-
Name
1-Naphthylboronic acid
- EINECS 604-119-4
- CAS No. 13922-41-3
- Article Data65
- CAS DataBase
- Density 1.21 g/cm3
- Solubility 2.5 g/100 mL in water
- Melting Point 208-214 °C(lit.)
- Formula C10H9BO2
- Boiling Point 381.9 °C at 760 mmHg
- Molecular Weight 171.991
- Flash Point 184.8 °C
- Transport Information
- Appearance Off-white to pink beige powder
- Safety 26-36-37/39
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms NSC78936;a-Naphthaleneboronic acid;a-Naphthylboronic acid;1-Naphthylboric acid;1-Naphthaleneboronicacid (6CI,7CI,8CI);Boronic acid, 1-naphthalenyl- (9CI);(1-Naphthalenyl)boronic acid;
- PSA 40.46000
- LogP 0.51960
Synthetic route
-
-
90-11-9
1-Bromonaphthalene

-
-
13922-41-3
1-Naphthylboronic acid

| Conditions | Yield |
|---|---|
| Stage #1: 1-Bromonaphthalene With tert.-butyl lithium In tetrahydrofuran at 78℃; for 0.75h; Stage #2: With Trimethyl borate In tetrahydrofuran at -78 - 20℃; for 4h; Stage #3: With hydrogenchloride; water In tetrahydrofuran; ethyl acetate at 20℃; | 96% |
| Stage #1: 1-Bromonaphthalene With diisobutylaluminium hydride; magnesium; lithium chloride In tetrahydrofuran; toluene at 20℃; for 15h; Inert atmosphere; Stage #2: With Trimethyl borate In tetrahydrofuran; toluene at 0℃; Inert atmosphere; Stage #3: With hydrogenchloride; water In tetrahydrofuran; toluene at 0℃; Inert atmosphere; | 86% |
| Stage #1: 1-Bromonaphthalene With n-butyllithium In tetrahydrofuran at -78℃; for 1h; Inert atmosphere; Stage #2: With Trimethyl borate In tetrahydrofuran for 2h; Inert atmosphere; Stage #3: With hydrogenchloride In tetrahydrofuran; water | 74% |

| Conditions | Yield |
|---|---|
| Stage #1: 1-Bromonaphthalene With n-butyllithium In tetrahydrofuran at -78 - 0℃; for 1h; Inert atmosphere; Stage #2: Trimethyl borate In tetrahydrofuran at -78 - 20℃; for 12h; Inert atmosphere; Stage #3: With hydrogenchloride In tetrahydrofuran; water for 0.5h; Inert atmosphere; | 84% |
| With n-butyllithium In tetrahydrofuran N2; BuLi dropping into org. compd. soln. at -78°C, mixt. keeping 3 h, B-compd. soln. addn. dropwise at -78°C, mixt. allowing to warm to room temp. overnight , stirring 1 h with 10% HCl, extn. (Et2O); org. extracts washing (water), drying, solvent vac. removal; | |
| Stage #1: 1-Bromonaphthalene With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 2h; Schlenk technique; Stage #2: Trimethyl borate In tetrahydrofuran; hexane at 20℃; Schlenk technique; |

| Conditions | Yield |
|---|---|
| Stage #1: Triisopropyl borate; 1-Bromonaphthalene With n-butyllithium In tetrahydrofuran; hexane; toluene at -30 - 20℃; for 6h; Inert atmosphere; Stage #2: With hydrogenchloride In tetrahydrofuran; hexane; water; toluene at 20℃; for 1h; | 83% |
| With n-butyllithium In tetrahydrofuran; hexane; toluene at -30 - 20℃; for 6h; Inert atmosphere; | 83% |
| With n-butyllithium In tetrahydrofuran; hexane; toluene at -30 - 20℃; for 6h; Inert atmosphere; | 83% |
-
-
5419-55-6
Triisopropyl borate

