Dayang Chem (Hangzhou) Co.,Ltd.
As a leading manufacturer and supplier of chemicals in China, DayangChem not only supply popular chemicals, but also DayangChem’s R&D center offer custom synthesis according to the contract research and development services for the fine c
Cas:688-73-3
Min.Order:1 Kilogram
FOB Price: $14.0 / 15.0
Type:Lab/Research institutions
inquirySimagchem Corporation
Welcome to Simagchem, your partner in China as a premier supply of bulk specialty chemicals for industry and life science. We introduce experienced quality product and exceptional JIT service with instant market intelligence in China to benefit our
Cas:688-73-3
Min.Order:1 Metric Ton
Negotiable
Type:Manufacturers
inquiryHenan Allgreen Chemical Co.,Ltd
The company has a professional R & D team, mature technology, very competitive prices and stable high quality products for customers to order high quality and low price product efforts! Appearance:White or off-white Solid Storage:Sealed, dr
Cas:688-73-3
Min.Order:1 Kilogram
Negotiable
Type:Manufacturers
inquiryAlity Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Chemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Cas:688-73-3
Min.Order:5 Kiloliter
FOB Price: $1.2 / 5.0
Type:Manufacturers
inquiryHenan Tianfu Chemical Co., Ltd.
Our company was built in 2009 with an ISO certificate.In the past 5 years, we have grown up as a famous fine chemicals supplier in China and we had established stable business relationships with Samsung,LG,Merck,Thermo Fisher Scientific and so on.O
Cas:688-73-3
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryChangchun Artel lmport and Export trade company
Minimum Order Qty. 10 Gram Supply Ability 500 Kilograms/Month Storage store in cool, dry, ventilated place 20℃ Delivery Time 3 business days after payment Payment Term TT,western union,Paypal,MoneyGram Package 10g,20g,50g,100g,500g,1KGS,
Cas:688-73-3
Min.Order:10 Gram
Negotiable
Type:Trading Company
inquiryHenan Wentao Chemical Product Co., Ltd.
Henan Wentao Chemical Product Co.,Ltd is Located in Zhengzhou High-tech Development Zone with import and export license, We passed ISO 9001:2008 as well, Henan Wentao has developed more than 1000 compounds, which are widely used in the fields of prod
Hangzhou Fandachem Co.,Ltd
Hangzhou Fanda Chemical Co.,Ltd (FandaChem) , a China-based chemical company, specialize in exporting Tributyltin Hydride; TBTH;Tributylstannane; CAS:688-73-3 , Please contact us by email freely. We are leading exporter in China.
Cas:688-73-3
Min.Order:1 Metric Ton
Negotiable
Type:Other
inquiryHangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Shanghai Massive Chemical Technology Co., Ltd.
Massive Chemical is certified with ISO9001 and ISO14001 manufacturer for this product. We will offer all documents as requirement for the materials which includes, Certificate of Analysis, Material Safety Data Sheet, and Method of Analysis and
Cas:688-73-3
Min.Order:1 Gram
FOB Price: $1.0
Type:Lab/Research institutions
inquiryZibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:688-73-3
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquiryHANGZHOU YUNUO CHEMICAL CO.,LTD
Superior quality, moderate price & quick delivery. Appearance:colourless liquid Storage:Stored in cool, dry and ventilation place; Away from fire and heat Package:25kg/drum, or as per your request. Application:Used as Pharmaceutical Intermediat
Zibo Dorne chemical technology co. LTD
Product Details Grade: pharmaceutical grade Purity:99%+ ProductionCapacity: 1000 Kilogram/Month Scope of use: For scientific research only(The product must be used legally) Our Advantage 1. Best quality with competitive price. 2. Quick shipping,
Cas:688-73-3
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquirySiwei Development Group Ltd.
Product name: Tri-N-Butyltin Hydride CAS No.:688-73-3 Molecule Formula:C12H27Sn Molecule Weight:290.05 Purity: 98.0% Package: 50kg/drum Description:Light yellow near colorless liquid Manufacture Standards:Enterprise Standard
Cas:688-73-3
Min.Order:1 Kilogram
Negotiable
Type:Trading Company
inquiryHangzhou Zhongqi chem Co.,Ltd.
Located in Hangzhou National Hi-Tech Industrial Development Zone, zhongqichem is a technical company mainly focus on the Custom synthesis, manufacturing, sales of chemicals to various industries. Benefiting from the outstanding customer service an
Hunan chemfish Pharmaceutical co.,Ltd
Appearance:95%+ Package:R&D,Pilot run Transportation:per client require Port:Express ,Air, Sea
Hubei Vanz Pharm Co.,Ltd
ISO/factory/goodqualityAppearance:off white Storage:Dry,cool place Package:drum Application:active pharmaceutical ingredients Transportation:by air/sea/express Port:shenzhen/shanghai
Kono Chem Co.,Ltd
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:drum and bag Application:for pharma use Transportation:by sea or air Port:Beijing or Guangzhou
Hangzhou Dingyan Chem Co., Ltd
R & D enterprises have their own stock in stock Package:1kg Application:pharmaceutical intermediates
Aecochem Corp.
Our clients, like BASF,CHEMO,Brenntag,ASR,Evonik,Merck and etc.Appearance:COA Storage:in stock Application:MSDS/TDS
Zhuozhou Wenxi import and Export Co., Ltd
Product Description Description & Specification Category Pharmaceutical Raw Materials, Fine Chemicals, Bulk drug Standard Medical standard
Cas:688-73-3
Min.Order:1 Kilogram
FOB Price: $112.0
Type:Trading Company
inquiryAntimex Chemical Limied
Ansciep Chemical is a professional enterprise manufacturing and distributing fine chemicals and speciality chemicals. We have been dedicated to heterocycle compounds and phenyl rings for tens of years. This is our mature product for export. Our quali
Xiamen AmoyChem Co.,Ltd
Amoychem is committed to providing the top-quality chemical products and services Internationally. We offer our customers with friendly, professional service and reliable, high performance products that have been manufactured according to the accredi
Hangzhou Ocean Chemical Co., Ltd.
★.Best quality according to requirement ★.Competitive price in China market ★.Mature Technical support ★.Professional logistic support
Hunan Russell Chemicals Technology Co.,Ltd
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:Foil bag; Drum; Plastic bottle Application:Pharma;Industry;Agricultural Transportation:by sea or air Port:any port in China
AstaTech ( Chengdu) BioPharmaceutical Corp.
Quality assurance, stable process and affordable priceAppearance:detailed see specifications Storage:enquiry Package:according to the clients requirement Application:Pharmaceutical intermediates Transportation:Normal Port:chengdu
Synthetic route

