-
Name
2,3-Epoxypropyltrimethylammonium chloride
- EINECS 221-221-0
- CAS No. 3033-77-0
- Article Data11
- CAS DataBase
- Density 1.13 g/mL at 20 °C(lit.)
- Solubility 852g/L at 20℃
- Melting Point 137-139 °C
- Formula C6H14ClNO
- Boiling Point
- Molecular Weight 151.636
- Flash Point 170 °C
- Transport Information
- Appearance solid
- Safety 53-14-26-36/37/39-45
- Risk Codes 45-21/22-41-43-48/22
-
Molecular Structure
-
Hazard Symbols
 T
T
- Synonyms (2,3-Epoxypropyl)trimethylammoniumchloride (7CI);Oxiranemethanaminium, N,N,N-trimethyl-, chloride (9CI);(b,g-Epoxypropyl)trimethylammonium chloride;1,2-Epoxy-3-(trimethylammonio)propyl chloride;1-(Trimethylammonio)-2,3-epoxypropane chloride;Catiomaster G;ETA (oniumcompound);G-MAC;Glycidyltrimethylammonium chloride;Glytac A;GlytacA 100;N-Glycidyl-N,N,N-trimethylammonium chloride;N-Glycidyltrimethylammoniumchloride;QUAB 151;Raisacat 151;Trimethylglycidylammonium chloride;Weisstex E 100;Wildye PTC;[(Trimethylammonio)methyl]oxiranechloride;
- PSA 12.53000
- LogP -2.90460
Synthetic route

| Conditions | Yield |
|---|---|
| With ethanol at 100℃; | |
| at 120℃; | |
| In water at 20℃; | |
| In chloroform; toluene at 20℃; Solvent; Temperature; Autoclave; | 405.5 g |

-
-
3033-77-0
glycidyltrimethylammonium chloride

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water at 20℃; for 0.5h; | |
| With sodium hydroxide In water | |
| With sodium hydroxide at 13 - 15℃; for 1.16667h; | |
| With sodium hydroxide |

| Conditions | Yield |
|---|---|
| With sodium chloride; sodium hydroxide In isopropyl alcohol at 20 - 40℃; for 7h; pH=11; | 96% |

-
-
3033-77-0
glycidyltrimethylammonium chloride

| Conditions | Yield |
|---|---|
| In water for 0.166667h; | 90% |

| Conditions | Yield |
|---|---|
| for 0.166667h; | 90% |


-
-
3033-77-0
glycidyltrimethylammonium chloride

| Conditions | Yield |
|---|---|
| In water for 0.166667h; | 90% |

-
-
3033-77-0
glycidyltrimethylammonium chloride

| Conditions | Yield |
|---|---|
| In water for 0.166667h; | 90% |
-
-
3033-77-0
glycidyltrimethylammonium chloride

| Conditions | Yield |
|---|---|
| In water for 10h; | 87.8% |

| Conditions | Yield |
|---|---|
| In water at 60℃; | 87% |

-
-
3033-77-0
glycidyltrimethylammonium chloride

-
-
68960-97-4
1,14-diamino-3,6,9,12-tetraoxatetradecane

| Conditions | Yield |
|---|---|
| Stage #1: C17H6Br2Cl2N4O2; 1,14-diamino-3,6,9,12-tetraoxatetradecane With sodium carbonate at 35 - 40℃; for 2h; pH=5-6; Stage #2: glycidyltrimethylammonium chloride With hydrogenchloride at 85℃; pH=4; | 82.46% |


| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate In isopropyl alcohol; acetone at 80℃; for 16h; | 79% |

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate In isopropyl alcohol; acetone at 80℃; for 16h; | 79% |
-
-
6522-75-4
1-(4,6-dichloro-[1,3,5]triazin-2-ylamino)-anthraquinone

-
-
3033-77-0
glycidyltrimethylammonium chloride

-
-
68960-97-4
1,14-diamino-3,6,9,12-tetraoxatetradecane

| Conditions | Yield |
|---|---|
| Stage #1: 1-(4,6-dichloro-[1,3,5]triazin-2-ylamino)-anthraquinone; 1,14-diamino-3,6,9,12-tetraoxatetradecane With sodium carbonate at 35 - 40℃; for 2h; pH=5-6; Stage #2: glycidyltrimethylammonium chloride With hydrogenchloride at 85℃; pH=4; | 79% |

-
-
1119-23-9
3-hydroxyethylammonium-n-propanesulfonate

-
-
3033-77-0
glycidyltrimethylammonium chloride

-
-
1379044-59-3
N-(2-hydroxy-3-trimethylammoniumpropyl)-N-(2-hydroxyethyl)homotaurine inner salt

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In ethanol; water at 5 - 20℃; for 21h; | 76% |

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate In isopropyl alcohol; acetone at 80℃; for 16h; | 76% |
-
-
14389-86-7
2-benzyloxybenzoic acid

-
-
3033-77-0
glycidyltrimethylammonium chloride

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate In isopropyl alcohol; acetone at 80℃; for 16h; | 73% |

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate In isopropyl alcohol; acetone at 80℃; for 16h; | 70% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water at 60℃; for 2h; | 60% |

| Conditions | Yield |
|---|---|
| With caesium carbonate In N,N-dimethyl-formamide at 120℃; for 16h; | 57% |
-
-
3033-77-0
glycidyltrimethylammonium chloride

