-
Name
1,3-Dimethylbenzene
- EINECS 203-576-3
- CAS No. 108-38-3
- Article Data486
- CAS DataBase
- Density 0.87 g/cm3
- Solubility Insoluble in water, soluble in alcohol and ether
- Melting Point -48 °C
- Formula C8H10
- Boiling Point 140.567 °C at 760 mmHg
- Molecular Weight 106.167
- Flash Point 25 °C
- Transport Information UN 1307 3/PG 3
- Appearance Colorless transparent liquid
- Safety 25-45-36/37-16-7
- Risk Codes 10-20/21-38-39/23/24/25-23/24/25-11-36/38
-
Molecular Structure
-
Hazard Symbols
 Xn,
Xn, T,
T, F
F
- Synonyms m-Xylene(8CI);1,3-Xylene;3-Methyltoluene;NSC 61769;m-Dimethylbenzene;m-Methyltoluene;m-xylol;M-Xylene;1,3-Dimethylbenzene;
- PSA 0.00000
- LogP 2.30340
Synthetic route
-
-
132636-71-6
3-(3,5-Dimethyl-phenoxy)-benzo[d]isothiazole 1,1-dioxide

-
A
-
108-38-3
m-xylene

-
B
-
81-07-2
saccharin

| Conditions | Yield |
|---|---|
| With sodium hypophosphite; palladium on activated charcoal In water; benzene for 2.5h; Heating; | A 100% B n/a |

| Conditions | Yield |
|---|---|
| With palladium dichloride In methanol at 40℃; for 1h; Green chemistry; chemoselective reaction; | 99% |
| With Perbenzoic acid; tri-n-butyl-tin hydride In benzene at 90℃; for 12h; Mechanism; in the presence of α-chlorotoluene (competitor), relative reactivity; |

| Conditions | Yield |
|---|---|
| With palladium dichloride In methanol at 40℃; for 18h; Inert atmosphere; Green chemistry; chemoselective reaction; | 99% |
| With 2,4,6-trimethyl-pyridine; 4,4'-dimethoxyphenyl disulfide; iridium(lll) bis[2-(2,4-difluorophenyl)-5-methylpyridine-N,C20]-4,40-di-tert-butyl-2,20-bipyridine hexafluorophosphate; triphenylphosphine In toluene for 24h; Irradiation; | 68 %Chromat. |

| Conditions | Yield |
|---|---|
| With n-butyllithium; potassium 2-methylbutan-2-olate Mechanism; 1) r.t., 16 h, 2) reflux, 6 h; further reagent: D2O; | 96% |
-
-
100379-00-8
2,6-dimethylbenzene boronic acid

-
-
108-38-3
m-xylene

| Conditions | Yield |
|---|---|
| With water at 20℃; for 48h; | 96% |
| With potassium methanolate In methanol at 120℃; for 12h; Catalytic behavior; Solvent; Sealed tube; Green chemistry; | 67 %Chromat. |
-
-
499-06-9
3,5-dimethylbenzoic acid

-
-
1552-94-9, 28010-12-0, 28010-13-1, 38446-98-9, 54352-97-5
5-phenyl-2,4-pentadienoic acid

-
-
108-38-3
m-xylene

| Conditions | Yield |
|---|---|
| With silver(I) acetate; potassium carbonate In N,N-dimethyl acetamide at 140℃; | 94% |

| Conditions | Yield |
|---|---|
| With tris-(dibenzylideneacetone)dipalladium(0); (S)-(1,1'-binaphthalene)-2,2'-diylbis(diphenylphosphine); sodium t-butanolate In toluene at 80℃; for 2h; Arylation; | A n/a B 88% C n/a |
| With 1,1'-bis-(diphenylphosphino)ferrocene; tris-(dibenzylideneacetone)dipalladium(0); sodium t-butanolate In toluene at 80℃; for 3h; Arylation; |


| Conditions | Yield |
|---|---|
| With formaldehyd; palladium diacetate; caesium carbonate In dimethyl sulfoxide at 80℃; for 12h; | 85% |
-
-
7452-79-1
ethyl 2-methylbutyrate