-
-
90-11-9
1-Bromonaphthalene

-
-
7732-18-5
water

-
-
13922-41-3
1-Naphthylboronic acid

| Conditions | Yield |
|---|---|
| Stage #1: Triisopropyl borate; 1-Bromonaphthalene With n-butyllithium In tetrahydrofuran; hexane; toluene at -30 - 20℃; for 6h; Inert atmosphere; Stage #2: water With hydrogenchloride In tetrahydrofuran; hexane; toluene at 20℃; for 1h; | 83% |


| Conditions | Yield |
|---|---|
| Stage #1: tri-n-propyl borate; 1-Bromonaphthalene With n-butyllithium In tetrahydrofuran; hexane; toluene at -30 - 20℃; for 6h; Inert atmosphere; Stage #2: With hydrogenchloride In tetrahydrofuran; hexane; water; toluene at 20℃; for 1h; | 83% |
-
-
67-56-1
methanol

-
-
131765-96-3
dicyclohexylamine borane complex

-
-
90-11-9
1-Bromonaphthalene

-
-
13922-41-3
1-Naphthylboronic acid

| Conditions | Yield |
|---|---|
| Stage #1: dicyclohexylamine borane complex; 1-Bromonaphthalene With magnesium In tetrahydrofuran at 70℃; Stage #2: methanol In tetrahydrofuran at 0℃; for 1h; Stage #3: With hydrogenchloride; water In methanol at 20℃; for 1h; | 82% |

-
-
67-56-1
methanol

-
-
55124-35-1
diisopropylamine borane

-
-
90-11-9
1-Bromonaphthalene

-
-
13922-41-3
1-Naphthylboronic acid

| Conditions | Yield |
|---|---|
| Stage #1: diisopropylamine borane With magnesium; phenylmagnesium bromide In tetrahydrofuran at 20℃; for 0.166667h; Stage #2: 1-Bromonaphthalene In tetrahydrofuran at 70℃; Stage #3: methanol Further stages; | 80% |


| Conditions | Yield |
|---|---|
| Stage #1: diisopropopylaminoborane With iodine; magnesium at 25℃; Barbier Coupling Reaction; Reflux; Inert atmosphere; Stage #2: 1-Bromonaphthalene at 65℃; for 4.083h; Stage #3: With hydrogenchloride In water at 25 - 65℃; for 0.416667h; | A n/a B 79% |

-
-
90-11-9
1-Bromonaphthalene

-
-
22092-92-8
diisopropopylaminoborane

-
-
7732-18-5
water

-
-
13922-41-3
1-Naphthylboronic acid

| Conditions | Yield |
|---|---|
| Stage #1: 1-Bromonaphthalene; diisopropopylaminoborane With magnesium In tetrahydrofuran at 65℃; for 4h; Inert atmosphere; Stage #2: water With hydrogenchloride In tetrahydrofuran at 65℃; for 0.25h; Barbier Coupling Reaction; Inert atmosphere; | 79% |


| Conditions | Yield |
|---|---|
| With [1,1'-bis(diphenylphosphino)ferrocene]dichloropalladium(II); potassium acetate In toluene at 110 - 120℃; Inert atmosphere; | 76.3% |
-
-
121-43-7
Trimethyl borate

-
-
90-11-9
1-Bromonaphthalene

-
-
7732-18-5
water

-
-
13922-41-3
1-Naphthylboronic acid

| Conditions | Yield |
|---|---|
| Stage #1: 1-Bromonaphthalene With n-butyllithium In tetrahydrofuran at -78℃; for 0.5h; Inert atmosphere; Stage #2: Trimethyl borate In tetrahydrofuran at -78 - 20℃; for 0.166667h; Inert atmosphere; Stage #3: water With hydrogenchloride In tetrahydrofuran for 0.333333h; | 73.1% |

-
-
972-09-8
1-(tributylstannyl)naphthalene

-
-
13922-41-3
1-Naphthylboronic acid

| Conditions | Yield |
|---|---|
| With borane; water In tetrahydrofuran for 4h; Heating; | 68% |

| Conditions | Yield |
|---|---|
| Stage #1: 1-Bromonaphthalene With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 1h; Stage #2: triethyl borate In tetrahydrofuran; hexane at -78℃; for 6h; | 62.24% |
| Stage #1: 1-Bromonaphthalene With iodine; magnesium In tetrahydrofuran at 75℃; for 17h; Stage #2: triethyl borate In tetrahydrofuran at 20℃; for 5h; Inert atmosphere; Cooling with acetone-dry ice; |

| Conditions | Yield |
|---|---|
| With fac-tris(2-phenylpyridinato-N,C2')iridium(III); tributyl-amine; water In acetonitrile at 25℃; for 24h; Schlenk technique; Inert atmosphere; Sealed tube; Irradiation; | 48% |
-
-
703-55-9
1-naphthylmagnesiumbromide

