Ality Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Cas:881386-01-2
Min.Order:1
Negotiable
Type:Other
inquiryXi'an Xszo Chem Co., Ltd.
1. Factory price and high quality must be guaranteed, base on 8 years of production and R&D experience2. Free samples will be provided,ensure specifications and quality are right for customer3. Customers will receive the most professional technical s
Cas:881386-01-2
Min.Order:1 Gram
FOB Price: $0.1
Type:Manufacturers
inquiryHebei Nengqian Chemical Import and Export Co., LTD
Our advantages: 1, High quality with competitive price: 1) Standard:BP/USP/EP/Enterprise standard 2) All Purity≥99% 3) We are manufacturer and can provide high quality products with factory price. 2, Fast and safe delivery 1) Parcel can be
Cas:881386-01-2
Min.Order:1 Gram
FOB Price: $1.0 / 2.0
Type:Trading Company
inquiryZhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:881386-01-2
Min.Order:10 Gram
FOB Price: $146.0 / 176.0
Type:Trading Company
inquiryHenan Sinotech Import&Export Corporation
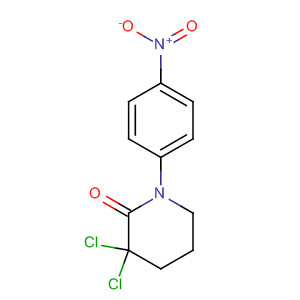
Name: 3,3-Dichloro-1-(4-nitrophenyl)-2-piperidinone CAS No.: 881386-01-2 MF: C11H10Cl2N2O3 Appearance: White Fine Powder purity > 99.0
Cas:881386-01-2
Min.Order:1 Kilogram
FOB Price: $1.0
Type:Other
inquiryShijiazhuang Sdyano Fine Chemical Co., Ltd
1. Best prices with satisfied quality; 2. It's clients' right to choose the package's Courier (EMS, DHL, FedEx, UPS); 3.It's clients' right to choose the packing way for his produccts from many recent effective packing ways;
Cas:881386-01-2
Min.Order:0 Metric Ton
Negotiable
Type:Trading Company
inquiryBaoji Guokang Healthchem co.,ltd
Our company has been in existence for 10 years since its establishment. We have our own unique team. The company integrates independent research and development, production and sales. We have established famous brands at home and abroad. At present,
Cas:881386-01-2
Min.Order:1 Kilogram
FOB Price: $40.0
Type:Trading Company
inquiryHubei Jiutian Bio-medical Technology Co., Ltd
1,we produce and sell good chemicals around the world. 2,our success rate is about 95%. this means, if customer order is accepted, the probability that the customer will obtain the ordered substances, is 95%. 3,our staff consists of highly qualifie
Cas:881386-01-2
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryAfine Chemicals Limited
Company Introduction 1. Established in 2005, with two independent business divisions: Fine chemicals division; Pharmaceutical division. 2. Main product: Optical brightener Textile auxiliary Dye stuff Pigments
SHANGHAI T&W PHARMACEUTICAL CO., LTD.
A substitute for perfluorooctanoic acid, mainly used as a surfactant, dispersant, additive, etc Appearance:White solid or Colorless liquid Purity:99.3 % We will ship the goods in a timely manner as required We can provide relevant documents acc
Cas:881386-01-2
Min.Order:4 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Cas:881386-01-2
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquirySiwei Development Group Ltd.
Product name: 3,3-Dichloro-1-(4-Nitrophenyl)-2-Piperidinone CAS No.: 881386-01-2 Molecule Formula:C11H10Cl2N2O3 Molecule Weight:289.11 Purity: 99.0% Package: 25kg/drum Description:White to off-white powder Manufacture Standards:Enterp
Cas:881386-01-2
Min.Order:1 Kilogram
Negotiable
Type:Trading Company
inquiryHenan Tianfu Chemical Co., Ltd.
Our company was built in 2009 with an ISO certificate.In the past 5 years, we have grown up as a famous fine chemicals supplier in China and we had established stable business relationships with Samsung,LG,Merck,Thermo Fisher Scientific and so on.O
Cas:881386-01-2
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryXiamen Jenny Chemical Technology Co., Ltd.
GMP standard, high purity, competitive price, in stock 1. Quick Response: within 6 hours after receiving your email. 2. Quality Guarantee: All products are strictly tested by our QC, confirmed by QA, and approved by a third-party lab in China, USA,
Cas:881386-01-2
Min.Order:1 Milligram
Negotiable
Type:Trading Company
inquiryShanghai Minstar Chemical Co., Ltd
3,3-Dichloro-1-(4-nitrophenyl)piperidin-2-one Chemical Properties Boiling point 520.2±50.0 °C(Predicted) density 1.50 storage temp. Sealed in dry,2-8°C pka -7.38±0.40(Predicted)
Cas:881386-01-2
Min.Order:1 Gram
FOB Price: $66.0
Type:Lab/Research institutions
