Dayang Chem (Hangzhou) Co.,Ltd.
DayangChem exported this product to many countries and regions at best price. If you are looking for the material's manufacturer or supplier in China, DayangChem is your best choice. Pls contact with us freely for getting detailed product spe
Cas:88-18-6
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquirySimagchem Corporation
Welcome to Simagchem, your partner in China as a premier supply of bulk specialty chemicals for industry and life science. We introduce experienced quality product and exceptional JIT service with instant market intelligence in China to benefit our
Cas:88-18-6
Min.Order:0 Metric Ton
Negotiable
Type:Manufacturers
inquiryAlity Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Chemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Cas:88-18-6
Min.Order:5 Kiloliter
FOB Price: $1.2 / 5.0
Type:Manufacturers
inquiryHenan Tianfu Chemical Co., Ltd.
High Quality Best price Fast Delivery Good Service Welcome to Henan Tianfu Chemical Co., Ltd. website. Our company engages in Sodium Tripolyphosphate (STPP) and Sodium Hexametabphosphate (SHMP) production; developm
Cas:88-18-6
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHenan Wentao Chemical Product Co., Ltd.
Henan Wentao Chemical Product Co.,Ltd is Located in Zhengzhou High-tech Development Zone with import and export license, We passed ISO 9001:2008 as well, Henan Wentao has developed more than 1000 compounds, which are widely used in the fields of prod
Hangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Zibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:88-18-6
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquiryHANGZHOU YUNUO CHEMICAL CO.,LTD
superior quality Appearance: CLEAR LIQUID Storage:Stored in cool, dry and ventilation place; Away from fire and heat Package:1kg/bag, 1kg/drum or 25kg/drum or as per your request. Application:Used as Pharmaceutical Intermediates Transportation:as p
Zibo Dorne chemical technology co. LTD
Product Details Grade: pharmaceutical grade Purity:99%+ ProductionCapacity: 1000 Kilogram/Month Scope of use: For scientific research only(The product must be used legally) Our Advantage 1. Best quality with competitive price. 2. Quick shipping,
Cas:88-18-6
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHunan chemfish Pharmaceutical co.,Ltd
Appearance:95%+ Package:R&D,Pilot run Transportation:per client require Port:Express ,Air, Sea
Kono Chem Co.,Ltd
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:drum and bag Application:for pharma use Transportation:by sea or air Port:Beijing or Guangzhou
Hangzhou Dingyan Chem Co., Ltd
R & D enterprises have their own stock in stockAppearance:To be subject to the object Package:Customized Application:pharmaceutical intermediates Transportation:Air Port:Shanghai;Guangzhou
GIHI CHEMICALS CO.,LIMITED
high purity,in stock Package:25kg/drum,or as per customers'demand Application:API,Pharmaceutical intermediates Transportation:air,sea,courier
Henan Allgreen Chemical Co.,Ltd
high quality Storage:Sealed, dry, microtherm , avoid light and smell. Package:According to the demand of customer Application:Organic synthesis Transportation:by air or by sea
Enke Pharma-tech Co.,Ltd. (Cangzhou, China )
high purity Storage:normal temperature Package:DRUM Application:mainly for medical use for R&Dpurpose use only Transportation:AIR,SEA Port:BEIJING,SHANGHAI,TIANJIN,SHENZHEN
Aecochem Corp.
Our clients, like BASF,CHEMO,Brenntag,ASR,Evonik,Merck and etc.Appearance:COA Storage:in stock Application:MSDS/TDS
