Dayang Chem (Hangzhou) Co.,Ltd.
As a leading manufacturer and supplier of chemicals in China, DayangChem not only supply popular chemicals, but also DayangChem's R&D center offer custom synthesis services. DayangChem can provide different quantities of custom synthesis ch
Simagchem Corporation
Welcome to Simagchem, your partner in China as a premier supply of bulk specialty chemicals for industry and life science. We introduce experienced quality product and exceptional JIT service with instant market intelligence in China to benefit our
Cas:108-38-3
Min.Order:0 Metric Ton
Negotiable
Type:Manufacturers
inquiryAlity Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Hangzhou Dingyan Chem Co., Ltd
Items Standard Result Appearance White to off-white powder Conforms Purity ≥98.0%
Cas:108-38-3
Min.Order:1 Gram
FOB Price: $100.0 / 500.0
Type:Trading Company
inquiryChemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Henan Tianfu Chemical Co., Ltd.
m-Xylene Basic information Product Name: m-Xylene Synonyms: [srp]m-methyltoluene;1,3-dimethyl-benzen;2,4-Xylene;3-methyltoluene;3-xylene;ai3-08916;Benzene,1,3-dimethyl-;m-Dimethylben
Cas:108-38-3
Min.Order:1 Metric Ton
Negotiable
Type:Lab/Research institutions
inquiryHenan Sinotech Import&Export Corporation
Product name m-Xylene Synonyms M-XYLENE, 1X1ML, MEOH, 200UG/ML;M-XYLENE, STANDARD FOR GC;M-XYLENE DISTILLED 1 L;M-XYLENE DISTILLED 5 L;M-XYLENE, 1X1ML, MEOH, 5000UG/ML;M-XYLENE, 5000MG, NEAT;M-XYLOL,
Hangzhou Keyingchem Co.,Ltd
Hangzhou KeyingChem Co., Ltd. exported this product to many countries and regions at best price. If you are looking for the material’s manufacturer or supplier in China, KeyingChem is your best choice. Pls contact with us freely for getting det
Cas:108-38-3
Min.Order:0 Metric Ton
Negotiable
Type:Lab/Research institutions
inquiryHangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Zibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:108-38-3
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquiryHANGZHOU YUNUO CHEMICAL CO.,LTD
Superior quality Appearance:clear colourless liquid Storage:Stored in cool, dry and ventilation place; Away from fire and heat Package:100g/bottle,1kg/bottle,25kg/drum or as per your request Application: Meta-Xylene is used for the production of is
Henan Wentao Chemical Product Co., Ltd.
1)quick response within 12 hours; 2)quality guarantee: all products are strictly tested by our qc, confirmed by qa and approved by third party lab in china, usa, canada, germany, uk, italy, france etc. 3) oem/odm available; 4) rea
Cas:108-38-3
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquiryHangzhou Huarong Pharm Co., Ltd.
Hangzhou Huarong Pharm Co., Ltd.established since 2006 , has been actively developing specialty products for Finished Dosages, APIs, Intermediates, and Fine chemicals markets in North America, Europe, Korea, Japan, Mid-East and all over the World. Hu
Hangzhou Zhongqi chem Co.,Ltd.
Located in Hangzhou National Hi-Tech Industrial Development Zone, zhongqichem is a technical company mainly focus on the Custom synthesis, manufacturing, sales of chemicals to various industries. Benefiting from the outstanding customer service and h
Kono Chem Co.,Ltd
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:drum and bag Application:for pharma use Transportation:by sea or air Port:Beijing or Guangzhou
Henan Allgreen Chemical Co.,Ltd
high quality Storage:Sealed, dry, microtherm , avoid light and smell. Package:According to the demand of customer Application:Organic synthesis Transportation:by air or by sea
Aecochem Corp.
Our clients, like BASF,CHEMO,Brenntag,ASR,Evonik,Merck and etc.Appearance:COA Storage:in stock Application:MSDS/TDS
Enke Pharma-tech Co.,Ltd. (Cangzhou, China )
Cangzhou Enke Pharma Tech Co.,ltd. is located in Cangzhou City, Hebei province ,where is a famous petroleum chemical industry city?in China. Enke Pharma a high-tech enterprise ,and we are dedicated to developing and manufacturing new api,?intermediat
Zhuozhou Wenxi import and Export Co., Ltd
Product Description Description & Specification Category Pharmaceutical Raw Materials, Fine Chemicals, Bulk drug Standard Medical standard
Cas:108-38-3
Min.Order:1 Kilogram
FOB Price: $112.0
Type:Trading Company
inquiryAntimex Chemical Limied
Ansciep Chemical is a professional enterprise manufacturing and distributing fine chemicals and speciality chemicals. We have been dedicated to heterocycle compounds and phenyl rings for tens of years. This is our mature product for export. Our quali
Xiamen AmoyChem Co.,Ltd
Amoychem is committed to providing the top-quality chemical products and services Internationally. We offer our customers with friendly, professional service and reliable, high performance products that have been manufactured according to the accredi
Hubei Langyou International Trading Co., Ltd
TIANFUCHEM--High purity 108-38-3 m-Xylene Application:TIANFUCHEM--High purity 108-38-3 m-Xylene
Hangzhou Fandachem Co.,Ltd
1,3-Dimethylbenzene cas 108-38-3Appearance:white crystalline powder Storage:Store in dry, dark and ventilated place Package:25KG drum Application:intermediate Transportation:by air, by sea, by express
Hunan Longxianng Runhui Trading Co.,Ltd
TIANFUCHEM--High purity 108-38-3 m-XyleneAppearance:powder Storage:room tempurature Package:As required Application:medical Transportation:By express (Door to door) such as FEDEX, DHL, EMS for small amount. By air(airport to airport) or by sea LCL/FC
Cas:108-38-3
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryChemsigma International Co.,Ltd.
bulk?production Application:Pharmaceutical intermediates
HENAN SUNLAKE ENTERPRISE CORPORATION
Our company is a professional production leading factory in China in pharmaceutical area of many years,our products have exported to USA, Greece, Spain, UK, Australia and other countries, and we have got very good feedback from our cust
Cas:108-38-3
Min.Order:100 Kilogram
Negotiable
Type:Trading Company
inquiryChangzhou Extraordinary Pharmatech co.,LTD
Changzhou Extraordinary Pharmatech co., LTD. As a leading chemical manufacturer and supplier in China.DAS authentication is passed.We can provide the popular precursor chemicals, we have our own strong R & D team, have our own laboratories and fa
Hebei Ruishun Trade Co.,Ltd
Hebei ruishun trade co.,ltd, registered capital one million,have a production of pharmaceutical raw materials, pharmaceutical raw materials factory reagent r&d center,seek development by credit reputation.our products have a large price advanta
Qingdao Sigma Chemical Co., Ltd.
Qingdao Sigma Chemical Ltd is is a global chemical industry manufacturers and suppliers of pharmaceuticals and intermediates, peptide,Nootropis etc API, food and feed additives, herbal extracts, agrochemicals and fine chemicals etc. Our Labo
Synthetic route
-
-
132636-71-6
3-(3,5-Dimethyl-phenoxy)-benzo[d]isothiazole 1,1-dioxide