inquiryHangzhou Huarong Pharm Co., Ltd.
We Huarong Pharm can provide Customized Synthesis & Process R&D & APIs and intermediates Production & Quality Research & Registration Application, especially our GMP validation service which complies with SFDA, FDA, WHO and EU EMPA. O
Cas:881386-01-2
Min.Order:0
Negotiable
Type:Trading Company
inquiryTAIZHOU ZHENYU BIOTECHNOLOGY CO., LTD
Zhenyu biotech exported this product to many countries and regions at best price. if you are looking for the material's manufacturer or supplier in china, zhenyu biotech is your best choice. pls contact with us freely for getting detailed
Cas:881386-01-2
Min.Order:1 Kilogram
FOB Price: $2.0
Type:Lab/Research institutions
inquirySAGECHEM LIMITED
SAGECHEM is a chemical R&D, manufacturing and distribution company in China since 2009, including pharmaceutical intermediates, agrochemical, dyestuff intermediates, organosilicone, API and etc. We also offer a full range of services in
Cas:881386-01-2
Min.Order:0 Metric Ton
Negotiable
Type:Lab/Research institutions
inquiryGolden Pharma Co., Limited
GOLDEN PHARMA CO.,LIMITED.is a professional pharmaceutical company,our team have more than 20years expereince in pharmaceutical production and sales. we are a professional technical enterprise specializing in the R & D, production,QA regulation
Hangzhou Dingyan Chem Co., Ltd
R & D enterprises have their own stock in stockAppearance:To be subject to the object Package:Customized Application:pharmaceutical intermediates Transportation:Air Port:Shanghai;Guangzhou
Cas:881386-01-2
Min.Order:0
Negotiable
Type:Manufacturers
inquiryShandong Jiling Chemical Co., Ltd
Appearance:Powder Storage:Sealed and preserved Package:200/Kilograms Application:Fine chemical intermediates, used as the main raw material for the synthesis of various pesticides, medicines, surfactants, polymer monomers, and antifungal agents Trans
Hubei Taiho Chemical Co.,LTD
TAIHO Unique Advantages:1.We're factory2.Free samples available3.Commodity inspection can be done4.ISO9001,Kosher certifications5.10 years experiences Storage:Store in cool &dry place Package:aluminium foil bag/fiber can/plastic drum Application:comp
Cas:881386-01-2
Min.Order:0
Negotiable
Type:Manufacturers
inquiryXi`an Eastling Biotech Co., Ltd.
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:Foil bag; Drum; Plastic bottle Application:Pharma;Industry;Agricultural Transportation:by sea or air Port:Beijing or Guangzhou
Cas:881386-01-2
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryAntimex Chemical Limied
Our own factory produces direct sales with absolute price advantage Application:Pharmaceutical industry Transportation:By sea Port:Shanghai/tianjin
Cas:881386-01-2
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryGuangdong Juda Chemical Industrial Co.,Limited
Factory supply high purity low priceAppearance:solid or liquid Storage:sealed in cool and dry place Package:As customer's requested Application:Pharma Intermediate Transportation:by courier/air/sea Port:Any port in China
Cas:881386-01-2
Min.Order:0
Negotiable
Type:Trading Company
inquiryWuhan Circle Star Chem-medical Technology co.,Ltd.
good quality, competitive price, thoughtful after sale serviceAppearance:white powder Storage:Keep it in dry,shady and cool place Package:25kg,50kg,180kg,200kg,250kg,1000kg,customization Application:Pharma;Industry;Agricultural;chemical reaserch Tran
Cas:881386-01-2
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryHangzhou Fandachem Co.,Ltd
2-Piperidinone, 3,3-dichloro-1-(4-nitrophenyl)-Appearance:white crystalline powder Storage:Store in dry, dark and ventilated place Package:25KG drum Application:pharmaceutical intermediate Transportation:by air, by sea, by express
Cas:881386-01-2
Min.Order:0
Negotiable
Type:Other
inquiryChemlyte Solutions
Stock products, own laboratory Package:Grams, Kilograms Application:For R&D Transportation:According to customer request Port:Shanghai
Cas:881386-01-2
Min.Order:0
Negotiable
Type:Other
inquiryShanghai Scochem Technology Co., Ltd.
High quality suppliers Application:healing drugs
Cas:881386-01-2
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryChemsigma International Co.,Ltd.
bulk?production Application:Pharmaceutical intermediates
Cas:881386-01-2
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquirySynthetic route
-
-
38560-30-4
1-(4-nitrophenyl)piperidine-2-one