Zhuozhou Wenxi import and Export Co., Ltd
Product Description Description & Specification Category Pharmaceutical Raw Materials, Fine Chemicals, Bulk drug Standard Medical standard
Cas:88-18-6
Min.Order:1 Kilogram
FOB Price: $112.0
Type:Trading Company
inquiryAntimex Chemical Limied
Ansciep Chemical is a professional enterprise manufacturing and distributing fine chemicals and speciality chemicals. We have been dedicated to heterocycle compounds and phenyl rings for tens of years. This is our mature product for export. Our quali
Cas:88-18-6
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryXiamen AmoyChem Co.,Ltd
Amoychem is committed to providing the top-quality chemical products and services Internationally. We offer our customers with friendly, professional service and reliable, high performance products that have been manufactured according to the accredi
Hunan Russell Chemicals Technology Co.,Ltd
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:Foil bag; Drum; Plastic bottle Application:Pharma;Industry;Agricultural Transportation:by sea or air Port:any port in China
Bide Pharmatech Ltd
Our mission is to provide high-quality and innovative products to our customers. By offering a broad range of products, custom synthesis and personalized services, Bide can help scientists speeding up their research in the chemical and pharmaceutical
Amadis Chemical Co., Ltd.
1.Professional synthesis laboratory and production base. 2.Strong synthesis team and service team. 3.Professional data management system. 4.We provide the professional test date and product information ,ex. HNMR ,CNMR,FNMR, HPLC/G
Cas:88-18-6
Min.Order:10 Milligram
Negotiable
Type:Lab/Research institutions
inquiryHENAN SUNLAKE ENTERPRISE CORPORATION
HENAN SUNLAKE ENTERPRISE CORPORATION Our company advantages: 1、The highest quality with the competitive price. 2、Professional human services. 3、The fastest and safest delivery service. 4、The faster and safest delivery service. 5、The hig
Zhengzhou Kingorgchem Chemical Technology Co., Ltd.
1. Subsidiary of Institute of Chemistry, Henan Academy of Sciences, national research platform;2. About 30 years’ experience in this field since 1983;3. An experienced R&D team consisting of Doctors and Masters;4. Various types of analytical instrume
Cas:88-18-6
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryXiamen BaiFuchem Co.,Ltd
BaiFuChem is a Professional chemical raw material supplier in China, our main products include Biochemical , Pharma Intermediate and Organic chemical etc. BaiFuChem have wealth of products,experience , expertise and state-of-the-art
JiYi Chemical (Beijing) Co., Ltd.
O-TERT–BUTYLPHENOLChemicalname:O-TERT–BUTYLPHENOLDomesticcommercialname:O-BUTYLPHENOLInternationalcommercialname:OTBPCASNO.:88-18-6Mol… Appearance:light yellow or orange oily liquid Storage:Protect against fire and water and avoi
HuBei Ipure Biotech CO.,ltd
HuBei ipure is a diversified product production and operation enterprise, with API, pharma intermediates, and other fine chemicals as well as R&D and pigments development and sales as one of the large enterprises, with more than 130 acres of pl
Cas:88-18-6
Min.Order:1 Gram
FOB Price: $1.0
Type:Trading Company
inquiryHangzhou KieRay Chem Co.,Ltd.
Hangzhou KieRaychem Co.,Ltd.is located in Yuhang District of Hangzhou City and specialized in the chemical product customization, development, sales, import and export. Current business is focused on fine chemicals, pharmaceutical materials and int
Synthetic route
-
-
373354-37-1
3-tert-butyl-bicyclo[3.1.0]hex-3-en-2-one

