-
Name
2,4'-Dichloroacetophenone
- EINECS 213-323-9
- CAS No. 937-20-2
- Article Data76
- CAS DataBase
- Density 1.31 g/cm3
- Solubility
- Melting Point 100-102 °C(lit.)
- Formula C8H6Cl2O
- Boiling Point 270 °C at 760 mmHg
- Molecular Weight 189.041
- Flash Point 119.1 °C
- Transport Information
- Appearance white to off-white crystalline powder
- Safety 26-36-37/39
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms 4-Chloroaphenacyl chloride;2,4-Dichloro Acetophenone;2-chloro-4-chloroacetophenone;2-chloro-4’-chloroacetophenone;Ethanone, 2-chloro-1- (4-chlorophenyl)-;ethanone, 2-chloro-1-(4-chlorophenyl)-;.alpha.-Chloro-p-chloroacetophenone;2-chloro-1-(4-chlorophenyl)ethanone;Acetophenone, 2,4-dichloro-;4-Chlorophenacyl chloride;p-Chlorophenacyl chloride;.omega.-Chloro-p-chloroacetophenone;
- PSA 17.07000
- LogP 2.76150
Synthetic route

| Conditions | Yield |
|---|---|
| With 1,3-dichloro-5,5-dimethylhydantoin; silica gel In methanol for 1h; Reflux; | 98% |
| With N,N,N-trimethylbenzenemethanaminium dichloroiodate In methanol; 1,2-dichloro-ethane for 6h; Heating; | 97% |
| With tetrachlorosilane; urea hydrogen peroxide adduct In dichloromethane at 0℃; for 1h; | 93% |

| Conditions | Yield |
|---|---|
| With [2,2]bipyridinyl; copper(l) chloride In 1,2-dichloro-ethane for 3h; Heating; | 93% |
-
-
80-11-5
N-methyl-N-nitrosotoluene-p-sulfonamide

-
-
122-01-0
4-chloro-benzoyl chloride

-
-
937-20-2
p-chlorophenacyl chloride

| Conditions | Yield |
|---|---|
| With potassium hydroxide In tetrahydrofuran; methanol; water at 20℃; Arndt-Eistert Homologation; | 93% |
-
-
33491-02-0
1-chloro-4-(chloroethynyl)benzene

-
-
937-20-2
p-chlorophenacyl chloride

| Conditions | Yield |
|---|---|
| With water In 1,2-dichloro-ethane at 20℃; for 14h; | 93% |
| With water; silver fluoride; trifluoroacetic acid at 40℃; for 6h; Green chemistry; | 88% |
| With tetrafluoroboric acid; water In 2,2,2-trifluoroethanol at 80℃; for 10h; | 87% |
| With indium(III) triflate; water In acetic acid at 100℃; Sealed tube; | 79% |

| Conditions | Yield |
|---|---|
| With magnesium(II) chloride hexahydrate; oxygen; lithium perchlorate; manganese(ll) chloride In dichloromethane; acetone at 40℃; for 40h; Electrochemical reaction; | 93% |
| With magnesium(II) chloride hexahydrate; cobalt(II) chloride hexahydrate; oxygen; lithium perchlorate In dichloromethane; acetone at 40℃; for 12h; Electrolysis; Sealed tube; chemoselective reaction; | 77% |
| With tris(2,2'-bipyridyl)ruthenium dichloride; chloroform; [bis(acetoxy)iodo]benzene In chloroform at 20℃; for 12h; Irradiation; | 77% |
| With iron(III) chloride; potassium chloride; oxygen; toluene-4-sulfonic acid In tert-butyl methyl ether at 20℃; Irradiation; Green chemistry; | 49% |
-
-
676486-15-0
1-(4-chlorophenyl)-2-(dimethyl(oxo)-λ6-sulfanylidene)ethan-1-one