-
-
881386-01-2
3,3-dichloro-1-(4-nitrophenyl)piperidine-2-one

| Conditions | Yield |
|---|---|
| With phosphorus pentachloride In chlorobenzene at 55℃; for 5h; | 94.9% |
| With phosphorus pentachloride In dichloromethane at 40℃; Cooling with ice; Inert atmosphere; | 93.1% |
| With phosphorus pentachloride In chloroform for 0.5h; Reflux; | 88.58% |
-
-
1575-61-7
5-Chlorovaleroyl chloride

-
-
881386-01-2
3,3-dichloro-1-(4-nitrophenyl)piperidine-2-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: triethylamine / dichloromethane / 0 - 20 °C / Inert atmosphere 2: phosphorus pentachloride / dichloromethane / Reflux View Scheme | |
| Multi-step reaction with 3 steps 1: N-ethyl-N,N-diisopropylamine / tetrahydrofuran / 5 - 25 °C / Inert atmosphere 2: sodium hydride / tetrahydrofuran / 10 - 25 °C / Inert atmosphere 3: phosphorus pentachloride / dichloromethane / 40 °C / Cooling with ice; Inert atmosphere View Scheme | |
| Multi-step reaction with 3 steps 1: trimethylamine / tetrahydrofuran / 0 - 20 °C 2: potassium carbonate; dimethyl sulfoxide / 6 h / 80 °C 3: phosphorus pentachloride / dichloromethane / 5 h / Reflux View Scheme |
-
-
100-01-6
4-nitro-aniline

-
-
881386-01-2
3,3-dichloro-1-(4-nitrophenyl)piperidine-2-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: triethylamine / dichloromethane / 0 - 20 °C / Inert atmosphere 2: phosphorus pentachloride / dichloromethane / Reflux View Scheme | |
| Multi-step reaction with 3 steps 1: N-ethyl-N,N-diisopropylamine / tetrahydrofuran / 5 - 25 °C / Inert atmosphere 2: sodium hydride / tetrahydrofuran / 10 - 25 °C / Inert atmosphere 3: phosphorus pentachloride / dichloromethane / 40 °C / Cooling with ice; Inert atmosphere View Scheme | |
| Multi-step reaction with 3 steps 1: trimethylamine / tetrahydrofuran / 0 - 20 °C 2: potassium carbonate; dimethyl sulfoxide / 6 h / 80 °C 3: phosphorus pentachloride / dichloromethane / 5 h / Reflux View Scheme |
-
-
1039914-85-6
5-chloro-N-(4-nitrophenyl)pentanamide

-
-
881386-01-2
3,3-dichloro-1-(4-nitrophenyl)piperidine-2-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: sodium hydride / tetrahydrofuran / 10 - 25 °C / Inert atmosphere 2: phosphorus pentachloride / dichloromethane / 40 °C / Cooling with ice; Inert atmosphere View Scheme | |
| Multi-step reaction with 2 steps 1: potassium carbonate; dimethyl sulfoxide / 6 h / 80 °C 2: phosphorus pentachloride / dichloromethane / 5 h / Reflux View Scheme | |
| Multi-step reaction with 2 steps 1: potassium carbonate / dimethyl sulfoxide / 6 h / 80 °C 2: phosphorus pentachloride / dichloromethane / 5 h / Reflux View Scheme | |
| Multi-step reaction with 2 steps 1: tetrabutylammomium bromide; potassium hydroxide / water; tetrahydrofuran / 0 - 20 °C 2: phosphorus pentachloride / toluene / 1 h / 25 - 80 °C View Scheme |
-
-
4509-90-4
5-bromovaleroyl chloride