-
-
88-18-6
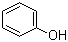
2-tert-Butylphenol

| Conditions | Yield |
|---|---|
| In chloroform Irradiation; | 98% |
-
-
75-65-0
tert-butyl alcohol

-
-
108-95-2
phenol

-
A
-
98-54-4
para-tert-butylphenol

-
B
-
88-18-6
2-tert-Butylphenol

| Conditions | Yield |
|---|---|
| With zeolite Beta at 40℃; for 6h; Reagent/catalyst; Temperature; Time; | A 91.74% B 5.08% |
| 30% TPA/ZrO2 at 80℃; for 1 - 10h; Product distribution / selectivity; | |
| 20% TSA/ZrO2 at 80℃; for 6h; Product distribution / selectivity; |

-
-
85686-09-5
2-t-butyl-1-isopropoxybenzene

-
-
88-18-6
2-tert-Butylphenol

| Conditions | Yield |
|---|---|
| With n-butyllithium; carbon dioxide | 87% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water at 25℃; | A 87% B n/a |
-
-
676-58-4
methylmagnesium chloride

-
-
346433-44-1
2-(tert-butoxycarbonyloxy)acetophenone

-
-
88-18-6
2-tert-Butylphenol

| Conditions | Yield |
|---|---|
| In diethyl ether at 0℃; Grignard reaction; | 75% |

| Conditions | Yield |
|---|---|
| In diethyl ether at 0℃; Grignard reaction; | 75% |
-
-
1571889-86-5
C19H34N3OPd(1+)*ClO4(1-)

-
-
88-18-6
2-tert-Butylphenol

| Conditions | Yield |
|---|---|
| In dimethylsulfoxide-d6 at 110℃; Concentration; Solvent; Inert atmosphere; Schlenk technique; Glovebox; Sealed tube; | 74.1% |
-
-
75-65-0
tert-butyl alcohol

-
-
108-95-2
phenol

-
A
-
98-54-4
para-tert-butylphenol

-
B
-
128-39-2
2,6-di-tert-butylphenol

-
C
-
88-18-6
2-tert-Butylphenol

| Conditions | Yield |
|---|---|
| With aluminium(III) phenoxide In various solvent(s) at 130℃; for 2h; | A 9.4% B 1.8% C 70.4% |


| Conditions | Yield |
|---|---|
| at 300℃; for 24h; | 69% |

| Conditions | Yield |
|---|---|
| at 300℃; for 24h; Product distribution; other temperatures; | A 69% B 6 % Chromat. |
-
-
115-11-7
isobutene

-
-
108-95-2
phenol

-
A
-
98-54-4
para-tert-butylphenol

-
B
-
128-39-2
2,6-di-tert-butylphenol

-
C
-
6669-13-2
t-butyl phenyl ether

-
D
-
88-18-6
2-tert-Butylphenol

-
E
-
96-76-4
2,4-di-tert-Butylphenol

| Conditions | Yield |
|---|---|
| 2-phenoxy-1,2-thialuminolane at 110℃; for 3h; Product distribution; Mechanism; other catalysts, other time, other temperature; | A 1.23% B 4.97% C 0.8% D 66.67% E 2.37% |

-
-
115-11-7
isobutene

-
-
108-95-2
phenol

-
A
-
128-39-2
2,6-di-tert-butylphenol

-
B
-
6669-13-2
t-butyl phenyl ether

-
C
-
88-18-6
2-tert-Butylphenol

-
D
-
96-76-4
2,4-di-tert-Butylphenol

| Conditions | Yield |
|---|---|
| 2-phenooxy-1,2-thialuminolane at 110℃; for 3h; Further byproducts given; | A 4.97% B 0.8% C 66.67% D 2.37% |

-
-
75-65-0
tert-butyl alcohol

-
-
108-95-2
phenol

-
A
-
98-54-4
para-tert-butylphenol

-
B
-
88-18-6
2-tert-Butylphenol

-
C
-
96-76-4
2,4-di-tert-Butylphenol

| Conditions | Yield |
|---|---|
| With carbon tetrabromide at 175℃; for 6h; Sealed tube; | A 65% B 6% C 29% |
| With 30 wtpercent pyrophosphoric acid onto mesoporous molecular sieve Al-MCM-41(70) at 189.84℃; for 8h; Inert atmosphere; | A 62.17% B 5.7 %Chromat. C 12.5 %Chromat. |
| With granular zeolite Y in the H-form at 175℃; for 6h; Sealed tube; | A 17% B 20% C 17% |


| Conditions | Yield |
|---|---|
| With aluminium(III) ion In chlorobenzene at 100℃; for 1h; | A 55% B 4% |
-
-
75-65-0
tert-butyl alcohol