| Conditions | Yield |
|---|---|
| In pentane -78°C; 3 h; | 100% |
| In not given | 99.8% |
| In not given | 99.8% |

| Conditions | Yield |
|---|---|
| In 1,2-dimethoxyethane formation at room temperature;; | 96% |
| In 1,2-dimethoxyethane formation at room temperature;; | 96% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran byproducts: LiCl, Ti(Sn(C4H9)3)1.7; addn. of Bu3SnLi to Ti-compound in soln. (-78°C), stirred, mixt. brought to 20°C, soln. became blue; evapn. in vac., residue washed with pentane, sublimation (10**-3 Torr, 90°C); elem. anal., chromy.; | A 90% B 0% C 30% D 96% |


| Conditions | Yield |
|---|---|
| 95% | |
| With aluminium amalgame In not given formation on reaction for 6 h at 10°C;; | 60% |
| With aluminium amalgame In not given formation on reaction for 6 h at 10°C;; | 60% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran byproducts: 1-naphthylboronic acid; to srirred soln. of Sn-compound in THF was added soln. of BH3 in THF under N2, mixt. was left at room temp. for 1 h, refluxed for 3 h, cooled, Et2O and water was added; dried with MgSO4, solvent was removed under vac., column chromy. on silica gel with hexane and hexane-Et2O, recrystd. from water; | 95% |
| In tetrahydrofuran byproducts: 1-naphthylboronic acid; to srirred soln. of Sn-compound in THF was added soln. of BH3 in THF under N2, mixt. was left at room temp. for 1 h, refluxed for 1 h, cooled, Et2O and water was added; dried with MgSO4, solvent was removed under vac., column chromy. on silica gel with hexane and hexane-Et2O, recrystd. from water; | 72% |

| Conditions | Yield |
|---|---|
| With (SiH(CH3)O)(n) exothermic react.; | 95% |

| Conditions | Yield |
|---|---|
| In pentane formation at -78°C;; | 94.4% |
| In pentane formation at -78°C;; | 94.4% |
-
-
1631-82-9
chloro(methyl)phenylsilane

