-
Name
Benzenesulfonyl hydrazide
- EINECS 201-255-2
- CAS No. 80-17-1
- Article Data89
- CAS DataBase
- Density 1.353 g/cm3
- Solubility Insoluble in water
- Melting Point 101-103 °C(lit.)
- Formula C6H8N2O2S
- Boiling Point 333.5 °C at 760 mmHg
- Molecular Weight 172.208
- Flash Point 155.5 °C
- Transport Information UN 3226 4.1
- Appearance white to pale yellow powder
- Safety 15-16-22-24/25
- Risk Codes 11
-
Molecular Structure
-
Hazard Symbols
 F
F
- Synonyms Benzenesulfonic acid, hydrazide;ChKhZ 9;N-Benzenesulfonylhydrazide;NSC 643;Nitropore OBSH;Phenylsulfohydrazide;Porofor-BSH-Pulver;Benzenesulfohydrazide;
- PSA 80.57000
- LogP 2.01060
Synthetic route

| Conditions | Yield |
|---|---|
| With hydrazine hydrate In chloroform at 0℃; for 2h; | 98% |
| With hydrazine hydrate In tetrahydrofuran at -8℃; for 0.5h; | 90% |
| With hydrazine hydrate In dichloromethane for 0.25h; | 90% |
-
-
74114-75-3
N-benzene sulfonyl-N'-carbo-tert-butoxy hydrazine

-
-
80-17-1
benzenesufonyl hydrazide

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid for 0.5h; | 84% |
| With trifluoroacetic acid for 1h; Ambient temperature; Yield given; |
-
-
126063-51-2
C13H19N2O2S(1-)*K(1+)

-
A
-
625-65-0
2,4-dimethyl-2-pentene

-
B
-
80-17-1
benzenesufonyl hydrazide

| Conditions | Yield |
|---|---|
| With Δ In dimethyl sulfoxide at 110℃; Rate constant; Kinetics; Thermodynamic data; other temp.; Ea, ΔH(excit.), ΔS(excit.) ΔG(excit.); | A 42% B 18% |

| Conditions | Yield |
|---|---|
| With hydrazine hydrate |
-
-
71202-80-7
D-glucose-benzenesulfonylhydrazone

-
-
7732-18-5
water

-
-
50-99-7, 59-23-4, 921-60-8, 1949-88-8, 1990-29-0, 2152-76-3, 2595-97-3, 2595-98-4, 3458-28-4, 4205-23-6, 5934-56-5, 5978-95-0, 5987-68-8, 6027-89-0, 6038-51-3, 7635-11-2, 10030-80-5, 15572-79-9, 19163-87-2, 23567-25-1, 26566-61-0, 30077-17-9, 31103-86-3, 39665-52-6, 40866-07-7, 58367-01-4, 58407-05-9, 58407-06-0, 83198-69-0, 83198-70-3, 83198-71-4, 93780-23-5, 145920-48-5
glucose

-
B
-
80-17-1
benzenesufonyl hydrazide


| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: triethylamine / acetonitrile / 0.01 h / 20 °C / Sonication 2: hydrazine hydrate / 0.02 h / Sonication View Scheme |
-
-
46248-01-5
benzenesulfonic acid imidazolide

-
-
80-17-1
benzenesufonyl hydrazide

| Conditions | Yield |
|---|---|
| With hydrazine hydrate for 0.0166667h; Sonication; |
-
-
51592-73-5
7,7-dideuteriobicyclo<3.2.0>hept-2-en-6-one

-
-
80-17-1
benzenesufonyl hydrazide

| Conditions | Yield |
|---|---|
| In methanol for 0.166667h; Heating; | 100% |
-
-
93750-00-6
Tricyclo[4.4.4.01,6]tetradeca-4,7,13-triene-3,9,12-trione

-
-
80-17-1
benzenesufonyl hydrazide

-
-
124820-82-2
C32H30N6O6S3

| Conditions | Yield |
|---|---|
| With hydrogenchloride In methanol for 144h; Ambient temperature; | 100% |
-
-
93775-15-6
Tricyclo[4.4.4.01,6]tetradeca-4,9,13-triene-3,8,12-trione

-
-
80-17-1
benzenesufonyl hydrazide

-
-
108189-51-1
C32H30N6O6S3

| Conditions | Yield |
|---|---|
| With hydrogenchloride In methanol for 144h; Ambient temperature; | 100% |
| Yield given; |
-
-
21794-55-8
(+)-Daunomycinone

-
-
80-17-1
benzenesufonyl hydrazide

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 20℃; for 18h; | 100% |
-
-
106-53-6
para-bromobenzenethiol

-
-
80-17-1
benzenesufonyl hydrazide

-
-
26974-36-7
benzenethiosulfonic acid S-(4-bromophenyl) ester

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; sodium iodide In acetonitrile at 25℃; for 8h; | 100% |
| With tert.-butylhydroperoxide; sodium iodide In acetonitrile at 20℃; for 12h; | 99% |
| With ammonium iodide In acetonitrile at 20℃; for 3h; Electrochemical reaction; | 65% |

