Dayang Chem (Hangzhou) Co.,Ltd.
DayangChem exported this product to many countries and regions at best price. If you are looking for the material's manufacturer or supplier in China, DayangChem is your best choice. Pls contact with us freely for getting detailed product spe
Cas:52415-29-9
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquiryChemwill Asia Co., Ltd.
Cas:52415-29-9
Min.Order:1 Metric Ton
FOB Price: $1.0
Type:Manufacturers
inquiryLIDE PHARMACEUTICALS LIMITED
Advantage : LIDE PHARMACEUTICALS LTD. is a mid-small manufacturing-type enterprise, engaged in pharmaceutical intermediates of R&D, custom-made and production, and also involving trading chemicals for export. We have established the R&
Cas:52415-29-9
Min.Order:1 Milligram
Negotiable
Type:Lab/Research institutions
inquiryHenan Tianfu Chemical Co., Ltd.
Our company was built in 2009 with an ISO certificate.In the past 6 years, we have grown up as a famous fine chemicals supplier in China and we had established stable business relationships with Samsung,LG,Merck,Thermo Fisher Scientific and so on.O
Cas:52415-29-9
Min.Order:1 Kilogram
FOB Price: $2000.0
Type:Lab/Research institutions
inquiryHebei Nengqian Chemical Import and Export Co., LTD
Our advantages: 1, High quality with competitive price: 1) Standard:BP/USP/EP/Enterprise standard 2) All Purity≥99% 3) We are manufacturer and can provide high quality products with factory price. 2, Fast and safe delivery 1) Parcel can be sent
Cas:52415-29-9
Min.Order:1 Kilogram
FOB Price: $9.0 / 99.0
Type:Trading Company
inquiryZhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:52415-29-9
Min.Order:1 Kilogram
FOB Price: $139.0 / 210.0
Type:Trading Company
inquiryHebei Sankai Chemical Technology Co., Ltd
1. Product advantages High purity, all above 98.5%, no impurities after dissolution We will test each batch to ensure quality OEM and private brand services designed for free Various cap colors available We can also provide MT1 peptide powd
Cas:52415-29-9
Min.Order:1 Kilogram
FOB Price: $50.0 / 100.0
Type:Trading Company
inquiryHenan Sinotech Import&Export Corporation
Product Name: 6-Bromo-1H-indole Synonyms: EOS-60560;6-Bromoindole in stock Factory;bromoindole;BROMOINDOLE-6;6-BROMO-1H-INDOLE;6-BROMOINDOLE;6-Bromoindole,98%;bromoindole 6-Bromoindole CAS:
Cas:52415-29-9
Min.Order:5 Metric Ton
FOB Price: $1.0 / 2.0
Type:Other
inquiryHenan Wentao Chemical Product Co., Ltd.
We are leading fine chemicals supplier in China with ISO certificate, Our main business covers the fields below: 1.Noble Metal Catalysts (Pt.Pd...) 2.Organic Phosphine Ligands (Tert-butyl-phosphine.Cyclohexyl-phosphine...) 3.OLED
Cas:52415-29-9
Min.Order:1 Gram
FOB Price: $2.0
Type:Lab/Research institutions
inquiryHubei Langyou International Trading Co., Ltd
1. Guaranteed purity; 2. Large quantity in stock; 3. Largest manufacturer; 4. Best service after shipment with email; 5. High quality & competitive price; Appearance:Powder Storage:Store in sealed containers at cool & dry pla
Cas:52415-29-9
Min.Order:100 Gram
Negotiable
Type:Other
inquiryShanghai Upbio Tech Co.,Ltd
1.No Less 8 years exporting experience. Clients can 100% received goods 2.Lower Price with higher quality 3,Free sample 4,We are sincerely responsible for the "product quality" and "After Service" Upbio is Specialized
Cas:52415-29-9
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryBaoji Guokang Healthchem co.,ltd
Our company has been in existence for 10 years since its establishment. We have our own unique team. The company integrates independent research and development, production and sales. We have established famous brands at home and abroad. At prese
Cas:52415-29-9
Min.Order:1 Kilogram
FOB Price: $56.0 / 58.0
Type:Trading Company
inquiryQingdao Beluga Import and Export Co., LTD
6-Bromo-1H-indole CAS:52415-29-9 Qingdao Belugas Import and Export Co., Ltd. is a scientific and technological company integrating research and development, production and trade of chemical intermediates, specializing in high quality organic interme
Cas:52415-29-9
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryWuhan Han Sheng New Material Technology Co.,Ltd
Our Advantage: high quality with competitive price High quality standard: BP/USP/EP Enterprise standard All purity customized Fast and safe delivery We have reliable forwarder who can help us deliver our goods more fast and safe. We