-
-
682-00-8
tributyltin ethoxide

-
A
-
775-56-4
diethoxy-methyl-phenyl-silane

-
B
-
1461-22-9
tributyltin chloride

-
C
-
688-73-3
tri-n-butyl-tin hydride

| Conditions | Yield |
|---|---|
| 2:1; room temp.; | A 94% B n/a C n/a |
| 2:1; room temp.; | A 94% B n/a C n/a |

-
-
1067-52-3
tributyltin methoxide

-
-
1025-08-7, 1025-09-8, 7427-12-5, 17037-10-4
(+)-methyl-(α-naphthyl)-phenylsilane

-
A
-
3553-88-6, 16544-83-5, 16952-14-0, 18544-02-0
methoxy(methyl)(1-naphthyl)phenylsilane

-
B
-
688-73-3
tri-n-butyl-tin hydride

| Conditions | Yield |
|---|---|
| In toluene Kinetics; 100°C; 12 h;; | A 92% B n/a |
| In toluene Kinetics; 100°C; 12 h;; | A 92% B n/a |

-
-
682-00-8
tributyltin ethoxide

-
-
1025-08-7, 1025-09-8, 7427-12-5, 17037-10-4
(+)-methyl-(α-naphthyl)-phenylsilane

-
A
-
143731-59-3
CH3(C6H5)(1-C10H7)SiOC2H5

-
B
-
688-73-3
tri-n-butyl-tin hydride

| Conditions | Yield |
|---|---|
| 100°C; 12 h; | A 92% B n/a |
| 100°C; 12 h; | A 92% B n/a |
| 100°C; 12 h; | A 92% B n/a |


| Conditions | Yield |
|---|---|
| In tetrahydrofuran byproducts: p-anisylboronic acid; to srirred soln. of Sn-compound in THF was added soln. of BH3 in THF under N2, mixt. was left at room temp. for 1 h, refluxed for 1 h, cooled, Et2O and water was added; dried with MgSO4, solvent was removed under vac., column chromy. on silica gel with hexane and hexane-Et2O, recrystd. from water; | 91% |
-
-
74-94-2
dimethylamine borane

-
-
68725-14-4
tributylstannyl trifluoromethanesulfonate

-
A
-
23273-02-1
(μ-dimethylamino)diborane

-
B
-
688-73-3
tri-n-butyl-tin hydride

-
C
-
23884-11-9
cyclic(dimethylamino borane) dimer

| Conditions | Yield |
|---|---|
| With 2,2,6,6-tetramethylpiperidine In further solvent(s) byproducts: H2; N2 or Ar; 1,2-dichlorobenzene soln. of nBuSnOTf (0.39 mmol) and 2,2,6,6-tetramethylpiperidine (0.4 mmol) added to soln. of B compd. (0.42 mmol), mixt. stirred at 20°c for 2 h; NMR; | A 2% B n/a C 91% |

-
-
16940-66-2
sodium tetrahydroborate

-
-
56-35-9
bis(tri-n-butyltin)oxide

-
-
688-73-3
tri-n-butyl-tin hydride

| Conditions | Yield |
|---|---|
| In ethanol Sn compd. dissolved in abs. ethanol at 0°C under Ar, then added NaBH4, allowed to warm to room temp., stirred for 1 h; EtOH removed in vac., residue dissolved in hexane, washed with water, dried, evapd. under reduced pressure; | 90% |
-
-
1067-52-3
tributyltin methoxide

-
-
1025-09-8
(S)-(-)-α-naphthylphenylmethylsilane

-
A
-
3553-88-6, 16544-83-5, 16952-14-0, 18544-02-0
methoxy(methyl)(1-naphthyl)phenylsilane

