Dayang Chem (Hangzhou) Co.,Ltd.
As a leading manufacturer and supplier of chemicals in China, DayangChem not only supply popular chemicals, but also DayangChem's R&D center offer custom synthesis services. DayangChem can provide different quantities of custom synthesis ch
Cas:576-26-1
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquiryHangzhou Dingyan Chem Co., Ltd
Items Standard Result Assay 98%min ----------------------------------------------------------------------------------------------
Cas:576-26-1
Min.Order:1 Gram
FOB Price: $100.0 / 500.0
Type:Manufacturers
inquiryHebei yanxi chemical co.,LTD.
Hebei Yanxi Chemical Co., Ltd. is a professional research, development and production of lead acetate benzene acetamide enterprise backbone members by local well-known entrepreneurs and professional senior engineers in the party's "low car
Cas:576-26-1
Min.Order:1 Metric Ton
FOB Price: $1.0 / 2.0
Type:Manufacturers
inquiryAlity Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Chemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Cas:576-26-1
Min.Order:5 Kiloliter
FOB Price: $1.2 / 5.0
Type:Manufacturers
inquiryHenan Tianfu Chemical Co., Ltd.
Our company was built in 2009 with an ISO certificate.In the past 5 years, we have grown up as a famous fine chemicals supplier in China and we had established stable business relationships with Samsung,LG,Merck,Thermo Fisher Scientific and so on.O
Cas:576-26-1
Min.Order:1 Metric Ton
Negotiable
Type:Lab/Research institutions
inquiryTriumph International Development Limilted
Appearance:off-white to white solid Storage:Store in a cool,dry place and keep away from direct strong light Package:As customer request Application:Used for Synthesis API and Research Transportation:As customer request Port:Qingda
Cas:576-26-1
Min.Order:1 Kilogram
FOB Price: $18.0 / 20.0
Type:Lab/Research institutions
inquiryHangzhou Keyingchem Co.,Ltd
Hangzhou KeyingChem Co., Ltd. exported this product to many countries and regions at best price. If you are looking for the material’s manufacturer or supplier in China, KeyingChem is your best choice. Pls contact with us freely for getting det
Cas:576-26-1
Min.Order:0 Metric Ton
Negotiable
Type:Lab/Research institutions
inquiryHangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
SHANGHAI T&W PHARMACEUTICAL CO., LTD.
A substitute for perfluorooctanoic acid, mainly used as a surfactant, dispersant, additive, etc Appearance:White solid or Colorless liquid Purity:99.3 % We will ship the goods in a timely manner as required We can provide relevant documents acc
Cas:576-26-1
Min.Order:4 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryShanghai Massive Chemical Technology Co., Ltd.
Massive Chemical is certified with ISO9001 and ISO14001 manufacturer for this product. We will offer all documents as requirement for the materials which includes, Certificate of Analysis, Material Safety Data Sheet, and Method of Analysis and
Cas:576-26-1
Min.Order:1 Gram
FOB Price: $1.0
Type:Lab/Research institutions
inquiryZibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:576-26-1
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquiryHANGZHOU YUNUO CHEMICAL CO.,LTD
moderate price & quick delivery. Appearance:colourless crystals or white powder Storage:Stored in cool, dry and ventilation place; Away from fire and heat Package:25kg/drum;200kg/drum as per your request Application:Similar to other xylenol com
Zibo Dorne chemical technology co. LTD
Product Details Grade: pharmaceutical grade Purity:99%+ ProductionCapacity: 1000 Kilogram/Month Scope of use: For scientific research only(The product must be used legally) Our Advantage 1. Best quality with competitive price. 2. Quick shipping,
Cas:576-26-1
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHenan Wentao Chemical Product Co., Ltd.