Hangzhou Keyingchem Co.,Ltd
Hangzhou KeyingChem Co., Ltd. exported this product to many countries and regions at best price. If you are looking for the material’s manufacturer or supplier in China, KeyingChem is your best choice. Pls contact with us freely for getting det
Cas:52415-29-9
Min.Order:0 Metric Ton
Negotiable
Type:Lab/Research institutions
inquiryHangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
SHANGHAI T&W PHARMACEUTICAL CO., LTD.
A substitute for perfluorooctanoic acid, mainly used as a surfactant, dispersant, additive, etc Appearance:White solid or Colorless liquid Purity:99.3 % We will ship the goods in a timely manner as required We can provide relevant documents acc
Cas:52415-29-9
Min.Order:4 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryAlity Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Shanghai Send Pharmaceutical Technology Co., Ltd.
low price high quality high purity good sevice stock Appearance:white Storage:Preserve In Well-Closed, Light-Resistant and Tight Containers. Store In Cool & Dry Place Package:1g,5g,10g...1kg,5kg...more Application:Pharmaceutical interme
Shanghai Massive Chemical Technology Co., Ltd.
Massive Chemical is certified with ISO9001 and ISO14001 manufacturer for this product. We will offer all documents as requirement for the materials which includes, Certificate of Analysis, Material Safety Data Sheet, and Method of Analysis and
Cas:52415-29-9
Min.Order:1 Gram
FOB Price: $1.0
Type:Lab/Research institutions
inquiryZibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:52415-29-9
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquirySHANGHAI SYSTEAM BIOCHEM CO., LTD
We are one of a few suppliers that can offer custom synthesis service of this product We are specialized in custom synthesis, chemical/pharmaceutical/ pesticides outsourcing and contract research. We are committed to prov
Cas:52415-29-9
Min.Order:100 Gram
FOB Price: $100.0 / 2000.0
Type:Lab/Research institutions
inquiryHANGZHOU YUNUO CHEMICAL CO.,LTD
moderate price & quick delivery. Appearance:white to brown powder Storage:Stored in cool, dry and ventilation place; Away from fire and heat Package:100g/bottle,1kg/bottle,25kg/drum or as per your request Application:6-Bromoindole is a starting
Shandong Hanjiang Chemical Co., Ltd.
Hello, dear friend! I'm Hansen and Allen from China. Welcome to my lookchem mall! The following is a brief introduction to our products and services. If you are interested in our products, please contact us via email product quality √ 1.
Cas:52415-29-9
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryXiamen Jenny Chemical Technology Co., Ltd.
GMP standard, high purity, competitive price, in stock 1. Quick Response: within 6 hours after receiving your email. 2. Quality Guarantee: All products are strictly tested by our QC, confirmed by QA, and approved by a third-party lab in China, USA,
Shanghai Minstar Chemical Co., Ltd
6-Bromo-1H-indole Basic information Product Name: 6-Bromo-1H-indole Synonyms: EOS-60560;6-Bromoindole in stock Factory;bromoindole;BROMOINDOLE-6;6-BROMO-1H-INDOLE;6-BROMOINDOLE;6-Bromoindole,98%;bromoindole 6-Bromoindole CAS: 52415-29-9 MF:
EAST CHEMSOURCES LIMITED
factory?direct?saleAppearance:White Powder Storage:Store In Dry, Cool And Ventilated Place Package:25kg/drum, also according to the clients requirement Application:It is widely used as a thickener, emulsifier and stabilizer Transportation:By Sea Or B
Cas:52415-29-9
Min.Order:1 Kilogram
FOB Price: $18.0 / 20.0
Type:Trading Company
inquiryHangzhou Huarong Pharm Co., Ltd.
We Huarong Pharm can provide Customized Synthesis & Process R&D & APIs and intermediates Production & Quality Research & Registration Application, especially our GMP validation service which complies with SFDA, FDA, WHO and EU EMPA. O
Shandong Mopai Biotechnology Co., LTD
Shandong Mopai Biotechnology Co., LTD is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemicals. W
KAISA GROUP INC
1.Applied in food field.it can improve the immune system and prolong life. 2.Appliedin cosmetic field.it can improve the skin care. 3.Applied in pharmaceutical field.it can treat various dieases. 4.Our product quality assurance will make our customer
Synthetic route
-
-
16732-65-3
6-bromo-indole-2-carboxylic acid(Ref.7.)