-
A
-
108-38-3
m-xylene

-
B
-
81-07-2
saccharin

| Conditions | Yield |
|---|---|
| With sodium hypophosphite; palladium on activated charcoal In water; benzene for 2.5h; Heating; | A 100% B n/a |

| Conditions | Yield |
|---|---|
| With palladium dichloride In methanol at 40℃; for 1h; Green chemistry; chemoselective reaction; | 99% |
| With Perbenzoic acid; tri-n-butyl-tin hydride In benzene at 90℃; for 12h; Mechanism; in the presence of α-chlorotoluene (competitor), relative reactivity; |

| Conditions | Yield |
|---|---|
| With palladium dichloride In methanol at 40℃; for 18h; Inert atmosphere; Green chemistry; chemoselective reaction; | 99% |
| With 2,4,6-trimethyl-pyridine; 4,4'-dimethoxyphenyl disulfide; iridium(lll) bis[2-(2,4-difluorophenyl)-5-methylpyridine-N,C20]-4,40-di-tert-butyl-2,20-bipyridine hexafluorophosphate; triphenylphosphine In toluene for 24h; Irradiation; | 68 %Chromat. |

| Conditions | Yield |
|---|---|
| With n-butyllithium; potassium 2-methylbutan-2-olate Mechanism; 1) r.t., 16 h, 2) reflux, 6 h; further reagent: D2O; | 96% |
-
-
100379-00-8
2,6-dimethylbenzene boronic acid

