Hunan Russell Chemicals Technology Co.,Ltd
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:Foil bag; Drum; Plastic bottle Application:Pharma;Industry;Agricultural Transportation:by sea or air Port:any port in China
Amadis Chemical Co., Ltd.
1.Professional synthesis laboratory and production base. 2.Strong synthesis team and service team. 3.Professional data management system. 4.We provide the professional test date and product information ,ex. HNMR ,CNMR,FNMR, HPLC/G
Cas:80-17-1
Min.Order:10 Milligram
Negotiable
Type:Lab/Research institutions
inquirySynthetic route

| Conditions | Yield |
|---|---|
| With hydrazine hydrate In chloroform at 0℃; for 2h; | 98% |
| With hydrazine hydrate In tetrahydrofuran at -8℃; for 0.5h; | 90% |
| With hydrazine hydrate In dichloromethane for 0.25h; | 90% |
-
-
74114-75-3
N-benzene sulfonyl-N'-carbo-tert-butoxy hydrazine

-
-
80-17-1
benzenesufonyl hydrazide

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid for 0.5h; | 84% |
| With trifluoroacetic acid for 1h; Ambient temperature; Yield given; |
-
-
126063-51-2
C13H19N2O2S(1-)*K(1+)

-
A
-
625-65-0
2,4-dimethyl-2-pentene

-
B
-
80-17-1
benzenesufonyl hydrazide

| Conditions | Yield |
|---|---|
| With Δ In dimethyl sulfoxide at 110℃; Rate constant; Kinetics; Thermodynamic data; other temp.; Ea, ΔH(excit.), ΔS(excit.) ΔG(excit.); | A 42% B 18% |

| Conditions | Yield |
|---|---|
| With hydrazine hydrate |
-
-
71202-80-7
D-glucose-benzenesulfonylhydrazone

-
-
7732-18-5
water

-
-
50-99-7, 59-23-4, 921-60-8, 1949-88-8, 1990-29-0, 2152-76-3, 2595-97-3, 2595-98-4, 3458-28-4, 4205-23-6, 5934-56-5, 5978-95-0, 5987-68-8, 6027-89-0, 6038-51-3, 7635-11-2, 10030-80-5, 15572-79-9, 19163-87-2, 23567-25-1, 26566-61-0, 30077-17-9, 31103-86-3, 39665-52-6, 40866-07-7, 58367-01-4, 58407-05-9, 58407-06-0, 83198-69-0, 83198-70-3, 83198-71-4, 93780-23-5, 145920-48-5
glucose

-
B
-
80-17-1
benzenesufonyl hydrazide


| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: triethylamine / acetonitrile / 0.01 h / 20 °C / Sonication 2: hydrazine hydrate / 0.02 h / Sonication View Scheme |
-
-
46248-01-5
benzenesulfonic acid imidazolide

-
-
80-17-1
benzenesufonyl hydrazide

| Conditions | Yield |
|---|---|
| With hydrazine hydrate for 0.0166667h; Sonication; |
-
-
51592-73-5
7,7-dideuteriobicyclo<3.2.0>hept-2-en-6-one

-
-
80-17-1
benzenesufonyl hydrazide

| Conditions | Yield |
|---|---|
| In methanol for 0.166667h; Heating; | 100% |
-
-
93750-00-6
Tricyclo[4.4.4.01,6]tetradeca-4,7,13-triene-3,9,12-trione

-
-
80-17-1
benzenesufonyl hydrazide

-
-
124820-82-2
C32H30N6O6S3

| Conditions | Yield |
|---|---|
| With hydrogenchloride In methanol for 144h; Ambient temperature; | 100% |
-
-
93775-15-6
Tricyclo[4.4.4.01,6]tetradeca-4,9,13-triene-3,8,12-trione

-
-
80-17-1
benzenesufonyl hydrazide

-
-
108189-51-1
C32H30N6O6S3

| Conditions | Yield |
|---|---|
| With hydrogenchloride In methanol for 144h; Ambient temperature; | 100% |
| Yield given; |
-
-
21794-55-8
(+)-Daunomycinone

-
-
80-17-1
benzenesufonyl hydrazide

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 20℃; for 18h; | 100% |
-
-
106-53-6
para-bromobenzenethiol

-
-
80-17-1
benzenesufonyl hydrazide

-
-
26974-36-7
benzenethiosulfonic acid S-(4-bromophenyl) ester

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; sodium iodide In acetonitrile at 25℃; for 8h; | 100% |
| With tert.-butylhydroperoxide; sodium iodide In acetonitrile at 20℃; for 12h; | 99% |
| With ammonium iodide In acetonitrile at 20℃; for 3h; Electrochemical reaction; | 65% |

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; sodium iodide In acetonitrile at 25℃; for 10h; | 100% |
| With tert.-butylhydroperoxide; sodium iodide In acetonitrile at 20℃; for 12h; | 99% |

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; sodium iodide In acetonitrile at 25℃; | 100% |
| With tert.-butylhydroperoxide; sodium iodide In acetonitrile at 20℃; for 12h; | 99% |
| With ammonium iodide In acetonitrile at 20℃; for 3h; Electrochemical reaction; | 78% |
-
-
80-17-1
benzenesufonyl hydrazide