-
-
201230-82-2
carbon monoxide

-
-
63509-96-6
2,6-xylyllithium

-
A
-
91873-92-6
1-(2,6-Dimethyl-phenyl)-3-methyl-pentane-1,2-dione

-
B
-
108-38-3
m-xylene

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; Dimethyl ether at -135℃; | A 84% B 13% |


| Conditions | Yield |
|---|---|
| With triethylamine; bis(dibenzylideneacetone)-palladium(0) In 1,4-dioxane at 100℃; for 6h; | A n/a B 82% |

-
-
100379-00-8
2,6-dimethylbenzene boronic acid

-
-
105-36-2
ethyl bromoacetate

-
A
-
105337-15-3
ethyl 2-(2,6-dimethylphenyl)acetate

-
B
-
108-38-3
m-xylene

| Conditions | Yield |
|---|---|
| With potassium fluoride; benzyltriethylammonium bromide; tris-(o-tolyl)phosphine; bis(dibenzylideneacetone)-palladium(0) In tetrahydrofuran for 24h; Product distribution / selectivity; Reflux; | A 81.9% B n/a |

-
-
22445-41-6
3,5-dimethylphenyl iodide

-
-
123-30-8
4-amino-phenol

-
A
-
86823-17-8
4-(3,5-dimethylphenoxy)aniline

-
B
-
108-38-3
m-xylene

| Conditions | Yield |
|---|---|
| With copper(l) iodide; potassium carbonate; (1S,2S)-N,N'-dimethyl-1,2-diaminocyclohexane In propyl cyanide at 70℃; for 24h; Inert atmosphere; | A 76% B 14 %Chromat. |

-
-
79-20-9
acetic acid methyl ester

-
-
201230-82-2
carbon monoxide

-
-
63509-96-6
2,6-xylyllithium

-
A
-
150457-77-5
1-(2,6-Dimethyl-phenyl)-propane-1,2-dione

-
B
-
108-38-3
m-xylene

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; diethyl ether; pentane at -135 - 25℃; | A 9% B 73% |
| In tetrahydrofuran; Dimethyl ether at -135℃; | A 9% B 48% |

-
-
137203-34-0
bis(trimethylaluminum)–1,4-diazabicyclo[2.2.2]octane adduct

-
-
625-95-6
3-Iodotoluene

-
-
108-38-3
m-xylene

| Conditions | Yield |
|---|---|
| With dichloro bis(acetonitrile) palladium(II); dicyclohexyl(2',6'-diisopropyl-4'-sulfobiphenyl-2-yl)phosphonium hydrogen sulfate In tetrahydrofuran at 65℃; for 4h; Inert atmosphere; Ionic liquid; chemoselective reaction; | 73% |
-
-
110-54-3
hexane

-
A
-
95-47-6
o-xylene

-
B
-
106-42-3
para-xylene

-
C
-
100-41-4
ethylbenzene

-
D
-
108-38-3
m-xylene

-
E
-
108-88-3
toluene

-
F
-
71-43-2
benzene

| Conditions | Yield |
|---|---|
| 540 degC; treated 16 h; U type zeolite at 450℃; Product distribution; | A n/a B n/a C n/a D n/a E 10.8% F 72.2% |
| 540 degC; treated 16 h; UHS type zeolite at 450℃; Product distribution; | A n/a B n/a C n/a D n/a E 33.3% F 50% |

-
-
201230-82-2
carbon monoxide

-
-
22445-41-6
3,5-dimethylphenyl iodide

-
A
-
5779-95-3
3,5-dimethylbenzaldehyde

-
B
-
108-38-3
m-xylene

| Conditions | Yield |
|---|---|
| With hydrogen; potassium carbonate In 1,4-dioxane at 120 - 140℃; under 30003 Torr; for 20h; Autoclave; | A 71% B 6% |

-
-
623-42-7
butanoic acid methyl ester

-
-
201230-82-2
carbon monoxide

-
-
63509-96-6
2,6-xylyllithium

-
A
-
91887-15-9
1-(2,6-Dimethyl-phenyl)-pentane-1,2-dione

-
B
-
108-38-3
m-xylene

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; Dimethyl ether at -135℃; | A 66% B 20% |