-
-
108-38-3
m-xylene

| Conditions | Yield |
|---|---|
| With water at 20℃; for 48h; | 96% |
| With potassium methanolate In methanol at 120℃; for 12h; Catalytic behavior; Solvent; Sealed tube; Green chemistry; | 67 %Chromat. |
-
-
499-06-9
3,5-dimethylbenzoic acid

-
-
1552-94-9, 28010-12-0, 28010-13-1, 38446-98-9, 54352-97-5
5-phenyl-2,4-pentadienoic acid

-
-
108-38-3
m-xylene

| Conditions | Yield |
|---|---|
| With silver(I) acetate; potassium carbonate In N,N-dimethyl acetamide at 140℃; | 94% |

| Conditions | Yield |
|---|---|
| With tris-(dibenzylideneacetone)dipalladium(0); (S)-(1,1'-binaphthalene)-2,2'-diylbis(diphenylphosphine); sodium t-butanolate In toluene at 80℃; for 2h; Arylation; | A n/a B 88% C n/a |
| With 1,1'-bis-(diphenylphosphino)ferrocene; tris-(dibenzylideneacetone)dipalladium(0); sodium t-butanolate In toluene at 80℃; for 3h; Arylation; |


| Conditions | Yield |
|---|---|
| With formaldehyd; palladium diacetate; caesium carbonate In dimethyl sulfoxide at 80℃; for 12h; | 85% |
-
-
7452-79-1
ethyl 2-methylbutyrate

-
-
201230-82-2
carbon monoxide

-
-
63509-96-6
2,6-xylyllithium

-
A
-
91873-92-6
1-(2,6-Dimethyl-phenyl)-3-methyl-pentane-1,2-dione

-
B
-
108-38-3
m-xylene

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; Dimethyl ether at -135℃; | A 84% B 13% |


| Conditions | Yield |
|---|---|
| With triethylamine; bis(dibenzylideneacetone)-palladium(0) In 1,4-dioxane at 100℃; for 6h; | A n/a B 82% |

-
-
100379-00-8
2,6-dimethylbenzene boronic acid

-
-
105-36-2
ethyl bromoacetate

-
A
-
105337-15-3
ethyl 2-(2,6-dimethylphenyl)acetate

-
B
-
108-38-3
m-xylene

| Conditions | Yield |
|---|---|
| With potassium fluoride; benzyltriethylammonium bromide; tris-(o-tolyl)phosphine; bis(dibenzylideneacetone)-palladium(0) In tetrahydrofuran for 24h; Product distribution / selectivity; Reflux; | A 81.9% B n/a |

-
-
22445-41-6
3,5-dimethylphenyl iodide

-
-
123-30-8
4-amino-phenol

-
A
-
86823-17-8
4-(3,5-dimethylphenoxy)aniline

-
B
-
108-38-3
m-xylene

| Conditions | Yield |
|---|---|
| With copper(l) iodide; potassium carbonate; (1S,2S)-N,N'-dimethyl-1,2-diaminocyclohexane In propyl cyanide at 70℃; for 24h; Inert atmosphere; | A 76% B 14 %Chromat. |

-
-
79-20-9
acetic acid methyl ester

-
-
201230-82-2
carbon monoxide

-
-
63509-96-6
2,6-xylyllithium

-
A
-
150457-77-5
1-(2,6-Dimethyl-phenyl)-propane-1,2-dione

-
B
-
108-38-3
m-xylene

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; diethyl ether; pentane at -135 - 25℃; | A 9% B 73% |
| In tetrahydrofuran; Dimethyl ether at -135℃; | A 9% B 48% |

