Hunan Russell Chemicals Technology Co.,Ltd
low price and high purityAppearance:solid or liquid Storage:in sealed air resistant place Package:As customer require Application:Pharma;Industry;Agricultural Transportation:by sea or by airplane Port:any port in China
Cas:78-95-5
Min.Order:0
Negotiable
Type:Trading Company
inquirySynthetic route

| Conditions | Yield |
|---|---|
| With chlorine; calcium carbonate In water at 110℃; Reagent/catalyst; Temperature; | 95.6% |
| With methanol; sulfuryl dichloride In dichloromethane | 85% |
| With sulfuryl dichloride |
-
-
35952-61-5
(C4H9)3SnOCH(CH2Cl)2

-
A
-
1461-22-9
tributyltin chloride

-
B
-
78-95-5
chloroacetone

-
C
-
106-89-8
epichlorohydrin

| Conditions | Yield |
|---|---|
| 63% decompn. at 210°C (1 h); | A n/a B 5% C 95% |


| Conditions | Yield |
|---|---|
| With trichloroisocyanuric acid; boron trifluoride diethyl etherate In acetic acid for 1h; Heating; Yields of byproduct given; | A n/a B 88% |
| With water; chlorine; calcium carbonate |

| Conditions | Yield |
|---|---|
| With sulfuric acid for 3h; Heating; | 85% |

| Conditions | Yield |
|---|---|
| With HT4-c823 In water at 149.84℃; under 750.075 Torr; for 0.5h; Catalytic behavior; Reagent/catalyst; Temperature; Flow reactor; chemoselective reaction; | A 18.9% B 80.5% |

| Conditions | Yield |
|---|---|
| With oxalyl dichloride; N,N-dimethyl-formamide In dichloromethane for 1h; Ambient temperature; | A 50% B 14% |
-
-
2684-62-0
diazoacetone

-
-
68-12-2, 33513-42-7
N,N-dimethyl-formamide

-
A
-
78-95-5
chloroacetone

-
B
-
177084-70-7
2-diazo-3-oxobutanal

| Conditions | Yield |
|---|---|
| With oxalyl dichloride In dichloromethane for 1h; Ambient temperature; | A 50% B 14% |

-
-
2684-62-0
diazoacetone

-
B
-
116-09-6
hydroxy-2-propanone

-
C
-
78-95-5
chloroacetone

-
D
-
76089-31-1
1,1'-oxydi(propan-2-one)

| Conditions | Yield |
|---|---|
| With hydrogenchloride; gallium(III) trichloride; water In dichloromethane at 20℃; for 0.0833333h; | A 34% B 8% C 20% D 31% |

-
-
173063-66-6
cis/trans-3,5-Bis-(chloromethyl)-3-fluoro-5-methyl-1,2,4-trioxolane

-
A
-
78-95-5
chloroacetone

-
B
-
79-04-9
chloroacetyl chloride

| Conditions | Yield |
|---|---|
| With triphenylphosphine In chloroform-d1 | A 28 % Spectr. B 23 % Spectr. C 33% |

-
-
115290-60-3
{Pd2(μ-H)(COCH3)2(μ-dppm)2}BPh4

-
-
98194-67-3
Pd2(CH3)2(Cl)2(μdppm)2

-
B
-
75-07-0
acetaldehyde

-
C
-
78-95-5
chloroacetone

| Conditions | Yield |
|---|---|
| In dichloromethane warming of {Pd2(μ-H)(COCH3)2(dppm)2}BPh4 in CH2Cl2 (23°C, 24 h); | A 32% B n/a C n/a |


| Conditions | Yield |
|---|---|
| With chlorine at 0 - 10℃; Kochen des Reaktionsprodukts mit Wasser; |

| Conditions | Yield |
|---|---|
| With sulfuric acid Destillieren der Loesung mit Wasser; |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; diethyl ether | |
| Stage #1: diazoacetone With ethylaluminum dichloride In hexane; dichloromethane at 20℃; for 0.0166667h; Stage #2: With methanol In hexane; dichloromethane |

| Conditions | Yield |
|---|---|
| With water; chlorine |
-
-
67-66-3
chloroform

-
-
29843-58-1
1,4-dichloro-2-methyl-2-butene

-
A
-
107-20-0
2-chloroethanal

-
B
-
78-95-5
chloroacetone

| Conditions | Yield |
|---|---|
| beim Ozonisierung und Behandeln des Ozonids mit Wasser bei 75-80grad; |


