-
Name
1,2-Cyclohexanedione
- EINECS 212-155-3
- CAS No. 765-87-7
- Article Data130
- CAS DataBase
- Density 1.127 g/cm3
- Solubility soluble in water
- Melting Point 35 °C
- Formula C6H8O2
- Boiling Point 194 °C at 760 mmHg
- Molecular Weight 112.128
- Flash Point 66.9 °C
- Transport Information
- Appearance clear yellow liquid after melting
- Safety 24/25
- Risk Codes 22
-
Molecular Structure
-
Hazard Symbols
 Xi,
Xi, Xn
Xn
- Synonyms 1,2-Dioxocyclohexane;NSC 32950;NSC 627435;
- PSA 34.14000
- LogP 0.69860
Synthetic route

| Conditions | Yield |
|---|---|
| Stage #1: 2-methoxycyclohexanone With propyl cyanide at 47℃; for 0.833333h; Stage #2: In dimethyl sulfoxide for 1.33333h; Stage #3: With sodium nitrate; platinum(IV) chloride In dimethyl sulfoxide at 56℃; for 3h; Temperature; | 98.3% |

| Conditions | Yield |
|---|---|
| With sodium bromate; sodium hydrogensulfite In water; acetonitrile at 20℃; for 2.5h; Oxidation; | A 1% B 95% |
| With fluorine In acetonitrile at 20℃; Baeyer-Villiger oxidation; | A 30% B 53% |
-
-
1072-86-2, 1460-57-7, 1792-81-0, 54383-22-1, 57794-08-8, 931-17-9
(1R,2R)-trans-1,2-cyclohexanediol

-
-
765-87-7
cyclohexane-1,2-dione

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide In decane; acetonitrile at 65℃; for 2.5h; | 93% |
| With tert.-butylhydroperoxide; thiourea S,S-dioxide In dodecane; acetonitrile at 65℃; for 4.5h; | 90% |
| With tert.-butylhydroperoxide In decane; acetonitrile at 65℃; for 0.5h; | 78% |

| Conditions | Yield |
|---|---|
| With N-methylpyrrolidine-2-one hydrotribromide; dihydrogen peroxide In acetonitrile for 0.5h; Heating; | 91% |
| With tert.-butylhydroperoxide; iron(III) chloride; thymidyl acetic acid In decane; acetonitrile at 80℃; for 16h; | 80% |
| With tetraphenylphosphonium monoperoxysulfate; 2-Iodobenzoic acid In 1,2-dichloro-ethane at 80℃; for 12h; | 79% |
-
-
63703-34-4
2,2-dimethoxycyclohexanol

-
A
-
38461-13-1
2,2-dimethoxycyclohexanone

-
B
-
765-87-7
cyclohexane-1,2-dione

| Conditions | Yield |
|---|---|
| With silver | A n/a B 88.5% |

| Conditions | Yield |
|---|---|
| With iron(III) chloride; dimethyl sulfoxide at 100℃; for 3h; | 85% |
| With iodine In water; acetonitrile at 40℃; for 2h; Solvent; Temperature; | 81% |
| With 2,2,6,6-tetramethylpiperidin-1-oxoammonium chloride; toluene-4-sulfonic acid | 75% |

| Conditions | Yield |
|---|---|
| With silica gel; copper(II) nitrate In tetrachloromethane at 25℃; for 0.833333h; | 85% |
| With [(hydrotris(3,5-diphenyl-pyrazol-1-yl)borate)FeII(benzilate)]; oxygen In benzene for 0.333333h; |

| Conditions | Yield |
|---|---|
| With oxygen In benzene at 10℃; for 1h; Kinetics; Reagent/catalyst; | A 85% B 13% |
-
-
85969-41-1
3-Chloro-N-(2-hydroxy-phenyl)-N-(6-oxo-cyclohex-1-enyl)-benzamide