-
-
937-20-2
p-chlorophenacyl chloride

| Conditions | Yield |
|---|---|
| With hydrogenchloride In tetrahydrofuran; 1,4-dioxane at 20 - 70℃; for 16h; | 92% |
| With hydrogenchloride In tetrahydrofuran; 1,4-dioxane at 20 - 70℃; | 78% |

| Conditions | Yield |
|---|---|
| With methanol; 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical; trichloroisocyanuric acid In dichloromethane at 20℃; for 2h; Reagent/catalyst; | 87% |
| With N,N-dichloro-p-toluenesulfonamide In acetonitrile at 55℃; for 3.5h; | 79% |
| With hydrogenchloride; 3-chloro-benzenecarboperoxoic acid In N,N-dimethyl-formamide at 25℃; for 6h; | 84 % Chromat. |

| Conditions | Yield |
|---|---|
| With methanol; 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical; trichloroisocyanuric acid In dichloromethane at 20℃; for 2h; Reagent/catalyst; | 87% |

| Conditions | Yield |
|---|---|
| aluminium trichloride In carbon disulfide for 2h; Heating; | 84% |
| With carbon disulfide; aluminium trichloride | |
| With aluminium trichloride In carbon disulfide |

| Conditions | Yield |
|---|---|
| With S4N4*SbCl5 In toluene for 3.5h; Heating; | 81% |
| With sodium chloride In acetonitrile | |
| With N-benzyl-N,N,N-triethylammonium chloride; benzenesulfonyl chloride In water at 20℃; for 1.3h; Solvent; Green chemistry; |

| Conditions | Yield |
|---|---|
| Stage #1: 4-n-chlorophenylacetylene With methanol; gold(III) chloride; silver(I) triflimide In 1,4-dioxane at 45℃; Stage #2: With N-chloro-succinimide In 1,4-dioxane regioselective reaction; | 75% |
| With hydrogenchloride; iodine In dimethyl sulfoxide at 20℃; for 16h; | 69% |

| Conditions | Yield |
|---|---|
| With iron(III) chloride; silica gel In dichloromethane at 20℃; for 0.166667h; | 75% |
-
-
55591-23-6
1,1,2,2,3,3,4,4,5,5,6,6,6-tridecafluorohexane-1-sulfonyl chloride

-
-
58518-76-6
((1-(4-chlorophenyl)vinyl)oxy)trimethylsilane

-
A
-
937-20-2
p-chlorophenacyl chloride

| Conditions | Yield |
|---|---|
| With tris(triphenylphosphine)ruthenium(II) chloride In benzene at 120℃; for 7h; | A 24% B 56% |


-
A
-
18362-49-7
1,3-bis(4-chlorophenyl)propane-1,3-dione

-
B
-
937-20-2
p-chlorophenacyl chloride

| Conditions | Yield |
|---|---|
| Stage #1: 2-chloro-1,3-bis(4′-chlorophenyl)-1,3-propanedione With [Cu(tris(2-pyridylmethyl)amine)(CH3CN)](ClO4)2; lithium hexamethyldisilazane In diethyl ether; acetonitrile for 48h; Inert atmosphere; Glovebox; Stage #2: With water In diethyl ether; acetonitrile | A n/a B 56% |
-
-
421-83-0
trifluoromethane sulfonyl chloride

-
-
58518-76-6
((1-(4-chlorophenyl)vinyl)oxy)trimethylsilane

-
A
-
121194-36-3
1-(4-chlorophenyl)-3,3,3-trifluoro-propan-1-one

-
B
-
937-20-2
p-chlorophenacyl chloride

| Conditions | Yield |
|---|---|
| With tris(triphenylphosphine)ruthenium(II) chloride In benzene at 120℃; for 7h; | A 51% B 15% |

-
-
2547-61-7
Trichloromethanesulfonyl chloride

-
-
58518-76-6
((1-(4-chlorophenyl)vinyl)oxy)trimethylsilane

-
A
-
41501-65-9
3,3-dichloro-1-(4-chlorophenyl)prop-2-en-1-one

-
B
-
937-20-2
p-chlorophenacyl chloride

| Conditions | Yield |
|---|---|
| With ruthenium(II) In benzene at 120℃; for 7h; sealed; | A 41% B 49% |