-
-
137203-34-0
bis(trimethylaluminum)–1,4-diazabicyclo[2.2.2]octane adduct

-
-
625-95-6
3-Iodotoluene

-
-
108-38-3
m-xylene

| Conditions | Yield |
|---|---|
| With dichloro bis(acetonitrile) palladium(II); dicyclohexyl(2',6'-diisopropyl-4'-sulfobiphenyl-2-yl)phosphonium hydrogen sulfate In tetrahydrofuran at 65℃; for 4h; Inert atmosphere; Ionic liquid; chemoselective reaction; | 73% |
-
-
110-54-3
hexane

-
A
-
95-47-6
o-xylene

-
B
-
106-42-3
para-xylene

-
C
-
100-41-4
ethylbenzene

-
D
-
108-38-3
m-xylene

-
E
-
108-88-3
toluene

-
F
-
71-43-2
benzene

| Conditions | Yield |
|---|---|
| 540 degC; treated 16 h; U type zeolite at 450℃; Product distribution; | A n/a B n/a C n/a D n/a E 10.8% F 72.2% |
| 540 degC; treated 16 h; UHS type zeolite at 450℃; Product distribution; | A n/a B n/a C n/a D n/a E 33.3% F 50% |

-
-
201230-82-2
carbon monoxide

-
-
22445-41-6
3,5-dimethylphenyl iodide

-
A
-
5779-95-3
3,5-dimethylbenzaldehyde

-
B
-
108-38-3
m-xylene

| Conditions | Yield |
|---|---|
| With hydrogen; potassium carbonate In 1,4-dioxane at 120 - 140℃; under 30003 Torr; for 20h; Autoclave; | A 71% B 6% |

-
-
623-42-7
butanoic acid methyl ester

-
-
201230-82-2
carbon monoxide

-
-
63509-96-6
2,6-xylyllithium

-
A
-
91887-15-9
1-(2,6-Dimethyl-phenyl)-pentane-1,2-dione

-
B
-
108-38-3
m-xylene

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; Dimethyl ether at -135℃; | A 66% B 20% |

-
-
22445-41-6
3,5-dimethylphenyl iodide

-
B
-
108-38-3
m-xylene

| Conditions | Yield |
|---|---|
| With bis(tri-t-butylphosphine)palladium(0); diisopropylamine In benzene at 20℃; for 24h; Glovebox; Sealed tube; | A 66% B n/a |


| Conditions | Yield |
|---|---|
| With aluminium trichloride; aluminum tri-bromide at 185 - 195℃; for 0.5h; | A 65% B n/a |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at -78℃; for 4h; Yields of byproduct given; | A 61% B n/a |

-
-
556-97-8
1-chloro-3,5-dimethylbenzene

-
-
73183-34-3
bis(pinacol)diborane

-
B
-
108-38-3
m-xylene

-
C
-
325142-93-6
2-(3,5-dimethylphenyl)-4,4,5,5-tetramethyl-1,3,2-dioxaborolane

| Conditions | Yield |
|---|---|
| With bis(1,3-dimesityl-1H-imidazol-2(3H)-ylidene)nickel(0); potassium methanolate In hexane at 25℃; for 6h; Inert atmosphere; Irradiation; | A 3% B 19 %Chromat. C 61% |

-
-
142-82-5
n-heptane

-
A
-
95-47-6
o-xylene

-
B
-
106-42-3
para-xylene

-
C
-
100-41-4
ethylbenzene

-
D
-
108-38-3
m-xylene

-
E
-
108-88-3
toluene

-
F
-
71-43-2
benzene

| Conditions | Yield |
|---|---|
| 540 degC; treated 16 h; U type zeolite at 450℃; Product distribution; | A n/a B n/a C n/a D n/a E 58.5% F 6% |
| 540 degC; treated 16 h; UHS type zeolite at 450℃; Product distribution; | A n/a B n/a C n/a D n/a E 42.8% F 8.8% |


| Conditions | Yield |
|---|---|
| With lithium; nickel dichloride In tetrahydrofuran at 20℃; Reduction; | A 58% B 15% |

-
-
73183-34-3
bis(pinacol)diborane

-
B
-
108-38-3
m-xylene

| Conditions | Yield |
|---|---|
| With cesium fluoride; 2-mercaptopyridine sodium salt In acetonitrile at 30 - 35℃; for 36h; Irradiation; Inert atmosphere; | A 57% B 13 %Chromat. |