-
-
13922-41-3
1-Naphthylboronic acid

| Conditions | Yield |
|---|---|
| With boric acid triisopropyl ester In diethyl ether | 39% |
-
-
4250-53-7
(1-naphthyl)dichloroborane

-
-
13922-41-3
1-Naphthylboronic acid

| Conditions | Yield |
|---|---|
| With water |

| Conditions | Yield |
|---|---|
| With diethyl ether und anschliessenden Hydrolyse; |

| Conditions | Yield |
|---|---|
| Grignard reaction; |

-
-
13922-41-3
1-Naphthylboronic acid

| Conditions | Yield |
|---|---|
| With hydrogenchloride In tetrahydrofuran; hexane at 5 - 20℃; | 1.5 g |
| With hydrogenchloride; water In tetrahydrofuran for 4h; | 3.74 g |
| With hydrogenchloride; water In tetrahydrofuran; hexane at 20℃; pH=3 - 5; |
-
-
607-51-2
di-[1]naphthyl-mercury

-
-
13922-41-3
1-Naphthylboronic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: boron trichloride / 120 - 150 °C 2: water View Scheme |
-
-
87199-18-6
3-hydroxyphenylboronic acid

-
-
13922-41-3
1-Naphthylboronic acid

| Conditions | Yield |
|---|---|
| With sulfuric acid; water |

| Conditions | Yield |
|---|---|
| With hydrogen bromide treatment with HBr, hydrolysis;; | |
| With HBr treatment with HBr, hydrolysis;; |

| Conditions | Yield |
|---|---|
| With water hot water; | |
| With H2O hot water; |

| Conditions | Yield |
|---|---|
| With water heating; | |
| With water heating; |

| Conditions | Yield |
|---|---|
| With water In water heating; | |
| With water In water heating; |
-
-
126508-82-5
1,2-bis(1-naphthyl)diborane(6)

-
-
13922-41-3
1-Naphthylboronic acid

| Conditions | Yield |
|---|---|
| With water | |
| With H2O |
-
-
696-62-8
para-iodoanisole

-
-
13922-41-3
1-Naphthylboronic acid

-
-
27331-33-5
1-(4-methoxyphenyl)naphthalene

| Conditions | Yield |
|---|---|
| With potassium phosphate; tetrabutylammomium bromide In 1,4-dioxane; water at 120℃; for 1.5h; Suzuki Coupling; Inert atmosphere; | 100% |
| With potassium carbonate; [PS-PEG-adppp-Pd(η3-C3H5)]Cl In water at 50℃; for 12h; Suzuki-Miyaura coupling; | 99% |
| With sodium carbonate; trisphosphine-based palladium In water at 100℃; for 3h; Suzuki-Miyaura cross-coupling; | 98% |
-
-
16932-45-9
1-bromo-2,6-dimethoxybenzene

-
-
13922-41-3
1-Naphthylboronic acid

-
-
173300-93-1
1-(2,6-dimethoxyphenyl)naphthalene

| Conditions | Yield |
|---|---|
| With potassium phosphate; tris-(dibenzylideneacetone)dipalladium(0); C31H27FeOP In toluene at 70℃; for 24h; Reagent/catalyst; Time; Suzuki-Miyaura Coupling; Schlenk technique; Inert atmosphere; Sealed tube; | 100% |
| With barium dihydroxide; tetrakis(triphenylphosphine) palladium(0) In 1,4-dioxane; water for 2h; Heating; | 92% |
| With tris-(dibenzylideneacetone)dipalladium(0); potassium phosphate monohydrate; C30H25FeP In toluene at 100℃; for 24h; Suzuki-Miyaura Coupling; Sealed tube; | 78% |
| With barium dihydroxide; tetrakis(triphenylphosphine) palladium(0) In 1,4-dioxane; water Heating; |
-
-
13922-41-3
1-Naphthylboronic acid

