|---|
| In toluene at 150℃; for 2h; Inert atmosphere; Sealed tube; | 52% |
| In toluene at 150℃; for 2h; Inert atmosphere; Sealed tube; | 52% |
-
-
3779-61-1
trans ocimene

-
-
22966-24-1
(E)-1-(3-methoxyphenyl)-3-phenylprop-2-en-1-one

| Conditions | Yield |
|---|---|
| With aluminum tri-bromide In dichloromethane at 28℃; for 6h; Diels-Alder Cycloaddition; Inert atmosphere; diastereoselective reaction; | 52% |
-
-
3779-61-1
trans ocimene

-
-
156723-02-3
(+/-)-12-hydroxy-9(10->20)-5aH-abeo-abieta-1(10),8(9),12(13)-triene-11,14-dione

| Conditions | Yield |
|---|---|
| With boron trifluoride diethyl etherate In dichloromethane at 0 - 50℃; for 8.5h; Inert atmosphere; | A 50% B 15% |

-
-
21264-30-2, 90243-59-7, 14592-56-4
dichloro bis(acetonitrile) palladium(II)

-
-
3779-61-1
trans ocimene

| Conditions | Yield |
|---|---|
| With water In acetone trans-ocimene was added under N2 to a soln. of (MeCN)2PdCl2 in water and acetone (1:5 v/v), mixt. was stirred at -5°C for 8 h; reaction mixt. was extd. with benzene, ext. washed with water, dried over CaCl2, solvent was removed under reduced pressure, residue purified by column chromy. on SiO2 (eluent 5/1 benzene-EtOAc); elem. anals.; | A 26% B 49% |
| With water In N,N,N,N,N,N-hexamethylphosphoric triamide trans-ocimene was added under N2 to a soln. of (MeCN)2PdCl2 in water and (Me2N)3PO (1:5 v/v), mixt. was stirred at -5°C for 8 h; reaction mixt. was extd. with benzene, ext. washed with water, dried over CaCl2, solvent was removed under reduced pressure, residue purified by column chromy. on SiO2 (eluent 5/1 benzene-EtOAc); elem. anals.; | A 37% B 12% |
| With water In N,N-dimethyl-formamide trans-ocimene was added under N2 to a soln. of (MeCN)2PdCl2 in water and DMF (1:5 v/v), mixt. was stirred at -5°C for 8 h; reaction mixt. was extd. with benzene, ext. washed with water, dried over CaCl2, solvent was removed under reduced pressure, residue purified by column chromy. on SiO2 (eluent 5/1 benzene-EtOAc); elem. anals.; | A 30% B 35% |

-
-
14371-10-9
(E)-3-phenylpropenal

-
-
3779-61-1
trans ocimene

| Conditions | Yield |
|---|---|
| In neat (no solvent) at 140℃; for 72h; Inert atmosphere; Sealed tube; | 45% |
-
-
3779-61-1
trans ocimene

-
-
22252-14-8, 72758-77-1, 4224-87-7
(E)-3-(4-methylphenyl)-1-phenyl-2-propen-1-one

| Conditions | Yield |
|---|---|
| With aluminum tri-bromide In toluene at 28℃; for 3h; Diels-Alder Cycloaddition; Inert atmosphere; diastereoselective reaction; | 44% |
-
-
920282-22-0, 53623-19-1
bis(acetonitrile)palladium(II) bromide

-
-
3779-61-1
trans ocimene

| Conditions | Yield |
|---|---|
| With water In N,N,N,N,N,N-hexamethylphosphoric triamide trans-ocimene was added under N2 to a soln. of (MeCN)2PdBr2 in water and (Me2N)3PO (1:10 v/v), mixt. was stirred at room temp. for 10 h; reaction mixt. was extd. with benzene, ext. washed with water, dried over CaCl2, solvent was removed under reduced pressure, residue purified by column chromy. on SiO2; | A 35% B 19% |

Related products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View