-
-
108-95-2
phenol

-
A
-
98-54-4
para-tert-butylphenol

-
B
-
88-18-6
2-tert-Butylphenol

-
C
-
96-76-4
2,4-di-tert-Butylphenol

-
D
-
732-26-3
2,4,6-tri-tert-butylphenoxol

| Conditions | Yield |
|---|---|
| With carbon tetrabromide at 175℃; for 6h; Sealed tube; | A 39% B 2% C 55% D 5% |
| With carbon tetrabromide at 175℃; for 6h; Reagent/catalyst; Temperature; Sealed tube; | A 39% B 4% C 52% D 5% |
| With carbon tetrabromide at 175℃; for 6h; Concentration; Time; Sealed tube; | A 41% B 8% C 44% D 2% |

-
-
513-35-9
2-methyl-but-2-ene

-
-
115-11-7
isobutene

-
-
108-95-2
phenol

-
A
-
98-54-4
para-tert-butylphenol

-
B
-
88-18-6
2-tert-Butylphenol

-
C
-
80-46-6
4-t-amylphenol

-
D
-
96-76-4
2,4-di-tert-Butylphenol

-
E
-
732-26-3
2,4,6-tri-tert-butylphenoxol

-
F
-
120-95-6
2,4-di-tert-amylphenol

-
G
-
122269-03-8
2-t-butyl-4-(1,1-dimethylpropyl)-phenol

-
H
-
122269-05-0
4-tert-butyl-2-(1,1-dimethyl-propyl)-phenol

| Conditions | Yield |
|---|---|
| Stage #1: isobutene; phenol; Fulcat 22B catalyst at 130 - 140℃; for 1.5h; Inert atmosphere; Stage #2: 2-methyl-but-2-ene at 130℃; for 3.25h; | A 50.8% B 1.4% C 15.3% D 17.6% E 0.3% F 1.3% G 10.7% H 10.7% |

-
-
75-65-0
tert-butyl alcohol

-
-
108-95-2
phenol

-
A
-
98-54-4
para-tert-butylphenol

-
B
-
128-39-2
2,6-di-tert-butylphenol

-
C
-
88-18-6
2-tert-Butylphenol

-
D
-
96-76-4
2,4-di-tert-Butylphenol

| Conditions | Yield |
|---|---|
| With carbon tetrabromide at 175℃; for 6h; Sealed tube; | A 33% B 3% C 9% D 49% |
| With 3-methyl-1-(4-sulfobutyl)imidazol-3-ium methanesulfonate at 70℃; for 2h; Temperature; Sealed tube; | |
| With 1-(4-sulfonic acid)butylpyridinium methane sulfonate at 80℃; for 2h; Reagent/catalyst; Sealed tube; | |
| With N-(4-sulfonic acid)butyl triethylammonium methane sulfonate at 80℃; for 2h; Catalytic behavior; Kinetics; Temperature; Reagent/catalyst; Sealed tube; |


| Conditions | Yield |
|---|---|
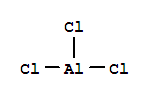
| With aluminium trichloride; potassium phenolate In chloroform at 25℃; for 10h; | 40% |
| at 330℃; under 83847.9 Torr; | |
| at 260 - 330℃; |

-
A
-
88-18-6
2-tert-Butylphenol

-
B
-
56846-74-3
1,1-dimethyl-1,2-dihydrocyclobutabenzene

| Conditions | Yield |
|---|---|
| With oxygen In [D3]acetonitrile; water-d2 at -5℃; for 0.0833333h; Inert atmosphere; Schlenk technique; | A 5% B 40% |
-
-
75-65-0
tert-butyl alcohol

-
-
108-95-2
phenol

-
A
-
98-54-4
para-tert-butylphenol

-
B
-
128-39-2
2,6-di-tert-butylphenol

-
C
-
88-18-6
2-tert-Butylphenol

-
D
-
96-76-4
2,4-di-tert-Butylphenol

-
E
-
732-26-3
2,4,6-tri-tert-butylphenoxol

| Conditions | Yield |
|---|---|
| With carbon tetrabromide at 150℃; for 6h; Sealed tube; | A 9% B 10% C 20% D 39% E 14% |
| With carbon tetrabromide at 175℃; for 6h; Sealed tube; | A 6% B 13% C 16% D 34% E 25% |
| With tetrachloromethane at 175℃; for 6h; Sealed tube; | A 17% B 4% C 21% D 23% E 7% |
| With Bromoform at 175℃; for 6h; Sealed tube; | A 21% B 1% C 21% D 22% E 1% |