-
A
-
78154-49-1
o-phenol

-
B
-
765-87-7
cyclohexane-1,2-dione

| Conditions | Yield |
|---|---|
| With silica gel In dichloromethane for 2.5h; Ambient temperature; | A 82% B 77% |

| Conditions | Yield |
|---|---|
| With 1-hydroxy-3H-benz[d][1,2]iodoxole-1,3-dione In dimethyl sulfoxide for 3.5h; Ambient temperature; | 81% |
-
-
52190-35-9
2-(methylthio)cyclohexanone

-
-
765-87-7
cyclohexane-1,2-dione

| Conditions | Yield |
|---|---|
| With CuCl2-CuO In water; acetone at 35℃; for 0.5h; | 80% |
| With copper(II) oxide; copper dichloride In water; acetone at 35℃; for 0.5h; | 80% |

-
-
765-87-7
cyclohexane-1,2-dione

| Conditions | Yield |
|---|---|
| With 1-hydroxy-3H-benz[d][1,2]iodoxole-1,3-dione In dimethyl sulfoxide for 5h; Ambient temperature; | 78% |
-
-
79532-62-0
1-(1-Ethoxy-2-oxocyclohexyl)-4-phenyl-1,2,4-triazolidin-3,5-dion

-
A
-
15988-11-1
4-Phenylurazole

-
B
-
765-87-7
cyclohexane-1,2-dione

| Conditions | Yield |
|---|---|
| With hydrogenchloride; water In 1,4-dioxane for 1.5h; Heating; | A 75% B 75% |
-
-
931-17-9
1,2-Cyclohexanediol

-
A
-
124-04-9
Adipic acid

-
B
-
765-87-7
cyclohexane-1,2-dione

-
C
-
533-60-8
cyclohexanone-2-ol

| Conditions | Yield |
|---|---|
| With sodium bromate; sodium hydrogensulfite In dichloromethane; water; acetonitrile at 20℃; for 3h; Oxidation; | A 13% B 74% C 8% |
| With sodium bromate; sodium hydrogensulfite In water; acetonitrile at 20℃; for 2h; Oxidation; | A 18% B 19% C 51% |
| With 2O34W9Zn(12-)*W(6+)*3Zn(2+)*2H2O; dihydrogen peroxide In water; acetonitrile at 135℃; under 2250.23 Torr; for 0.25h; Microwave irradiation; |


-
-
765-87-7
cyclohexane-1,2-dione

| Conditions | Yield |
|---|---|
| With 1,1'-carbonyldiimidazole In dichloromethane for 5h; Ambient temperature; | 74% |

| Conditions | Yield |
|---|---|
| With oxygen; copper(II) bis(trifluoromethanesulfonate); dimethyl sulfoxide; bismuth at 100℃; for 2h; | 74% |

| Conditions | Yield |
|---|---|
| With pyridine; N-Bromosuccinimide In tetrachloromethane for 1.5h; Heating; | 74% |
-
-
24858-28-4
cyclohexane-1,2-dione monooxime

-
-
765-87-7
cyclohexane-1,2-dione

| Conditions | Yield |
|---|---|
| With silica gel; copper(II) nitrate In tetrachloromethane for 0.833333h; Heating; | 72% |
-
-
1056477-06-5
2-bromocyclohexanone

-
-
765-87-7
cyclohexane-1,2-dione

| Conditions | Yield |
|---|---|
| With potassium fluoride; dimethyl sulfoxide at 85℃; for 5h; | 65% |
-
-
158-97-4
1,4,7,10-tetrathia-dispiro[4.0.4.4]tetradecane

-
A
-
27694-08-2
6-oxospiro

-
B
-
765-87-7
cyclohexane-1,2-dione

| Conditions | Yield |
|---|---|
| With water; 1-fluoro-2,4,6-trimethylpyridinium trifluoromethanesulfonate In tetrahydrofuran; dichloromethane at 23℃; | A 65% B 15% |
-
-
1792-81-0
cis-1,2-cyclohexane

