-
Name
2-ETHYL-2-METHYL-1,3-PROPANEDIOL
- EINECS 201-062-3
- CAS No. 77-84-9
- Article Data14
- CAS DataBase
- Density 0.963 g/cm3
- Solubility almost transparency
- Melting Point 43 ºC
- Formula C6H14O2
- Boiling Point 225.8 °C at 760 mmHg
- Molecular Weight 118.176
- Flash Point 96.1 °C
- Transport Information
- Appearance
- Safety 24/25
- Risk Codes
-
Molecular Structure
- Hazard Symbols
- Synonyms 2-Methyl-2-ethyl-1,3-propanediol;
- PSA 40.46000
- LogP 0.38730
Synthetic route

| Conditions | Yield |
|---|---|
| With lithium aluminium tetrahydride In diethyl ether at 20℃; for 4h; | 94% |
| With lithium aluminium tetrahydride; diethyl ether | |
| With lithium aluminium tetrahydride In diethyl ether for 3h; Heating; |
-
-
123-91-1
1,4-dioxane

-
-
922-63-4
ethylacrolein

-
A
-
137-32-6
(+/-)-2-methyl-1-butanol

-
B
-
77-84-9
2-ethyl-2-methyl-1,3-propanediol

| Conditions | Yield |
|---|---|
| With formaldehyd; hydroquinone | A 61.5% B n/a |


| Conditions | Yield |
|---|---|
| With formaldehyd; hydroquinone In 1,4-dioxane; ethanol | A 57.5% B n/a |
-
-
123-91-1
1,4-dioxane

-
-
922-63-4
ethylacrolein

-
A
-
6032-29-7
(+/-)-2-pentanol

-
B
-
77-84-9
2-ethyl-2-methyl-1,3-propanediol

| Conditions | Yield |
|---|---|
| With formaldehyd; hydroquinone | A 55.9% B n/a |

-
-
1629-58-9
4-penten-3-one

-
A
-
137-32-6
(+/-)-2-methyl-1-butanol

-
B
-
77-84-9
2-ethyl-2-methyl-1,3-propanediol

| Conditions | Yield |
|---|---|
| With sodium hydroxide; formaldehyd; hydroquinone In 1,4-dioxane; ethanol | A 55.7% B n/a |
-
-
50-00-0
formaldehyd

-
-
96-17-3, 57456-98-1
2-Methylbutyraldehyde

-
-
77-84-9
2-ethyl-2-methyl-1,3-propanediol

| Conditions | Yield |
|---|---|
| With potassium carbonate | |
| With potassium hydroxide; water |

| Conditions | Yield |
|---|---|
| With potassium hydroxide |
-
-
96-17-3, 57456-98-1
2-Methylbutyraldehyde

-
-
77-84-9
2-ethyl-2-methyl-1,3-propanediol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: K2CO3 2: alcoholic KOH-solution View Scheme |

| Conditions | Yield |
|---|---|
| In diethyl ether |
-
-
1445-45-0
Trimethyl orthoacetate

-
-
77-84-9
2-ethyl-2-methyl-1,3-propanediol

-
-
656241-03-1
2-acetoxymethyl-2-methylbutanol

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In dichloromethane at 20℃; for 1h; | 91% |
-
-
77-84-9
2-ethyl-2-methyl-1,3-propanediol

-
-
10312-83-1
methoxyacetaldehyde

| Conditions | Yield |
|---|---|
| With pyridinium p-toluenesulfonate In chloroform | 89% |
-
-
77-84-9
2-ethyl-2-methyl-1,3-propanediol

-
-
31643-49-9
4-Nitrophthalonitrile

-
A
-
130326-30-6
4-(2-(hydroxymethyl)-2-methylbutoxy)benzene-1,2-dicarbonitrile

-
B
-
93673-02-0
1,3-bis(3',4'-dicyanophenyl)-2-ethyl-2-methylpropane

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide for 48h; Ambient temperature; Yields of byproduct given; | A 86% B n/a |
| With potassium carbonate In N,N-dimethyl-formamide for 48h; Ambient temperature; Yield given. Yields of byproduct given; |