-
B
-
688-73-3
tri-n-butyl-tin hydride

| Conditions | Yield |
|---|---|
| In toluene Kinetics; 100°C; 12 h;; | A 90% B n/a |
| In toluene Kinetics; 100°C; 12 h;; | A 90% B n/a |


| Conditions | Yield |
|---|---|
| In tetrahydrofuran byproducts: o-anisylboronic acid; to srirred soln. of Sn-compound in THF was added soln. of BH3 in THF under N2, mixt. was left at room temp. for 1 h, refluxed for 1 h, cooled, Et2O and water was added; dried with MgSO4, solvent was removed under vac., column chromy. on silica gel with hexane and hexane-Et2O, recrystd. from water; | 90% |
-
-
682-00-8
tributyltin ethoxide

-
-
1025-09-8
(S)-(-)-α-naphthylphenylmethylsilane

-
A
-
143731-59-3
CH3(C6H5)(1-C10H7)SiOC2H5

-
B
-
688-73-3
tri-n-butyl-tin hydride

| Conditions | Yield |
|---|---|
| 100°C; 12 h; | A 89% B n/a |
| 100°C; 12 h; | A 89% B n/a |
| 100°C; 12 h; | A 89% B n/a |


| Conditions | Yield |
|---|---|
| With poly(methylhydrosiloxane) In not given to Bu3SnOMe poly(methylhydrosiloxane) was added and stirred at room temp. for 8 h; distillation under reduced pressure; | 89% |
| Multi-step reaction with 2 steps 2: C6H5(CH3)2SiH View Scheme | |
| Multi-step reaction with 2 steps 2: C6H5(CH3)2SiH View Scheme |
-
-
16524-87-1
Tri-n-butyl-tri-n-butyl-siloxy-zinn

-
-
43435-60-5
poly(methylhydrosiloxane)

-
-
688-73-3
tri-n-butyl-tin hydride

| Conditions | Yield |
|---|---|
| In not given formation at room temperature;; distillation;; | 88% |
| In not given formation at room temperature;; distillation;; | 88% |

-
-
16524-87-1
Tri-n-butyl-tri-n-butyl-siloxy-zinn

-
-
688-73-3
tri-n-butyl-tin hydride

| Conditions | Yield |
|---|---|
| 1:2; 150°C; | 88% |
| 1:2; 150°C; | 88% |
-
-
16853-85-3
lithium aluminium tetrahydride

-
-
1461-22-9
tributyltin chloride

-
-
688-73-3
tri-n-butyl-tin hydride

| Conditions | Yield |
|---|---|
| In diethyl ether heating on reflux for 2-4 h; decomposition of excess LiAlH4 on addn. of ice-water;; distillation;; | 87% |
| In diethyl ether heating on reflux for 2-4 h; decomposition of excess LiAlH4 on addn. of ice-water;; distillation;; | 87% |
| In diethyl ether heating on reflux for 2-4 h; decomposition of excess LiAlH4 on addn. of ice-water;; distillation;; | 74% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran byproducts: o-tolylboronic acid; to srirred soln. of Sn-compound in THF was added soln. of BH3 in THF under N2, mixt. was left at room temp. for 1 h, refluxed for 1 h, cooled, Et2O and water was added; dried with MgSO4, solvent was removed under vac., column chromy. on silica gel with hexane and hexane-Et2O, recrystd. from water; | 86% |

| Conditions | Yield |
|---|---|
| In not given formation at room temperature;; distillation;; | 86% |
| In not given formation at room temperature;; distillation;; | 86% |
-
-
17198-96-8, 147377-23-9, 147377-34-2
Methyl-phenyl-ethoxy-silan

-
-
682-00-8
tributyltin ethoxide

-
A
-
775-56-4
diethoxy-methyl-phenyl-silane

-
B
-
688-73-3
tri-n-butyl-tin hydride

| Conditions | Yield |
|---|---|
| 1:1; room temp.; | A 86% B n/a |
| 1:1; room temp.; | A 86% B n/a |


| Conditions | Yield |
|---|---|
| 1:1; 30 min; little contaminated with siloxane fragments; | 86% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran byproducts: phenylboronic acid; to srirred soln. of Sn-compound in THF was added soln. of BH3 in THF under N2, mixt. was left at room temp. for 1 h, refluxed for 1 h, cooled, Et2O and water was added; dried with MgSO4, solvent was removed under vac., column chromy. on silica gel with hexane and hexane-Et2O, recrystd. from water; | 85% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran byproducts: p-tolylboronic acid; to srirred soln. of Sn-compound in THF was added soln. of BH3 in THF under N2, mixt. was left at room temp. for 1 h, refluxed for 1 h, cooled, Et2O and water was added; dried with MgSO4, solvent was removed under vac., column chromy. on silica gel with hexane and hexane-Et2O, recrystd. from water; | 84% |
-
-
43435-60-5
poly(methylhydrosiloxane)