-
-
881386-01-2
3,3-dichloro-1-(4-nitrophenyl)piperidine-2-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: pyridine; dmap / tetrahydrofuran / 1 h / 0 - 100 °C 2: phosphorus pentachloride / chloroform / 0.5 h / Reflux View Scheme | |
| Multi-step reaction with 2 steps 1.1: water; tetrahydrofuran / 1 h / 20 °C / Green chemistry 1.2: 4 h / 20 °C / Green chemistry 2.1: phosphorus pentachloride / chlorobenzene / 2 h / 53 °C / Green chemistry View Scheme |
-
-
62-53-3
aniline

-
-
881386-01-2
3,3-dichloro-1-(4-nitrophenyl)piperidine-2-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: sodium hydroxide; tetrabutylammomium bromide / dichloromethane; water / 1 h / 0 - 5 °C 1.2: 20 °C 2.1: nitric acid; sulfuric acid / 0 - 5 °C 3.1: phosphorus pentachloride / toluene / 1 h / 25 - 80 °C View Scheme | |
| Multi-step reaction with 4 steps 1: sodium hydroxide; tetrabutylammomium bromide / dichloromethane; water / 1 h / 25 - 30 °C 2: potassium hydroxide / dichloromethane; N,N-dimethyl-formamide / 1 h / 0 - 20 °C / Inert atmosphere 3: nitric acid; sulfuric acid / 0 - 5 °C 4: phosphorus pentachloride / toluene / 1 h / 25 - 80 °C View Scheme |
-
-
4789-09-7
1-phenylpiperidin-2-one

-
-
881386-01-2
3,3-dichloro-1-(4-nitrophenyl)piperidine-2-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: nitric acid; sulfuric acid / 0 - 5 °C 2: phosphorus pentachloride / toluene / 1 h / 25 - 80 °C View Scheme |
-
-
91131-23-6
5-chloro-pentanoic acid phenylamide

-
-
881386-01-2
3,3-dichloro-1-(4-nitrophenyl)piperidine-2-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: potassium hydroxide / dichloromethane; N,N-dimethyl-formamide / 1 h / 0 - 20 °C / Inert atmosphere 2: nitric acid; sulfuric acid / 0 - 5 °C 3: phosphorus pentachloride / toluene / 1 h / 25 - 80 °C View Scheme |
-
-
881386-01-2
3,3-dichloro-1-(4-nitrophenyl)piperidine-2-one

| Conditions | Yield |
|---|---|
| With tin(II) chloride dihdyrate In N,N-dimethyl-formamide at 60 - 65℃; for 0.5h; Solvent; Temperature; Reagent/catalyst; | 90.89% |
-
-
110-91-8
morpholine

-
-
881386-01-2
3,3-dichloro-1-(4-nitrophenyl)piperidine-2-one

-
-
503615-03-0
3-(morpholin-4-yl)-1-(4-nitrophenyl)-5,6-dihydropyridin-2(1H)-one

| Conditions | Yield |
|---|---|
| at 120℃; for 2h; Inert atmosphere; | 90.4% |
| at 130℃; for 1.5h; | 87.6% |
| at 130℃; for 5h; | 64.5% |
| at 130℃; for 1.5h; | |
| In N,N-dimethyl-formamide for 1h; Reflux; | 85.73 g |
-
-
881386-01-2
3,3-dichloro-1-(4-nitrophenyl)piperidine-2-one