-
-
22445-41-6
3,5-dimethylphenyl iodide

-
B
-
108-38-3
m-xylene

| Conditions | Yield |
|---|---|
| With bis(tri-t-butylphosphine)palladium(0); diisopropylamine In benzene at 20℃; for 24h; Glovebox; Sealed tube; | A 66% B n/a |


| Conditions | Yield |
|---|---|
| With aluminium trichloride; aluminum tri-bromide at 185 - 195℃; for 0.5h; | A 65% B n/a |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at -78℃; for 4h; Yields of byproduct given; | A 61% B n/a |

-
-
556-97-8
1-chloro-3,5-dimethylbenzene

-
-
73183-34-3
bis(pinacol)diborane

-
B
-
108-38-3
m-xylene

-
C
-
325142-93-6
2-(3,5-dimethylphenyl)-4,4,5,5-tetramethyl-1,3,2-dioxaborolane

| Conditions | Yield |
|---|---|
| With bis(1,3-dimesityl-1H-imidazol-2(3H)-ylidene)nickel(0); potassium methanolate In hexane at 25℃; for 6h; Inert atmosphere; Irradiation; | A 3% B 19 %Chromat. C 61% |

-
-
142-82-5
n-heptane

-
A
-
95-47-6
o-xylene

-
B
-
106-42-3
para-xylene

-
C
-
100-41-4
ethylbenzene

-
D
-
108-38-3
m-xylene

-
E
-
108-88-3
toluene

-
F
-
71-43-2
benzene

| Conditions | Yield |
|---|---|
| 540 degC; treated 16 h; U type zeolite at 450℃; Product distribution; | A n/a B n/a C n/a D n/a E 58.5% F 6% |
| 540 degC; treated 16 h; UHS type zeolite at 450℃; Product distribution; | A n/a B n/a C n/a D n/a E 42.8% F 8.8% |


| Conditions | Yield |
|---|---|
| With lithium; nickel dichloride In tetrahydrofuran at 20℃; Reduction; | A 58% B 15% |

-
-
73183-34-3
bis(pinacol)diborane

-
B
-
108-38-3
m-xylene

| Conditions | Yield |
|---|---|
| With cesium fluoride; 2-mercaptopyridine sodium salt In acetonitrile at 30 - 35℃; for 36h; Irradiation; Inert atmosphere; | A 57% B 13 %Chromat. |

-
-
5441-52-1
3,5-dimethylcyclohexan-1-ol

-
A
-
2320-30-1
3,5-dimethylcyclohexanone

-
B
-
108-38-3
m-xylene

-
C
-
108-68-9
3,5-Dimethylphenol

| Conditions | Yield |
|---|---|
| aluminum oxide; nickel at 350℃; Product distribution; effect of temperature on conversion; various temperatures, catalysts various origin and amount; | A 34.5% B 56% C 0.7% |

-
-
31689-43-7
1-methyl-3-methylcyclohexa-2,5-dienecarboxylic acid

-
A
-
108-38-3
m-xylene

| Conditions | Yield |
|---|---|
| With chlorosulfonic acid In dichloromethane at 0℃; for 0.166667h; | A 32% B 53% |
-
-
31689-43-7
1-methyl-3-methylcyclohexa-2,5-dienecarboxylic acid

-
A
-
643749-27-3
C9H12O2

-
B
-
108-38-3
m-xylene

| Conditions | Yield |
|---|---|
| With chlorosulfonic acid In dichloromethane at 0℃; for 0.166667h; | A 53% B 32% |

| Conditions | Yield |
|---|---|
| With PEG-400; sodium hydroxide; zinc; palladium on activated charcoal In water at 100℃; for 2h; coupling; reduction; | A 51% B 38 % Chromat. |

| Conditions | Yield |
|---|---|
| With potassium phosphate; bis(tri-t-butylphosphine)palladium(0) In 1-methyl-pyrrolidin-2-one at 20℃; for 60h; | A 51% B 36 % Chromat. |