-
-
5441-52-1
3,5-dimethylcyclohexan-1-ol

-
A
-
2320-30-1
3,5-dimethylcyclohexanone

-
B
-
108-38-3
m-xylene

-
C
-
108-68-9
3,5-Dimethylphenol

| Conditions | Yield |
|---|---|
| aluminum oxide; nickel at 350℃; Product distribution; effect of temperature on conversion; various temperatures, catalysts various origin and amount; | A 34.5% B 56% C 0.7% |

-
-
31689-43-7
1-methyl-3-methylcyclohexa-2,5-dienecarboxylic acid

-
A
-
108-38-3
m-xylene

| Conditions | Yield |
|---|---|
| With chlorosulfonic acid In dichloromethane at 0℃; for 0.166667h; | A 32% B 53% |
-
-
31689-43-7
1-methyl-3-methylcyclohexa-2,5-dienecarboxylic acid

-
A
-
643749-27-3
C9H12O2

-
B
-
108-38-3
m-xylene

| Conditions | Yield |
|---|---|
| With chlorosulfonic acid In dichloromethane at 0℃; for 0.166667h; | A 53% B 32% |

| Conditions | Yield |
|---|---|
| With PEG-400; sodium hydroxide; zinc; palladium on activated charcoal In water at 100℃; for 2h; coupling; reduction; | A 51% B 38 % Chromat. |

| Conditions | Yield |
|---|---|
| With potassium phosphate; bis(tri-t-butylphosphine)palladium(0) In 1-methyl-pyrrolidin-2-one at 20℃; for 60h; | A 51% B 36 % Chromat. |

-
-
156-60-5
trans-1,2-dichloroethylene

-
-
172975-69-8
3,5-dimethylphenyl boronic acid

-
B
-
13863-27-9
(E)-1,2-bis(3,5-dimethylphenyl)ethene

-
C
-
108-38-3
m-xylene

| Conditions | Yield |
|---|---|
| With cesium fluoride; tris-(dibenzylideneacetone)dipalladium(0); 2,2'-bis(di-tert-butylphosphino)biphenyl In 1,4-dioxane at 70℃; Suzuki coupling; | A 50% B n/a C n/a |


| Conditions | Yield |
|---|---|
| With gallium tri(nonafluorobutane sulfonate); lithium perchlorate In nitromethane at 20℃; for 6h; Acetylation; Friedel-Crafts reaction; | 100% |
| With lithium perchlorate; hafnium tetrakis(trifluoromethanesulfonate) In nitromethane for 6h; Product distribution; Ambient temperature; effect of var. Lewis acid catalysts, LiClO4 and different solvents; | 91% |
| With hafnium(IV) trifluoromethanesulfonate; lithium perchlorate In nitromethane for 6h; Product distribution; Ambient temperature; other aromatic compounds, acylation and alkylation agents, Lewis acids, var. concetration of LiClO4; | 91% |

| Conditions | Yield |
|---|---|
| With gallium(III) triflate; 1-dodecyloxy-4-(heptadecafluorooctyl)benzene In carbon dioxide at 100℃; under 60004.8 Torr; for 6h; | 100% |
| Hf[N(SO2C8F17)2]4 In various solvent(s) at 120℃; for 3h; | 96% |
| With VNU-1 In nitrobenzene at 120℃; for 0.0833333h; Friedel-Crafts Acylation; Microwave irradiation; | 92% |

| Conditions | Yield |
|---|---|
| With trifluoroacetic anhydride; bismuth(lll) trifluoromethanesulfonate at 30℃; for 6h; Product distribution; Further Variations:; Catalysts; Reagents; Temperatures; reaction times; Friedel-Crafts acetylation; | 100% |
| With trifluoromethylsulfonic anhydride at 20℃; for 0.0833333h; Friedel-Crafts acylation; | 97% |
| Stage #1: acetic acid; m-xylene With trifluoroacetic anhydride In dichloromethane at 20℃; for 0.25h; Stage #2: With trifluorormethanesulfonic acid In dichloromethane at 20℃; for 1h; | 88% |
-
-
108-38-3
m-xylene