We are leading fine chemicals supplier in China with ISO certificate, Our main business covers the fields below: 1.Noble Metal Catalysts (Pt.Pd...) 2.Organic Phosphine Ligands (Tert-butyl-phosphine.Cyclohexyl-phosphine...) 3.OLED
Cas:576-26-1
Min.Order:1 Gram
FOB Price: $2.0
Type:Lab/Research institutions
inquiryHangzhou Zhongqi chem Co.,Ltd.
Located in Hangzhou National Hi-Tech Industrial Development Zone, zhongqichem is a technical company mainly focus on the Custom synthesis, manufacturing, sales of chemicals to various industries. Benefiting from the outstanding customer service an
Hunan chemfish Pharmaceutical co.,Ltd
Appearance:95%+ Package:R&D,Pilot run Transportation:per client require Port:Express ,Air, Sea
SAGECHEM LIMITED
SAGECHEM is a chemical R&D, manufacturing and distribution company in China since 2009, including pharmaceutical intermediates, agrochemical, dyestuff intermediates, organosilicone, API and etc. We also offer a full range of services in custom synthe
Cas:576-26-1
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryBeijing Greenchem Technology Co.,Ltd. ( Panjin Greenchem Technology Co.Ltd .)
Capability on chemical synthesis1. Beijing High-Tech Enterprises2. Strong R&D Team3. 8 years of experiences in R & D of high-tech Catalyst;4. 5000 production techniques, 69 items of national patents, and 360 kinds of products on sales;5. The producti
Kono Chem Co.,Ltd
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:drum and bag Application:for pharma use Transportation:by sea or air Port:Beijing or Guangzhou
GIHI CHEMICALS CO.,LIMITED
high purity,in stock Package:25kg/drum,or as per customers'demand Application:API,Pharmaceutical intermediates Transportation:air,sea,courier
Wuhan Fortuna Chemical Co.,Ltd
1.High quality : the purity is 99% min . through multiple producing procedures. 2.Competitive price : low price because of our skilled production technolpgy ,save the production cost at most , and give big profit room to our customers321.Safe and fa
Henan Allgreen Chemical Co.,Ltd
high quality Storage:Sealed, dry, microtherm , avoid light and smell. Package:According to the demand of customer Application:Organic synthesis Transportation:by air or by sea
Nanjing Yuance Industry&Trade Co., Ltd.
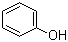
product Name 2,6-Dimethylphenol Synonyms 2,6-Xylenol; 2,6-dimethyl phenol; 2,6-Xylenol(OH=1) Molecular Formula C8H10O Molecular Weight 122.17 InChI
Cas:576-26-1
Min.Order:0 Metric Ton
Negotiable
Type:Trading Company
inquiryAecochem Corp.
Our clients, like BASF,CHEMO,Brenntag,ASR,Evonik,Merck and etc.Appearance:COA Storage:in stock Application:MSDS/TDS
Zhuozhou Wenxi import and Export Co., Ltd
Product Description Description & Specification Category Pharmaceutical Raw Materials, Fine Chemicals, Bulk drug Standard Medical standard
Cas:576-26-1
Min.Order:1 Kilogram
FOB Price: $112.0
Type:Trading Company
inquiryAntimex Chemical Limied
Ansciep Chemical is a professional enterprise manufacturing and distributing fine chemicals and speciality chemicals. We have been dedicated to heterocycle compounds and phenyl rings for tens of years. This is our mature product for export. Our quali
Xiamen AmoyChem Co.,Ltd
Amoychem is committed to providing the top-quality chemical products and services Internationally. We offer our customers with friendly, professional service and reliable, high performance products that have been manufactured according to the accredi
Wuhan ZeShanCheng Biomedical Technology Co., Ltd.
1,we produce and sell good chemicals around the world. 2,our success rate is about 95%. this means, if customer order is accepted, the probability that the customer will obtain the ordered substances, is 95%. 3,our staff consists of highl
Synthetic route