-
-
56-35-9
bis(tri-n-butyltin)oxide

-
-
688-73-3
tri-n-butyl-tin hydride

| Conditions | Yield |
|---|---|
| In not given formation at room temperature;; distillation;; | 79% |
| In not given formation at room temperature;; distillation;; | 79% |
-
-
1749-41-3
Cyclohexyloxy-tributyl-stannan

-
-
1025-08-7, 1025-09-8, 7427-12-5, 17037-10-4
(+)-methyl-(α-naphthyl)-phenylsilane

-
B
-
688-73-3
tri-n-butyl-tin hydride

| Conditions | Yield |
|---|---|
| for 88 h at 120°C; retention >87%; | A 79% B n/a |
| for 88 h at 120°C; retention >87%; | A 79% B n/a |
| for 88 h at 120°C; retention >87%; | A 79% B n/a |

-
-
5847-51-8
tributyltin formate

-
A
-
688-73-3
tri-n-butyl-tin hydride

-
B
-
813-19-4
bis(tri-n-butyltin)

-
C
-
1461-25-2
tetra-n-butyltin(IV)

| Conditions | Yield |
|---|---|
| heating 11.5 h at 150 to 185°C; | A 6.9% B 77.5% C 5.2% |
| heating 11.5 h at 150 to 185°C; | A 6.9% B 77.5% C 5.2% |
| In neat (no solvent) byproducts: CO2; heating for 11.5 h at 150-185°C under N2;; | A 6.9% B n/a C n/a |

-
-
16524-88-2
tributyl(tris(phenyl)siloxy)tin

-
-
43435-60-5
poly(methylhydrosiloxane)

-
-
688-73-3
tri-n-butyl-tin hydride

| Conditions | Yield |
|---|---|
| In not given formation at room temperature;; distillation;; | 76% |
| In not given formation at room temperature;; distillation;; | 76% |
-
-
688-73-3
tri-n-butyl-tin hydride

-
-
23326-27-4
Methyl 2-butynoate

-
-
151568-81-9, 151568-82-0
methyl (2E)-3-(tributylstannyl)but-2-enoate

| Conditions | Yield |
|---|---|
| Stage #1: tri-n-butyl-tin hydride With n-butyllithium In tetrahydrofuran at -78℃; for 0.166667h; Stage #2: With methanol In tetrahydrofuran at -78℃; for 0.05h; Stage #3: Methyl 2-butynoate In tetrahydrofuran at -78℃; for 1.5h; | 100% |
-
-
688-73-3
tri-n-butyl-tin hydride

-
-
1165952-91-9
cyclohexa-1,3-diene

-
-
112520-99-7, 1177092-71-5
tributyl(cyclohex-2-en-1-yl)stannane

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0) In benzene at 20℃; for 1h; Inert atmosphere; | 100% |
| With tetrakis(triphenylphosphine) palladium(0) In benzene at 20℃; for 0.166667h; Inert atmosphere; | 78% |
| tetrakis(triphenylphosphine) palladium(0) In benzene at 20℃; for 0.25h; | 78.6% |
-
-
688-73-3
tri-n-butyl-tin hydride

-
-
762-21-0
acetylenedicarboxylic acid diethyl ester

-
-
3422-68-2, 4276-31-7, 6150-36-3
(CH3CH2CH2CH2)3SnC(COOC2H5)CHCOOC2H5

| Conditions | Yield |
|---|---|
| 0°C;; cis-trans-ratio = 10:1;; | 100% |
| 0°C;; cis-trans-ratio = 10:1;; | 100% |
| In not given |

| Conditions | Yield |
|---|---|
| With diisobutylaluminium hydride 75-80°C;; | 100% |
| With (iso-C4H9)2AlH 75-80°C;; | 100% |
| With diisobutylaluminium hydride 75-80°C;; | 100% |
-
-
688-73-3
tri-n-butyl-tin hydride