-
-
638-04-0, 2207-03-6, 69685-67-2, 69685-68-3, 129939-72-6, 129939-73-7, 591-21-9
1,3-dimethylcyclohexane

| Conditions | Yield |
|---|---|
| With hydrogen; [(norbornadiene)rhodium(I)chloride]2; phosphinated polydiacetylene In n-heptane at 30℃; under 60800 Torr; for 1.5h; | 100% |
| With 5% Ru/MgO; hydrogen In tetrahydrofuran at 120℃; under 7600.51 Torr; for 2.9h; | 100% |
| With Ti8O8(14+)*6C8H4O4(2-)*4O(2-)*3.3Li(1+)*0.7Co(2+)*0.7C4H8O*0.7H(1-); hydrogen In neat (no solvent) at 120℃; under 37503.8 Torr; for 18h; | 100% |
-
-
5565-36-6
1,4-bis-(5,5-dimethyl-1,3,2-dioxaborinan-2-yl)benzene

-
-
108-38-3
m-xylene

-
-
2712-78-9
bis-[(trifluoroacetoxy)iodo]benzene

-
-
1417418-28-0
C14H14I(1+)*C2F3O2(1-)

| Conditions | Yield |
|---|---|
| With air In dichloromethane at 20℃; for 6h; regioselective reaction; | 100% |


| Conditions | Yield |
|---|---|
| With chlorine; 1-butyl-3-methylimidazolium chloride at 58℃; for 3h; Reagent/catalyst; Temperature; Irradiation; Ionic liquid; | 99.18% |
| With chlorine Sonnenlicht; | |
| With phosphorus pentachloride at 190℃; |

| Conditions | Yield |
|---|---|
| With silica gel supported sodium hydrogen sulfate at 60℃; for 0.5h; Friedel-Crafts type alkylation; | 99% |
| With boron trifluoride diethyl etherate; water at 80℃; for 2h; regioselective reaction; | 92% |
| With phosphorus pentoxide |
-
-
18734-63-9
perfluoro-o-phenylenemercury trimer

-
-
108-38-3
m-xylene

-
-
649559-07-9
perfluoro-ortho-phenylene mercury trimer - meta-xylene (1:1)

| Conditions | Yield |
|---|---|
| In further solvent(s) Hg-compound was dissolved by boiling in m-xylene, cooled; slow evapd. in a well-aerated fume hood; elem. anal.; | 99% |
-
-
60430-43-5
norborna-2,5-diene(1,2-bis(diphenylphosphino)ethane)rhodium(I) tetrafluoroborate

-
-
108-38-3
m-xylene

-
-
188659-74-7
(C6H5)2PCH2CH2P(C6H5)2RhC6H4(CH3)2(1+)*BF4(1-)=[(C6H5)2PCH2CH2P(C6H5)2RhC6H4(CH3)2]BF4

| Conditions | Yield |
|---|---|
| With H2 In methanol (N2); solution of Rh complex was bubbled with H2 (5 min), concn. of the solution, addn. of 1,3-dimethylbenzene, stirring (12 h, room temp.); removal of the solvent; | 99% |
-
-
531490-86-5
[Ir(2,6-bis(di-tert-butylphosphinomethylene)pyridine)(cyclooctene)][PF6]

-
-
108-38-3
m-xylene

-
-
898225-76-8
[Ir(2,6-bis(di-tert-butylphosphinomethylene)pyridine)(H)(m-xylyl)][PF6]

| Conditions | Yield |
|---|---|
| In m-xylene under N2 atm. soln. Ir complex in m-xylene was heated at 60°C for1 h; solvent was evapd., residue was washed with pentane and ether and dried in vacuo overnight; elem. anal.; | 99% |
-
-
108-38-3
m-xylene