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; sodium iodide In acetonitrile at 25℃; for 10h; | 100% |
| With tert.-butylhydroperoxide; sodium iodide In acetonitrile at 20℃; for 12h; | 99% |

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; sodium iodide In acetonitrile at 25℃; | 100% |
| With tert.-butylhydroperoxide; sodium iodide In acetonitrile at 20℃; for 12h; | 99% |
| With ammonium iodide In acetonitrile at 20℃; for 3h; Electrochemical reaction; | 78% |
-
-
80-17-1
benzenesufonyl hydrazide

-
-
137-06-4
2-thiocresol

-
-
96097-67-5
benzenethiosulfonic acid S-o-tolyl ester

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; sodium iodide In acetonitrile at 25℃; for 6h; | 100% |
| With tert.-butylhydroperoxide; sodium iodide In acetonitrile at 20℃; for 12h; | 99% |
| With ammonium iodide In acetonitrile at 20℃; for 3h; Electrochemical reaction; | 75% |

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; sodium iodide In acetonitrile at 25℃; for 6h; | 100% |
| With tert.-butylhydroperoxide; sodium iodide In acetonitrile at 20℃; for 12h; | 99% |
| With ammonium iodide In acetonitrile at 20℃; for 3h; Electrochemical reaction; | 78% |

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; sodium iodide In acetonitrile at 25℃; for 12h; | 100% |
| With tert.-butylhydroperoxide; sodium iodide In acetonitrile at 20℃; for 12h; | 99% |

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; sodium iodide In acetonitrile at 25℃; for 6h; Reagent/catalyst; Solvent; | 100% |
| With tert.-butylhydroperoxide; sodium iodide In acetonitrile at 20℃; for 12h; Reagent/catalyst; Solvent; | 99% |
| With ammonium iodide In acetonitrile at 20℃; for 3h; Solvent; Reagent/catalyst; Electrochemical reaction; | 82% |

| Conditions | Yield |
|---|---|
| With acetic acid In ethanol Reflux; | 100% |
-
-
334-99-6
trifluoronitrosomethane

-
-
80-17-1
benzenesufonyl hydrazide

-
-
88978-51-2
N-trifluoromethyl-N-hydroxybenzenesulfonamide

| Conditions | Yield |
|---|---|
| In tetrahydrofuran for 1.5h; Product distribution; Ambient temperature; oth. arenesulfonohydrazides, oth. solvent,; | 99% |
-
-
1005008-04-7
(4bR,8aS,9S)-4b-methoxy-7-oxo-4b,7,8,8a,9,10-hexahydrophenanthrene-9-carbaldehyde

-
-
80-17-1
benzenesufonyl hydrazide

-
-
1005008-21-8
(E)-N'-(((4bS,8aR,9R)-4b-methoxy-7-oxo-4b,7,8,8a,9,10-hexahydrophenanthrene-9-yl)methylene)benzenesulfonylhydrazide

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; for 0.25h; | 99% |
-
-
135-02-4
ortho-anisaldehyde

-
-
80-17-1
benzenesufonyl hydrazide

-
-
145260-32-8
(E)-N'-(2-methoxybenzylidene)benzenesulfonohydrazide

| Conditions | Yield |
|---|---|
| In ethanol at 20℃; for 1h; | 99% |
| In ethanol at 20℃; for 3h; | 51% |
-
-
4843-42-9
3-(1-naphthyl)propiolic acid

-
-
80-17-1
benzenesufonyl hydrazide

-
-
4044-57-9
1-phenylethynyl-naphthalene

| Conditions | Yield |
|---|---|
| With palladium(II) trifluoroacetate; 1,3-bis-(diphenylphosphino)propane; copper diacetate In N,N-dimethyl-formamide at 100℃; for 0.5h; | 99% |
-
-
106-54-7
p-Chlorothiophenol

-
-
80-17-1
benzenesufonyl hydrazide

-
-
1213-40-7
S-(4-chlorophenyl)benzene thiosulfonate

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; sodium iodide In acetonitrile at 20℃; for 12h; | 99% |
| With ammonium iodide In acetonitrile at 20℃; for 3h; Electrochemical reaction; | 72% |

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; sodium iodide In acetonitrile at 20℃; for 12h; | 99% |
| With ammonium iodide In acetonitrile at 20℃; for 3h; Electrochemical reaction; | 70% |
-
-
696-63-9
4-Methoxybenzenethiol

-
-
80-17-1
benzenesufonyl hydrazide

-
-
1146-48-1
benzenethiosulfonic acid S-(4-methoxyphenyl) ester

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; sodium iodide In acetonitrile at 20℃; for 12h; | 99% |
| With ammonium iodide In acetonitrile at 20℃; for 3h; Electrochemical reaction; | 85% |

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; sodium iodide In acetonitrile at 20℃; for 12h; | 99% |
| With ammonium iodide In acetonitrile at 20℃; for 3h; Electrochemical reaction; | 78% |
-
-
15570-12-4
3-methoxybenzenethiol

-
-
80-17-1
benzenesufonyl hydrazide

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; sodium iodide In acetonitrile at 20℃; for 12h; | 99% |

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; sodium iodide In acetonitrile at 20℃; for 12h; | 99% |