-
-
536-74-3
phenylacetylene

-
-
66680-87-3, 66680-88-4, 79159-76-5
(E)-1-phenyl-(2-tributylstannyl)ethene

| Conditions | Yield |
|---|---|
| With 2,2'-azobis(isobutyronitrile); oxygen In benzene-d6 at 80℃; for 2h; diastereoselective reaction; | 100% |
| In not given 100-150°C, 48 h; | 93% |
| With C44H72O4Ru2(2-) In tetrahydrofuran at 30℃; for 0.166667h; Solvent; UV-irradiation; Inert atmosphere; stereoselective reaction; | 92% |
-
-
653-37-2
perfluorobenzaldehyde

-
-
688-73-3
tri-n-butyl-tin hydride

-
A
-
20749-43-3
Perfluorphenylmethoxytributylzinn

-
B
-
20428-47-1
Perfluorphenylmethyl-(perfluorphenyl-tributylstannoxymethyl)-ether

| Conditions | Yield |
|---|---|
| In neat (no solvent) 2:3 mole ratio of (C4H9)3SnH and aldehyde, 20. degree.C; 2.6:1 mixt. of (C4H9)3SnOCH2C6F5 and the 1:2 adduct (C4H9)3SnOCH(C6F5)OCH2C5F6; | A 100% B n/a |
| In neat (no solvent) 2:3 mole ratio of (C4H9)3SnH and aldehyde, 20. degree.C; 2.6:1 mixt. of (C4H9)3SnOCH2C6F5 and the 1:2 adduct (C4H9)3SnOCH(C6F5)OCH2C5F6; | A 100% B n/a |
| In not given reaction at 20°C;; |

-
-
75-77-4
chloro-trimethyl-silane

-
-
688-73-3
tri-n-butyl-tin hydride

-
-
17955-46-3
trimethylsilyltributyltin

| Conditions | Yield |
|---|---|
| In tetrahydrofuran 1) DIPA, THF, -78°C; 2)TMSCl, room temp.; | 100% |
| With n-butyllithium; diisopropylamine In tetrahydrofuran cooling of soln. of diisopropylamine in THF to -78°C for 10 min; addn. of n-BuLi for 5 min; after 5 min heating to room temp.; addn. of SnH(n-Bu)3 for 5 min; stirring at room temp. for 20 min; addn. of Me3SiCl; stirring at room temp. for >10 min; evapn., chromy. on silica gel (hexanes); | 99% |
| In neat (no solvent) deprotonation of (C4H9)3HSn with LDA followed by quench with (CH3)3SiCl; |
-
-
688-73-3
tri-n-butyl-tin hydride

-
-
2845-62-7
benzenesulphonyl isocyanate

-
-
37755-83-2
N-<(Tri-n-butylstannyloxy)-methylen>-benzolsulfonamid

| Conditions | Yield |
|---|---|
| In cyclohexane 1:1 mole ratio of educts, at room temp.; | 100% |
| In cyclohexane 1:1 mole ratio of educts, at room temp.; | 100% |

| Conditions | Yield |
|---|---|
| In cyclohexane 1:1 mole ratio of educts, at room temp.; | 100% |
| In cyclohexane 1:1 mole ratio of educts, at room temp.; | 100% |
-
-
76425-45-1
CH2CHC(CH3)(SO2C6H4CH3)CH2CH2COOCH3

-
-
688-73-3
tri-n-butyl-tin hydride

| Conditions | Yield |
|---|---|
| With 2,2'-azobis(isobutyronitrile) In benzene under N2 a mixture of Sn-compound, sulfone, and AIBN in dry benzene wasrefluxed for 5 h; evapd.; | 100% |
-
-
7018-76-0
4-methoxybenzenesulfonyl isocyanate

-
-
688-73-3
tri-n-butyl-tin hydride

-
-
37755-85-4
(C4H9)3SnOCHNSO2C6H4OCH3

| Conditions | Yield |
|---|---|
| In cyclohexane 1:1 mole ratio of educts, at room temp.; | 100% |
| In cyclohexane 1:1 mole ratio of educts, at room temp.; | 100% |
-
-
688-73-3
tri-n-butyl-tin hydride