-
A
-
1072-21-5
hexanedial

-
B
-
765-87-7
cyclohexane-1,2-dione

-
C
-
533-60-8
cyclohexanone-2-ol

| Conditions | Yield |
|---|---|
| With oxygen; [(n-Bu)4N]3H2[IMo6O24] In acetonitrile at 80℃; under 1520.1 Torr; for 5h; | A 5% B 31% C 64% |


| Conditions | Yield |
|---|---|
| With sulfuric acid; silica gel In dichloromethane for 24h; Ambient temperature; | 63% |
-
-
67-56-1
methanol

-
-
3242-56-6
2-diazocyclohexanone

-
A
-
627-93-0
hexanedioic acid dimethyl ester

-
B
-
765-87-7
cyclohexane-1,2-dione

| Conditions | Yield |
|---|---|
| With oxygen; methylene blue Irradiation; | A 60% B 25% |

-
-
3242-56-6
2-diazocyclohexanone

-
A
-
627-93-0
hexanedioic acid dimethyl ester

-
B
-
765-87-7
cyclohexane-1,2-dione

| Conditions | Yield |
|---|---|
| With oxygen; methylene blue In methanol Irradiation; | A 60% B 25% |
-
-
1460-57-7
trans-1,2-cyclohexandiol

-
A
-
1072-21-5
hexanedial

-
B
-
765-87-7
cyclohexane-1,2-dione

-
C
-
533-60-8
cyclohexanone-2-ol

| Conditions | Yield |
|---|---|
| With oxygen; [(n-Bu)4N]3H2[IMo6O24] In acetonitrile at 80℃; under 1520.1 Torr; for 5h; | A 3% B 56% C 41% |


| Conditions | Yield |
|---|---|
| With oxygen In acetonitrile at 25℃; for 6h; Kinetics; | A 55% B 38% |
-
-
533-60-8
cyclohexanone-2-ol

-
A
-
124-04-9
Adipic acid

-
B
-
2035-75-8
adipic anhydride

-
C
-
765-87-7
cyclohexane-1,2-dione

| Conditions | Yield |
|---|---|
| With air; chromium(III)naphthenate In chlorobenzene at 50℃; Product distribution; var. catalyst; | A 54% B 43% C 3% |


-
-
765-87-7
cyclohexane-1,2-dione

| Conditions | Yield |
|---|---|
| Stage #1: trans-cyclohexane-1,2-diol; platinum at 90℃; for 0.166667h; Stage #2: With dihydrogen peroxide In water at 90℃; for 20h; Product distribution / selectivity; | 51% |
-
-
42738-99-8
11-hydroxytetrahydrocarbazolenine

-
-
937-14-4
3-chloro-benzenecarboperoxoic acid

-
A
-
78154-49-1
o-phenol

-
B
-
765-87-7
cyclohexane-1,2-dione

-
C
-
85969-41-1
3-Chloro-N-(2-hydroxy-phenyl)-N-(6-oxo-cyclohex-1-enyl)-benzamide

-
D
-
4669-18-5
spiro[cyclopentane-1,2'-indolin]-3'-one

| Conditions | Yield |
|---|---|
| In dichloromethane at -60℃; for 1h; | A 42% B 24% C 24% D 8% |

-
-
42738-99-8
11-hydroxytetrahydrocarbazolenine

-
A
-
78154-49-1
o-phenol

-
B
-
765-87-7
cyclohexane-1,2-dione

-
C
-
85969-41-1
3-Chloro-N-(2-hydroxy-phenyl)-N-(6-oxo-cyclohex-1-enyl)-benzamide

-
D
-
4669-18-5
spiro[cyclopentane-1,2'-indolin]-3'-one

| Conditions | Yield |
|---|---|
| With 3-chloro-benzenecarboperoxoic acid In dichloromethane at -60℃; for 1h; | A 42% B 24% C 24% D 8% |