-
-
576-26-1
2.6-dimethylphenol

| Conditions | Yield |
|---|---|
| With sodium methylate In methanol; dichloromethane at 20℃; for 3h; | 98% |
| With hydrogenchloride In 1,4-dioxane for 3h; Product distribution; Heating; deprotection; | 91% |
| In 2,2,2-trifluoroethanol at 100℃; for 1h; Microwave irradiation; | 96 %Spectr. |

| Conditions | Yield |
|---|---|
| aluminum oxide; nickel(II) sulphate In various solvent(s) at 305℃; Product distribution; fixed, quartz powder bed tubular reactor; gaseous phase in N2 flow; varied: temperature 280 to 330 deg C; N2 flow; quartz powder/catalyst ratio; residence time; substrates other alkylated phenols too; | A 97.5% B n/a |

| Conditions | Yield |
|---|---|
| 97% |
-
-
15827-34-6
2,6-bis-<(dimethyamino)methyl>phenol

-
-
576-26-1
2.6-dimethylphenol

| Conditions | Yield |
|---|---|
| With hydrogen; nickel-titanium-aluminum In various solvent(s) at 160℃; for 3h; | 96.2% |
| With hydrogen; nickel-titanium-aluminum In various solvent(s) at 160℃; for 3h; Product distribution; Rate constant; Thermodynamic data; other temperatures; other concentrations of title compound; | 96.2% |

| Conditions | Yield |
|---|---|
| With diethyl dibromomalonate at 100℃; for 48h; Dehydrogenation; | 96% |
| at 330 - 335℃; Leiten ueber Palladium/Kohle; |

| Conditions | Yield |
|---|---|
| With cesiumhydroxide monohydrate; bis[(trimethylsilyl)methyl](1,5-cyclooctadiene)palladium(II); 2-((di-adamantan-1-yl)phosphaneyl)-1-(2,6-diisopropylphenyl)-1H-imidazole In tetrahydrofuran at 24℃; for 20h; Inert atmosphere; | 96% |
| With potassium hydroxide; tris-(dibenzylideneacetone)dipalladium(0); tert-butyl XPhos In 1,4-dioxane; water at 100℃; for 8h; | 94% |
| With 5-(di-tert-butylphosphino)-1′, 3′, 5′-triphenyl-1′H-[1,4′]bipyrazole; tris-(dibenzylideneacetone)dipalladium(0); cesiumhydroxide monohydrate In tetrahydrofuran at 24℃; Inert atmosphere; Glovebox; Sealed tube; | 92% |
| Stage #1: 2,6-dimethyl-1-chlorobenzene With potassium hydroxide; tris-(dibenzylideneacetone)dipalladium(0); 2-((di-adamantan-1-yl)phosphaneyl)-1-(2,6-diisopropylphenyl)-1H-imidazole In 1,4-dioxane; water at 100℃; for 20h; Inert atmosphere; Stage #2: With hydrogenchloride In 1,4-dioxane; water at 20℃; Inert atmosphere; | 90% |
-
-
100379-00-8
2,6-dimethylbenzene boronic acid

-
-
576-26-1
2.6-dimethylphenol

| Conditions | Yield |
|---|---|
| With water; caesium carbonate; hydrazine hydrate at 80℃; for 12h; | 96% |
| With water; 3-chloro-benzenecarboperoxoic acid In ethanol at 20℃; for 6h; | 94% |
| With 2,5-dimethylfuran; zinc(II) phthalocyanine; oxygen; citric acid In tetrahydrofuran at 25℃; under 760.051 Torr; for 1.5h; Irradiation; Sealed tube; Schlenk technique; | 93% |
-
-
100379-00-8
2,6-dimethylbenzene boronic acid

-
-
7722-84-1
dihydrogen peroxide

-
-
576-26-1
2.6-dimethylphenol

| Conditions | Yield |
|---|---|
| With ammonium bicarbonate In water at 20℃; for 2h; Schlenk technique; | 95% |
-
-
438247-33-7
Ni(CH2C(CH3)2C6H5)OC6H3(CH3)2(((CH3)2CH)2P(CH2)2P(CH(CH3)2)2)

-
A
-
576-26-1
2.6-dimethylphenol

-
-
438247-37-1
Ni(CH2C(CH3)2C6H4)(((CH3)2CH)2P(CH2)2P(CH(CH3)2)2)

| Conditions | Yield |
|---|---|
| In tetrahydrofuran (N2); stirring a soln. of nickel complex in THF at room temp. for 12 h; evapn., extn. with Et2O, filtration, concn., addn. of petroleum ether, cooling to -30°C; elem. anal.; | A n/a B 94% |