-
-
156-60-5
trans-1,2-dichloroethylene

-
-
172975-69-8
3,5-dimethylphenyl boronic acid

-
B
-
13863-27-9
(E)-1,2-bis(3,5-dimethylphenyl)ethene

-
C
-
108-38-3
m-xylene

| Conditions | Yield |
|---|---|
| With cesium fluoride; tris-(dibenzylideneacetone)dipalladium(0); 2,2'-bis(di-tert-butylphosphino)biphenyl In 1,4-dioxane at 70℃; Suzuki coupling; | A 50% B n/a C n/a |


| Conditions | Yield |
|---|---|
| With gallium tri(nonafluorobutane sulfonate); lithium perchlorate In nitromethane at 20℃; for 6h; Acetylation; Friedel-Crafts reaction; | 100% |
| With lithium perchlorate; hafnium tetrakis(trifluoromethanesulfonate) In nitromethane for 6h; Product distribution; Ambient temperature; effect of var. Lewis acid catalysts, LiClO4 and different solvents; | 91% |
| With hafnium(IV) trifluoromethanesulfonate; lithium perchlorate In nitromethane for 6h; Product distribution; Ambient temperature; other aromatic compounds, acylation and alkylation agents, Lewis acids, var. concetration of LiClO4; | 91% |

| Conditions | Yield |
|---|---|
| With gallium(III) triflate; 1-dodecyloxy-4-(heptadecafluorooctyl)benzene In carbon dioxide at 100℃; under 60004.8 Torr; for 6h; | 100% |
| Hf[N(SO2C8F17)2]4 In various solvent(s) at 120℃; for 3h; | 96% |
| With VNU-1 In nitrobenzene at 120℃; for 0.0833333h; Friedel-Crafts Acylation; Microwave irradiation; | 92% |

| Conditions | Yield |
|---|---|
| With trifluoroacetic anhydride; bismuth(lll) trifluoromethanesulfonate at 30℃; for 6h; Product distribution; Further Variations:; Catalysts; Reagents; Temperatures; reaction times; Friedel-Crafts acetylation; | 100% |
| With trifluoromethylsulfonic anhydride at 20℃; for 0.0833333h; Friedel-Crafts acylation; | 97% |
| Stage #1: acetic acid; m-xylene With trifluoroacetic anhydride In dichloromethane at 20℃; for 0.25h; Stage #2: With trifluorormethanesulfonic acid In dichloromethane at 20℃; for 1h; | 88% |
-
-
108-38-3
m-xylene

-
-
638-04-0, 2207-03-6, 69685-67-2, 69685-68-3, 129939-72-6, 129939-73-7, 591-21-9
1,3-dimethylcyclohexane

| Conditions | Yield |
|---|---|
| With hydrogen; [(norbornadiene)rhodium(I)chloride]2; phosphinated polydiacetylene In n-heptane at 30℃; under 60800 Torr; for 1.5h; | 100% |
| With 5% Ru/MgO; hydrogen In tetrahydrofuran at 120℃; under 7600.51 Torr; for 2.9h; | 100% |
| With Ti8O8(14+)*6C8H4O4(2-)*4O(2-)*3.3Li(1+)*0.7Co(2+)*0.7C4H8O*0.7H(1-); hydrogen In neat (no solvent) at 120℃; under 37503.8 Torr; for 18h; | 100% |
-
-
5565-36-6
1,4-bis-(5,5-dimethyl-1,3,2-dioxaborinan-2-yl)benzene

-
-
108-38-3
m-xylene

-
-
2712-78-9
bis-[(trifluoroacetoxy)iodo]benzene

-
-
1417418-28-0
C14H14I(1+)*C2F3O2(1-)

| Conditions | Yield |
|---|---|
| With air In dichloromethane at 20℃; for 6h; regioselective reaction; | 100% |


| Conditions | Yield |
|---|---|
| With chlorine; 1-butyl-3-methylimidazolium chloride at 58℃; for 3h; Reagent/catalyst; Temperature; Irradiation; Ionic liquid; | 99.18% |
| With chlorine Sonnenlicht; | |
| With phosphorus pentachloride at 190℃; |

| Conditions | Yield |
|---|---|
| With silica gel supported sodium hydrogen sulfate at 60℃; for 0.5h; Friedel-Crafts type alkylation; | 99% |
| With boron trifluoride diethyl etherate; water at 80℃; for 2h; regioselective reaction; | 92% |
| With phosphorus pentoxide |
-
-
18734-63-9
perfluoro-o-phenylenemercury trimer