-
-
18162-48-6
tert-butyldimethylsilyl chloride

-
-
765-87-7
cyclohexane-1,2-dione

-
-
116608-34-5
2-{(tert-butyldimethylsilyl)oxy}cyclohex-2-en-1-one

| Conditions | Yield |
|---|---|
| With 1H-imidazole In dichloromethane at 20℃; for 0.5h; | 99% |
| With 1H-imidazole In dichloromethane at 23℃; for 24h; Inert atmosphere; | 98% |
| With 1H-imidazole In dichloromethane at 20℃; | 95% |
-
-
765-87-7
cyclohexane-1,2-dione

| Conditions | Yield |
|---|---|
| With [15N]hydroxylamine hydrochloride; potassium carbonate In water at 20℃; for 24h; | 99% |
-
-
1826-67-1
vinyl magnesium bromide

-
-
765-87-7
cyclohexane-1,2-dione

-
-
27332-53-2
2‐hydroxy‐2‐vinylcyclohexan‐1‐one

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 0℃; | 98% |
| In tetrahydrofuran at 20℃; Cooling with ice; | 85% |
| In tetrahydrofuran | |
| In tetrahydrofuran at 0 - 20℃; Schlenk technique; Inert atmosphere; |

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid Reflux; | 98% |
| With toluene-4-sulfonic acid In toluene for 48h; Reflux; | 69% |
| With toluene-4-sulfonic acid In toluene for 48h; Reflux; | 66% |
-
-
5153-69-5, 706-08-1
4-fluoro-β-nitrostyrene

-
-
765-87-7
cyclohexane-1,2-dione

-
-
1382997-49-0
(1S,5S,6R,7S)-6-(4-fluorophenyl)-1-hydroxy-7-nitrobicyclo[3.2.1]octan-8-one

| Conditions | Yield |
|---|---|
| With nickel diacetate; triethylamine; (1R,2R)-N,N'-di(4'-bromobenzyl)-1,2-diaminocyclohexane In tetrahydrofuran at 20℃; Michael-Henry reaction; optical yield given as %ee; enantioselective reaction; | 98% |
-
-
765-87-7
cyclohexane-1,2-dione

-
-
16452-01-0
ortho-cresidine

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In toluene at 130℃; for 4.5h; Sealed tube; Molecular sieve; Microwave irradiation; | 98% |

| Conditions | Yield |
|---|---|
| With sodium chloride; zinc In methanol; water Heating; | 97% |
| With ammonium chloride; zinc In tetrahydrofuran at 20℃; for 0.75h; | 92% |
| With trimethylphosphane In tetrahydrofuran at 20℃; for 2h; | 80% |
-
-
765-87-7
cyclohexane-1,2-dione

-
-
1439-36-7
1-triphenylphosphoranylidene-2-propanone

-
-
33553-25-2
2-(2-oxopropyl)cyclohex-2-enone

| Conditions | Yield |
|---|---|
| In dichloromethane for 25h; Wittig-Horner reaction; Heating; | 97% |
| at 130 - 140℃; |

| Conditions | Yield |
|---|---|
| With ozone In tetrachloromethane; water at 20℃; for 1h; UV-irradiation; | 97% |
| With water; ozone at 25℃; under 760.051 Torr; for 1h; Irradiation; | 95% |
| With oxygen; sodium hydroxide In water at 90℃; under 3000.3 Torr; for 3h; pH=7; | 21% |
-
-
622-88-8
4-bromophenylhydrazine hydrochloride

-
-
765-87-7
cyclohexane-1,2-dione

-
-
108606-59-3
(E)-2-(2-(4-bromophenyl)hydrazono)cyclohexan-1-one

| Conditions | Yield |
|---|---|
| In water at 20℃; for 16h; | 97% |

| Conditions | Yield |
|---|---|
| With sulfuric acid In ethanol at 65℃; for 4.08333h; | 96.3% |
-
-
593-56-6
N-methoxylamine hydrochloride