-
-
576-26-1
2.6-dimethylphenol

| Conditions | Yield |
|---|---|
| With Oxone; water In acetone at 20℃; for 0.0333333h; | 94% |

| Conditions | Yield |
|---|---|
| With N,N,N,N,N,N-hexamethylphosphoric triamide; lithium In tetrahydrofuran; ethanol at -30℃; for 2h; | 93% |
| With 1,3-dimethyl-2-imidazolidinone; lithium diisopropyl amide In tetrahydrofuran; n-heptane; ethylbenzene at 185℃; for 12h; further reagent: NaN(SiMe3)2; | 91% |
| With lithium; ethylenediamine In tetrahydrofuran at -10℃; for 1h; | 83% |
| With sodium methylate; 1-dodecylthiol In N,N-dimethyl-formamide at 100℃; for 1h; | 75% |
| With boron trichloride; tetra-(n-butyl)ammonium iodide In dichloromethane at -78 - 20℃; for 2h; dealkylation; | 70% |
-
-
117658-42-1
2-(2,6-dimethylphenoxy)tetrahydro-2H-pyran

-
-
576-26-1
2.6-dimethylphenol

| Conditions | Yield |
|---|---|
| With boron trifluoride diethyl etherate; sodium cyanoborohydride In tetrahydrofuran for 8h; Ambient temperature; | 92% |
-
-
100379-00-8
2,6-dimethylbenzene boronic acid

-
-
576-26-1
2.6-dimethylphenol

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; | 92% |

| Conditions | Yield |
|---|---|
| With haggite at 300℃; for 6h; Autoclave; Inert atmosphere; | A 89.1% B 5.6% |


| Conditions | Yield |
|---|---|
| With potassium hydroxide; water; polyaniline-supported palladium In 1,4-dioxane at 100℃; for 12h; | 89% |
| With copper(l) iodide; lithium pipecolinate; tetrabutyl ammonium fluoride; sodium hydroxide In water at 130℃; for 24h; | 45% |
| Multi-step reaction with 3 steps 1.1: tert.-butyl lithium / tetrahydrofuran; hexane / 1 h / -78 °C / Inert atmosphere 1.2: -78 - 20 °C / Inert atmosphere 2.1: palladium 10% on activated carbon / 0.08 h / 20 °C / Inert atmosphere 3.1: tetrabutyl ammonium fluoride; water; dihydrogen peroxide; potassium hydrogencarbonate / tetrahydrofuran; methanol / 3 h / 20 °C View Scheme |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water; dimethyl sulfoxide at 120℃; for 16h; | 89% |
| Stage #1: 2,6-dimethyliodobenzene With copper(l) iodide; 2-methyl-8-quinolinol; tetra(n-butyl)ammonium hydroxide In water; dimethyl sulfoxide at 100℃; for 10h; Stage #2: With hydrogenchloride In water; N,N-dimethyl-formamide at 20℃; | 88% |
| With glycolic Acid; copper hydroxide; sodium hydroxide In water; dimethyl sulfoxide at 120℃; for 6h; Inert atmosphere; Schlenk technique; | 87% |

-
-
576-26-1
2.6-dimethylphenol

| Conditions | Yield |
|---|---|
| With zirconocene dichloride In tetrahydrofuran at 20℃; Inert atmosphere; | 88% |
-
-
67-56-1
methanol