-
-
5769-15-3
4-Chlorophenylsulfonyl isocyanate

-
-
38421-33-9
(C4H9)3SnOCHNSO2C6H4Cl

| Conditions | Yield |
|---|---|
| In cyclohexane 1:1 mole ratio of educts, at room temp.; | 100% |
| In cyclohexane 1:1 mole ratio of educts, at room temp.; | 100% |
-
-
688-73-3
tri-n-butyl-tin hydride

-
-
93059-38-2
(+)-(1S,3S,4R)-menthyl pyruvate

| Conditions | Yield |
|---|---|
| Irradiation (UV/VIS); accelerated by UV light; two diastereomeric isomers in equal amts.; | 100% |
| Irradiation (UV/VIS); accelerated by UV light; two diastereomeric isomers in equal amts.; | 100% |

-
-
688-73-3
tri-n-butyl-tin hydride

-
-
83591-03-1
(but-2-yn-1-yloxy)(tert-butyl)dimethylsilane

-
-
1403390-08-8, 1403390-34-0, 1416133-99-7
C22H48OSiSn

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; benzene-d6 at 50℃; for 18h; | 100% |

-
-
1451073-41-8
5-O-benzyl-1,3-O-methyl-2-O-[(methylthio)thiocarbonyl]-α-D-ribo-furanose

-
-
688-73-3
tri-n-butyl-tin hydride

-
-
1451073-58-7
5-O-benzyl-1,3-O-dimethyl-2-O-tributylstannylmethylthio-α-D-ribo-furanose

| Conditions | Yield |
|---|---|
| Stage #1: 5-O-benzyl-1,3-O-methyl-2-O-[(methylthio)thiocarbonyl]-α-D-ribo-furanose With triethyl borane In benzene at 20℃; for 0.0833333h; Stage #2: tri-n-butyl-tin hydride In benzene for 1.3h; | 100% |

| Conditions | Yield |
|---|---|
| With 2,2'-azobis(isobutyronitrile); oxygen In benzene-d6 at 80℃; for 2h; diastereoselective reaction; | 100% |
| With C44H72O4Ru2(2-) at 30℃; for 0.166667h; Reagent/catalyst; Time; UV-irradiation; stereoselective reaction; | 99 %Spectr. |
-
-
688-73-3
tri-n-butyl-tin hydride

| Conditions | Yield |
|---|---|
| With triethyl borane In tetrahydrofuran; hexane at 80℃; for 4h; | 100% |
| With triethyl borane In tetrahydrofuran; hexane at 80℃; for 4h; Temperature; | 100% |
-
-
86318-61-8
(tert-butyldimethylsilyl)acetylene

-
-
688-73-3
tri-n-butyl-tin hydride

| Conditions | Yield |
|---|---|
| Stage #1: (tert-butyldimethylsilyl)acetylene With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 0.5h; Stage #2: tri-n-butyl-tin hydride In tetrahydrofuran; hexane at -78℃; for 3h; | 100% |
-
-
917-92-0
2,2-dimethyl-3-butyne

-
-
688-73-3
tri-n-butyl-tin hydride

-
-
58064-10-1
tributyl(3,3-dimethyl-1-butyn-1-yl)stannane

| Conditions | Yield |
|---|---|
| Stage #1: 3,3-Dimethylbut-1-yne With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 1h; Stage #2: tri-n-butyl-tin hydride In tetrahydrofuran; hexane at -78℃; for 1h; | 100% |
| With pyridine; 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical; zinc trifluoromethanesulfonate at 70℃; for 8h; Schlenk technique; Inert atmosphere; chemoselective reaction; | 57% |
-
-
57443-39-7
2-methyl-6-phenylhex-3-yn-2-ol

-
-
688-73-3
tri-n-butyl-tin hydride

| Conditions | Yield |
|---|---|
| With [RuCl2pentamethylcyclopentadiene]n In dichloromethane for 0.0833333h; Inert atmosphere; | 100% |
| With [RuCl2pentamethylcyclopentadiene]n In dichloromethane for 1.08333h; Schlenk technique; Inert atmosphere; | 5.16 g |
-
-
688-73-3
tri-n-butyl-tin hydride