-
-
128-37-0
2,6-di-tert-butyl-4-methyl-phenol

-
-
108-95-2
phenol

-
A
-
106-44-5
p-cresol

-
B
-
98-54-4
para-tert-butylphenol

-
C
-
2409-55-4
2-tert-Butyl-4-methylphenol

-
D
-
128-39-2
2,6-di-tert-butylphenol

-
E
-
88-18-6
2-tert-Butylphenol

-
F
-
96-76-4
2,4-di-tert-Butylphenol

| Conditions | Yield |
|---|---|
| C24H20ClNbO4 at 140℃; for 3h; Product distribution; Rate constant; Mechanism; also with 2-methylphenol, other catalysts, other products; | A 14% B 8% C 30.5% D 2% E 20% F 15% |

-
-
75-65-0
tert-butyl alcohol

-
-
108-95-2
phenol

-
A
-
98-54-4
para-tert-butylphenol

-
B
-
6669-13-2
t-butyl phenyl ether

-
C
-
88-18-6
2-tert-Butylphenol

-
D
-
96-76-4
2,4-di-tert-Butylphenol

| Conditions | Yield |
|---|---|
| With 5 wtpercent pyrophosphoric acid onto mesoporous molecular sieve Al-MCM-41(70) at 109.84℃; for 8h; Inert atmosphere; | A 22.7% B 30.1% C 25.5% D 21.2% |
| H7P2Mo17VO62 at 80℃; for 4h; Product distribution; Further Variations:; Catalysts; | |
| With Sulfanilic acid immobilized onto rice husk ash via 3-(chloropropyl)triethoxy-silane at 120℃; for 9h; Mechanism; Concentration; Temperature; Time; chemoselective reaction; |


| Conditions | Yield |
|---|---|
| With lithium diisopropyl amide In tetrahydrofuran; hexane 1.) -78 deg C, 2 h, 2.) r.t., 10 h; Yields of byproduct given; | A 20% B n/a |
| With lithium diisopropyl amide In tetrahydrofuran; hexane 1.) -78 deg C, 2 h, 2.) r.t., 10 h; Yield given; | A 20% B n/a |


| Conditions | Yield |
|---|---|
| Ku-2-8chS at 80℃; for 16h; Equilibrium constant; Product distribution; other catalysis, other temperature, other time; | |
| In neat (no solvent) at 79.9 - 209.9℃; Equilibrium constant; effect of temperature; | |
| With aluminum oxide; silica gel at 190 - 230℃; |
-
-
108-90-7
chlorobenzene

-
-
513-36-0
isobutyryl chloride

-
A
-
585-34-2
3-tert-butylphenyol

-
B
-
88-18-6
2-tert-Butylphenol

| Conditions | Yield |
|---|---|
| With aluminium trichloride Erhitzen des Reaktionsprodukts mit wss. NaOH und Cu2O auf 250grad; |


| Conditions | Yield |
|---|---|
| With aluminum oxide; silica gel at 150 - 200℃; under 66195.7 Torr; |


| Conditions | Yield |
|---|---|
| KU-23 sulfonated cation-exchanger at 59.9℃; Product distribution; relative reactivity, other olefin, mixture of butylenes, other temperature, other concentrations; | |
| With aluminium trichloride; potassium phenolate In chloroform at 25℃; for 10h; Product distribution; different amounts of PhOK; | |
| With sulfuric acid at 100 - 110℃; Erhitzen des Reaktionsprodukts mit geringen Mengen konz. Schwefelsaeure unter 25-30 Torr; | |
| With boric acid; oxalic acid at 45℃; |