-
-
108-95-2
phenol

-
A
-
576-26-1
2.6-dimethylphenol

-
B
-
100-66-3
methoxybenzene

-
C
-
95-48-7
ortho-cresol

| Conditions | Yield |
|---|---|
| zirconium(IV) oxide at 320℃; under 112509 Torr; for 1.5h; Product distribution; Kinetics; Further Variations:; Catalysts; Pressures; Temperatures; | A 87% B 0.5% C 6% |
| aluminum oxide at 290℃; under 195.02 - 600.05 Torr; Kinetics; Product distribution; Rate constant; | |
| samarium(III) oxide; tin(IV) oxide at 349.85℃; for 1h; Product distribution; Further Variations:; Catalysts; Alkylation; |


| Conditions | Yield |
|---|---|
| With sodium sulfite In water at 130℃; for 10h; Sealed tube; Microwave irradiation; Green chemistry; | 87% |

| Conditions | Yield |
|---|---|
| In methanol at 21.85℃; Irradiation; | 85% |
| In isopropyl alcohol for 15h; Irradiation; Inert atmosphere; | 92 %Chromat. |
| With dehaloperoxidase; dihydrogen peroxide In ethanol at 4℃; pH=7; Kinetics; aq. phosphate buffer; Enzymatic reaction; |
-
-
845786-38-1
tert-butyl-[2-(2,6-dimethyl-phenoxy)-ethyl]-diphenyl-silane

-
-
576-26-1
2.6-dimethylphenol

| Conditions | Yield |
|---|---|
| With tetrabutyl ammonium fluoride In tetrahydrofuran at 40℃; | 83% |
-
-
54058-00-3
diethyl phosphite potassium salt

-
-
86364-02-5
2,6-dimethylphenyl trifluoromethanesulphonate

-
A
-
576-26-1
2.6-dimethylphenol

-
B
-
39604-15-4
2,6-dimethylphenyl diethyl phosphate

| Conditions | Yield |
|---|---|
| In ammonia at -33℃; for 21h; | A 12% B 82% |


| Conditions | Yield |
|---|---|
| With decarboxylase at 25℃; for 15h; pH=6.5; Enzymatic reaction; | 80.3% |
| With hydrogenchloride at 200℃; |

| Conditions | Yield |
|---|---|
| Stage #1: m-xylene With spiro[bicyclo[2.2.1]heptane-2,4′-[1,2]dioxolane]-3′,5′-dione at 40℃; for 84h; Sealed tube; Stage #2: With methylamine In ethanol | A 5% B 78% |
| Stage #1: m-xylene With phthaloyl peroxide Heating; Stage #2: With water; sodium hydrogencarbonate In methanol at 40℃; Overall yield = 94 %; | |
| With perfluorodecanoic acid; cytochrome P450Bm3 KT2 (A191T/N239H/I259V/A276T/L353I); oxygen; NADPH In dimethyl sulfoxide at 30℃; pH=7.4; Reagent/catalyst; Enzymatic reaction; | |
| With C27H19N3O7OsS; dihydrogen peroxide; acetic acid In dichloromethane at 23℃; for 0.416667h; Inert atmosphere; Overall yield = 95 %; |

| Conditions | Yield |
|---|---|
| With sodium hydroxide; tetra(n-butyl)ammonium hydrogensulfate In tetrahydrofuran at 20℃; | 75% |
-
-
100379-00-8
2,6-dimethylbenzene boronic acid

-
A
-
576-26-1
2.6-dimethylphenol

-
B
-
87-62-7
2,6-dimethylaniline

| Conditions | Yield |
|---|---|
| With ammonium hydroxide; potassium nitrate In water at 20℃; for 2h; Electrochemical reaction; chemoselective reaction; | A n/a B 73% |
| With ammonium hydroxide; potassium nitrate In water at 20℃; for 4h; Electrochemical reaction; chemoselective reaction; | A 70% B n/a |

| Conditions | Yield |
|---|---|
| With phosphovanadomolybdic acid; oxygen under 750.075 Torr; | 100% |
| With Cu2(ophen)2 In methanol at 28℃; for 12h; | 100% |
| With oxygen In chloroform at 50℃; under 3800.26 Torr; for 24h; Catalytic behavior; Reagent/catalyst; Pressure; Solvent; regioselective reaction; | 97% |
-
-
576-26-1
2.6-dimethylphenol

-
-
94669-47-3, 94669-48-4, 76085-17-1
C25H26ClN2OP

-
-
94669-51-9, 94669-54-2
C33H35N2O2P

| Conditions | Yield |
|---|---|
| With triethylamine at -76℃; | 100% |
-
-
576-26-1
2.6-dimethylphenol