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; sodium iodide In acetonitrile at 20℃; for 12h; | 99% |

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; sodium iodide In acetonitrile at 20℃; for 12h; | 99% |

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; sodium iodide In acetonitrile at 20℃; for 12h; | 99% |
-
-
2557-78-0
2-fluorothiophenol

-
-
80-17-1
benzenesufonyl hydrazide

-
-
1394124-47-0
S-(2-fluorophenyl) benzenesulfonothioate

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; sodium iodide In acetonitrile at 20℃; for 12h; | 99% |

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; sodium iodide In acetonitrile at 20℃; for 12h; | 99% |

| Conditions | Yield |
|---|---|
| In ethanol at 20℃; for 1h; | 99% |

| Conditions | Yield |
|---|---|
| With tetra-(n-butyl)ammonium iodide; propylene glycol dimethyl ether dimer at 100℃; for 12h; Schlenk technique; | 99% |
Benzenesulfonyl hydrazide Consensus Reports
Benzenesulfonyl hydrazide Standards and Recommendations
Benzenesulfonyl hydrazide Specification
The Benzenesulfonic acid, hydrazide, with the CAS registry number 80-17-1, is also known as Phenylsulfonyl hydrazide. It belongs to the product category of Pharmaceutical Intermediates. Its EINECS number is 201-255-2. This chemical's molecular formula is C6H8N2O2S and molecular weight is 172.20. What's more, its systematic name is benzenesulfonohydrazide. It should be sealed and stored in a cool, ventilated and dry place. Moreover, it should be protected from light and moisture. It is mainly used as foaming agent in the manufacturing of foam plastics and foam rubber.
Physical properties of Benzenesulfonic acid, hydrazide are: (1)ACD/LogP: -0.14; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): -0.14; (4)ACD/LogD (pH 7.4): -0.15; (5)ACD/BCF (pH 5.5): 1; (6)ACD/BCF (pH 7.4): 1; (7)ACD/KOC (pH 5.5): 19.99; (8)ACD/KOC (pH 7.4): 19.75; (9)#H bond acceptors: 4; (10)#H bond donors: 3; (11)#Freely Rotating Bonds: 2; (12)Polar Surface Area: 49 Å2; (13)Index of Refraction: 1.588; (14)Molar Refractivity: 42.84 cm3; (15)Molar Volume: 127.1 cm3; (16)Polarizability: 16.98×10-24cm3; (17)Surface Tension: 51.8 dyne/cm; (18)Density: 1.353 g/cm3; (19)Flash Point: 155.5 °C; (20)Enthalpy of Vaporization: 57.64 kJ/mol; (21)Boiling Point: 333.5 °C at 760 mmHg; (22)Vapour Pressure: 0.000136 mmHg at 25°C.
Preparation of Benzenesulfonic acid, hydrazide: this chemical can be prepared by benzenesulfonyl chloride by heating. This reaction will need reagents triethylamine, Me3Si-NHNH2. The yield is about 88%.

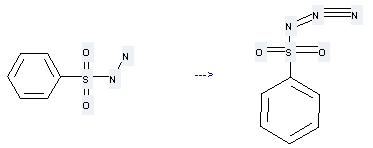
Uses of Benzenesulfonic acid, hydrazide: it can be used to produce benzenesulfonyl azide at the ambient temperature. It will need reagent SHNC and solvent H2O with the reaction time of 1.5 hours. The yield is about 92%.
When you are using this chemical, please be cautious about it as the following:
This chemical is highly flammable, so you should keep it away from sources of ignition - No smoking. You must keep it away from heat. You should not breathe dust. When using it, you must avoid contact with eyes.
You can still convert the following datas into molecular structure:
(1)SMILES: O=S(=O)(NN)c1ccccc1
(2)Std. InChI: InChI=1S/C6H8N2O2S/c7-8-11(9,10)6-4-2-1-3-5-6/h1-5,8H,7H2
(3)Std. InChIKey: VJRITMATACIYAF-UHFFFAOYSA-N
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| rat | LDLo | oral | 50mg/kg (50mg/kg) | International Polymer Science and Technology. Vol. 3, Pg. 93, 1976. |
Related Products
- Benzenesulfonyl bromide
- Benzenesulfonyl chloride
- Benzenesulfonyl chloride, 2,3,5,6-tetramethyl-
- Benzenesulfonyl chloride, 2,4-dichloro-3-methyl-
- Benzenesulfonyl chloride, 2,4-dimethoxy-
- Benzenesulfonyl chloride, 2,5-dibromo-3,6-difluoro-
- Benzenesulfonyl chloride, 2,6-dimethyl-
- Benzenesulfonyl chloride, 3-(1-methyl-1H-pyrazol-3-yl)-
- Benzenesulfonyl chloride, 4-(1H-pyrazol-1-yl)-
- Benzenesulfonyl chloride, 4-(2-thienyl)-
- 8017-16-1
- 80171-68-2
- 80172-04-9
- 80173-27-9
- 80174-64-7
- 8018-01-7
- 80181-31-3
- 80-18-2
- 80189-07-7
- 80192-55-8
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View