-
-
108-38-3
m-xylene

-
-
649559-07-9
perfluoro-ortho-phenylene mercury trimer - meta-xylene (1:1)

| Conditions | Yield |
|---|---|
| In further solvent(s) Hg-compound was dissolved by boiling in m-xylene, cooled; slow evapd. in a well-aerated fume hood; elem. anal.; | 99% |
-
-
60430-43-5
norborna-2,5-diene(1,2-bis(diphenylphosphino)ethane)rhodium(I) tetrafluoroborate

-
-
108-38-3
m-xylene

-
-
188659-74-7
(C6H5)2PCH2CH2P(C6H5)2RhC6H4(CH3)2(1+)*BF4(1-)=[(C6H5)2PCH2CH2P(C6H5)2RhC6H4(CH3)2]BF4

| Conditions | Yield |
|---|---|
| With H2 In methanol (N2); solution of Rh complex was bubbled with H2 (5 min), concn. of the solution, addn. of 1,3-dimethylbenzene, stirring (12 h, room temp.); removal of the solvent; | 99% |
-
-
531490-86-5
[Ir(2,6-bis(di-tert-butylphosphinomethylene)pyridine)(cyclooctene)][PF6]

-
-
108-38-3
m-xylene

-
-
898225-76-8
[Ir(2,6-bis(di-tert-butylphosphinomethylene)pyridine)(H)(m-xylyl)][PF6]

| Conditions | Yield |
|---|---|
| In m-xylene under N2 atm. soln. Ir complex in m-xylene was heated at 60°C for1 h; solvent was evapd., residue was washed with pentane and ether and dried in vacuo overnight; elem. anal.; | 99% |
-
-
108-38-3
m-xylene

-
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

-
-
325142-93-6
2-(3,5-dimethylphenyl)-4,4,5,5-tetramethyl-1,3,2-dioxaborolane

| Conditions | Yield |
|---|---|
| With RhH{κ3-P,O,P-[9,9-dimethyl-4,5-bis-(diisopropylphosphino)xanthene2]}; cyclohexene at 110℃; for 48h; Inert atmosphere; Glovebox; | 99% |
| With IrH2(Bpin){κ3-P,O,P-[9,9-dimethyl-4,5-bis(diisopropylphosphino)xanthene]}; cyclohexene at 110℃; for 18h; Inert atmosphere; Glovebox; | 95% |
| With (N,N'-bis(2,6-diisopropylphenyl)imidazol-2-ylidene)CuFp at 25℃; for 48h; Inert atmosphere; Glovebox; UV-irradiation; Sealed tube; | 80% |
-
-
14104-20-2
silver tetrafluoroborate

-
-
12354-84-6, 12354-85-7
bis[dichloro(pentamethylcyclopentadienyl)iridium(III)]

-
-
108-38-3
m-xylene

| Conditions | Yield |
|---|---|
| Stage #1: silver tetrafluoroborate; bis[dichloro(pentamethylcyclopentadienyl)iridium(III)] In acetone at 24℃; for 0.25h; Stage #2: m-xylene With trifluoroacetic acid at 24℃; | 99% |


-
-
108-38-3
m-xylene

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide at 100℃; for 24h; Schlenk technique; Inert atmosphere; Green chemistry; | 99% |

| Conditions | Yield |
|---|---|
| With 2-Ethylhexanoic acid; [Fe(CF3SO3)2((S,S)-N,N’-bis(2-pyridylmethyl)-2,2’-bipyrrolidine)]; dihydrogen peroxide In acetonitrile at 0℃; for 2.5h; | 99% |

| Conditions | Yield |
|---|---|
| With 2,2'-azobis(isobutyronitrile); chlorine; aluminum oxide; molybdenum(VI) oxide at 380 - 400℃; for 0.00277778h; | A 95.15% B 98.06% |
-
-
108-38-3
m-xylene