-
-
199609-54-6
4,6-Dibromo-2,3,7-trimethoxy-cyclohepta-2,4,6-trienone

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); sodium carbonate In ethanol; toluene at 100℃; for 16h; | 100% |
-
-
3401-47-6
1-bromo-2-methoxynaphthalene

-
-
13922-41-3
1-Naphthylboronic acid

-
-
93603-10-2, 80317-71-1, 126451-61-4, 86596-61-4
2-methoxy-1,1'-binaphthalene

| Conditions | Yield |
|---|---|
| With dichloro[1,1'-bis(di-t-butylphosphino)ferrocene]palladium(II); triethylamine In water at 20℃; for 24h; Suzuki-Miyaura reaction; Inert atmosphere; Micellar solution; | 100% |
| With tris-(dibenzylideneacetone)dipalladium(0); caesium carbonate In tetrahydrofuran at 20℃; Suzuki coupling; Inert atmosphere; | 100% |
| With potassium phosphate; tris-(dibenzylideneacetone)dipalladium(0); C31H27FeOP In toluene at 70℃; for 24h; Reagent/catalyst; Time; Suzuki-Miyaura Coupling; Schlenk technique; Inert atmosphere; Sealed tube; | 100% |
-
-
7507-86-0
2-bromo-5-methoxy-benzaldehyde

-
-
13922-41-3
1-Naphthylboronic acid

-
-
199117-07-2
5-methoxy-2-(naphthalen-1-yl)benzaldehyde

| Conditions | Yield |
|---|---|
| With cesium fluoride; tetrakis(triphenylphosphine) palladium(0) In 1,2-dimethoxyethane for 18h; Suzuki reaction; Heating; | 100% |
| With tetrakis(triphenylphosphine) palladium(0); sodium carbonate In methanol; 1,2-dimethoxyethane; water at 100℃; for 12h; | |
| With tetrakis(triphenylphosphine) palladium(0); sodium carbonate In 1,2-dimethoxyethane; water at 100℃; for 12h; |
-
-
13922-41-3
1-Naphthylboronic acid

-
-
99-91-2
para-chloroacetophenone

| Conditions | Yield |
|---|---|
| With C38H53Cl2N2O2PPd*CH2Cl2; tetrabutylammomium bromide; potassium hydroxide In water at 80℃; for 6h; Suzuki-Miyaura Coupling; Schlenk technique; | 100% |
| With potassium phosphate; 1,2-bis(diphenylphosphino)ethane nickel(II) chloride; trisodium tris(3-sulfophenyl)phosphine; zinc In 1,4-dioxane; water at 50℃; | 99% |
| With potassium fluoride; 18-crown-6 ether; bis-phenanthrylimidazolium chloride; palladium diacetate In tetrahydrofuran at 50℃; for 3h; Suzuki-Miyaura coupling; | 99% |
-
-
13922-41-3
1-Naphthylboronic acid

-
-
84539-35-5
(3-bromopyridin-4-yl)-dimethyl-amine

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); sodium carbonate In ethanol; water; toluene Arylation; cross-coupling; Suzuki reaction; Heating; | 100% |
-
-
104-92-7
1-bromo-4-methoxy-benzene

-
-
13922-41-3
1-Naphthylboronic acid

-
-
27331-33-5
1-(4-methoxyphenyl)naphthalene

| Conditions | Yield |
|---|---|
| With di-tert-butyl(4-sulfonatobenzyl)phosphonium; palladium diacetate; sodium carbonate In water; acetonitrile at 50℃; Reagent/catalyst; Suzuki Coupling; Inert atmosphere; Glovebox; | 100% |
| With di-tert-butyl(4-sulfonatobenzyl)phosphonium; palladium diacetate; sodium carbonate In water; acetonitrile at 50℃; Reagent/catalyst; Suzuki Coupling; Inert atmosphere; Glovebox; | 100% |
| With C38H53Cl2N2O2PPd*CH2Cl2; tetrabutylammomium bromide; potassium hydroxide In water at 80℃; for 12h; Suzuki-Miyaura Coupling; Schlenk technique; | 100% |
-
-
13922-41-3
1-Naphthylboronic acid