-
-
52415-29-9
6-bromo-1H-indole

| Conditions | Yield |
|---|---|
| With quinoline; copper Heating; | 98% |
| With copper In quinoline Heating; | 95% |
| With quinoline; copper(I) bromide |
-
-
99474-21-2
(E)-2-(4-bromo-2-nitrophenyl)-N,N-dimethylethyleneamine

-
-
52415-29-9
6-bromo-1H-indole

| Conditions | Yield |
|---|---|
| With iron(III) chloride hexahydrate; pyrographite; hydrazine hydrate In ethanol at 75℃; for 7h; | 93% |
| With zinc In acetic acid at 85℃; for 2h; | 62% |
| With acetic acid; zinc In methanol; dichloromethane Heating; | 52% |
| With acetic acid; zinc at 75 - 85℃; for 3.5h; | 2.675 g |
-
-
60956-26-5
4-bromo-2-nitrotoluene

-
-
4637-24-5
N,N-dimethyl-formamide dimethyl acetal

-
-
52415-29-9
6-bromo-1H-indole

| Conditions | Yield |
|---|---|
| Stage #1: 4-bromo-2-nitrotoluene; N,N-dimethyl-formamide dimethyl acetal With pyrrolidine In N,N-dimethyl-formamide at 110℃; for 2h; Stage #2: With hydrogenchloride; titanium(III) chloride; ammonium acetate In N,N-dimethyl-formamide at 0℃; for 0.25h; | 90% |
| Stage #1: 4-bromo-2-nitrotoluene; N,N-dimethyl-formamide dimethyl acetal With pyrrolidine In 1,4-dioxane at 102℃; for 3.5h; Leimgruber-Batcho Indole Synthesis; Reflux; Inert atmosphere; Stage #2: With hydrazine hydrate In 1,4-dioxane at 45℃; for 1h; Leimgruber-Batcho Indole Synthesis; | 82% |
| Stage #1: 4-bromo-2-nitrotoluene; N,N-dimethyl-formamide dimethyl acetal With pyrrolidine In N,N-dimethyl-formamide at 105 - 110℃; for 21h; Leimgruber-Batcho Indole Synthesis; Stage #2: With acetic acid; zinc at 70 - 90℃; for 3h; Temperature; Reagent/catalyst; Leimgruber-Batcho Indole Synthesis; | 56% |
-
-
99365-40-9
6-bromo-2,3-dihydro-1H-indole-2-one

-
-
52415-29-9
6-bromo-1H-indole

| Conditions | Yield |
|---|---|
| With bis(trimethylsilyl)amide yttrium(III); 4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane In toluene at 120℃; for 36h; Inert atmosphere; | 90% |
-
-
189265-99-4
6-bromo-1-(4-methylphenylsulfonyl)-1H-indole

-
-
52415-29-9
6-bromo-1H-indole

| Conditions | Yield |
|---|---|
| With sodium hydride In N,N-dimethyl acetamide at 60℃; for 5h; Inert atmosphere; | 85% |
-
-
496-15-1
1-indoline

-
-
52415-29-9
6-bromo-1H-indole

| Conditions | Yield |
|---|---|
| Stage #1: 1-indoline With silver(II) sulfate; sulfuric acid for 0.5h; Stage #2: With bromine for 0.5h; Stage #3: With chloranil In 5,5-dimethyl-1,3-cyclohexadiene for 5h; Reflux; | 73% |
-
-
52448-17-6
(2S)-2-amino-3-(6-bromo-1H-indol-3-yl)propanoic acid

-
-
52415-29-9
6-bromo-1H-indole

| Conditions | Yield |
|---|---|
| With dipotassium hydrogenphosphate; tryptophanase TnaA at 25℃; pH=7.4; Enzymatic reaction; | 70.4% |

-
-
52415-29-9
6-bromo-1H-indole

| Conditions | Yield |
|---|---|
| With ammonium hydroxide; iron(II) sulfate Heating; | 31% |
-
-
78508-22-2
β-dimethylamino-2-nitrostyrene

-
-
52415-29-9
6-bromo-1H-indole

| Conditions | Yield |
|---|---|
| With acetic acid; zinc In water at 75℃; for 1h; Stirred; | 27% |
| With hydrogen; W-2 Raney nickel In ethanol | 340 mg |
| With acetic acid; zinc | |
| With hydrogen In benzene under 2585.81 Torr; for 26h; | 0.29 g |
-
-
123-75-1
pyrrolidine

-
-
60956-26-5
4-bromo-2-nitrotoluene

-
-
4637-24-5
N,N-dimethyl-formamide dimethyl acetal

-
-
52415-29-9
6-bromo-1H-indole

| Conditions | Yield |
|---|---|
| With ammonium acetate; titanium(III) chloride 1) DMF, 100-110 deg C, 2 h, 2) water, DMF; Yield given. Multistep reaction; | |
| With zinc In hexane; acetic acid; N,N-dimethyl-formamide | |
| With zinc In hexane; acetic acid; N,N-dimethyl-formamide | |
| With zinc In hexane; acetic acid; N,N-dimethyl-formamide |

-
-
63839-24-7
6-bromo-indoline

-
-
52415-29-9
6-bromo-1H-indole

| Conditions | Yield |
|---|---|
| With dimethylsulfide; tert-butylhypochlorite; sodium ethanolate; triethylamine 1.) -65 deg C, CH2Cl2, 1 h, 2.) -65 deg C --> RT, 2 h; Yield given. Multistep reaction; |
-
-
112671-54-2
(5-Bromo-2-trimethylsilanylethynyl-phenyl)-carbamic acid ethyl ester

-
-
52415-29-9
6-bromo-1H-indole

| Conditions | Yield |
|---|---|
| With sodium ethanolate In ethanol Heating; Yield given; |
-
-
108061-73-0
(E)-4-bromo-2-nitro-β-piperidinostyrene