-
-
765-87-7
cyclohexane-1,2-dione

-
-
1312566-43-0
cyclohexane-1,2-dione-bis-O-methyldioxime

| Conditions | Yield |
|---|---|
| With pyridine In methanol | 96% |

| Conditions | Yield |
|---|---|
| With indium(III) chloride; 1-butyl-3-methylimidazolium Tetrafluoroborate at 100℃; for 2h; | 96% |
| With sulfuric acid; silica gel In methanol for 2h; Friedlaender Quinoline Synthesis; Reflux; | 93% |
| With eaton’s reagent In neat (no solvent) at 90℃; for 3h; | 86% |

| Conditions | Yield |
|---|---|
| In water; toluene | 95.34% |
| In toluene for 5h; Inert atmosphere; Dean-Stark trap; Reflux; | 95.34% |
| In toluene for 5h; Inert atmosphere; Reflux; | 95.34% |
-
-
18306-29-1
bis(trimethylsilyl)sulphate

-
-
765-87-7
cyclohexane-1,2-dione

-
-
6838-67-1
1,2-bis(trimethylsilyloxy)cyclohex-1-ene

| Conditions | Yield |
|---|---|
| With calcium In toluene at 100℃; for 2h; Solvent; Reagent/catalyst; | 95.18% |

-
-
765-87-7
cyclohexane-1,2-dione

| Conditions | Yield |
|---|---|
| With calcium In toluene at 125℃; for 2h; Solvent; Reagent/catalyst; | 95.1% |

| Conditions | Yield |
|---|---|
| In dichloromethane for 1h; Ambient temperature; | 95% |

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; for 1h; | 95% |
-
-
5291-08-7
N-(2-methyl-1-oximino-1-phenyl-2-propyl)hydroxylamine

-
-
765-87-7
cyclohexane-1,2-dione

-
-
82811-27-6
5,5-dimethyl-1-hydroxy-2-(spirocyclohexan-2-one 2)-4-phenyl-3-imidazoline 3-oxide

| Conditions | Yield |
|---|---|
| In methanol for 6h; Heating; | 95% |

| Conditions | Yield |
|---|---|
| With acetic acid In ethanol; acetonitrile at 0 - 5℃; for 2h; Condensation; | 95% |
| In ethanol at 0℃; for 3h; | 80% |

-
-
765-87-7
cyclohexane-1,2-dione

| Conditions | Yield |
|---|---|
| With borate buffer at 37℃; for 18h; pH=9.0; | 95% |
-
-
104-94-9
4-methoxy-aniline

-
-
765-87-7
cyclohexane-1,2-dione

-
-
105553-63-7
2-[(4-methoxyphenyl)amino]cyclohex-2-en-1-one

| Conditions | Yield |
|---|---|
| In toluene for 12h; Inert atmosphere; Reflux; | 95% |
| With toluene-4-sulfonic acid In toluene at 130℃; for 4.5h; Sealed tube; Molecular sieve; Microwave irradiation; | 72% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 20℃; for 48h; Solvent; Concentration; regioselective reaction; | 95% |


| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In toluene at 130℃; for 4.5h; Sealed tube; Molecular sieve; Microwave irradiation; | 95% |

| Conditions | Yield |
|---|---|
| With hydroxylamine hydrochloride; silica gel at 50℃; for 3h; Temperature; Green chemistry; | 94% |
| With hydroxylamine hydrochloride; potassium hydroxide In ethanol; water at 0 - 20℃; for 48h; | 87% |
| With hydroxylamine | |
| With potassium hydroxide; hydroxylamine hydrochloride |
-
-
765-87-7
cyclohexane-1,2-dione