-
-
445417-44-7
(R)-Nα-benzyloxycarbonyl-(1-bromo-2-napthyl)alanine methyl ester

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); sodium carbonate In water; benzene at 80℃; for 36h; Suzuki cross coupling; | 100% |
| With water; tetrakis(triphenylphosphine) palladium(0) In benzene at 80℃; for 36h; Suzuki cross-coupling; | 100% |
-
-
13922-41-3
1-Naphthylboronic acid

-
-
604-53-5
1,1'-bisnaphthalene

| Conditions | Yield |
|---|---|
| With 1-bromo-2-methylnaphtalene; bis[1-((1R,2S,5R)-2-isopropyl-5-methylcyclohexyl)-3-methyl-4-phenyl-1,2,3-triazol-5-ylidene]palladium(II) iodide; potassium tert-butylate In ethanol at 20℃; for 2h; Inert atmosphere; | 100% |
| With C54H74Cl2N4O2Pd2 In ethanol at 22℃; for 6h; | 98% |
| With 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical; palladium diacetate; tetraethylammonium perchlorate; potassium carbonate In water; acetonitrile at 20℃; Inert atmosphere; Electrochemical reaction; | 97% |

| Conditions | Yield |
|---|---|
| With tris-(dibenzylideneacetone)dipalladium(0); caesium carbonate In tetrahydrofuran at 20℃; for 6h; Suzuki coupling; Inert atmosphere; | 100% |
| With C64H76Br2N4O2Pd; potassium carbonate In N,N-dimethyl acetamide at 20 - 140℃; for 24h; Suzuki coupling; Inert atmosphere; | 99% |
| With potassium fluoride; monophosphine 1,2,3,4,5-pentaphenyl-1'-(di-tert-butylphosphino)ferrocene; bis(dibenzylideneacetone)-palladium(0) In tetrahydrofuran at 20℃; for 48h; Suzuki coupling; | 97% |
-
-
6781-98-2
2,6-dimethyl-1-chlorobenzene

-
-
13922-41-3
1-Naphthylboronic acid

-
-
147424-62-2
1-(2,6-dimethylphenyl)naphthalene

| Conditions | Yield |
|---|---|
| With (3-phenylallyl)(chloro)-[1,3-bis(2,6-diisopropylphenyl)-4,5-dihydroimidazol-2-ylidene]palladium(II); triethylamine In water at 23℃; for 24h; Suzuki-Miyaura reaction; Inert atmosphere; Micellar solution; | 100% |
| With naphthalene; Pd(η3-cinnamyl)(1,3-bis(2,6-diisopropylphenyl)imidazol-2-ylidene)Cl; potassium tert-butylate In tetrahydrofuran; methanol; isopropyl alcohol at 40℃; for 0.5h; Reagent/catalyst; Temperature; Time; Suzuki-Miyaura Coupling; Inert atmosphere; Schlenk technique; | 99% |
| With potassium phosphate; C29H37O2P; palladium diacetate In toluene at 110℃; for 24h; Suzuki-Miyaura Coupling; Inert atmosphere; Schlenk technique; | 99% |
-
-
13922-41-3
1-Naphthylboronic acid

-
-
99-90-1
para-bromoacetophenone

| Conditions | Yield |
|---|---|
| With caesium carbonate; palladium diacetate In toluene at 80℃; for 10h; Suzuki-Miyaura cross-coupling; | 100% |
| With potassium carbonate In ethanol; water at 20℃; for 4h; Suzuki-Miyaura reaction; Inert atmosphere; | 100% |
| With potassium phosphate; Pd[1,3,5,7-Me4-2,4,8-trioxa-6-Ph-6-phosphaadamantane]2*dba In toluene at 20℃; for 0.166667h; Suzuki cross-coupling; | 99% |
-
-
5600-21-5
2-amino-6-methyl-4-chloropyrimidine