-
-
507-20-0
tertiary butyl chloride

-
-
108-95-2
phenol

-
A
-
98-54-4
para-tert-butylphenol

-
B
-
88-18-6
2-tert-Butylphenol

| Conditions | Yield |
|---|---|
| at 125 - 170℃; | |
| With silica chloride at 70℃; for 3h; | |
| With o-tetrachloroquinone; C16H21IMoO3 In 1,2-dichloro-ethane at 80℃; for 12h; Friedel-Crafts Alkylation; Inert atmosphere; Schlenk technique; Green chemistry; Overall yield = 90.3 %; regioselective reaction; | |
| at 125 - 170℃; |


| Conditions | Yield |
|---|---|
| With oil at 60 - 120℃; | |
| at 125℃; |

| Conditions | Yield |
|---|---|
| With μ-Oxo-I,I'-bis(trifluoroacetato-O)-I,I'-diphenyldiiodine(III) In water; acetonitrile at 0℃; for 2h; | 100% |
| With titanium superoxide; dihydrogen peroxide; acetic acid In water at 50℃; for 2h; | 98% |
| With oxygen In water; acetonitrile at 40℃; under 15001.5 Torr; for 1h; Green chemistry; | 91% |
-
-
88-18-6
2-tert-Butylphenol

-
-
10323-39-4
4-bromo-2-tert-butylphenol

| Conditions | Yield |
|---|---|
| With tetra-N-butylammonium tribromide In chloroform at 20℃; for 3h; | 100% |
| With ammonium metavanadate; aluminum tri-bromide; oxygen In diethyl ether at 20℃; for 8h; | 99% |
| With tetra-N-butylammonium tribromide In dichloromethane at 23℃; for 2h; | 98% |

| Conditions | Yield |
|---|---|
| With tris(triphenylphosphine)rhodium(I) chloride; caesium carbonate; diisopropyl(2-tert-butyl)phenoxyphosphine In toluene for 18h; Heating; | 100% |
| With caesium carbonate; diisopropyl(2-tert-butyl)phenoxyphosphine; RhCl(PPh3)3 In toluene for 18h; Heating; | 100% |
| With caesium carbonate; chloro(1,5-cyclooctadiene)rhodium(I) dimer In toluene for 18h; Heating; | 94 % Spectr. |

| Conditions | Yield |
|---|---|
| With caesium carbonate; diisopropyl(2-tert-butyl)phenoxyphosphine; RhCl(PPh3)3 In toluene for 18h; Heating; | 100% |

| Conditions | Yield |
|---|---|
| With caesium carbonate; diisopropyl(2-tert-butyl)phenoxyphosphine; RhCl(PPh3)3 In toluene for 18h; Heating; | 100% |
-
-
1498-42-6
phosphorodichloridous acid ethyl ester

-
-
88-18-6
2-tert-Butylphenol

-
-
851348-76-0
di-(2-tert-butylphenyl) ethyl phosphite

| Conditions | Yield |
|---|---|
| With triethylamine In toluene at 0 - 20℃; for 3.5h; | 100% |
| With triethylamine In diethyl ether; toluene at 0 - 20℃; for 3.5h; | 94% |
-
-
88-18-6
2-tert-Butylphenol

-
-
1795-48-8
Isopropyl isocyanate

-
-
899427-15-7
2-tert-butylphenyl isopropylcarbamate

| Conditions | Yield |
|---|---|
| 100% |
-
-
88-18-6
2-tert-Butylphenol

-
-
106-96-7
propargyl bromide

-
-
55619-88-0
1-(tert-butyl)-2-(prop-2-yn-1-yloxy)benzene

| Conditions | Yield |
|---|---|
| With caesium carbonate In acetonitrile | 100% |
| With potassium carbonate In acetonitrile at 20℃; for 48h; | 90% |
| With potassium carbonate In acetonitrile at 20℃; for 48h; | 90% |
-
-
7647-01-0
hydrogenchloride

-
-
88-18-6
2-tert-Butylphenol

-
-
20942-68-1
2-bromo-5-(1,1-dimethylethyl)phenol

| Conditions | Yield |
|---|---|
| With tetra-n-butylammonium tribromide In methanol; dichloromethane; water | 100% |
-
-
88-18-6
2-tert-Butylphenol