-
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

-
-
325142-93-6
2-(3,5-dimethylphenyl)-4,4,5,5-tetramethyl-1,3,2-dioxaborolane

| Conditions | Yield |
|---|---|
| With RhH{κ3-P,O,P-[9,9-dimethyl-4,5-bis-(diisopropylphosphino)xanthene2]}; cyclohexene at 110℃; for 48h; Inert atmosphere; Glovebox; | 99% |
| With IrH2(Bpin){κ3-P,O,P-[9,9-dimethyl-4,5-bis(diisopropylphosphino)xanthene]}; cyclohexene at 110℃; for 18h; Inert atmosphere; Glovebox; | 95% |
| With (N,N'-bis(2,6-diisopropylphenyl)imidazol-2-ylidene)CuFp at 25℃; for 48h; Inert atmosphere; Glovebox; UV-irradiation; Sealed tube; | 80% |
-
-
14104-20-2
silver tetrafluoroborate

-
-
12354-84-6, 12354-85-7
bis[dichloro(pentamethylcyclopentadienyl)iridium(III)]

-
-
108-38-3
m-xylene

| Conditions | Yield |
|---|---|
| Stage #1: silver tetrafluoroborate; bis[dichloro(pentamethylcyclopentadienyl)iridium(III)] In acetone at 24℃; for 0.25h; Stage #2: m-xylene With trifluoroacetic acid at 24℃; | 99% |


-
-
108-38-3
m-xylene

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide at 100℃; for 24h; Schlenk technique; Inert atmosphere; Green chemistry; | 99% |

| Conditions | Yield |
|---|---|
| With 2-Ethylhexanoic acid; [Fe(CF3SO3)2((S,S)-N,N’-bis(2-pyridylmethyl)-2,2’-bipyrrolidine)]; dihydrogen peroxide In acetonitrile at 0℃; for 2.5h; | 99% |

| Conditions | Yield |
|---|---|
| With 2,2'-azobis(isobutyronitrile); chlorine; aluminum oxide; molybdenum(VI) oxide at 380 - 400℃; for 0.00277778h; | A 95.15% B 98.06% |
-
-
108-38-3
m-xylene

-
-
98-59-9
p-toluenesulfonyl chloride

-
-
13249-97-3
2,4-dimethyl-1-[(4-methylphenyl)sulfonyl]benzene

| Conditions | Yield |
|---|---|
| With tin(II) trifluoromethanesulfonate at 120℃; for 8h; Friedel-Crafts sulfonylation; | 98% |
| With [BTBA]Cl-FeCl3 at 60℃; for 0.0166667h; Friedel-Crafts sulfonylation; | 97% |
| With peroxodisulfate ion; zirconium(IV) oxide at 150℃; for 0.5h; Friedel-Crafts reaction; | 89% |

| Conditions | Yield |
|---|---|
| With sulfuric acid; dihydrogen peroxide; sodium bromide In water at 49.84℃; | 98% |
| With N-Bromosuccinimide In acetonitrile at 0 - 20℃; Inert atmosphere; | 98% |
| With N-Bromosuccinimide In 1,2-dichloro-ethane at 60℃; for 10h; Sealed tube; | 97% |

| Conditions | Yield |
|---|---|
| With trifluoroacetic anhydride; bismuth(lll) trifluoromethanesulfonate at 30℃; for 12h; Friedel-Crafts acetylation; | 98% |
| With pyridin-2-yl trifluoromethanesulfonate; trifluoroacetic acid for 5h; Heating; | 95% |
| With trifluoromethylsulfonic anhydride In nitromethane at 20℃; for 0.0833333h; Friedel-Crafts acylation; | 90% |
-
-
108-38-3
m-xylene

-
-
13369-81-8
2-iodo-4-nitro-1(3)H-imidazole

| Conditions | Yield |
|---|---|
| In acetonitrile for 4h; Irradiation; | 98% |
-
-
108-38-3
m-xylene