| Conditions | Yield |
|---|---|
| With lithium carbonate; lithium chloride In N,N-dimethyl-formamide at 105℃; for 4h; Green chemistry; | 90% |
| With lithium carbonate; lithium chloride In N,N-dimethyl-formamide at 45 - 110℃; | |
| With lithium carbonate In N,N-dimethyl-formamide at 100 - 105℃; for 5h; Inert atmosphere; | 257.2 g |
-
-
881386-01-2
3,3-dichloro-1-(4-nitrophenyl)piperidine-2-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: lithium carbonate / N,N-dimethyl-formamide / 5 h / 100 - 105 °C / Inert atmosphere 2: triethylamine / tetrahydrofuran / 46 h / Inert atmosphere; Reflux 3: triethylamine / toluene / 5 h / 90 °C / Inert atmosphere 4: trichloroacetic acid / dichloromethane / 1.5 h / 20 °C View Scheme | |
| Multi-step reaction with 3 steps 1: 1.5 h / 130 °C 2: triethylamine / ethyl acetate / 6 h / 80 °C 3: trifluoroacetic acid / dichloromethane / 1 h / 20 °C View Scheme | |
| Multi-step reaction with 2 steps 1.1: 5 h / 130 °C 2.1: triethylamine; potassium iodide / ethyl acetate / 4.5 h / Reflux 2.2: 4 h / 5 - 20 °C View Scheme | |
| Multi-step reaction with 2 steps 1: lithium chloride; lithium carbonate / N,N-dimethyl-formamide / 4 h / 105 °C / Green chemistry 2: triethylamine / toluene / 2 h / 96 °C / Green chemistry View Scheme |
-
-
881386-01-2
3,3-dichloro-1-(4-nitrophenyl)piperidine-2-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: lithium carbonate / N,N-dimethyl-formamide / 5 h / 100 - 105 °C / Inert atmosphere 2: triethylamine / tetrahydrofuran / 46 h / Inert atmosphere; Reflux 3: triethylamine / toluene / 5 h / 90 °C / Inert atmosphere View Scheme |
-
-
881386-01-2
3,3-dichloro-1-(4-nitrophenyl)piperidine-2-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: lithium carbonate / N,N-dimethyl-formamide / 5 h / 100 - 105 °C / Inert atmosphere 2: triethylamine / tetrahydrofuran / 46 h / Inert atmosphere; Reflux 3: triethylamine / toluene / 5 h / 90 °C / Inert atmosphere 4: trichloroacetic acid / dichloromethane / 1.5 h / 20 °C 5: 5%-palladium/activated carbon; hydrogen / 1-methyl-pyrrolidin-2-one / 23 h / 20 °C / 4500.45 Torr / Autoclave; Inert atmosphere View Scheme | |
| Multi-step reaction with 4 steps 1: 1.5 h / 130 °C 2: triethylamine / ethyl acetate / 6 h / 80 °C 3: trifluoroacetic acid / dichloromethane / 1 h / 20 °C 4: hydrogenchloride; iron; ammonium chloride / water; ethanol / 2.5 h / 80 °C View Scheme | |
| Multi-step reaction with 3 steps 1.1: 5 h / 130 °C 2.1: triethylamine; potassium iodide / ethyl acetate / 4.5 h / Reflux 2.2: 4 h / 5 - 20 °C 3.1: acetic acid; zinc / ethanol; dichloromethane / 3 h / 20 °C / Cooling with ice View Scheme | |
| Multi-step reaction with 3 steps 1: lithium chloride; lithium carbonate / N,N-dimethyl-formamide / 4 h / 105 °C / Green chemistry 2: triethylamine / toluene / 2 h / 96 °C / Green chemistry 3: 5%-palladium/activated carbon; hydrogen / ethanol / 4 h / 50 - 55 °C / 2250.23 - 3000.3 Torr / Green chemistry View Scheme |
-
-
881386-01-2
3,3-dichloro-1-(4-nitrophenyl)piperidine-2-one

-
-
1423803-24-0
1-(4-methoxyphenyl)-7-oxo-6-(4-aminophenyl)-4,5,6,7-tetrahydro-1H-pyrazolo[3,4-c]pyridine-3-carboxamide

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1: lithium carbonate / N,N-dimethyl-formamide / 5 h / 100 - 105 °C / Inert atmosphere 2: triethylamine / tetrahydrofuran / 46 h / Inert atmosphere; Reflux 3: triethylamine / toluene / 5 h / 90 °C / Inert atmosphere 4: trichloroacetic acid / dichloromethane / 1.5 h / 20 °C 5: 5%-palladium/activated carbon; hydrogen / 1-methyl-pyrrolidin-2-one / 23 h / 20 °C / 4500.45 Torr / Autoclave; Inert atmosphere 6: ammonia / methanol / 18 h / 100 °C / Autoclave View Scheme |
-
-
881386-01-2
3,3-dichloro-1-(4-nitrophenyl)piperidine-2-one

-
-
1437577-94-0
6-(4-(5-bromopentanamido)phenyl)-1-(4-methoxyphenyl)-7-oxo-4,5,6,7-tetrahydro-1H-pyrazolo[3,4-c]pyridine-3-carboxamide