-
-
99-91-2
para-chloroacetophenone

-
A
-
5157-57-3
2,2-dichloro-1-(4-chlorophenyl)ethanone

-
B
-
937-20-2
p-chlorophenacyl chloride

| Conditions | Yield |
|---|---|
| With chloro-trimethyl-silane; potassium nitrate In dichloromethane at 60℃; for 16h; | A n/a B 42% |
| With aluminum (III) chloride; oxygen; vanadium(V) oxytriisopropoxide; tetra-(n-butyl)ammonium iodide In acetonitrile at 80℃; for 25h; | A 13 %Spectr. B 66 %Spectr. |

| Conditions | Yield |
|---|---|
| With tetrabutyl-ammonium chloride In dichloromethane for 72h; Ambient temperature; | A 36% B 21% |
-
-
3391-10-4
1-(p-chlorophenyl)ethyl alcohol

-
A
-
103966-61-6, 137408-28-7, 137491-94-2, 19759-43-4
1-(4-chlorophenyl)ethyl acetate

-
B
-
99-91-2
para-chloroacetophenone

-
C
-
937-20-2
p-chlorophenacyl chloride

| Conditions | Yield |
|---|---|
| With *5H2O; lithium chloride In acetic acid for 0.3h; Heating; | A 11% B 18% C 2% |

-
-
3391-10-4
1-(p-chlorophenyl)ethyl alcohol

-
A
-
103966-61-6, 137408-28-7, 137491-94-2, 19759-43-4
1-(4-chlorophenyl)ethyl acetate

-
B
-
99-91-2
para-chloroacetophenone

-
C
-
937-20-2
p-chlorophenacyl chloride

| Conditions | Yield |
|---|---|
| With *5H2O; lithium chloride In acetic acid for 0.3h; Heating; | A 11% B 18% C 2% |


| Conditions | Yield |
|---|---|
| With diazomethane; benzene Einleiten von HCl in die Reaktionsloesung unter Kuehlung; |
-
-
5333-82-4
2,2,2-trichloro-1-(4-chloro-phenyl)-ethanol

-
A
-
58369-61-2
2-chloro-2-(4-chlorophenyl)acetaldehyde

-
B
-
937-20-2
p-chlorophenacyl chloride

| Conditions | Yield |
|---|---|
| With n-butyllithium |
-
-
1073-67-2
4-vinylbenzyl chloride

-
A
-
18684-82-7
Z-1-chloro-4-(2-chloroethenyl)benzene

-
B
-
18684-97-4
(E)-1-(2-chlorovinyl)-4-chlorobenzene

-
C
-
104-88-1
4-chlorobenzaldehyde

-
D
-
937-20-2
p-chlorophenacyl chloride

| Conditions | Yield |
|---|---|
| With chlorine dioxide In tetrachloromethane at 52 - 54℃; for 1.66667h; Further byproducts given. Title compound not separated from byproducts; | A 3.2 % Chromat. B 17.6 % Chromat. C 1.0 % Chromat. D 37.5 % Chromat. |
| With chlorine dioxide In tetrachloromethane at 52 - 54℃; for 1.66667h; Further byproducts given; | A 3.2 % Chromat. B 17.6 % Chromat. C 1.0 % Chromat. D 37.5 % Chromat. |

-
-
1073-67-2
4-vinylbenzyl chloride

-
A
-
5157-57-3
2,2-dichloro-1-(4-chlorophenyl)ethanone

-
B
-
18684-82-7
Z-1-chloro-4-(2-chloroethenyl)benzene

-
C
-
18684-97-4
(E)-1-(2-chlorovinyl)-4-chlorobenzene

-
D
-
937-20-2
p-chlorophenacyl chloride

| Conditions | Yield |
|---|---|
| With chlorine dioxide In tetrachloromethane at 52 - 54℃; for 1.66667h; Further byproducts given; | A 1.7 % Chromat. B 3.2 % Chromat. C 17.6 % Chromat. D 37.5 % Chromat. |