-
-
98-59-9
p-toluenesulfonyl chloride

-
-
13249-97-3
2,4-dimethyl-1-[(4-methylphenyl)sulfonyl]benzene

| Conditions | Yield |
|---|---|
| With tin(II) trifluoromethanesulfonate at 120℃; for 8h; Friedel-Crafts sulfonylation; | 98% |
| With [BTBA]Cl-FeCl3 at 60℃; for 0.0166667h; Friedel-Crafts sulfonylation; | 97% |
| With peroxodisulfate ion; zirconium(IV) oxide at 150℃; for 0.5h; Friedel-Crafts reaction; | 89% |

| Conditions | Yield |
|---|---|
| With sulfuric acid; dihydrogen peroxide; sodium bromide In water at 49.84℃; | 98% |
| With N-Bromosuccinimide In acetonitrile at 0 - 20℃; Inert atmosphere; | 98% |
| With N-Bromosuccinimide In 1,2-dichloro-ethane at 60℃; for 10h; Sealed tube; | 97% |

| Conditions | Yield |
|---|---|
| With trifluoroacetic anhydride; bismuth(lll) trifluoromethanesulfonate at 30℃; for 12h; Friedel-Crafts acetylation; | 98% |
| With pyridin-2-yl trifluoromethanesulfonate; trifluoroacetic acid for 5h; Heating; | 95% |
| With trifluoromethylsulfonic anhydride In nitromethane at 20℃; for 0.0833333h; Friedel-Crafts acylation; | 90% |
-
-
108-38-3
m-xylene

-
-
13369-81-8
2-iodo-4-nitro-1(3)H-imidazole

| Conditions | Yield |
|---|---|
| In acetonitrile for 4h; Irradiation; | 98% |
-
-
108-38-3
m-xylene

-
-
73183-34-3
bis(pinacol)diborane

-
-
325142-93-6
2-(3,5-dimethylphenyl)-4,4,5,5-tetramethyl-1,3,2-dioxaborolane

| Conditions | Yield |
|---|---|
| Stage #1: bis(pinacol)diborane With (1,5-cyclooctadiene)(methoxy)iridium(I) dimer; 4,4'-di-tert-butyl-2,2'-bipyridine In tert-butyl methyl ether Inert atmosphere; Stage #2: m-xylene In tert-butyl methyl ether at 80℃; for 6h; Inert atmosphere; | 98% |
| With 6Zr(4+)*4O(2-)*4HO(1-)*4C18H12O4(2-)*2C16H10N2O4(2-)*C8H12*Ir(1+)*CH3O(1-) In tetrahydrofuran; n-heptane at 115℃; for 16h; Catalytic behavior; Reagent/catalyst; Temperature; Glovebox; Schlenk technique; Inert atmosphere; | 97% |
| With Ir(1+)*C8H12*CH3O(1-)*6C12H6N2O4(2-)*4Zr(4+)*4HO(1-)*4O(2-) In neat (no solvent) at 100℃; for 7h; Time; Reagent/catalyst; Glovebox; Inert atmosphere; Sealed tube; | 94% |
-
-
108-38-3
m-xylene

-
A
-
626-17-5
benzene-1,3-dicarbonitrile

-
B
-
3441-01-8
3-cyanobenzamide

-
C
-
620-22-4
3-Methylbenzonitrile

| Conditions | Yield |
|---|---|
| With ammonia; oxygen; B0.5CrK0.02Mo0086Na0009P0.007V at 350 - 420℃; under 1500.15 Torr; Gas phase; Heating / reflux; | A 98% B n/a C n/a |

-
-
64881-40-9
(3S)-3-methoxycarbonyl-3-trifluoroacetylaminopropanoyl chloride

-
-
108-38-3
m-xylene

-
-
1088806-53-4
methyl 4-(2,4-dimethylphenyl)-4-oxo-2-(2,2,2-trifluoroacetamido)butanoate

| Conditions | Yield |
|---|---|
| With trifluorormethanesulfonic acid at 0℃; for 1h; Friedel Crafts acylation; Neat (no solvent); | 98% |
-
-
108-38-3
m-xylene

-
-
161553-08-8
(R)-N-trifluoroacetylaspartic acid-4-chloride-1-methyl ester

-
-
1088806-55-6
methyl 4-(2,4-dimethylphenyl)-4-oxo-2-(2,2,2-trifluoroacetamido)butanoate

| Conditions | Yield |
|---|---|
| With trifluorormethanesulfonic acid at 0℃; for 1h; Friedel Crafts acylation; Neat (no solvent); | 98% |

| Conditions | Yield |
|---|---|
| With di-tert-butyl peroxide; [copper(II)(trifluoroacetylacetonate)2]; salicylic acid at 120℃; for 24h; Inert atmosphere; regioselective reaction; | 98% |
-
-
108-38-3
m-xylene