-
-
180779-80-0
(4R,5R,E)-5-hydroxy-4,6-dimethyl-N-(prop-2-yn- 1-yl)hept-2-enamide

| Conditions | Yield |
|---|---|
| Stage #1: tri-n-butyl-tin hydride With n-butyllithium; copper(l) cyanide In tetrahydrofuran; hexane at -78℃; for 0.583333h; Inert atmosphere; Sealed tube; Stage #2: (4R,5R,E)-5-hydroxy-4,6-dimethyl-N-(prop-2-yn- 1-yl)hept-2-enamide In tetrahydrofuran; hexane at -78℃; for 1.25h; Inert atmosphere; Sealed tube; | 100% |
| Stage #1: tri-n-butyl-tin hydride With n-butyllithium; copper(I) cyanide In tetrahydrofuran; hexane at -78℃; for 0.5h; Inert atmosphere; Stage #2: (4R,5R,E)-5-hydroxy-4,6-dimethyl-N-(prop-2-yn- 1-yl)hept-2-enamide In tetrahydrofuran; hexane at -78℃; for 1h; Inert atmosphere; | 100% |
| With n-butyllithium; copper(l) cyanide | 100% |
| Stage #1: tri-n-butyl-tin hydride With n-butyllithium; copper(l) cyanide In tetrahydrofuran; hexane for 0.583333h; Inert atmosphere; Stage #2: (4R,5R,E)-5-hydroxy-4,6-dimethyl-N-(prop-2-yn- 1-yl)hept-2-enamide In tetrahydrofuran; hexane for 1.25h; Inert atmosphere; | 100% |

-
-
688-73-3
tri-n-butyl-tin hydride

| Conditions | Yield |
|---|---|
| With tris-(dibenzylideneacetone)dipalladium(0); N-ethyl-N,N-diisopropylamine; tricyclohexylphosphine tetrafluoroborate In dichloromethane at 0℃; for 2.5h; Inert atmosphere; | 100% |

| Conditions | Yield |
|---|---|
| With n-butyllithium; copper(I) cyanide | 100% |

| Conditions | Yield |
|---|---|
| With n-butyllithium; copper(I) cyanide | 100% |

| Conditions | Yield |
|---|---|
| at 20℃; for 24h; Temperature; Inert atmosphere; Schlenk technique; Glovebox; | 100% |

| Conditions | Yield |
|---|---|
| Stage #1: tri-n-butyl-tin hydride With n-butyllithium; copper(l) cyanide In tetrahydrofuran; hexane for 0.583333h; Inert atmosphere; Stage #2: C20H27NO4 In tetrahydrofuran; hexane for 1.25h; Inert atmosphere; | 100% |

| Conditions | Yield |
|---|---|
| Stage #1: tri-n-butyl-tin hydride With n-butyllithium; copper(l) cyanide In tetrahydrofuran; hexane for 0.583333h; Inert atmosphere; Stage #2: C13H21NO2 In tetrahydrofuran; hexane for 1.25h; Inert atmosphere; | 100% |

| Conditions | Yield |
|---|---|
| Stage #1: tri-n-butyl-tin hydride With n-butyllithium; copper(l) cyanide In tetrahydrofuran; hexane for 0.583333h; Inert atmosphere; Stage #2: C13H21NO2 In tetrahydrofuran; hexane for 1.25h; Inert atmosphere; | 100% |
-
-
111-34-2
-butyl vinyl ether

-
-
688-73-3
tri-n-butyl-tin hydride

-
-
51283-48-8
<2-Butyloxy-ethyl>-triisobutyl-zinn

| Conditions | Yield |
|---|---|
| In neat (no solvent) other Radiation; under Ar; (60)Co-irradiation; 1.5:1 molar ratio (olefine:Sn-compound) ;25°C; GLC; | 99% |
| other Radiation; gamma-radiation;; | 99% |
| other Radiation; gamma-radiation;; | 99% |
| In not given other Radiation; γ-radiation;; |
Related products
Raw Materials
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View










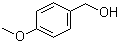




 T,
T, N,
N, F
F