-
-
1073-67-2
4-vinylbenzyl chloride

-
A
-
5157-57-3
2,2-dichloro-1-(4-chlorophenyl)ethanone

-
B
-
18684-97-4
(E)-1-(2-chlorovinyl)-4-chlorobenzene

-
C
-
937-20-2
p-chlorophenacyl chloride

-
D
-
74-11-3
para-chlorobenzoic acid

| Conditions | Yield |
|---|---|
| With chlorine dioxide In tetrachloromethane at 52 - 54℃; for 1.66667h; Further byproducts given; | A 1.7 % Chromat. B 17.6 % Chromat. C 37.5 % Chromat. D 6.4 % Chromat. |

-
-
873-73-4
4-n-chlorophenylacetylene

-
-
64-19-7
acetic acid

-
A
-
33491-02-0
1-chloro-4-(chloroethynyl)benzene

-
B
-
5157-57-3
2,2-dichloro-1-(4-chlorophenyl)ethanone

-
C
-
39561-82-5
4-chlorophenacyl acetate

-
D
-
99-91-2
para-chloroacetophenone

-
E
-
84553-28-6
1-Chloro-4-((E)-1,2-dichloro-vinyl)-benzene

-
F
-
937-20-2
p-chlorophenacyl chloride

| Conditions | Yield |
|---|---|
| With perchloric acid at 40℃; Rate constant; Product distribution; Mechanism; | A 4 % Chromat. B 23 % Chromat. C 5 % Chromat. D 10 % Chromat. E 21 % Chromat. F 29 % Chromat. |

-
-
873-73-4
4-n-chlorophenylacetylene

-
-
64-19-7
acetic acid

-
A
-
33491-02-0
1-chloro-4-(chloroethynyl)benzene

-
B
-
5157-57-3
2,2-dichloro-1-(4-chlorophenyl)ethanone

-
C
-
99-91-2
para-chloroacetophenone

-
D
-
84553-28-6
1-Chloro-4-((E)-1,2-dichloro-vinyl)-benzene

-
E
-
84553-23-1
2-chloro-1-(4-chlorophenyl)butane-1,3-dione

-
F
-
937-20-2
p-chlorophenacyl chloride

| Conditions | Yield |
|---|---|
| With perchloric acid at 0 - 40℃; Kinetics; Mechanism; |


| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: t-BuOK / tetrahydrofuran / 2 h / Heating 1.2: 53 percent / tetrahydrofuran / 48 h / 20 °C 2.1: 92 percent / HCl / tetrahydrofuran; dioxane / 16 h / 20 - 70 °C View Scheme |
-
-
78704-49-1
1-(4-chlorophenyl)-2-trimethylsilylacetylene

-
-
937-20-2
p-chlorophenacyl chloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 2: 21 percent / n-Bu4NCl / CH2Cl2 / 72 h / Ambient temperature View Scheme |
-
-
937-20-2
p-chlorophenacyl chloride

-
-
178460-78-1
(S)-2-chloro-1-(4-chlorophenyl)ethan-1-ol

| Conditions | Yield |
|---|---|
| With bis(1,5-cyclooctadiene)diiridium(I) dichloride; f-amphox; hydrogen; potassium carbonate In ethanol; hexane at 25 - 30℃; under 38002.6 Torr; for 4h; Inert atmosphere; Glovebox; Autoclave; enantioselective reaction; | 99% |
| With D-glucose; D-glucose dehydrogenase; NADPH; potassium phosphate buffer; recombinant ketoreductase In water at 20℃; pH=7.0; | 97% |
| With glucose dehydrogenase; pET28-bmgdh-dhcr; sodium carbonate In aq. phosphate buffer; toluene at 30℃; for 24h; pH=6.5; Green chemistry; Enzymatic reaction; enantioselective reaction; | 89% |
-
-
937-20-2
p-chlorophenacyl chloride

| Conditions | Yield |
|---|---|
| With hydrogen; Ru(OTf)[(S,S)-TsDPEN](η6-mesitylene) In methanol at 30℃; under 7600 Torr; for 15h; | 99% |
| With formic acid; C25H29N2O3RuS; potassium formate In water; ethyl acetate at 60℃; for 4h; Inert atmosphere; enantioselective reaction; | 94% |
| With dimethylsulfide borane complex; (1R,2S,3R,5R)-2-(1',3',2'-dioxaborolan-2'-yloxy)apopinan-3-amine In tetrahydrofuran at 20℃; for 1h; enantioselective reaction; | 92% |