-
-
264882-00-0
methyl 2-(2-chlorophenyl)-2-diazoacetate

| Conditions | Yield |
|---|---|
| With (triphenylphosphine)gold(I) chloride; silver trifluoromethanesulfonate at 20℃; for 0.0666667h; Schlenk technique; | 98% |
| With MCM-41-PPh2-AuOTf at 20℃; for 0.1h; Schlenk technique; Inert atmosphere; regioselective reaction; | 87% |

| Conditions | Yield |
|---|---|
| With 4-Chloropyridine; nickel(II) oxide; aluminum (III) chloride; barium titanate; 4-trifluoromethylpyridine; copper(II) sulfate In ethanol at 25℃; for 0.0666667h; Temperature; | 97.7% |
| With aluminum (III) chloride In dichloromethane at 20℃; for 1.5h; | 70% |
| montmorillonite doped with TiCl4 for 0.333333h; Heating; | 60% |

| Conditions | Yield |
|---|---|
| Stage #1: phthalic anhydride; m-xylene With aluminum (III) chloride at 55℃; for 5h; cooling with salt-ice; Stage #2: With hydrogenchloride In water; m-xylene cooling with ice; | 97% |
| With aluminum (III) chloride at 60℃; for 0.416667h; Catalytic behavior; Reagent/catalyst; Time; Concentration; Friedel-Crafts Acylation; Ionic liquid; Irradiation; | 85% |
| With aluminium trichloride |
1,3-Dimethylbenzene Chemical Properties
.
1,3-Dimethylbenzene Consensus Reports
1,3-Dimethylbenzene Standards and Recommendations
ACGIH TLV: TWA 100 ppm; STEL 150 ppm; BEI: methyl hippuric acids in urine at end of shift 1.5 g/g creatinine; Not Classifiable as a Human Carcinogen
NIOSH REL: (Xylene) TWA 100 ppm; CL 200 ppm/10M
DOT Classification: 3; Label: Flammable Liquid
1,3-Dimethylbenzene Specification
The 1,3-Dimethylbenzene with CAS registry number of 108-38-3 is also known as 1,3-Dimethylbenzene. The IUPAC name is 1,3-Xylene. It belongs to product categories of Analytical Chemistry; Standard Solution of Volatile Organic Compounds for Water & Soil Analysis; Standard Solutions (VOC). Its EINECS registry number is 203-576-3. In addition, the formula is C8H10 and the molecular weight is 106.17. This chemical is a colorless transparent liquid and should be stored in cool, dry room away from fire and oxidizing agents.
Physical properties about 1,3-Dimethylbenzene are:
(1)ACD/LogP: 3.27; (2)ACD/LogD (pH 5.5): 3.271; (3)ACD/LogD (pH 7.4): 3.271; (4)ACD/BCF (pH 5.5): 180.187; (5)ACD/BCF (pH 7.4): 180.187; (6)ACD/KOC (pH 5.5): 1433.027; (7)ACD/KOC (pH 7.4): 1433.027; (8)Index of Refraction: 1.5; (9)Molar Refractivity: 35.903 cm3; (10)Molar Volume: 121.984 cm3; (11)Polarizability: 14.233×10-24cm3; (12)Surface Tension: 28.791 dyne/cm; (13)Density: 0.87 g/cm3; (14)Flash Point: 25 °C; (15)Enthalpy of Vaporization: 35.66 kJ/mol; (16)Boiling Point: 140.567 °C at 760 mmHg; (17)Vapour Pressure: 7.613 mmHg at 25 °C.
Preparation of 1,3-Dimethylbenzene :
It can be prepared by sulfonated hydrolysis. Firstly, mixed xylene is distillated. Then the fraction of 130-140 °C is collected and sulfonated with sulfuric acid at the temperature of 80-90 °C for 3.5 hours. At last, the product is obtained by collecting fraction of 140-150 °C after hydrolysis and distillation.
Uses of 1,3-Dimethylbenzene :
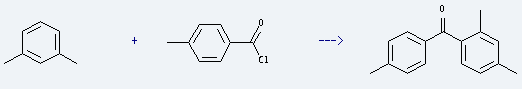