-
-
77-84-9
2-ethyl-2-methyl-1,3-propanediol

-
-
57648-76-7
N-(1,2-dimethylethenylenedioxyphosphoryl)imidazole

| Conditions | Yield |
|---|---|
| In dichloromethane at 25℃; for 1h; | 85% |

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In benzene Heating; | 80% |

| Conditions | Yield |
|---|---|
| In dichloromethane byproducts: HCl; (Ar or N2); to a soln. of 2-ethyl-2-methyl-1,3-propanediol added a soln. of VOCl3 and stirred for 30 min; hexane added; evapd.; filtered; recrystd. from CH2Cl2/pentane; elem. anal.; | 79% |

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In benzene Heating; | 72% |

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In benzene Heating; | 70% |

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In benzene Heating; | 62% |

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In benzene Heating; | 60% |

| Conditions | Yield |
|---|---|
| With antimonypentachloride In dichloromethane for 0.05h; Heating; | 40% |
-
-
77-84-9
2-ethyl-2-methyl-1,3-propanediol

-
-
31643-49-9
4-Nitrophthalonitrile

-
A
-
30757-50-7
4-hydroxy-1,2-benzenedicarbonitrile

-
B
-
93673-02-0
1,3-bis(3',4'-dicyanophenyl)-2-ethyl-2-methylpropane

| Conditions | Yield |
|---|---|
| With potassium carbonate In dimethyl sulfoxide | A 560 mg B 37% |

-
-
50-00-0
formaldehyd

-
-
77-84-9
2-ethyl-2-methyl-1,3-propanediol

-
-
13432-74-1
5-ethyl-5-methyl-[1,3]dioxane

| Conditions | Yield |
|---|---|
| With hydrogenchloride |
-
-
77-84-9
2-ethyl-2-methyl-1,3-propanediol

-
-
500541-53-7
2,2-bis-bromomethyl-butane

| Conditions | Yield |
|---|---|
| With phosphorus tribromide | |
| With phosphorus tribromide |
-
-
77-84-9
2-ethyl-2-methyl-1,3-propanediol

-
-
40894-01-7
2-bromomethyl-2-methyl-butan-1-ol

| Conditions | Yield |
|---|---|
| With hydrogen bromide at 100℃; |

| Conditions | Yield |
|---|---|
| With potassium permanganate |
-
-
77-84-9
2-ethyl-2-methyl-1,3-propanediol

-
-
3124-54-7
4-ethyl-4-methyl-2,6-dioxa-heptanedioic acid diamide

| Conditions | Yield |
|---|---|
| /BRN= 1803039/; |

| Conditions | Yield |
|---|---|
| With alkaline aqueous potassium permanganate |
-
-
77-84-9
2-ethyl-2-methyl-1,3-propanediol

-
-
75-07-0
acetaldehyde

-
-
39087-06-4
5-ethyl-2,5-dimethyl-[1,3]dioxane

| Conditions | Yield |
|---|---|
| With hydrogenchloride |

| Conditions | Yield |
|---|---|
| With hydrogen bromide at 100℃; |
-
-
77-84-9
2-ethyl-2-methyl-1,3-propanediol

| Conditions | Yield |
|---|---|
| With triphenyl phosphite; methyl iodide Heating; |
-
-
77-84-9
2-ethyl-2-methyl-1,3-propanediol

-
-
1885-14-9
phenyl chloroformate

-
-
3124-54-7
4-ethyl-4-methyl-2,6-dioxa-heptanedioic acid diamide

| Conditions | Yield |
|---|---|
| (i) Py, (ii) liq. NH3; Multistep reaction; |

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In benzene Heating; Yield given. Yields of byproduct given. Title compound not separated from byproducts; |