| Conditions | Yield |
|---|---|
| Multi-step reaction with 7 steps 1: lithium carbonate / N,N-dimethyl-formamide / 5 h / 100 - 105 °C / Inert atmosphere 2: triethylamine / tetrahydrofuran / 46 h / Inert atmosphere; Reflux 3: triethylamine / toluene / 5 h / 90 °C / Inert atmosphere 4: trichloroacetic acid / dichloromethane / 1.5 h / 20 °C 5: 5%-palladium/activated carbon; hydrogen / 1-methyl-pyrrolidin-2-one / 23 h / 20 °C / 4500.45 Torr / Autoclave; Inert atmosphere 6: ammonia / methanol / 18 h / 100 °C / Autoclave 7: potassium carbonate / tetrahydrofuran; water / 1.33 h / Cooling View Scheme |
-
-
881386-01-2
3,3-dichloro-1-(4-nitrophenyl)piperidine-2-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 8 steps 1: lithium carbonate / N,N-dimethyl-formamide / 5 h / 100 - 105 °C / Inert atmosphere 2: triethylamine / tetrahydrofuran / 46 h / Inert atmosphere; Reflux 3: triethylamine / toluene / 5 h / 90 °C / Inert atmosphere 4: trichloroacetic acid / dichloromethane / 1.5 h / 20 °C 5: 5%-palladium/activated carbon; hydrogen / 1-methyl-pyrrolidin-2-one / 23 h / 20 °C / 4500.45 Torr / Autoclave; Inert atmosphere 6: ammonia / methanol / 18 h / 100 °C / Autoclave 7: potassium carbonate / tetrahydrofuran; water / 1.33 h / Cooling 8: potassium ethoxide / tetrahydrofuran; ethanol / 0.5 h View Scheme | |
| Multi-step reaction with 6 steps 1: 120 - 130 °C 2: sodiumsulfide nonahydrate / water / 35 - 40 °C 3: triethylamine / dichloromethane / 25 - 35 °C 4: potassium iodide; triethylamine / ethyl acetate / Reflux 5: hydrogenchloride / 25 - 35 °C 6: ammonia / methanol / 45 °C / 6000.6 Torr / Autoclave View Scheme | |
| Multi-step reaction with 7 steps 1: 1.5 h / 130 °C 2: triethylamine / ethyl acetate / 6 h / 80 °C 3: trifluoroacetic acid / dichloromethane / 1 h / 20 °C 4: hydrogenchloride; iron; ammonium chloride / water; ethanol / 2.5 h / 80 °C 5: trimethylamine / tetrahydrofuran / 6.5 h / 0 - 25 °C 6: sodium hydride / N,N-dimethyl-formamide / 1 h / 0 °C 7: sodium methylate; formamide / N,N-dimethyl-formamide / 5 h / 50 °C View Scheme |
-
-
881386-01-2
3,3-dichloro-1-(4-nitrophenyl)piperidine-2-one

-
-
1449510-64-8
6-(4-(5-chloropentanamido)phenyl)-1-(4-methoxyphenyl)-7-oxo-4,5,6,7-tetrahydro-1H-pyrazolo[3,4-c]pyridine-3-carboxamide

| Conditions | Yield |
|---|---|
| Multi-step reaction with 7 steps 1: lithium carbonate / N,N-dimethyl-formamide / 5 h / 100 - 105 °C / Inert atmosphere 2: triethylamine / tetrahydrofuran / 46 h / Inert atmosphere; Reflux 3: triethylamine / toluene / 5 h / 90 °C / Inert atmosphere 4: trichloroacetic acid / dichloromethane / 1.5 h / 20 °C 5: 5%-palladium/activated carbon; hydrogen / 1-methyl-pyrrolidin-2-one / 23 h / 20 °C / 4500.45 Torr / Autoclave; Inert atmosphere 6: ammonia / methanol / 18 h / 100 °C / Autoclave 7: potassium carbonate / 1-methyl-pyrrolidin-2-one View Scheme |
-
-
881386-01-2
3,3-dichloro-1-(4-nitrophenyl)piperidine-2-one

-
-
503615-03-0
3-(morpholin-4-yl)-1-(4-nitrophenyl)-5,6-dihydropyridin-2(1H)-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: lithium carbonate / N,N-dimethyl-formamide / 5 h / 100 - 105 °C / Inert atmosphere 2: triethylamine / tetrahydrofuran / 46 h / Inert atmosphere; Reflux View Scheme |
-
-
881386-01-2
3,3-dichloro-1-(4-nitrophenyl)piperidine-2-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 120 - 130 °C 2: sodiumsulfide nonahydrate / water / 35 - 40 °C View Scheme |
-
-
881386-01-2
3,3-dichloro-1-(4-nitrophenyl)piperidine-2-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 120 - 130 °C 2: sodiumsulfide nonahydrate / water / 35 - 40 °C 3: triethylamine / dichloromethane / 25 - 35 °C View Scheme |
-
-
881386-01-2
3,3-dichloro-1-(4-nitrophenyl)piperidine-2-one