-
-
762-21-0
acetylenedicarboxylic acid diethyl ester

-
-
109-77-3
malononitrile

-
-
1325716-33-3
ethyl 2-amino-3-cyano-4-(2-ethoxy-2-oxoethyl)-5-oxo-5,6,7,8-tetrahydro-4H-chromene-4-carboxylate

| Conditions | Yield |
|---|---|
| With copper(II) ferrite In water at 60℃; for 2h; | 94% |


| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In toluene at 130℃; for 4.5h; Sealed tube; Molecular sieve; Microwave irradiation; | 94% |
1,2-Cyclohexanedione Chemical Properties
Chemical Name: 1,2-Cyclohexanedione
IUPAC NAME: Cyclohexane-1,2-dione
CAS No.: 765-87-7
EINECS: 212-155-3
RTECS: GV0340000
Molecular Formula: C6H8O2
Molecular Weight: 112.13 g/mol
Melting Point: 35 °C
Density: 1.127 g/cm3
Flash Point: 66.9 °C
Boiling Point: 194 °C at 760 mmHg
Storage temp.: 0-6°C
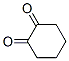
Following is the structure of 1,2-Cyclohexanedione (765-87-7):
Product Categories about 1,2-Cyclohexanedione (765-87-7) are Ketone ; Pharmaceutical intermediate ; C3 to C6Protein Modification ; Reagents for arginine modification ; Carbonyl Compounds ; Ketones ; Specific Amino Acid Modification
The chemical synonymous of 1,2-Cyclohexanedione (765-87-7) are 1,2-Cyclohexadione ; 1,2-Dioxocyclohexane ; Cyclohexane-1,2-dione ; Dihydrocatechol ; 1,2-Cyclohexandione ; 1,2-Cyclohexanedione ; 2-Hydroxy-2-cyclohexen-1-one ; 2-Hydroxy-cyclohex-2-enone
1,2-Cyclohexanedione Uses
1,2-Cyclohexanedione (765-87-7) is mainly used in organic synthesis.
1,2-Cyclohexanedione Toxicity Data With Reference
| 1. | unr-mus LD50:440 mg/kg | PCJOAU Pharmaceutical Chemistry Journal (English Translation). Translation of KHFZAN. 12 (1978),227. |
1,2-Cyclohexanedione Consensus Reports
Reported in EPA TSCA Inventory.
1,2-Cyclohexanedione Safety Profile
Moderately toxic by unspecified route. When heated to decomposition it emits acrid smoke and irritating vapors.
Hazard Note: Irritant
Hazard Codes:
Xn: Harmful ![]()
Xi: Irritant ![]()
Risk Statements about 1,2-Cyclohexanedione (765-87-7):
R22 Harmful if swallowed.
Safety Statements about 1,2-Cyclohexanedione (765-87-7):
S24/25: Avoid contact with skin and eyes.
Attentions:
1. Storage: Keep away from sources of ignition. Store in a tightly closed container. Store in a dry area. Keep refrigerated. (Store below 4°C/39°F.) Store under an inert atmosphere.
2. Handling: Avoid breathing dust, vapor, mist, or gas. Avoid contact with skin and eyes. Avoid ingestion and inhalation.
Related Products
- 10,10'-Bis([1,1'-biphenyl]-4-yl)-9,9'-bianthracene
- 10,10'-Dibromo-9,9'-bianthryl
- 10,10-Dimethylanthrone
- 10,10-Oxybisphenoxarsine
- 10-[1,1'-Biphenyl]-4-yl-2-(1-methylethyl)-9-oxo-9H-thioxanthenium hexafluorophosphate
- 10,11-Dehydroimipramine
- 10,11-Dihydro-11-oxodibenzo[b,f][1,4]thiazepine
- 10,11-Dihydro-2-(4-methyl-1-piperazinyl)-11-(2-athiazolyl)-pyridazino(3,4-b)(1,4)benzoxazepine
- 10,11-DIHYDRO-2-(4-METHYL-1-PIPERAZINYL)-11-(3,4-XYLYL)PYRIDAZINO(3,4b)(1,4)-BENZOXAZEPINE MALEATE
- 10,11-Dihydro-5-(3-dimethylamino-2-methylpropyl)-5h-dibenz (b,f)azepine
- 765877-97-2
- 7658-80-2
- 765895-65-6
- 765908-38-1
- 765910-73-4
- 76594-49-5
- 765945-04-8
- 765948-78-5
- 76-59-5
- 7659-86-1
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View