-
-
52415-29-9
6-bromo-1H-indole

| Conditions | Yield |
|---|---|
| With titanium(III) chloride; ammonium acetate buffer In water; acetone for 0.166667h; Yield given; |
-
-
5318-27-4
6-amino-1H-indole

-
-
52415-29-9
6-bromo-1H-indole

| Conditions | Yield |
|---|---|
| With hydrogenchloride; copper(I) bromide; sodium nitrite 1.) 0 deg C, 5 min, 2.) H2O, -10 deg C, 3 h; Yield given. Multistep reaction; |
-
-
60956-26-5
4-bromo-2-nitrotoluene

-
-
52415-29-9
6-bromo-1H-indole

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: CuI; DMF / 0.33 h / 180 °C / 6000.6 - 7500.75 Torr / microwave irradiation 2: 52 percent / Zn; AcOH / methanol; CH2Cl2 / Heating View Scheme | |
| Multi-step reaction with 2 steps 1: pyrrolidine / dimethylformamide / 1 h / 110 °C 2: 2.675 g / Zn; aq. HOAc / 3.5 h / 75 - 85 °C View Scheme | |
| Multi-step reaction with 2 steps 1: 4 h / 110 °C 2: 20percent TiCl3, 4M ammonium acetate buffer / H2O; acetone / 0.17 h View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 99 percent / 2 h / Heating 2: 64 percent / aluminum amalgam / H2O / 5 h / Irradiation 3: 1.) 1M aq. HCl, sodium nitrite, 2.) CuBr / 1.) 0 deg C, 5 min, 2.) H2O, -10 deg C, 3 h View Scheme |
-
-
61293-29-6
(E)-2-(2,4-dinitrophenyl)-N,N-dimethyl-1-ethenamine

-
-
52415-29-9
6-bromo-1H-indole

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 64 percent / aluminum amalgam / H2O / 5 h / Irradiation 2: 1.) 1M aq. HCl, sodium nitrite, 2.) CuBr / 1.) 0 deg C, 5 min, 2.) H2O, -10 deg C, 3 h View Scheme |
-
-
119-32-4
4-Methyl-3-nitroanilin

-
-
52415-29-9
6-bromo-1H-indole

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 1.) sodium nitrite, 16percent HBr, 2.) copper(I) bromide / 1.) water, -1 deg C, 2.) water, 70 deg C, 1 h 2: 4 h / 110 °C 3: 20percent TiCl3, 4M ammonium acetate buffer / H2O; acetone / 0.17 h View Scheme | |
| Multi-step reaction with 2 steps 1.1: hydrogen bromide / water / 0.33 h / Reflux 1.2: 0.25 h / 0 °C 1.3: 0 °C / Heating 2.1: pyrrolidine / N,N-dimethyl-formamide / 21 h / 105 - 110 °C 2.2: 3 h / 70 - 90 °C View Scheme | |
| Multi-step reaction with 3 steps 1.1: hydrogen bromide / water / 0.33 h / Reflux 1.2: 0.25 h / 0 °C 1.3: 0 °C / Heating 2.1: 5.5 h / 110 °C 3.1: hydrogen / benzene / 26 h / 2585.81 Torr View Scheme | |
| Multi-step reaction with 3 steps 1.1: hydrogen bromide / water / 0.33 h / Reflux 1.2: 0.25 h / 0 °C 1.3: 0 °C / Heating 2.1: N,N-dimethyl-formamide / 21 h / 105 - 110 °C 3.1: acetic acid; zinc / 3 h / 70 - 90 °C View Scheme |
-
-
881-50-5
N-(4-bromo-2-nitrophenyl)acetamide

-
-
52415-29-9
6-bromo-1H-indole

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1: 3N KOH / 24 h / Heating 2: 1.) sodium nitrite, 5N HCl; 2.) aq. KI / 1.) 0-10 deg C; 2.) RT, 30 min 3: 69 percent / SnCl2, conc. HCl / ethanol / 0.25 h / 50 - 60 °C 4: 72 percent / pyridine / 3 h / 0-10 deg C 5: CuI, Et3N / Pd(PPh3)2Cl2 / 60 °C 6: NaOEt / ethanol / Heating View Scheme |
-
-
103-88-8
4-bromoacetanilide

-
-
52415-29-9
6-bromo-1H-indole

| Conditions | Yield |
|---|---|
| Multi-step reaction with 7 steps 1: HNO3 / 8 min below 0 deg C, 8 min, RT. 2: 3N KOH / 24 h / Heating 3: 1.) sodium nitrite, 5N HCl; 2.) aq. KI / 1.) 0-10 deg C; 2.) RT, 30 min 4: 69 percent / SnCl2, conc. HCl / ethanol / 0.25 h / 50 - 60 °C 5: 72 percent / pyridine / 3 h / 0-10 deg C 6: CuI, Et3N / Pd(PPh3)2Cl2 / 60 °C 7: NaOEt / ethanol / Heating View Scheme |
-
-
875-51-4
4-Bromo-2-nitroaniline