-
-
73183-34-3
bis(pinacol)diborane

-
-
325142-93-6
2-(3,5-dimethylphenyl)-4,4,5,5-tetramethyl-1,3,2-dioxaborolane

| Conditions | Yield |
|---|---|
| Stage #1: bis(pinacol)diborane With (1,5-cyclooctadiene)(methoxy)iridium(I) dimer; 4,4'-di-tert-butyl-2,2'-bipyridine In tert-butyl methyl ether Inert atmosphere; Stage #2: m-xylene In tert-butyl methyl ether at 80℃; for 6h; Inert atmosphere; | 98% |
| With 6Zr(4+)*4O(2-)*4HO(1-)*4C18H12O4(2-)*2C16H10N2O4(2-)*C8H12*Ir(1+)*CH3O(1-) In tetrahydrofuran; n-heptane at 115℃; for 16h; Catalytic behavior; Reagent/catalyst; Temperature; Glovebox; Schlenk technique; Inert atmosphere; | 97% |
| With Ir(1+)*C8H12*CH3O(1-)*6C12H6N2O4(2-)*4Zr(4+)*4HO(1-)*4O(2-) In neat (no solvent) at 100℃; for 7h; Time; Reagent/catalyst; Glovebox; Inert atmosphere; Sealed tube; | 94% |
-
-
108-38-3
m-xylene

-
A
-
626-17-5
benzene-1,3-dicarbonitrile

-
B
-
3441-01-8
3-cyanobenzamide

-
C
-
620-22-4
3-Methylbenzonitrile

| Conditions | Yield |
|---|---|
| With ammonia; oxygen; B0.5CrK0.02Mo0086Na0009P0.007V at 350 - 420℃; under 1500.15 Torr; Gas phase; Heating / reflux; | A 98% B n/a C n/a |

-
-
64881-40-9
(3S)-3-methoxycarbonyl-3-trifluoroacetylaminopropanoyl chloride

-
-
108-38-3
m-xylene

-
-
1088806-53-4
methyl 4-(2,4-dimethylphenyl)-4-oxo-2-(2,2,2-trifluoroacetamido)butanoate

| Conditions | Yield |
|---|---|
| With trifluorormethanesulfonic acid at 0℃; for 1h; Friedel Crafts acylation; Neat (no solvent); | 98% |
-
-
108-38-3
m-xylene

-
-
161553-08-8
(R)-N-trifluoroacetylaspartic acid-4-chloride-1-methyl ester

-
-
1088806-55-6
methyl 4-(2,4-dimethylphenyl)-4-oxo-2-(2,2,2-trifluoroacetamido)butanoate

| Conditions | Yield |
|---|---|
| With trifluorormethanesulfonic acid at 0℃; for 1h; Friedel Crafts acylation; Neat (no solvent); | 98% |

| Conditions | Yield |
|---|---|
| With di-tert-butyl peroxide; [copper(II)(trifluoroacetylacetonate)2]; salicylic acid at 120℃; for 24h; Inert atmosphere; regioselective reaction; | 98% |
-
-
108-38-3
m-xylene

-
-
264882-00-0
methyl 2-(2-chlorophenyl)-2-diazoacetate

| Conditions | Yield |
|---|---|
| With (triphenylphosphine)gold(I) chloride; silver trifluoromethanesulfonate at 20℃; for 0.0666667h; Schlenk technique; | 98% |
| With MCM-41-PPh2-AuOTf at 20℃; for 0.1h; Schlenk technique; Inert atmosphere; regioselective reaction; | 87% |

| Conditions | Yield |
|---|---|
| With 4-Chloropyridine; nickel(II) oxide; aluminum (III) chloride; barium titanate; 4-trifluoromethylpyridine; copper(II) sulfate In ethanol at 25℃; for 0.0666667h; Temperature; | 97.7% |
| With aluminum (III) chloride In dichloromethane at 20℃; for 1.5h; | 70% |
| montmorillonite doped with TiCl4 for 0.333333h; Heating; | 60% |

| Conditions | Yield |
|---|---|
| Stage #1: phthalic anhydride; m-xylene With aluminum (III) chloride at 55℃; for 5h; cooling with salt-ice; Stage #2: With hydrogenchloride In water; m-xylene cooling with ice; | 97% |
| With aluminum (III) chloride at 60℃; for 0.416667h; Catalytic behavior; Reagent/catalyst; Time; Concentration; Friedel-Crafts Acylation; Ionic liquid; Irradiation; | 85% |
| With aluminium trichloride |
Related products
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View
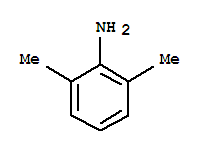













 Xn,
Xn, T,
T, F
F