| Conditions | Yield |
|---|---|
| bei der Ozonspaltung; |

| Conditions | Yield |
|---|---|
| beim Destillieren unter gewoehnlichem Druck; |

| Conditions | Yield |
|---|---|
| With water |

| Conditions | Yield |
|---|---|
| at -20℃; |


| Conditions | Yield |
|---|---|
| With N-oxo-N-nitrosoamine at 280℃; under 367754 Torr; |

| Conditions | Yield |
|---|---|
| With chromic acid | |
| With ruthenium(IV) oxide; sodium chloride In water; ethyl acetate electrooxidation; | |
| With sodium hydroxide; N-bromoacetamide In water at 24.9℃; Mechanism; Kinetics; Thermodynamic data; var. of temp. (Tab. VI); ΔH(excit.), ΔS(excit.), ΔF(excit.); |
-
-
75-56-9, 16033-71-9
methyloxirane

-
-
78-95-5
chloroacetone

| Conditions | Yield |
|---|---|
| With chlorine at 0℃; |

| Conditions | Yield |
|---|---|
| With hydrogenchloride 1.) ether, 2 h, -20 to 0 deg C, 2.) -20 deg C to room temp.; Multistep reaction; |

| Conditions | Yield |
|---|---|
| for 48h; |
-
-
96-23-1
1,3-Dichloro-2-propanol

-
A
-
616-23-9
2,3-Dichloro-1-propanol

-
B
-
96-18-4
1,2,3-trichloropropane

-
C
-
23916-48-5
1-chloro-3-(2-chloro-1-chloromethyl-ethoxy)-propan-2-ol

-
D
-
78-95-5
chloroacetone

| Conditions | Yield |
|---|---|
| at 180℃; Product distribution; also in the presence of 1.5 percent HCl or 5percent epichlorhydrin; |


| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 60℃; for 4h; | 100% |

| Conditions | Yield |
|---|---|
| With triethylamine In tetrahydrofuran at 20℃; for 1h; | 100% |
| With potassium carbonate In tetrahydrofuran |
-
-
2687-43-6
O-benzylhydoxylamine hydrochloride

-
-
78-95-5
chloroacetone

-
-
83200-26-4
1-chloropropan-2-one O-benzyloxime

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate In water at 20℃; for 5h; | 100% |
| With sodium hydroxide In methanol 2.) room temp., 5 h; | 91% |
-
-
123979-29-3
2,6-dimethyl-1,5-dihydroxynaphthalene

-
-
78-95-5
chloroacetone

-
-
137517-19-2
2,6-Dimethyl-1,5-bis(2-oxopropoxy)naphthalene

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide Ambient temperature; | 100% |
-
-
137517-25-0
2,6-Dipropyl-1,5-naphthalenediol

-
-
78-95-5
chloroacetone

-
-
137517-22-7
2,6-Dipropyl-1,5-bis(2-oxopropoxy)naphthalene

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide Ambient temperature; | 100% |
-
-
132483-78-4
Diethyl 2-methyl-2-(4-thiocarbamoylphenyl)malonate

-
-
78-95-5
chloroacetone

-
-
138569-11-6
Diethyl 2-methyl-2-<4-(4-methylthiozol-2-yl)phenyl>malonate

| Conditions | Yield |
|---|---|
| In benzene for 3.25h; Heating; | 100% |
-
-
78-95-5
chloroacetone

-
-
18156-75-7
1-trimethylsilylpyrazole

| Conditions | Yield |
|---|---|
| at 80℃; for 1h; | 100% |
-
-
78-95-5
chloroacetone

-
-
18293-54-4
1-trimethylsilyl-1,2,4-triazole

| Conditions | Yield |
|---|---|
| at 100℃; for 1h; | 100% |
-
-
78-95-5
chloroacetone

-
-
43183-36-4
1-(trimethylsilyl)-1H-benzotriazole

| Conditions | Yield |
|---|---|
| at 100℃; for 1h; | 100% |
-
-
78-95-5
chloroacetone