It can be used for the production of isophthalic acid, methyl benzoate, isophthaloyl nitrile, and also can be used as raw material of pharmaceutical, dye, spice and color film purity. It is used to produce 2,4,4'-trimethyl-benzophenone by reaction with 4-methyl-benzoyl chloride. The reaction needs reagent AlCl3 and solvent CH2Cl2. The yield is about 66%.
Safety Information of 1,3-Dimethylbenzene:
When you are using 1,3-Dimethylbenzene, please be cautious about it. As a chemical, it is irritating to eyes and skin. It has danger of very serious irreversible effects through inhalation, in contact with skin and if swallowed. Besides, it is highly flammable. During using it, wear suitable protective clothing and gloves. Keep container tightly closed and away from sources of ignition. Avoid contact with eyes. In case of accident or if you feel unwell seek medical advice immediately.
You can still convert the following datas into molecular structure:
1. Canonical SMILES: CC1=CC(=CC=C1)C
2. InChI: InChI=1S/C8H10/c1-7-4-3-5-8(2)6-7/h3-6H,1-2H3
3. InChIKey: IVSZLXZYQVIEFR-UHFFFAOYSA-N
The toxicity data of 1,3-Dimethylbenzene is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mammal (species unspecified) | LDLo | intraperitoneal | 2gm/kg (2000mg/kg) | Journal of Pathology and Bacteriology. Vol. 46, Pg. 95, 1938. | |
| mammal (species unspecified) | LDLo | subcutaneous | 5gm/kg (5000mg/kg) | Journal of Pathology and Bacteriology. Vol. 46, Pg. 95, 1938. | |
| man | TCLo | inhalation | 424mg/m3/6H/6 (424mg/m3) | BEHAVIORAL: CHANGES IN MOTOR ACTIVITY (SPECIFIC ASSAY) | Toxicology Letters. Vol. 1000(Sp, |
| man | TCLo | inhalation | 870mg/m3/4H-I (870mg/m3) | BEHAVIORAL: ATAXIA BEHAVIORAL: IRRITABILITY | Archives of Toxicology, Supplement. Vol. 7, Pg. 412, 1984. |
| mouse | LCLo | inhalation | 2010ppm/24H (2010ppm) | Journal of Pathology and Bacteriology. Vol. 46, Pg. 95, 1938. | |
| mouse | LD50 | intraperitoneal | 2003uL/kg (2.003mL/kg) | Archives of Toxicology. Vol. 58, Pg. 106, 1985. | |
| rabbit | LD50 | skin | 14100uL/kg (14.1mL/kg) | American Industrial Hygiene Association Journal. Vol. 23, Pg. 95, 1962. | |
| rat | LCLo | inhalation | 8000ppm/4H (8000ppm) | American Industrial Hygiene Association Journal. Vol. 23, Pg. 95, 1962. | |
| rat | LD50 | oral | 5gm/kg (5000mg/kg) | Gekkan Yakuji. Pharmaceuticals Monthly. Vol. 22, Pg. 883, 1980. |
Related Products
- 10,10'-Bis([1,1'-biphenyl]-4-yl)-9,9'-bianthracene
- 10,10'-Dibromo-9,9'-bianthryl
- 10,10-Dimethylanthrone
- 10,10-Oxybisphenoxarsine
- 10-[1,1'-Biphenyl]-4-yl-2-(1-methylethyl)-9-oxo-9H-thioxanthenium hexafluorophosphate
- 10,11-Dehydroimipramine
- 10,11-Dihydro-11-oxodibenzo[b,f][1,4]thiazepine
- 10,11-Dihydro-2-(4-methyl-1-piperazinyl)-11-(2-athiazolyl)-pyridazino(3,4-b)(1,4)benzoxazepine
- 10,11-DIHYDRO-2-(4-METHYL-1-PIPERAZINYL)-11-(3,4-XYLYL)PYRIDAZINO(3,4b)(1,4)-BENZOXAZEPINE MALEATE
- 10,11-Dihydro-5-(3-dimethylamino-2-methylpropyl)-5h-dibenz (b,f)azepine
- 108-39-4
- 108-40-7
- 108412-04-0
- 108-41-8
- 108-42-9
- 108-43-0
- 1084334-28-0
- 108433-95-0
- 108433-99-4
- 108436-80-2
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View