-
-
13922-41-3
1-Naphthylboronic acid

-
-
92554-55-7
4-methyl-6-(naphthalen-1-yl)-pyrimidin-2-ylamine

| Conditions | Yield |
|---|---|
| With potassium phosphate monohydrate; C66H84O10P2Pd In water; ethyl acetate; toluene at 45℃; for 18h; Suzuki-Miyaura Coupling; Inert atmosphere; | 100% |
| With dicyclohexyl-(2',6'-dimethoxybiphenyl-2-yl)-phosphane; potassium phosphate; palladium diacetate In 1,4-dioxane at 100℃; for 14h; Suzuki-Miyaura cross-coupling; | 92% |

| Conditions | Yield |
|---|---|
| With potassium phosphate monohydrate; C66H84O10P2Pd In water; toluene at 45℃; for 4.5h; Reagent/catalyst; Suzuki-Miyaura Coupling; Inert atmosphere; | 100% |
| With (3-phenylallyl)(chloro)-[1,3-bis(2,6-diisopropylphenyl)-4,5-dihydroimidazol-2-ylidene]palladium(II); potassium carbonate In ethanol at 40℃; for 4h; Suzuki-Miyaura Coupling; Green chemistry; | 99% |
| With potassium carbonate In water; toluene at 90℃; for 0.5h; Suzuki-Miyaura Coupling; | 97% |
-
-
656800-67-8
4-bromo-2-methyl-3,5,6,7-tetrahydro-s-indacen-1(2H)-one

-
-
13922-41-3
1-Naphthylboronic acid

-
-
656800-71-4
3,5,6,7-tetrahydro-2-methyl-4-(1-naphthalenyl)-s-hydraindacen-1(2H)-one

| Conditions | Yield |
|---|---|
| With sodium carbonate; tetrakis(triphenylphosphine) palladium(0) In 1,2-dimethoxyethane; water for 16h; Heating / reflux; | 100% |
-
-
1011736-47-2
3-benzyl-2-chloro-6-p-tolylfuro[2,3-b]pyrazine

-
-
13922-41-3
1-Naphthylboronic acid

-
-
1011736-54-1
3-benzyl-2-(naphthalen-1-yl)-6-p-tolylfuro[2,3-b]pyrazine

| Conditions | Yield |
|---|---|
| With sodium carbonate; tetrakis(triphenylphosphine) palladium(0) In water; N,N-dimethyl-formamide at 100℃; for 0.166667h; Suzuki coupling; microwave irradiation; | 100% |
| With tetrakis(triphenylphosphine) palladium(0); sodium carbonate In water; N,N-dimethyl-formamide at 100℃; for 0.166667h; Suzuki-Miyaura reaction; Microwave irradiation; regioselective reaction; | 100% |
-
-
1162652-16-5
(mesityl)C((C4H2N)(C6H4)OH)((C4H3N)(C6H4)OH)

-
-
13922-41-3
1-Naphthylboronic acid

-
-
1171817-78-9
C40H31BN2O2

| Conditions | Yield |
|---|---|
| In chloroform for 3h; Reflux; | 100% |
-
-
13922-41-3
1-Naphthylboronic acid

-
-
90-15-3
α-naphthol

| Conditions | Yield |
|---|---|
| With water; 3-chloro-benzenecarboperoxoic acid In ethanol at 20℃; for 6h; | 100% |
| With dihydrogen peroxide at 30℃; for 5h; Green chemistry; | 98% |
| With water; dihydrogen peroxide at 20℃; for 0.166667h; Green chemistry; | 97% |
-
-
45644-21-1
6-chloropyridin-2-amine

-
-
13922-41-3
1-Naphthylboronic acid

-
-
1028834-19-6
6-naphthalen-1-yl-pyridin-2-ylamine

| Conditions | Yield |
|---|---|
| With potassium phosphate monohydrate; Ethyl 4-bromobenzoate; C52H49PRu; bis(dibenzylideneacetone)-palladium(0) In 1,4-dioxane at 100℃; for 1.16667h; Suzuki-Miyaura Coupling; Inert atmosphere; | 100% |
-
-
13922-41-3
1-Naphthylboronic acid