-
-
611-19-8
1-chloro-2-(chloromethyl)benzene

-
-
937-20-2
p-chlorophenacyl chloride

-
-
133001-02-2
1-chloro-2-(4-chlorophenyl)-3-(2-chlorophenyl)propan-2-ol

| Conditions | Yield |
|---|---|
| With magnesium In toluene | 99% |

-
-
937-20-2
p-chlorophenacyl chloride

-
-
1569467-56-6
1-(4-(4-chlorophenyl)thiazol-2-yl)-2-cyclooctylidenehydrazine

| Conditions | Yield |
|---|---|
| In ethanol at 20℃; | 99% |

| Conditions | Yield |
|---|---|
| In ethanol; water at 20℃; for 4h; | 99% |

| Conditions | Yield |
|---|---|
| With methanol; sodium at 20℃; for 0.25h; Darzens Condensation; | 99% |
-
-
937-20-2
p-chlorophenacyl chloride

-
-
636-26-0
5-methyl-2-thiouracil

-
-
119760-69-9
2-(4-chlorobenzoylmethylthio)-5-methylpyrimidin-4(1H)-one sodium salt

| Conditions | Yield |
|---|---|
| With sodium hydroxide In ethanol for 0.333333h; Heating; | 98% |
-
-
288-88-0
1,2,4-Triazole

-
-
937-20-2
p-chlorophenacyl chloride

-
-
58905-19-4
1-(4-chlorophenyl)-2-(1H-1,2,4-triazol-1-yl)ethanone

| Conditions | Yield |
|---|---|
| Stage #1: 1,2,4-Triazole With sodium carbonate In toluene at 20℃; for 0.5h; Stage #2: p-chlorophenacyl chloride In toluene Solvent; Reflux; | 98% |
| Stage #1: 1,2,4-Triazole With sodium carbonate In toluene at 20℃; for 0.5h; Stage #2: p-chlorophenacyl chloride In toluene Solvent; Reflux; | 98% |
| With potassium carbonate In acetonitrile at 85℃; for 0.833333h; microwave irradiation; | 90% |
-
-
108-90-7
chlorobenzene

-
-
937-20-2
p-chlorophenacyl chloride

-
-
51490-05-2
1,2-bis(4-chlorophenyl)ethanone

| Conditions | Yield |
|---|---|
| Stage #1: chlorobenzene; p-chlorophenacyl chloride With aluminum (III) chloride at 20 - 50℃; for 14.25h; Friedel-Crafts Acylation; Cooling with ice; Inert atmosphere; Stage #2: With hydrogenchloride In dichloromethane; water | 98% |
| With aluminum (III) chloride In dichloromethane at 20℃; | 89% |
-
-
937-20-2
p-chlorophenacyl chloride

-
-
1752-30-3
Acetone thiosemicarbazone

-
-
163107-37-7
1-(4-(4-chlorophenyl)thiazol-2-yl)-2-(propan-2-ylidene)hydrazine

| Conditions | Yield |
|---|---|
| In ethanol at 20℃; | 98% |
| In ethanol at 20 - 50℃; |

-
-
937-20-2
p-chlorophenacyl chloride

-
-
1569467-26-0
1-(4-(4-chlorophenyl)thiazol-2-yl)-2-(hex-5-en-2-ylidene)hydrazine

| Conditions | Yield |
|---|---|
| In ethanol at 20℃; | 98% |
-
-
5419-96-5
furan-2-aldehyde thiosemicarbazone

-
-
937-20-2
p-chlorophenacyl chloride

-
-
307325-12-8
1-(4-(4-chlorophenyl)thiazol-2-yl)-2-(furan-2-ylmethylene)hydrazine

| Conditions | Yield |
|---|---|
| In ethanol at 20℃; | 98% |
-
-
937-20-2
p-chlorophenacyl chloride

-
-
6868-28-6
2-((1H-indol-3-yl)methylene)hydrazine-1-carbothioamide

-
-
307544-76-9
1-((1H-indol-3-yl)methylene)-2-(4-(4-chlorophenyl)thiazol-2-yl)hydrazine

| Conditions | Yield |
|---|---|
| In ethanol at 20℃; | 98% |
-
-
133477-39-1
2-[1-(2-naphthenyl)ethyledene]hydrazinecarbothioamide