-
-
52415-29-9
6-bromo-1H-indole

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: 1.) sodium nitrite, 5N HCl; 2.) aq. KI / 1.) 0-10 deg C; 2.) RT, 30 min 2: 69 percent / SnCl2, conc. HCl / ethanol / 0.25 h / 50 - 60 °C 3: 72 percent / pyridine / 3 h / 0-10 deg C 4: CuI, Et3N / Pd(PPh3)2Cl2 / 60 °C 5: NaOEt / ethanol / Heating View Scheme |
-
-
112671-42-8
1-iodo-4-bromo-2-nitrobenzene

-
-
52415-29-9
6-bromo-1H-indole

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: 69 percent / SnCl2, conc. HCl / ethanol / 0.25 h / 50 - 60 °C 2: 72 percent / pyridine / 3 h / 0-10 deg C 3: CuI, Et3N / Pd(PPh3)2Cl2 / 60 °C 4: NaOEt / ethanol / Heating View Scheme |
-
-
64085-52-5
5-bromo-2-iodo-aniline

-
-
52415-29-9
6-bromo-1H-indole

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 72 percent / pyridine / 3 h / 0-10 deg C 2: CuI, Et3N / Pd(PPh3)2Cl2 / 60 °C 3: NaOEt / ethanol / Heating View Scheme |
-
-
112671-48-4
ethyl (5-bromo-2-iodophenyl)carbamate

-
-
52415-29-9
6-bromo-1H-indole

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: CuI, Et3N / Pd(PPh3)2Cl2 / 60 °C 2: NaOEt / ethanol / Heating View Scheme |
-
-
99365-39-6
3-(4-bromo-2-nitrophenyl)-2-oxopropanoic acid

-
-
52415-29-9
6-bromo-1H-indole

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: Fe(OH)2; H2O 2: quinoline; copper (I)-bromide View Scheme |

-
-
52415-29-9
6-bromo-1H-indole

| Conditions | Yield |
|---|---|
| With acetic acid; zinc at 75 - 85℃; for 3h; | 1.7 g |
| With acetic acid; zinc at 70 - 90℃; for 3h; Reagent/catalyst; | 7.19 g |
-
-
25408-61-1
Ν,Ν-dimethylacetamide dimethyl acetal

-
-
60956-26-5
4-bromo-2-nitrotoluene

-
-
52415-29-9
6-bromo-1H-indole

| Conditions | Yield |
|---|---|
| Stage #1: Ν,Ν-dimethylacetamide dimethyl acetal; 4-bromo-2-nitrotoluene With pyrrolidine In N,N-dimethyl-formamide at 110℃; for 3h; Stage #2: With acetic acid; zinc In water at 75 - 85℃; for 2h; |
-
-
73-22-3
L-Tryptophan

-
-
52415-29-9
6-bromo-1H-indole

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: sodium bromide; disodium hydrogenphosphate; tryptophan halogenase / 72 h / 25 °C / pH 7.4 / Enzymatic reaction 2: tryptophanase TnaA; dipotassium hydrogenphosphate / 25 °C / pH 7.4 / Enzymatic reaction View Scheme |
-
-
120-72-9
indole

-
-
52415-29-9
6-bromo-1H-indole

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: acetic acid; sodium cyanoborohydride / 2 h / 15 °C 2.1: sulfuric acid; silver(II) sulfate / 0.5 h 2.2: 0.5 h 2.3: 5 h / Reflux View Scheme |
-
-
52415-29-9
6-bromo-1H-indole

-
-
68-12-2, 33513-42-7
N,N-dimethyl-formamide

-
-
17826-04-9
6-bromo-1H-indole-3-carbaldehyde

| Conditions | Yield |
|---|---|
| With trichlorophosphate at 0 - 40℃; for 2.5h; Inert atmosphere; | 100% |
| With trichlorophosphate at 0 - 20℃; Vilsmeier-Haack Formylation; Inert atmosphere; | 99% |
| With sodium hydroxide; trichlorophosphate Vilsmeier-Haack reaction; | 98% |
-
-
52415-29-9
6-bromo-1H-indole