-
-
503614-91-3
1-(4-methoxy-phenyl)-7-oxo-6-[4-(2-oxo-piperidin-1-yl)-phenyl]-4,5,6,7-tetrahydro-1H-pyrazolo[3,4-c]pyridine-3-carboxylic acid ethyl ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: 120 - 130 °C 2: sodiumsulfide nonahydrate / water / 35 - 40 °C 3: triethylamine / dichloromethane / 25 - 35 °C 4: potassium iodide; triethylamine / ethyl acetate / Reflux 5: hydrogenchloride / 25 - 35 °C View Scheme | |
| Multi-step reaction with 6 steps 1: 1.5 h / 130 °C 2: triethylamine / ethyl acetate / 6 h / 80 °C 3: trifluoroacetic acid / dichloromethane / 1 h / 20 °C 4: hydrogenchloride; iron; ammonium chloride / water; ethanol / 2.5 h / 80 °C 5: trimethylamine / tetrahydrofuran / 6.5 h / 0 - 25 °C 6: sodium hydride / N,N-dimethyl-formamide / 1 h / 0 °C View Scheme | |
| Multi-step reaction with 5 steps 1: tin(II) chloride dihdyrate / N,N-dimethyl-formamide / 0.5 h / 60 - 65 °C 2: pyridine; dmap / dichloromethane / 0.5 h / 20 °C 3: lithium tert-butoxide / dichloromethane / 3 h / 20 °C 4: lithium carbonate / N,N-dimethyl-formamide / 3 h / 105 - 110 °C 5: triethylamine / toluene / 3.5 h / 95 - 100 °C View Scheme |
-
-
881386-01-2
3,3-dichloro-1-(4-nitrophenyl)piperidine-2-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: 120 - 130 °C 2: sodiumsulfide nonahydrate / water / 35 - 40 °C 3: triethylamine / dichloromethane / 25 - 35 °C 4: potassium iodide; triethylamine / ethyl acetate / Reflux View Scheme |
-
-
109-01-3
1-methyl-piperazine

-
-
881386-01-2
3,3-dichloro-1-(4-nitrophenyl)piperidine-2-one

| Conditions | Yield |
|---|---|
| at 120 - 130℃; |
-
-
881386-01-2
3,3-dichloro-1-(4-nitrophenyl)piperidine-2-one

-
-
1267610-26-3
1-(4-aminophenyl)-3-(morpholin-4-yl)-5,6-dihydropyridin-2(1H)-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 2 h / 120 °C / Inert atmosphere 2: water; sodium sulfide / ethanol / 50 °C / Inert atmosphere View Scheme | |
| Multi-step reaction with 2 steps 1: N,N-dimethyl-formamide / 1 h / Reflux 2: hydrazine hydrate / water; ethanol / 0.5 h / 60 - 65 °C View Scheme |
-
-
881386-01-2
3,3-dichloro-1-(4-nitrophenyl)piperidine-2-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 2 h / 120 °C / Inert atmosphere 2: water; sodium sulfide / ethanol / 50 °C / Inert atmosphere 3: N-ethyl-N,N-diisopropylamine / tetrahydrofuran / 10 - 25 °C / Inert atmosphere View Scheme | |
| Multi-step reaction with 3 steps 1: N,N-dimethyl-formamide / 1 h / Reflux 2: hydrazine hydrate / water; ethanol / 0.5 h / 60 - 65 °C 3: sodium hydroxide; tetrabutylammomium bromide / dichloromethane; water / 1 h / 0 - 5 °C View Scheme |
-
-
881386-01-2
3,3-dichloro-1-(4-nitrophenyl)piperidine-2-one

-
-
545445-44-1
5,6‑dihydro‑3‑(4‑morpholinyl)‑1‑[4‑(2‑oxo‑1‑piperidinyl)phenyl]‑2(1H)‑pyridone