-
-
15264-63-8
5-(4-pyridinyl)-1,3,4-oxadiazole-2-thiol

-
-
81555-96-6
1-(5-Pyridin-4-yl-[1,3,4]oxadiazol-2-ylsulfanyl)-propan-2-one

| Conditions | Yield |
|---|---|
| With sodium hydroxide for 12h; Ambient temperature; | 100% |
-
-
110766-90-0
8-Chloro-2-methyl-2,3-dihydro-5H-benzo[b][1,4]thiazepin-4-one

-
-
78-95-5
chloroacetone

| Conditions | Yield |
|---|---|
| With potassium hydroxide; tetrabutylammomium bromide In tetrahydrofuran Ambient temperature; | 100% |
-
-
78-95-5
chloroacetone

-
-
216776-76-0
6-Fluoro-4-methyl-2-phenyl-thieno[2,3-d]pyrimidin-5-ylamine

-
-
115073-27-3
6-acetyl-5-amino-4-methyl-2-phenylthieno[2,3-d]pyrimidine

| Conditions | Yield |
|---|---|
| With potassium carbonate | 100% |

| Conditions | Yield |
|---|---|
| at 60℃; for 12h; Solid phase reaction; substitution; | 100% |
-
-
78-95-5
chloroacetone

-
-
239131-42-1
5-(2-chlorophenyl)-3-mercapto-2-cyclohexen-1-one

-
-
239131-43-2
5-(2-chlorophenyl)-3-[(2-oxopropyl)thio]-2-cyclohexen-1-one

| Conditions | Yield |
|---|---|
| With sodium ethanolate In ethanol at 20℃; for 5h; | 100% |
| With sodium ethanolate In ethanol at 20℃; for 5h; |

| Conditions | Yield |
|---|---|
| With sodium hydride In dimethyl sulfoxide | 100% |
| With sodium hydride In dimethyl sulfoxide at 20℃; | 23% |
-
-
496-69-5
2-bromo-4-fluoro-phenol

-
-
78-95-5
chloroacetone

-
-
286836-12-2
1-(2-bromo-4-fluorophenoxy)propan-2-one

| Conditions | Yield |
|---|---|
| With potassium carbonate; potassium iodide In tetrahydrofuran for 4h; Heating / reflux; | 100% |
| With potassium carbonate; potassium iodide In tetrahydrofuran for 4h; Heating / reflux; | 100% |
| With potassium carbonate In N,N-dimethyl-formamide at 0 - 20℃; | 59% |
| With potassium carbonate; potassium iodide In acetone at 55℃; for 2h; | |
| With potassium iodide; potassium carbonate In tetrahydrofuran; ethyl acetate | 2.7 gm (100%) |

| Conditions | Yield |
|---|---|
| With potassium carbonate; potassium iodide In acetone at 0 - 20℃; for 1h; Heating / reflux; | 100% |
-
-
872707-11-4
N-[4-({[tert-butyl(dimethyl)silyl]oxy}methyl)pyridin-2-yl]thiourea

-
-
78-95-5
chloroacetone

-
-
872707-12-5
{2-[(4-methyl-1,3-thiazol-2-yl)amino]pyridin-4-yl}methanol

| Conditions | Yield |
|---|---|
| In ethanol for 16h; Heating / reflux; | 100% |
| In ethanol for 16h; Heating / reflux; | 100% |
| In ethanol for 16h; Heating / reflux; Under nitrogen; | 100% |
-
-
1059686-59-7
2-Cyclopropylamino-4,6-difluoro-benzoic acid

-
-
78-95-5
chloroacetone

-
-
1059686-60-0
2-Cyclopropylamino-4,6-difluoro-benzoic acid 2-oxo-propyl ester

| Conditions | Yield |
|---|---|
| Stage #1: 2-Cyclopropylamino-4,6-difluoro-benzoic acid With potassium carbonate In N,N-dimethyl-formamide at 90℃; for 1h; Stage #2: chloroacetone In N,N-dimethyl-formamide at 20 - 55℃; for 1.5h; | 100% |
-
-
953045-29-9
1-(5-bromo-3-(phenylthio)pyridin-2-yl)thiourea