-
-
18162-48-6
tert-butyldimethylsilyl chloride

-
-
184637-11-4
6-bromo-1-(tert-butyldimethylsilyl)-1H-indole

| Conditions | Yield |
|---|---|
| Stage #1: 6-bromo-1H-indole With sodium hydride In N,N-dimethyl-formamide; mineral oil at 0 - 20℃; for 0.5h; Stage #2: tert-butyldimethylsilyl chloride In N,N-dimethyl-formamide; mineral oil at 0 - 20℃; for 1h; | 100% |
| Stage #1: 6-bromo-1H-indole With sodium hydride In tetrahydrofuran; mineral oil at 0 - 20℃; for 0.75h; Inert atmosphere; Stage #2: tert-butyldimethylsilyl chloride In tetrahydrofuran; mineral oil at 0 - 20℃; for 12h; Inert atmosphere; | 90% |
| Stage #1: 6-bromo-1H-indole With sodium hydride In N,N-dimethyl-formamide at 0 - 10℃; for 0.5h; Inert atmosphere; Stage #2: tert-butyldimethylsilyl chloride In N,N-dimethyl-formamide at 20℃; Inert atmosphere; | 73% |
| With n-butyllithium 1.) THF, hexane, -78 deg C, 15 min, 2.) THF, hexane, RT, 4 h; Yield given. Multistep reaction; |
-
-
52415-29-9
6-bromo-1H-indole

-
-
372077-73-1
3-iodo-6-bromoindole

| Conditions | Yield |
|---|---|
| With potassium hydroxide; iodine In N,N-dimethyl-formamide at 20℃; | 100% |
| With potassium hydroxide; iodine In N,N-dimethyl-formamide Ambient temperature; | |
| Stage #1: 6-bromo-1H-indole With sodium hydroxide In methanol at 20℃; for 0.166667h; Stage #2: With iodine; potassium iodide In methanol at 20℃; for 3h; | 134 mg |

| Conditions | Yield |
|---|---|
| With potassium hydroxide In acetone at 0 - 25℃; | 100% |
| Stage #1: 6-bromo-1H-indole With sodium hydride In 1,2-dimethoxyethane; dimethyl sulfoxide at 20℃; for 0.5h; Stage #2: methyl iodide In 1,2-dimethoxyethane; dimethyl sulfoxide at 20℃; | 95% |
| Stage #1: 6-bromo-1H-indole With sodium hydride In N,N-dimethyl-formamide at 0℃; for 0.5h; Inert atmosphere; Stage #2: methyl iodide In N,N-dimethyl-formamide at 20℃; for 2h; Inert atmosphere; | 93.1% |
-
-
52415-29-9
6-bromo-1H-indole

-
-
73430-27-0
(E)-N,N-dimethyl-2-nitroethenamine

-
-
222173-67-3
3-<(E)-2-nitroethenyl>-6-bromoindole

| Conditions | Yield |
|---|---|
| Stage #1: (E)-N,N-dimethyl-2-nitroethenamine In trifluoroacetic acid at 20℃; for 0.0833333h; Michael Addition; Inert atmosphere; Stage #2: 6-bromo-1H-indole In dichloromethane at 20℃; for 0.75h; Inert atmosphere; | 100% |
| With trifluoroacetic acid at 20℃; for 0.5h; | 96% |
-
-
52415-29-9
6-bromo-1H-indole

-
-
375853-82-0, 286961-14-6
tert-Butyl 4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-3,6-dihydro-1(2H)-pyridinecarboxylate

| Conditions | Yield |
|---|---|
| With (1,1'-bis(diphenylphosphino)ferrocene)palladium(II) dichloride; potassium carbonate In 1,4-dioxane; water for 2h; Inert atmosphere; | 100% |
-
-
52415-29-9
6-bromo-1H-indole

-
-
98-59-9
p-toluenesulfonyl chloride

-
-
189265-99-4
6-bromo-1-(4-methylphenylsulfonyl)-1H-indole

| Conditions | Yield |
|---|---|
| Stage #1: 6-bromo-1H-indole With sodium hydride In acetonitrile at 0℃; for 0.166667h; Stage #2: p-toluenesulfonyl chloride In acetonitrile at 0 - 20℃; for 4h; | 99% |
| With sodium hydride In acetonitrile at 0 - 20℃; for 4h; | 99% |
| With potassium hydroxide; tetra(n-butyl)ammonium hydrogensulfate In toluene for 4h; | 98% |
-
-
52415-29-9
6-bromo-1H-indole

-
-
13154-24-0
triisopropylsilyl chloride

-
-
299432-99-8
6-bromo-1-(triisopropylsilyl)-1H-indole

| Conditions | Yield |
|---|---|
| Stage #1: 6-bromo-1H-indole With sodium hydride In tetrahydrofuran at 20℃; for 0.166667h; Stage #2: triisopropylsilyl chloride In tetrahydrofuran at 20℃; for 0.5h; | 99% |
| Stage #1: 6-bromo-1H-indole With sodium hydride In tetrahydrofuran at 20℃; for 0.166667h; Stage #2: triisopropylsilyl chloride In tetrahydrofuran at 20℃; for 0.5h; | 99% |
| Stage #1: 6-bromo-1H-indole With sodium hydride In tetrahydrofuran; mineral oil at 0 - 20℃; for 0.5h; Stage #2: triisopropylsilyl chloride In tetrahydrofuran; mineral oil at 0 - 20℃; | 96% |
-
-
52415-29-9
6-bromo-1H-indole