-
-
141793-36-4
dichloro(2,6-dimethylphenoxy)phosphine

| Conditions | Yield |
|---|---|
| With lithium chloride; phosphorus trichloride In neat (no solvent) Inert atmosphere; Schlenk technique; Reflux; | 100% |
| With phosphorus trichloride for 5h; Heating; | 85% |
| With phosphorus trichloride | |
| With phosphorus trichloride for 5h; Substitution; Heating; |

| Conditions | Yield |
|---|---|
| With n-butyllithium In tetrahydrofuran at 0℃; | 100% |
| With tetrabutyl-ammonium chloride; sodium hydroxide In dichloromethane; water at 0℃; for 0.0833333h; | 97% |
| With triethylamine In dichloromethane | |
| With triethylamine In dichloromethane | |
| With pyridine In dichloromethane at 0℃; for 4h; |
-
-
576-26-1
2.6-dimethylphenol

-
-
105-36-2
ethyl bromoacetate

-
-
6279-47-6
ethyl 2-(2,6-dimethylphenoxy)acetate

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone at 20℃; for 24h; | 100% |
| With caesium carbonate In 1,4-dioxane for 48h; Reflux; | 93% |
| With potassium carbonate In N,N-dimethyl-formamide at 20℃; | 89% |

| Conditions | Yield |
|---|---|
| [Ir(1,5-cyclooctadiene)(CH3CN)2]PF6 In toluene at 0℃; for 15h; | 100% |
-
-
576-26-1
2.6-dimethylphenol

-
-
96-32-2
bromoacetic acid methyl ester

-
-
38930-22-2
methyl 2-(2,6-dimethylphenoxy)acetate

| Conditions | Yield |
|---|---|
| With caesium carbonate In acetonitrile at 20℃; for 24h; | 100% |
| In ethyl acetate; N,N-dimethyl-formamide; mineral oil |
-
-
286371-46-8
4-[(6,7-dimethoxy-4-quinolyl)oxy]-2,5-dimethylaniline

-
-
576-26-1
2.6-dimethylphenol

-
-
32315-10-9
bis(trichloromethyl) carbonate

-
-
144-55-8
sodium hydrogencarbonate

| Conditions | Yield |
|---|---|
| With triethylamine In methanol; dichloromethane; chloroform; toluene | 100% |

-
-
576-26-1
2.6-dimethylphenol

-
-
190728-25-7
4-((6,7-dimethoxyquinolin-4-yl)oxy)aniline

-
-
32315-10-9
bis(trichloromethyl) carbonate

-
-
144-55-8
sodium hydrogencarbonate

| Conditions | Yield |
|---|---|
| With triethylamine In methanol; dichloromethane; chloroform; toluene | 100% |

-
-
576-26-1
2.6-dimethylphenol

-
-
166903-87-3
(pentamethylcyclopentadienyl)TaMe(N(2,6-Me2C6H3))(η(1)-N(2,6-Me2C6H3)C(Me)=CMe2)

-
-
180386-44-1
((CH3)5C5)Ta(CH3)(OC6H3(CH3)2)(NC6H3(CH3)2)

| Conditions | Yield |
|---|---|
| In benzene-d6 Ar-atmosphere, sealed NMR tube; 60°C (3 d); not isolated, detd. by NMR spectroscopy; | 100% |
-
-
576-26-1
2.6-dimethylphenol