-
-
13491-79-7
2-tert-butylcyclohexan-1-ol

| Conditions | Yield |
|---|---|
| With hydrogen In water at 40℃; under 7500.75 Torr; for 6h; Autoclave; | 99.2% |
| With nickel(II) oxide; hydrogen; palladium In hexane at 100℃; under 22502.3 Torr; for 8h; | 92% |
| With 5%-palladium/activated carbon; hydrogen at 90 - 100℃; under 15001.5 Torr; |
-
-
3888-00-4
nitropentafluoroacetone

-
-
88-18-6
2-tert-Butylphenol

-
-
123557-88-0
2-(2-hydroxy-1-nitropentafluoroisopropyl)-6-tert-butylphenol

| Conditions | Yield |
|---|---|
| In tetrachloromethane at 25℃; for 48h; various phenols and phenolates, other polyfluorophenyl compounds; | 99.1% |
| In tetrachloromethane at 25℃; for 48h; | 99.1% |

| Conditions | Yield |
|---|---|
| Stage #1: 2-tert-Butylphenol With potassium hydroxide In tetrahydrofuran at 10 - 20℃; Inert atmosphere; Stage #2: methyl iodide In tetrahydrofuran at 0 - 20℃; Inert atmosphere; | 99% |
| Stage #1: 2-tert-Butylphenol With sodium hydroxide In N,N-dimethyl-formamide; mineral oil for 1h; Stage #2: methyl iodide In N,N-dimethyl-formamide; mineral oil for 2h; | 80% |
| With potassium carbonate In N,N-dimethyl-formamide at 80℃; for 24h; | 33% |
-
-
88-18-6
2-tert-Butylphenol

-
-
23159-87-7
2-bromo-6-(tert-butyl)phenol

| Conditions | Yield |
|---|---|
| With N-Bromosuccinimide; diisopropylamine In dichloromethane at 20℃; for 1.5h; | 99% |
| With N-Bromosuccinimide; diisopropylamine In dichloromethane for 16h; Inert atmosphere; Reflux; | 96% |
| With N-Bromosuccinimide; diisopropylamine In dichloromethane for 16h; Reflux; Inert atmosphere; | 95% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 100℃; for 4h; | 99% |
| With potassium carbonate In N,N-dimethyl-formamide at 100℃; for 4h; | 99% |

| Conditions | Yield |
|---|---|
| With triethylamine In acetonitrile at 80℃; for 20h; | 99% |

| Conditions | Yield |
|---|---|
| With triethylamine In toluene for 18h; Heating; | 98.5% |
-
-
88-18-6
2-tert-Butylphenol

-
-
13395-86-3
2-(tert-butyl)-4,6-dichlorophenol

| Conditions | Yield |
|---|---|
| With N-chloro-succinimide; triphenylphosphine sulfide In dichloromethane at 20℃; for 12h; | 98% |
| With pyridine; sulfuryl dichloride | |
| With hydrogenchloride; manganese(II) sulfate; dihydrogen peroxide In water at 80℃; | 81 %Chromat. |
-
-
88-18-6
2-tert-Butylphenol

-
-
24424-99-5
di-tert-butyl dicarbonate

-
-
95932-30-2
Carbonic acid tert-butyl ester 2-tert-butyl-phenyl ester

| Conditions | Yield |
|---|---|
| With potassium carbonate; 18-crown-6 ether In tetrahydrofuran at 0℃; for 20h; | 98% |
| With 6,7-dimethoxyisoquinoline In dichloromethane at 20℃; for 10h; Inert atmosphere; chemoselective reaction; | 93% |
-
-
88-18-6
2-tert-Butylphenol