-
-
24424-99-5
di-tert-butyl dicarbonate

-
-
147621-26-9
6-bromoindole-1-carboxylic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| With dmap In tetrahydrofuran at 20℃; for 4h; | 99% |
| With dmap In tetrahydrofuran at 20℃; Inert atmosphere; | 99% |
| With dmap In dichloromethane | 98% |

| Conditions | Yield |
|---|---|
| With Ti0.97Pd0.03O1.97; potassium carbonate In water at 100℃; for 6h; Suzuki-Miyaura Coupling; | 99% |
| With C20H20N2O2Pd; sodium hydrogencarbonate In water at 80℃; for 6h; Suzuki-Miyaura Coupling; | 94% |
| With potassium carbonate In water for 0.5h; Suzuki-Miyaura Coupling; Heating; | 90% |
-
-
52415-29-9
6-bromo-1H-indole

-
-
6482-24-2
2-Bromoethyl methyl ether

| Conditions | Yield |
|---|---|
| Stage #1: 6-bromo-1H-indole With sodium hydride In 1,2-dimethoxyethane; dimethyl sulfoxide at 20℃; for 0.5h; Stage #2: 2-Bromoethyl methyl ether In 1,2-dimethoxyethane; dimethyl sulfoxide at 20℃; | 99% |
-
-
52415-29-9
6-bromo-1H-indole

-
-
106-95-6
allyl bromide

-
-
945399-52-0
6-bromo-1-(prop-2-en-1-yl)-1H-indole

| Conditions | Yield |
|---|---|
| Stage #1: 6-bromo-1H-indole With sodium hydride In N,N-dimethyl-formamide at 0℃; for 0.5h; Stage #2: allyl bromide In N,N-dimethyl-formamide at 20℃; for 0.5h; | 99% |
| Stage #1: 6-bromo-1H-indole With sodium hydride In N,N-dimethyl-formamide at 20℃; Stage #2: allyl bromide In N,N-dimethyl-formamide for 2h; | 98% |
| Stage #1: 6-bromo-1H-indole With sodium hydride In N,N-dimethyl-formamide; mineral oil at 20℃; for 1h; Stage #2: allyl bromide In N,N-dimethyl-formamide; mineral oil at 20℃; for 0.75h; | 3.2 g |

| Conditions | Yield |
|---|---|
| With iodine; potassium iodide In ethanol; water at 20 - 60℃; | 99% |
| With iodine; potassium iodide In ethanol; water at 60℃; for 72h; Schlenk technique; Inert atmosphere; | 77% |
-
-
52415-29-9
6-bromo-1H-indole

-
-
63839-24-7
6-bromo-indoline

| Conditions | Yield |
|---|---|
| With sodium cyanoborohydride; acetic acid at 20℃; | 99% |
| With sodium cyanoborohydride; acetic acid for 1h; | 79% |
| With sodium cyanoborohydride; acetic acid at 10℃; for 1h; | 70% |
-
-
52415-29-9
6-bromo-1H-indole

-
-
115666-47-2
6-iodo-1 H-indole

| Conditions | Yield |
|---|---|
| With copper(l) iodide; sodium iodide; N,N`-dimethylethylenediamine In 1,4-dioxane at 110℃; for 22h; Inert atmosphere; Sealed tube; | 99% |
| With copper(l) iodide; sodium iodide; N,N`-dimethylethylenediamine In 1,4-dioxane at 110℃; for 22h; Inert atmosphere; | 99% |
| With copper(l) iodide; sodium iodide; N,N`-dimethylethylenediamine In 1,4-dioxane at 110℃; for 22h; Microwave irradiation; Sealed tube; Inert atmosphere; | 97% |
-
-
52415-29-9
6-bromo-1H-indole

-
-
1533443-74-1
ethyl 5-methyl-2-oxo-3,4-dihydro-2H-benzo[b][1,4]oxazine-3-carboxylate

-
-
1533444-23-3
ethyl (R)-5-methyl-3-(5-methyl-1H-indol-3-yl)-2-oxo-3,4-dihydro-2H-benzo[b][1,4]oxazine-3-carboxylate

| Conditions | Yield |
|---|---|
| With C38H33O4P In toluene at -40℃; for 4h; enantioselective reaction; | 99% |
-
-
52415-29-9
6-bromo-1H-indole

| Conditions | Yield |
|---|---|
| With C53H40N2O2; copper(II) bis(trifluoromethanesulfonate) In ethylbenzene at 20℃; for 24h; Friedel-Crafts Alkylation; Inert atmosphere; enantioselective reaction; | 99% |
-
-
52415-29-9
6-bromo-1H-indole

-
-
530-93-8
1,2,3,4-tetrahydronaphthalen-2-one

| Conditions | Yield |
|---|---|
| With L-Tartaric acid; 1,1-Dimethylurea at 70℃; for 3h; | 99% |

| Conditions | Yield |
|---|---|
| With (2-dicyclohexylphosphino-2’,6’-diisopropoxy-1,1‘-biphenyl)[2-(2’-methylamino-1,1’-biphenyl)]palladium(II) methanesulfonate; caesium carbonate In tetrahydrofuran; water at 80℃; Suzuki Coupling; | 99% |
-
-
52415-29-9
6-bromo-1H-indole