-
-
77664-95-0
1-chloro-7-methoxy-3,4-dihydronaphthalene-2-carboxaldehyde

| Conditions | Yield |
|---|---|
| With tetrabutylammomium bromide; palladium diacetate; potassium carbonate In water at 45℃; for 1.5h; Suzuki-Miyaura Coupling; Inert atmosphere; | 100% |
-
-
13922-41-3
1-Naphthylboronic acid

| Conditions | Yield |
|---|---|
| With dicyclohexyl-(2',6'-dimethoxybiphenyl-2-yl)-phosphane; potassium phosphate; palladium diacetate In tetrahydrofuran; water for 3h; Suzuki-Miyaura Coupling; Reflux; Inert atmosphere; | 100% |

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); potassium carbonate In ethanol; water; toluene at 90℃; for 2h; Reflux; | 100% |
-
-
624-38-4
para-diiodobenzene

-
-
13922-41-3
1-Naphthylboronic acid

-
-
64065-97-0
1,4-di(naphthalen-1-yl)benzene

| Conditions | Yield |
|---|---|
| With potassium carbonate In ethanol; water at 80℃; for 4h; Suzuki-Miyaura Coupling; | 100% |
-
-
13922-41-3
1-Naphthylboronic acid

-
-
5597-04-6
2-cyanomethylbenzoic acid methyl ester

-
-
130369-98-1
3-(naphthalen-1-yl)isoquinolin-1-(2H)-one

| Conditions | Yield |
|---|---|
| With [2,2]bipyridinyl; palladium(II) trifluoroacetate; trifluoroacetic acid In tetrahydrofuran at 85℃; for 20h; Catalytic behavior; Reagent/catalyst; Solvent; | 99.4% |
| With [2,2]bipyridinyl; palladium(II) trifluoroacetate; trifluoroacetic acid In tetrahydrofuran at 80℃; for 24h; Schlenk technique; | 99% |
| With 1,3-bis[(diphenylphosphino)propane]dichloronickel(II); zinc(II) chloride In 2-methyltetrahydrofuran at 120℃; for 48h; Schlenk technique; | 86% |
-
-
40288-69-5
N-(2-cyanophenyl)benzamide

-
-
13922-41-3
1-Naphthylboronic acid

| Conditions | Yield |
|---|---|
| With [2,2]bipyridinyl; palladium diacetate; trifluoroacetic acid In tetrahydrofuran at 90℃; for 16h; Reagent/catalyst; Solvent; | 99.4% |
| With [2,2]bipyridinyl; palladium diacetate; trifluoroacetic acid In tetrahydrofuran at 80℃; for 24h; Schlenk technique; | 91% |
-
-
57774-35-3
4'-bromobiphenyl-4-carbonitrile

-
-
13922-41-3
1-Naphthylboronic acid

| Conditions | Yield |
|---|---|
| Stage #1: 4'-bromobiphenyl-4-carbonitrile; 1-Naphthylboronic acid With potassium carbonate In toluene Inert atmosphere; Stage #2: With palladium diacetate; tri(m-tolyl)phosphine In toluene at 90℃; | 99.3% |
-
-
556-96-7
5-bromo-1,3-xylene

-
-
13922-41-3
1-Naphthylboronic acid

-
-
164172-87-6
1-(3,5-dimethylphenyl)naphthalene

| Conditions | Yield |
|---|---|
| With potassium phosphate; tris-(dibenzylideneacetone)dipalladium(0); dicyclohexyl(1-(naphtho[2,3-b]furan-9-yl)naphthalen-2-yl)phosphine In toluene at 80℃; for 0.5h; Suzuki-Miyaura Coupling; Inert atmosphere; Schlenk technique; | 99% |
| With palladium diacetate; 5A molecular sieve; triethylamine; tris-(o-tolyl)phosphine In N,N-dimethyl-formamide at 100℃; for 4h; | 73% |