-
-
30754-24-6
heptakis(6-amino-6-deoxy)-β-cyclodextrin

| Conditions | Yield |
|---|---|
| In aq. buffer at 50℃; for 24h; pH=10; | 51% |
-
-
50979-18-5
guanidine sulfamate

-
-
3033-77-0
glycidyltrimethylammonium chloride

| Conditions | Yield |
|---|---|
| With water; 4-pyrrolidin-1-ylpyridine at 45℃; for 2h; Temperature; | 50.5% |

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate In isopropyl alcohol; acetone at 60℃; for 16h; | 26% |

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate In isopropyl alcohol; acetone at 60℃; for 16h; | 25% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide at 50℃; | 23% |
-
-
35717-98-7
bis(2-propyl)-p-toluenesulfonyloxymethylphosphonate

-
-
3033-77-0
glycidyltrimethylammonium chloride


| Conditions | Yield |
|---|---|
| Multistep reaction; |

-
-
35717-98-7
bis(2-propyl)-p-toluenesulfonyloxymethylphosphonate

-
-
3033-77-0
glycidyltrimethylammonium chloride

-
-
1904-98-9
2,6-diaminopurine

| Conditions | Yield |
|---|---|
| Multistep reaction; |


| Conditions | Yield |
|---|---|
| With sodium hydroxide Product distribution; var. conditions; also degradation of γ-cyclodextrin; |

-
-
35717-98-7
bis(2-propyl)-p-toluenesulfonyloxymethylphosphonate

-
-
3033-77-0
glycidyltrimethylammonium chloride

-
-
4637-24-5
N,N-dimethyl-formamide dimethyl acetal


| Conditions | Yield |
|---|---|
| With ammonium hydroxide; trimethylsilyl bromide; sodium hydride; triethylamine Yield given. Multistep reaction; | |
| With ammonium hydroxide; trimethylsilyl bromide; sodium hydride; triethylamine Multistep reaction; |

-
-
35717-98-7
bis(2-propyl)-p-toluenesulfonyloxymethylphosphonate

-
-
3033-77-0
glycidyltrimethylammonium chloride

-
-
4637-24-5
N,N-dimethyl-formamide dimethyl acetal


| Conditions | Yield |
|---|---|
| With ammonium hydroxide; sodium hydride Multistep reaction. Title compound not separated from byproducts; |

-
-
35717-98-7
bis(2-propyl)-p-toluenesulfonyloxymethylphosphonate

-
-
3033-77-0
glycidyltrimethylammonium chloride

-
-
4637-24-5
N,N-dimethyl-formamide dimethyl acetal



| Conditions | Yield |
|---|---|
| With sodium hydride Multistep reaction. Title compound not separated from byproducts; |


| Conditions | Yield |
|---|---|
| With 1.) CsCO3 1.) DMF, 120 deg C, 16 h, 2.) DMF, 20 deg C, 24 h; Multistep reaction. Title compound not separated from byproducts; |

2,3-Epoxypropyltrimethylammonium chloride Chemical Properties
Glycidyl trimethyl ammonium chloride(3033-77-0)'s molecular formula is C6H14NOCl and its formula weight is 151.6.
The density of glycidyl trimethyl ammonium chloride(3033-77-0) is 1.13 g/mL at 20 °C(lit.)
Its flash point is 170°C.
The molecular structure of glycidyl trimethyl ammonium chloride(3033-77-0):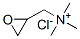
2,3-Epoxypropyltrimethylammonium chloride Uses
2,3-Epoxypropyltrimethylammonium chloride Toxicity Data With Reference
| 1. | mmo-klp 2 mmol/L | MUREAV Mutation Research. 89 (1981),269. | ||
| 2. | cyt-rat:lvr 10 mg/L | MUREAV Mutation Research. 153 (1985),57. | ||
| 3. | scu-mus LD50:90 mg/kg | JCSOA9 Journal of the Chemical Society .(London, England.: )1947,176. |
2,3-Epoxypropyltrimethylammonium chloride Consensus Reports
2,3-Epoxypropyltrimethylammonium chloride Safety Profile
2,3-Epoxypropyltrimethylammonium chloride Standards and Recommendations
Related Products
- 20,22-DIHYDRODIGITOXIN
- 20,29,30-Trinorlupane,(17alpha)-
- 20-ETHYL-6-β,8-DIHYDROXY-1-α-METHOXY-4-METHYLHETERATISAN-14-ONE
- 20-Ethylprostaglandin F2-alpha
- 20-Isopropylcholanthrene
- 20-METHYLCHOLANTHREN-15-ONE
- 20-METHYLCHOLANTHRENE PICRATE
- 20-METHYLCHOLANTHRENE-TRINITRO-BENZENE
- 20(S)-Ginsenoside C-K
- 2,10-DIFLUOROBENZO(rst)PENTAPHENE
- 3033-82-7
- 303-38-8
- 30339-30-1
- 3034-19-3
- 3034-22-8
- 303-42-4
- 30342-62-2
- 3034-31-9
- 3034-34-2
- 303-43-5
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View