-
-
5520-77-4
3,10-dihydroxy-3,5,8,10-tetramethyltricyclo[6.2.2.0(2,7)]dodeca-5,11-diene-4,9-dione

| Conditions | Yield |
|---|---|
| Stage #1: 2.6-dimethylphenol With (R)-2-(2-iodoxyphenyl)propanoic acid In tetrahydrofuran at -78℃; Inert atmosphere; Stage #2: With trifluoroacetic acid In tetrahydrofuran at 20℃; for 1h; Inert atmosphere; | 100% |

| Conditions | Yield |
|---|---|
| Stage #1: C17H20N2O8 With 2,4,6-triisopropylphenylsulfonyl chloride; phosphorus pentoxide; potassium carbonate In acetonitrile at 60℃; Inert atmosphere; Stage #2: 2.6-dimethylphenol With 1,4-diaza-bicyclo[2.2.2]octane In acetonitrile at 50℃; for 1h; Inert atmosphere; | 100% |
| Stage #1: C17H20N2O8 With 2,4,6-triisopropylphenylsulfonyl chloride; potassium carbonate In acetonitrile at 60℃; for 2.5h; Inert atmosphere; Stage #2: 2.6-dimethylphenol With 1,4-diaza-bicyclo[2.2.2]octane In acetonitrile at 50℃; for 1h; | 100% |
-
-
576-26-1
2.6-dimethylphenol

-
-
112-71-0
1-Bromotetradecane

-
-
1335029-37-2
2-(tetradecyloxy)-1,3-dimethylbenzene

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetonitrile for 48h; Williamson synthesis; Inert atmosphere; Reflux; | 99.3% |
| Stage #1: 2.6-dimethylphenol With sodium hydroxide In ethanol at 25℃; for 2h; Stage #2: 1-Bromotetradecane In ethanol; N,N-dimethyl-formamide at 60℃; for 6h; | 79% |
-
-
576-26-1
2.6-dimethylphenol

-
-
81639-99-8
di(2,6-xylyl)phosphorochloridate

| Conditions | Yield |
|---|---|
| With hydrogenchloride; trichlorophosphate; magnesium chloride In 5,5-dimethyl-1,3-cyclohexadiene | 99.2% |
| With magnesium chloride; trichlorophosphate at 150℃; for 4h; | 92% |
| With trichlorophosphate | 53% |

| Conditions | Yield |
|---|---|
| With benzyltrimethylammonium tribromide In methanol; dichloromethane | 99% |
| With phosphotungstic acid; N-bromosaccharin In acetonitrile at 0℃; regioselective reaction; | 98% |
| With 2,4,4,6-tetrabromo-3-n-pentadecylcyclohexa-2,5-dienone In cyclohexane at 20℃; for 24h; | 96% |

| Conditions | Yield |
|---|---|
| With tetrabutylammonium chromate; sodium nitrite In dichloromethane for 23h; Reflux; chemoselective reaction; | 99% |
| With trichloroisocyanuric acid; bismuth subnitrate/charcoal In dichloromethane at 20℃; for 2h; regioselective reaction; | 85% |
| With silica-acetate; dinitrogen tetraoxide In dichloromethane at 20℃; for 0.0833333h; | 83% |

| Conditions | Yield |
|---|---|
| Stage #1: 2.6-dimethylphenol With oxygen In tert-butyl alcohol at 70℃; under 760.051 Torr; for 18h; Green chemistry; Stage #2: With hydrogen In tert-butyl alcohol at 50℃; under 760.051 Torr; for 4h; Catalytic behavior; Temperature; Solvent; Time; Green chemistry; | 99% |
| With CuCl(OH)*TMEDA In dichloromethane at 20℃; for 5h; | 93% |
| Stage #1: 2.6-dimethylphenol In dichloromethane at 20℃; for 5h; Stage #2: With sodium dithionite In ethanol for 2h; Heating; | 93% |

| Conditions | Yield |
|---|---|
| With silver trifluoromethanesulfonate at 60℃; for 0.15h; neat (no solvent); | 99% |
| With 4-(N,N-dimethylamino)pyridine based 1,3,5,7-tetraphenyladamantane polymer In neat (no solvent) at 25℃; for 1.5h; Schlenk technique; | 98% |
| With sulfuric acid at 0 - 20℃; for 1h; | 94% |
-
-
576-26-1
2.6-dimethylphenol