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); sodium carbonate In 1,2-dimethoxyethane; water for 0.416667h; Ambient temperature; | 99% |
| With copper(l) iodide; sodium carbonate In 1,2-dimethoxyethane; water at 35℃; for 0.333333h; | 96% |
-
-
13922-41-3
1-Naphthylboronic acid

-
-
166328-07-0
potassium trifluoro(naphth-1-yl)borate

| Conditions | Yield |
|---|---|
| Stage #1: 1-Naphthylboronic acid With potassium fluoride In water; acetonitrile at 21℃; Stage #2: With L-Tartaric acid In tetrahydrofuran; water; acetonitrile | 99% |
| With potassium hydrogen bifluoride In methanol; water at 20℃; Fluorination; | 94% |
| With potassium hydrogen bifluoride In methanol; water for 0.5h; | 81% |
1-Naphthaleneboronic acid Specification
The 1-Naphthaleneboronic acid is an organic compound with the formula C10H9BO2. The IUPAC name of this chemical is naphthalen-1-ylboronic acid. With the CAS registry number 13922-41-3, it is also named as Boronic acid, B-1-naphthalenyl-. The product's categories are Blocks; Boronic Acids; Boronic Acid Series; Naphthalene Derivatives; Heterocyclic Compounds; Naphthalene; Organoborons; B (Classes of Boron Compounds). It is off-white to pink beige powder which is stable under normal temperature and pressure. Additionally, this chemical should be sealed in the container, kept away from light and stored in thewell-ventilated and dry place.
The other characteristics of this product can be summarized as: (1)ACD/LogP: 2.82; (2)# of Rule of 5 Violations: 0; (3)#H bond acceptors: 2; (4)#H bond donors: 2; (5)#Freely Rotating Bonds: 3; (6)Polar Surface Area: 18.46 Å2; (7)Index of Refraction: 1.638; (8)Molar Refractivity: 50.84 cm3; (9)Molar Volume: 141.3 cm3; (10)Polarizability: 20.15×10-24 cm3; (11)Surface Tension: 50.4 dyne/cm; (12)Density: 1.21 g/cm3; (13)Flash Point: 184.8 °C; (14)Enthalpy of Vaporization: 66.48 kJ/mol; (15)Boiling Point: 381.9 °C at 760 mmHg; (16)Vapour Pressure: 1.63E-06 mmHg at 25°C.
Preparation of 1-Naphthaleneboronic acid: It can be obtained by tributyl-naphthalen-1-yl-stannane. This reaction needs reagents BH3, H2O and solvent tetrahydrofuran by heating. The reaction time is 4 hours. The yield is 68%.
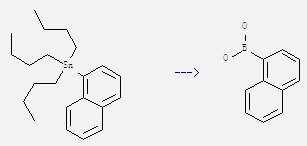
Uses of 1-Naphthaleneboronic acid: It is used as pharmaceutical intermediate. And it can react with 4-iodo-3,5-dimethyl-isoxazole to get 4-(1-naphthyl)-3,5-dimethylisoxazole. This reaction needs reagents (PPh3)2PdCl2, NaHCO3 and solvents 1,2-dimethoxy-ethane, H2O at temperature of 80 °C. The reaction time is 18 hours. The yield is 75%.
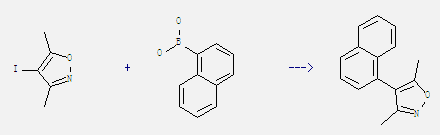
When you are using this chemical, please be cautious about it as the following:
It is irritating to eyes, respiratory system and skin. In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. If you want to contact this product, you must wear suitable protective clothing, gloves and eye/face protection.
People can use the following data to convert to the molecule structure.
1. SMILES:OB(O)c2cccc1ccccc12
2. InChI:InChI=1/C10H9BO2/c12-11(13)10-7-3-5-8-4-1-2-6-9(8)10/h1-7,12-13H
3. InChIKey:HUMMCEUVDBVXTQ-UHFFFAOYAQ
Related Products
- 1-Naphthaleneboronic acid
- 139226-28-1
- 139226-30-5
- 139227-42-2
- 139237-76-6
- 139238-61-2
- 139244-00-1
- 13924-50-0
- 13924-94-2
- 13924-95-3
- 13924-99-7
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View