-
-
78-95-5
chloroacetone

-
-
953040-73-8
N-(5-bromo-3-(phenylthio)pyridin-2-yl)-4-methylthiazol-2-amine

| Conditions | Yield |
|---|---|
| With triethylamine In ethanol at 70℃; for 6h; | 100% |
-
-
1189115-86-3
tert-butyl (S)-1-amino-3-(1H-indol-3-yl)-1-thioxopropan-2-yl carbamate

-
-
78-95-5
chloroacetone

-
-
1189115-87-4
tert-butyl [(S)-1-(4-hydroxy-4-methyl-4,5-dihydro-thiazol-2-yl)-2-(1H-indol-3-yl)-ethyl]-carbamate

| Conditions | Yield |
|---|---|
| With potassium hydrogencarbonate In 1,2-dimethoxyethane at 45 - 70℃; | 100% |
-
-
1186033-44-2
C9H9ClOS

-
-
78-95-5
chloroacetone

-
-
66490-34-4
1-(4-chloro-1H-benzo[b]thiophen-2-yl)ethanone

| Conditions | Yield |
|---|---|
| With calcium oxide In acetone at 59℃; | 100% |
| With calcium oxide for 3h; Reflux; |
-
-
53606-33-0
2-(ethylsulfanyl)benzenecarbaldehyde

-
-
78-95-5
chloroacetone

-
-
22720-75-8
2-acetyl benzo[b]thiophene

| Conditions | Yield |
|---|---|
| With calcium oxide In acetone at 59℃; | 100% |
| With calcium oxide for 3h; Reflux; |

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone at 60℃; for 20h; | 100% |
| With sodium hydrogencarbonate In toluene at 110 - 120℃; for 3h; Inert atmosphere; | 22.2% |

| Conditions | Yield |
|---|---|
| With N,N-dimethyl-formamide In water at 0 - 5℃; Inert atmosphere; | 100% |
-
-
78-95-5
chloroacetone

-
-
188115-27-7
(S)-11-thioxo-2,3,11,11a-tetrahydro-1H-benzo[e]-pyrrolo[1,2-a][1,4]diazepin-5(10H)-one

-
-
1271450-06-6
C15H16N2O2S

| Conditions | Yield |
|---|---|
| Stage #1: (S)-11-thioxo-2,3,11,11a-tetrahydro-1H-benzo[e]-pyrrolo[1,2-a][1,4]diazepin-5(10H)-one With sodium hydride In dimethyl sulfoxide at 20℃; for 0.5h; Inert atmosphere; Stage #2: chloroacetone In dimethyl sulfoxide at 20℃; for 0.666667h; Inert atmosphere; | 100% |
-
-
1152617-24-7
2-amino-3-chloro-4-iodopyridine

-
-
78-95-5
chloroacetone

-
-
1416551-60-4
8-chloro-7-iodo-2-methylimidazo[1,2-a]pyridine

| Conditions | Yield |
|---|---|
| In ethanol at 65℃; | 100% |
-
-
576-23-8
2,3-dimethylbromobenzene

-
-
78-95-5
chloroacetone

-
-
42432-45-1
2-(2,3-dimethylphenyl)-2-methyloxirane

| Conditions | Yield |
|---|---|
| Stage #1: 2,3-dimethylbromobenzene With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 0.5h; Stage #2: chloroacetone In tetrahydrofuran; hexane; toluene at -78 - 20℃; for 1.33333h; Product distribution / selectivity; | 100% |
| Stage #1: 2,3-dimethylbromobenzene With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 0.5h; Stage #2: chloroacetone In tetrahydrofuran; hexane; toluene at -78 - 20℃; Product distribution / selectivity; | 100% |
-
-
59108-90-6
1H-indole-3-carbothioamide

-
-
78-95-5
chloroacetone

-
-
93588-10-4
4-methyl-2-(3-indolyl)thiazole

| Conditions | Yield |
|---|---|
| In ethanol at 80℃; for 1.5h; Reflux; | 100% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In dichloromethane; acetonitrile at 20℃; for 8h; Reflux; | 100% |
| With potassium carbonate In dichloromethane; acetonitrile for 8h; Reflux; | 100% |
| With potassium carbonate In dichloromethane; acetonitrile for 8h; Reflux; |
Related products
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View

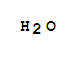

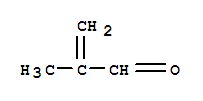











 T,
T,  F,
F,  Xi,
Xi,  T+,
T+,  N
N