-
-
31502-36-0
tri(o-t-butylphenyl)phosphite

| Conditions | Yield |
|---|---|
| With triethylamine; phosphorus trichloride In tetrahydrofuran at 0 - 20℃; for 24h; Inert atmosphere; | 98% |
| With 2,3-Dimethylaniline; phosphorus trichloride In xylene 1.) room temperature, 2.) reflux, 2 h; | 90% |
| With phosphorus trichloride at 160℃; | 70% |

| Conditions | Yield |
|---|---|
| With potassium fluoride on basic alumina; 18-crown-6 ether In acetonitrile for 84h; Heating; | 98% |
| With cesium fluoride/clinoptilolite In dimethyl sulfoxide at 110℃; for 0.75h; Ullmann Condensation; | 98% |
| With potassium fluoride supported on Clinoptilolite In dimethyl sulfoxide at 110 - 115℃; for 2h; | 87% |
-
-
88-18-6
2-tert-Butylphenol

-
-
107-14-2
chloroacetonitrile

-
-
202821-16-7
(2-tert-butylphenoxy)acetonitrile

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone Heating; | 98% |
-
-
88-18-6
2-tert-Butylphenol

-
-
1493-27-2
ortho-nitrofluorobenzene

-
-
212691-73-1
1-(tert-butyl)-2-(2-nitrophenoxy)benzene

| Conditions | Yield |
|---|---|
| With potassium fluoride on basic alumina; 18-crown-6 ether In acetonitrile for 68h; Heating; | 97% |
-
-
88-18-6
2-tert-Butylphenol

-
-
60803-26-1
2-(tert-butyl)-4,6-diiodophenol

| Conditions | Yield |
|---|---|
| Stage #1: 2-tert-Butylphenol With sodium hydroxide; sodium hypochlorite; sodium iodide In methanol at -2 - 2℃; Stage #2: With hydrogenchloride; water In methanol pH=3; Product distribution / selectivity; | 97% |
| Stage #1: 2-tert-Butylphenol With sodium hydroxide; sodium hypochlorite; sodium iodide In methanol; water at -2 - 1.3℃; Stage #2: With hydrogenchloride; water In methanol pH=2; Product distribution / selectivity; | 93% |
| Stage #1: 2-tert-Butylphenol With sodium hydroxide In methanol Stage #2: With sodium hypochlorite; sodium iodide In methanol; water at -2 - 1.3℃; Stage #3: With hydrogenchloride In methanol; water at 1.2 - 4.1℃; for 1.61667h; pH=3; Product distribution / selectivity; | 93% |
-
-
88-18-6
2-tert-Butylphenol

-
A
-
3602-55-9
2-tert-butyl-1,4-benzoquinone

-
B
-
1948-33-0
tert-butylhydroquinone

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide; Ti-superoxide In water; acetic acid at 50 - 60℃; for 1.25h; Product distribution / selectivity; | A 97% B 0.7% |

| Conditions | Yield |
|---|---|
| In [D3]acetonitrile at 20℃; Inert atmosphere; | 97% |

| Conditions | Yield |
|---|---|
| With copper(l) iodide; caesium carbonate In 1-methyl-pyrrolidin-2-one at 160℃; for 24h; Inert atmosphere; Schlenk technique; | 97% |
-
-
88-18-6
2-tert-Butylphenol

-
-
728-87-0
4,4'-Dimethoxybenzhydrol

-
-
77464-14-3
4-[Bis-(4-methoxy-phenyl)-methyl]-2-tert-butyl-phenol

| Conditions | Yield |
|---|---|
| aluminium trichloride In diethyl ether at 20℃; for 72h; | 96% |

| Conditions | Yield |
|---|---|
| With tris(triphenylphosphine)rhodium(I) chloride; P(iPr)2(O-C6H4-2-tBu); caesium carbonate In toluene for 18h; Heating; | 96% |
| With caesium carbonate; diisopropyl(2-tert-butyl)phenoxyphosphine; RhCl(PPh3)3 In toluene for 18h; Heating; | 96% |
-
-
88-18-6
2-tert-Butylphenol

-
-
40244-90-4
Chlorodiisopropylphosphane

-
-
521273-02-9
diisopropyl(2-tert-butyl)phenoxyphosphine

| Conditions | Yield |
|---|---|
| With triethylamine In toluene for 18h; Heating; | 96% |
Related products
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View













 T,
T, N
N