-
-
1189-71-5
isocyanate de chlorosulfonyle

-
-
224434-83-7
6-bromo-1H-indole-3-carbonitrile

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide at -50 - 20℃; Inert atmosphere; | 99% |
-
-
52415-29-9
6-bromo-1H-indole

-
-
198012-02-1
4-methyl-N-(4-trifluoromethylbenzylidene)-benzenesulfonamide

| Conditions | Yield |
|---|---|
| With (Rp)-4,12-di(4-(3',5'-bis(trifluoromethyl))-phenyl-3-yl)-[2.2]paracyclophane-hydrogenphosphate In toluene at -20℃; for 12h; Molecular sieve; enantioselective reaction; | 99% |
-
-
52415-29-9
6-bromo-1H-indole

-
-
224434-83-7
6-bromo-1H-indole-3-carbonitrile

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide at -50 - 20℃; for 1.5h; Inert atmosphere; | 99% |
-
-
52415-29-9
6-bromo-1H-indole

| Conditions | Yield |
|---|---|
| Stage #1: 3-(1-phenylvinyl)-1H-indole With C76H57F12NO6P2 In 1,2-dichloro-ethane at 0℃; for 0.166667h; Molecular sieve; Stage #2: 6-bromo-1H-indole In 1,2-dichloro-ethane at 0℃; for 24h; Molecular sieve; enantioselective reaction; | 99% |
-
-
52415-29-9
6-bromo-1H-indole

-
-
1033591-96-6
1-(1,3-benzothiazol-2-yl)-2,2,2-trifluoroethane-1,1-diol

| Conditions | Yield |
|---|---|
| With (R)-6,6'-bis(2,4,6-triisopropylphenyl)-1,1'-spirobiindanyl-7,7'-diyl hydrogen phosphate In dichloromethane at 20℃; Friedel-Crafts Alkylation; Molecular sieve; enantioselective reaction; | 99% |

| Conditions | Yield |
|---|---|
| With Ti0.97Pd0.03O1.97; potassium carbonate In water at 100℃; for 6h; Suzuki-Miyaura Coupling; | 98% |
-
-
52415-29-9
6-bromo-1H-indole

-
-
13081-18-0
ethyl-3,3,3-trifluoropyruvate

| Conditions | Yield |
|---|---|
| With (3aR,11aR,14aS,16bS)-2,2,13,13-tetra-isopropyl-3a,11a,14a,16b-tetrahydro-bis(1,3-dioxolano[4',5':3,4]pyrrolo)[1,2-a:1',2'-a]naphtho[1,2-d:8,7-d']diimidazole; copper(II) bis(trifluoromethanesulfonate) at 25℃; for 1h; Friedel-Crafts Alkylation; Inert atmosphere; Schlenk technique; stereoselective reaction; | 97.3% |
-
-
52415-29-9
6-bromo-1H-indole

-
-
98-09-9
benzenesulfonyl chloride

-
-
679794-03-7
1‐(benzenesulfonyl)‐6‐bromo‐1H‐indole

| Conditions | Yield |
|---|---|
| With sodium hydroxide; tetra(n-butyl)ammonium hydrogensulfate In dichloromethane at 0 - 20℃; for 3h; | 97% |
| With tetra(n-butyl)ammonium hydrogensulfate; sodium hydroxide In dichloromethane at 20℃; for 3.16667h; Cooling with ice; | 95% |
| With sodium hydride In N,N-dimethyl-formamide at 0 - 20℃; | 92% |
-
-
52415-29-9
6-bromo-1H-indole

-
-
24424-99-5
di-tert-butyl dicarbonate

-
-
182344-70-3
5-bromo-indole-1-carboxylic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| With dmap In dichloromethane at 20℃; | 97% |
| With dmap In dichloromethane at 20℃; for 1h; Addition; | 90% |
-
-
52415-29-9
6-bromo-1H-indole

-
-
56-45-1
L-serin

-
-
52448-17-6
(2S)-2-amino-3-(6-bromo-1H-indol-3-yl)propanoic acid

| Conditions | Yield |
|---|---|
| With Pf0A9 enzyme In aq. phosphate buffer; water; dimethyl sulfoxide at 75℃; for 12h; pH=8; Enzymatic reaction; | 97% |
| With ammonium sulfate; pyridoxal 5'-phosphate; sodium sulfite In phosphate buffer; dimethyl sulfoxide at 37℃; for 6h; pH=7.8; | |
| Stage #1: 6-bromo-1H-indole; L-serin With acetic anhydride; acetic acid at 73℃; for 3h; Inert atmosphere; Stage #2: With cobalt(II) chloride; sodium hydroxide In aq. buffer at 37℃; for 48h; pH=8; Enzymatic reaction; |
Related products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View
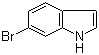
 Xi
Xi