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: 2 h / 120 °C / Inert atmosphere 2: water; sodium sulfide / ethanol / 50 °C / Inert atmosphere 3: N-ethyl-N,N-diisopropylamine / tetrahydrofuran / 10 - 25 °C / Inert atmosphere 4: sodium hydride / tetrahydrofuran / 10 - 25 °C / Inert atmosphere View Scheme | |
| Multi-step reaction with 3 steps 1.1: N,N-dimethyl-formamide / 1 h / Reflux 2.1: hydrazine hydrate / water; ethanol / 0.5 h / 60 - 65 °C 3.1: sodium hydroxide; tetrabutylammomium bromide / dichloromethane; water / 1 h / 0 - 5 °C 3.2: 20 °C View Scheme |
-
-
881386-01-2
3,3-dichloro-1-(4-nitrophenyl)piperidine-2-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: 2 h / 120 °C / Inert atmosphere 2: water; sodium sulfide / ethanol / 50 °C / Inert atmosphere 3: N-ethyl-N,N-diisopropylamine / tetrahydrofuran / 10 - 25 °C / Inert atmosphere 4: sodium hydride / tetrahydrofuran / 10 - 25 °C / Inert atmosphere 5: triethylamine / ethyl acetate / 80 °C / Inert atmosphere View Scheme |
-
-
881386-01-2
3,3-dichloro-1-(4-nitrophenyl)piperidine-2-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1: 2 h / 120 °C / Inert atmosphere 2: water; sodium sulfide / ethanol / 50 °C / Inert atmosphere 3: N-ethyl-N,N-diisopropylamine / tetrahydrofuran / 10 - 25 °C / Inert atmosphere 4: sodium hydride / tetrahydrofuran / 10 - 25 °C / Inert atmosphere 5: triethylamine / ethyl acetate / 80 °C / Inert atmosphere 6: ammonium hydroxide / methanol / 55 °C / Inert atmosphere View Scheme |
-
-
881386-01-2
3,3-dichloro-1-(4-nitrophenyl)piperidine-2-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: 2 h / 120 °C / Inert atmosphere 2: water; sodium sulfide / ethanol / 50 °C / Inert atmosphere 3: N-ethyl-N,N-diisopropylamine / tetrahydrofuran / 10 - 25 °C / Inert atmosphere 4: sodium hydride / tetrahydrofuran / 10 - 25 °C / Inert atmosphere 5: triethylamine / ethyl acetate / 80 °C / Inert atmosphere View Scheme |
-
-
881386-01-2
3,3-dichloro-1-(4-nitrophenyl)piperidine-2-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1: 2 h / 120 °C / Inert atmosphere 2: water; sodium sulfide / ethanol / 50 °C / Inert atmosphere 3: N-ethyl-N,N-diisopropylamine / tetrahydrofuran / 10 - 25 °C / Inert atmosphere 4: sodium hydride / tetrahydrofuran / 10 - 25 °C / Inert atmosphere 5: triethylamine / ethyl acetate / 80 °C / Inert atmosphere 6: ammonium hydroxide / methanol / 55 °C / Inert atmosphere View Scheme |
-
-
881386-01-2
3,3-dichloro-1-(4-nitrophenyl)piperidine-2-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: 2 h / 120 °C / Inert atmosphere 2: water; sodium sulfide / ethanol / 50 °C / Inert atmosphere 3: N-ethyl-N,N-diisopropylamine / tetrahydrofuran / 10 - 25 °C / Inert atmosphere 4: sodium hydride / tetrahydrofuran / 10 - 25 °C / Inert atmosphere 5: triethylamine / ethyl acetate / 80 °C / Inert atmosphere View Scheme |
-
-
881386-01-2
3,3-dichloro-1-(4-nitrophenyl)piperidine-2-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1: 2 h / 120 °C / Inert atmosphere 2: water; sodium sulfide / ethanol / 50 °C / Inert atmosphere 3: N-ethyl-N,N-diisopropylamine / tetrahydrofuran / 10 - 25 °C / Inert atmosphere 4: sodium hydride / tetrahydrofuran / 10 - 25 °C / Inert atmosphere 5: triethylamine / ethyl acetate / 80 °C / Inert atmosphere 6: ammonium hydroxide / methanol / 55 °C / Inert atmosphere View Scheme |
-
-
881386-01-2
3,3-dichloro-1-(4-nitrophenyl)piperidine-2-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: 2 h / 120 °C / Inert atmosphere 2: water; sodium sulfide / ethanol / 50 °C / Inert atmosphere 3: N-ethyl-N,N-diisopropylamine / tetrahydrofuran / 10 - 25 °C / Inert atmosphere 4: sodium hydride / tetrahydrofuran / 10 - 25 °C / Inert atmosphere 5: triethylamine / ethyl acetate / 80 °C / Inert atmosphere View Scheme |
Related products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View