-
-
937-20-2
p-chlorophenacyl chloride

-
-
36941-40-9
1-(4-(4-chlorophenyl)thiazol-2-yl)-2-(1-(naphthalen-2-yl)ethylidene)hydrazine

| Conditions | Yield |
|---|---|
| In ethanol at 20℃; | 98% |
-
-
52831-78-4
2-(1-(2-oxo-2H-chromen-3-yl)ethylidene)hydrazine-1-carbothioamide

-
-
937-20-2
p-chlorophenacyl chloride

-
-
1359132-64-1
1-(4-(4-chlorophenyl)thiazol-2-yl)-2-(1-(2H-chromen-2-oxo-3-yl)ethylidene)hydrazine

| Conditions | Yield |
|---|---|
| In ethanol at 20℃; | 98% |

| Conditions | Yield |
|---|---|
| With methanol; sodium at 20℃; for 0.25h; Darzens Condensation; | 98% |
-
-
16629-40-6
4-phenyl-5-(pyridin-4-yl)-2,4-dihydro-3H-1,2,4-triazole-3-thione

-
-
937-20-2
p-chlorophenacyl chloride

-
-
325693-94-5
1-(4-chlorophenyl)-2-[(4-phenyl-5-pyridin-4-yl-4H-1,2,4-triazol-3-yl)thio]ethanone

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone for 3h; Heating / reflux; | 97% |
-
-
1752-38-1
2-octanone thiosemicarbazone

-
-
937-20-2
p-chlorophenacyl chloride

-
-
384808-58-6
1-(4-(4-chlorophenyl)thiazol-2-yl)-2-(octan-2-ylidene)hydrazine

| Conditions | Yield |
|---|---|
| In ethanol at 20℃; | 97% |
-
-
1448514-14-4
C9H15N3S

-
-
937-20-2
p-chlorophenacyl chloride

-
-
1569467-49-7
1-(4-(4-chlorophenyl)thiazol-2-yl)-2-(4,4-dimethylcyclohex-2-enylidene)hydrazine

| Conditions | Yield |
|---|---|
| In ethanol at 20℃; | 97% |
-
-
387847-19-0
C9H17N3S

-
-
937-20-2
p-chlorophenacyl chloride

-
-
1569467-62-4
1-(4-(4-chlorophenyl)thiazol-2-yl)-2-(1-cyclohexylethylidene)hydrazine

| Conditions | Yield |
|---|---|
| In ethanol at 20℃; | 97% |
-
-
5351-91-7
thiophene-2-carbaldehyde thiosemicarbazone

-
-
937-20-2
p-chlorophenacyl chloride

-
-
464224-97-3
1-(4-(4-chlorophenyl)thiazol-2-yl)-2-(thiophen-2-ylmethylene)hydrazine

| Conditions | Yield |
|---|---|
| In ethanol at 20℃; | 97% |
-
-
2302-93-4
acetophenonethiosemicarbazone

-
-
937-20-2
p-chlorophenacyl chloride

-
-
36937-52-7
1-(4-(4-chlorophenyl)thiazol-2-yl)-2-(1-phenylethylidene)hydrazine

| Conditions | Yield |
|---|---|
| In ethanol at 20℃; | 97% |
-
-
5351-81-5
1-(naphthalen-1-ylmethylene)thiosemicarbazide

-
-
937-20-2
p-chlorophenacyl chloride

-
-
464224-98-4
1-(4-(4-chlorophenyl)thiazol-2-yl)-2-(naphthalen-1-ylmethylene)hydrazine

| Conditions | Yield |
|---|---|
| In ethanol at 20℃; | 97% |
-
-
95-54-5
1,2-diamino-benzene