-
-
358-23-6
trifluoromethylsulfonic anhydride

-
-
86364-02-5
2,6-dimethylphenyl trifluoromethanesulphonate

| Conditions | Yield |
|---|---|
| With triethylamine In chloroform at 20℃; for 25.5h; | 99% |
| With 2,4,6-trimethyl-pyridine In dichloromethane | 93% |
| With triethylamine In dichloromethane at -78 - 20℃; for 14h; Inert atmosphere; | 90% |
-
-
576-26-1
2.6-dimethylphenol

-
-
616-38-6
carbonic acid dimethyl ester

-
-
1004-66-6
2-methoxy-1,3-dimethylbenzene

| Conditions | Yield |
|---|---|
| With dimanganese decacarbonyl at 180℃; for 1h; Reagent/catalyst; | 99% |
| With N-butyl-4-methylpyridinium bromide at 170℃; for 2h; Inert atmosphere; Ionic liquid; Green chemistry; chemoselective reaction; | 70% |
| N,N,N',N'-tetrabutyl-N''-methylguanidine at 180℃; for 4.5h; | 91 % Chromat. |

| Conditions | Yield |
|---|---|
| for 0.5h; Ambient temperature; | 99% |

-
-
576-26-1
2.6-dimethylphenol

-
-
24424-99-5
di-tert-butyl dicarbonate

| Conditions | Yield |
|---|---|
| With dmap In dichloromethane at 20℃; | 99% |
| With dmap In hexane for 0.05h; Product distribution; protection of var. hindered phenols; var. time; | 98% |
| With dmap In hexane for 0.0833333h; | 98% |
-
-
576-26-1
2.6-dimethylphenol

-
-
54974-07-1
phthalimidesulfenyl chloride

-
-
233600-07-2
N-(3,5-Dimethyl-4-hydroxyphenylthio)phthalimide

| Conditions | Yield |
|---|---|
| In chloroform at 20℃; Substitution; | 99% |

| Conditions | Yield |
|---|---|
| In dichloromethane for 24h; Heating; | 99% |
| In dichloromethane for 5h; Heating; | 91% |

-
-
576-26-1
2.6-dimethylphenol

| Conditions | Yield |
|---|---|
| In benzene byproducts: H2; React. of starting materials in benzene (anhydrous conditions, dry Ar) provides gas evolution. Stirring of mixt.; Removal of solvent by trap-to-trap distn., filtn., elem. anal.; | 99% |

| Conditions | Yield |
|---|---|
| In benzene byproducts: isopropanol; anhyd. conditions; Co(Al(OC3H7)4)2 : 2,6-dimethylphenol = 1 : 4; refluxed (2 h); fractionated isopropanol; solvent removed (reduced pressure); elem.anal.; | 99% |

| Conditions | Yield |
|---|---|
| In hexane byproducts: CH4; dry N2-atmosphere; slight excess of Al2Me6; evapn.; elem. anal.; | 99% |

-
-
576-26-1
2.6-dimethylphenol

-
-
1044744-31-1
(E)-4-(1,3-bis(4-bromophenyl)allyl)-2,6-dimethylphenol

| Conditions | Yield |
|---|---|
| With gold(III) chloride In dichloromethane at 20℃; for 15h; Friedel Crafts alkylation; Molecular sieve; Inert atmosphere; regioselective reaction; | 99% |
-
-
576-26-1
2.6-dimethylphenol

-
-
1192167-42-2
4-(1-hydroxy-1,3-diphenylprop-2-ynyl)benzonitrile

-
-
1192167-48-8
4-(6-hydroxy-5,7-dimethyl-1,3-diphenyl-1H-inden-1-yl)benzonitrile

| Conditions | Yield |
|---|---|
| With ytterbium(III) triflate In nitromethane at 80℃; Inert atmosphere; regioselective reaction; | 99% |
-
-
576-26-1
2.6-dimethylphenol

-
-
3393-96-2
p-nitrobenzanilide

-
-
5336-56-1
(4‐hydroxy‐3,5‐dimethylphenyl)(phenyl)methanone

| Conditions | Yield |
|---|---|
| With trifluorormethanesulfonic acid In dichloromethane at 50℃; for 3h; Friedel Crafts acylation; Inert atmosphere; | 99% |
Related products
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View














 T,
T,  N
N