-
-
137-06-4
2-thiocresol

-
-
96097-67-5
benzenethiosulfonic acid S-o-tolyl ester

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; sodium iodide In acetonitrile at 25℃; for 6h; | 100% |
| With tert.-butylhydroperoxide; sodium iodide In acetonitrile at 20℃; for 12h; | 99% |
| With ammonium iodide In acetonitrile at 20℃; for 3h; Electrochemical reaction; | 75% |

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; sodium iodide In acetonitrile at 25℃; for 6h; | 100% |
| With tert.-butylhydroperoxide; sodium iodide In acetonitrile at 20℃; for 12h; | 99% |
| With ammonium iodide In acetonitrile at 20℃; for 3h; Electrochemical reaction; | 78% |

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; sodium iodide In acetonitrile at 25℃; for 12h; | 100% |
| With tert.-butylhydroperoxide; sodium iodide In acetonitrile at 20℃; for 12h; | 99% |

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; sodium iodide In acetonitrile at 25℃; for 6h; Reagent/catalyst; Solvent; | 100% |
| With tert.-butylhydroperoxide; sodium iodide In acetonitrile at 20℃; for 12h; Reagent/catalyst; Solvent; | 99% |
| With ammonium iodide In acetonitrile at 20℃; for 3h; Solvent; Reagent/catalyst; Electrochemical reaction; | 82% |

| Conditions | Yield |
|---|---|
| With acetic acid In ethanol Reflux; | 100% |
-
-
334-99-6
trifluoronitrosomethane

-
-
80-17-1
benzenesufonyl hydrazide

-
-
88978-51-2
N-trifluoromethyl-N-hydroxybenzenesulfonamide

| Conditions | Yield |
|---|---|
| In tetrahydrofuran for 1.5h; Product distribution; Ambient temperature; oth. arenesulfonohydrazides, oth. solvent,; | 99% |
-
-
1005008-04-7
(4bR,8aS,9S)-4b-methoxy-7-oxo-4b,7,8,8a,9,10-hexahydrophenanthrene-9-carbaldehyde

-
-
80-17-1
benzenesufonyl hydrazide

-
-
1005008-21-8
(E)-N'-(((4bS,8aR,9R)-4b-methoxy-7-oxo-4b,7,8,8a,9,10-hexahydrophenanthrene-9-yl)methylene)benzenesulfonylhydrazide

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; for 0.25h; | 99% |
-
-
135-02-4
ortho-anisaldehyde

-
-
80-17-1
benzenesufonyl hydrazide

-
-
145260-32-8
(E)-N'-(2-methoxybenzylidene)benzenesulfonohydrazide

| Conditions | Yield |
|---|---|
| In ethanol at 20℃; for 1h; | 99% |
| In ethanol at 20℃; for 3h; | 51% |
-
-
4843-42-9
3-(1-naphthyl)propiolic acid

-
-
80-17-1
benzenesufonyl hydrazide

-
-
4044-57-9
1-phenylethynyl-naphthalene

| Conditions | Yield |
|---|---|
| With palladium(II) trifluoroacetate; 1,3-bis-(diphenylphosphino)propane; copper diacetate In N,N-dimethyl-formamide at 100℃; for 0.5h; | 99% |
-
-
106-54-7
p-Chlorothiophenol

-
-
80-17-1
benzenesufonyl hydrazide

-
-
1213-40-7
S-(4-chlorophenyl)benzene thiosulfonate

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; sodium iodide In acetonitrile at 20℃; for 12h; | 99% |
| With ammonium iodide In acetonitrile at 20℃; for 3h; Electrochemical reaction; | 72% |

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; sodium iodide In acetonitrile at 20℃; for 12h; | 99% |
| With ammonium iodide In acetonitrile at 20℃; for 3h; Electrochemical reaction; | 70% |
-
-
696-63-9
4-Methoxybenzenethiol

-
-
80-17-1
benzenesufonyl hydrazide

-
-
1146-48-1
benzenethiosulfonic acid S-(4-methoxyphenyl) ester

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; sodium iodide In acetonitrile at 20℃; for 12h; | 99% |
| With ammonium iodide In acetonitrile at 20℃; for 3h; Electrochemical reaction; | 85% |

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; sodium iodide In acetonitrile at 20℃; for 12h; | 99% |
| With ammonium iodide In acetonitrile at 20℃; for 3h; Electrochemical reaction; | 78% |
-
-
15570-12-4
3-methoxybenzenethiol

-
-
80-17-1
benzenesufonyl hydrazide

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; sodium iodide In acetonitrile at 20℃; for 12h; | 99% |

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; sodium iodide In acetonitrile at 20℃; for 12h; | 99% |

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; sodium iodide In acetonitrile at 20℃; for 12h; | 99% |

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; sodium iodide In acetonitrile at 20℃; for 12h; | 99% |

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; sodium iodide In acetonitrile at 20℃; for 12h; | 99% |
-
-
2557-78-0
2-fluorothiophenol

-
-
80-17-1
benzenesufonyl hydrazide

-
-
1394124-47-0
S-(2-fluorophenyl) benzenesulfonothioate

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; sodium iodide In acetonitrile at 20℃; for 12h; | 99% |

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; sodium iodide In acetonitrile at 20℃; for 12h; | 99% |

| Conditions | Yield |
|---|---|
| In ethanol at 20℃; for 1h; | 99% |

| Conditions | Yield |
|---|---|
| With tetra-(n-butyl)ammonium iodide; propylene glycol dimethyl ether dimer at 100℃; for 12h; Schlenk technique; | 99% |
Related products
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View




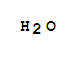








 F
F