1,3-Propanediol,2-ethyl-2-methyl- Specification
The 1,3-Propanediol,2-ethyl-2-methyl-, with the CAS registry number 77-84-9, is also known as 2-Methyl-2-ethyl-1,3-propanediol. Its EINECS registry number is 201-062-3. This chemical's molecular formula is C6H14O2 and molecular weight is 118.1742. What's more, both its IUPAC name and systematic name are the same which is 2-Ethyl-2-methylpropane-1,3-diol.
Physical properties about 1,3-Propanediol,2-ethyl-2-methyl- are: (1)ACD/LogP: 0.19; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 0.19; (4)ACD/LogD (pH 7.4): 0.19; (5)ACD/BCF (pH 5.5): 1; (6)ACD/BCF (pH 7.4): 1; (7)ACD/KOC (pH 5.5): 30.17; (8)ACD/KOC (pH 7.4): 30.17; (9)#H bond acceptors: 2; (10)#H bond donors: 2; (11)#Freely Rotating Bonds: 5; (12)Polar Surface Area: 18.46 Å2; (13)Index of Refraction: 1.448; (14)Molar Refractivity: 32.88 cm3; (15)Molar Volume: 122.6 cm3; (16)Polarizability: 13.03×10-24 cm3; (17)Surface Tension: 35.5 dyne/cm; (18)Density: 0.963 g/cm3; (19)Flash Point: 96.1 °C; (20)Enthalpy of Vaporization: 53.75 kJ/mol; (21)Boiling Point: 225.8 °C at 760 mmHg; (22)Vapour Pressure: 0.0168 mmHg at 25 °C.
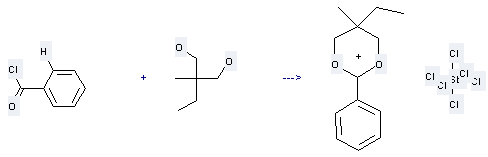
Use of 1,3-Propanediol,2-ethyl-2-methyl-: it is used to produce other chemicals. For example, it is used to produce 2-Phenyl-5-methyl-5-ethyl-1,3-dioxanium hexachloroantimonate. The reaction occurs with reagent SbCl5 and solvent CH2Cl2. This reaction will occur at the condition of heating for 3 mins. The yield is 40%.
When you are dealing with this chemical, you should be very careful. You must avoid contacting with skin and eyes.
You can still convert the following datas into molecular structure:
(1) SMILES: OCC(C)(CC)CO
(2) InChI: InChI=1/C6H14O2/c1-3-6(2,4-7)5-8/h7-8H,3-5H2,1-2H3
(3) InChIKey: VNAWKNVDKFZFSU-UHFFFAOYAD
Related Products
- 10,10'-Bis([1,1'-biphenyl]-4-yl)-9,9'-bianthracene
- 10,10'-Dibromo-9,9'-bianthryl
- 10,10-Dimethylanthrone
- 10,10-Oxybisphenoxarsine
- 10-[1,1'-Biphenyl]-4-yl-2-(1-methylethyl)-9-oxo-9H-thioxanthenium hexafluorophosphate
- 10,11-Dehydroimipramine
- 10,11-Dihydro-11-oxodibenzo[b,f][1,4]thiazepine
- 10,11-Dihydro-2-(4-methyl-1-piperazinyl)-11-(2-athiazolyl)-pyridazino(3,4-b)(1,4)benzoxazepine
- 10,11-DIHYDRO-2-(4-METHYL-1-PIPERAZINYL)-11-(3,4-XYLYL)PYRIDAZINO(3,4b)(1,4)-BENZOXAZEPINE MALEATE
- 10,11-Dihydro-5-(3-dimethylamino-2-methylpropyl)-5h-dibenz (b,f)azepine
- 77-85-0
- 7785-20-8
- 7785-21-9
- 7785-23-1
- 7785-26-4
- 7785-33-3
- 77855-73-3
- 778-56-3
- 7785-70-8
- 778571-57-6
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View