-
-
937-20-2
p-chlorophenacyl chloride

-
-
17286-63-4
2-(4-chlorophenyl)quinoxaline

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate In dimethyl sulfoxide at 120℃; for 24h; | 97% |
| With Saccharomyces cerevisiae In acetonitrile at 25℃; for 18h; Green chemistry; | 82% |

| Conditions | Yield |
|---|---|
| With sodium methylate In methanol at 15℃; for 0.25h; Darzens Condensation; | 97% |

| Conditions | Yield |
|---|---|
| With bis[dichloro(pentamethylcyclopentadienyl)iridium(III)]; sodium fluoride In methanol at 90℃; for 24h; | 97% |

| Conditions | Yield |
|---|---|
| In isopropyl alcohol for 0.0833333h; Heating; | 96% |

-
-
937-20-2
p-chlorophenacyl chloride

-
-
19339-59-4
1-(4-chloro-phenyl)-2-thiocyanato-ethanone

| Conditions | Yield |
|---|---|
| In water; isopropyl alcohol at 20℃; for 27h; | 96% |
-
-
555-90-8
pyridine-3-carbaldehyde thiosemicarbazone

-
-
937-20-2
p-chlorophenacyl chloride

-
-
328127-98-6
1-(4-(4-chlorophenyl)thiazol-2-yl)-2-(pyridin-3-ylmethylene)hydrazine

| Conditions | Yield |
|---|---|
| In ethanol at 20℃; | 96% |
2,4'-Dichloroacetophenone Chemical Properties
IUPAC Name: 2-Chloro-1-(4-chlorophenyl)ethanone
Following is the structure of 2,4'-Dichloroacetophenone (CAS NO.937-20-2):
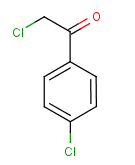
Empirical Formula: C8H6Cl2O
Molecular Weight: 189.0386 g/mol
EINECS: 213-323-9
Index of Refraction: 1.55
Molar Refractivity: 46.02 cm3
Molar Volume: 144.2 cm3
Density: 1.31 g/cm3
Flash Point: 119.1 °C
Melting point 100-102 °C
Surface Tension: 41.7 dyne/cm
Enthalpy of Vaporization: 50.82 kJ/mol
Boiling Point: 270 °C at 760 mmHg
Vapour Pressure: 0.00702 mmHg at 25 °C
Product Categories of 2,4'-Dichloroacetophenone (CAS NO.937-20-2): Benzene series; C7 to C8; Carbonyl Compounds; Ketones
Solubility in water of 2,4'-Dichloroacetophenone (CAS NO.937-20-2): Insoluble
Canonical SMILES: C1=CC(=CC=C1C(=O)CCl)Cl
InChI: InChI=1S/C8H6Cl2O/c9-5-8(11)6-1-3-7(10)4-2-6/h1-4H,5H2
InChIKey: FWDFNLVLIXAOMX-UHFFFAOYSA-N
2,4'-Dichloroacetophenone Toxicity Data With Reference
2,4'-Dichloroacetophenone (CAS NO.937-20-2) hasn't been listed as a carcinogen by ACGIH, IARC, NTP, or CA Prop 65. And the toxicological properties have not been fully investigated.
2,4'-Dichloroacetophenone Safety Profile
Hazard Codes:  Xi
Xi
Risk Statements: 36/37/38
R36/37/38:Irritating to eyes, respiratory system and skin.
Safety Statements: 26-36-37/39
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S36/37/39:Wear suitable protective clothing, gloves and eye/face protection.
WGK Germany: 3
2,4'-Dichloroacetophenone Specification
2,4'-Dichloroacetophenone , its cas register number is 937-20-2. It also can be called 2-Chloro-1-(4-chlorophenyl)ethanone ; Ethanone, 2-chloro-1-(4-chlorophenyl)- .
2,4'-Dichloroacetophenone (CAS NO.937-20-2) is hazardous, so the first aid measures and others should be known. Such as: When on the skin: first, should flush skin with plenty of water immediately for at least 15 minutes while removing contaminated clothing. Secondly, get medical aid. Or in the eyes: Flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids. Then get medical aid soon. While, it's inhaled: Remove from exposure and move to fresh air immediately. Give artificial respiration while not breathing. When breathing is difficult, give oxygen. And as soon as to get medical aid. Then you have the ingesting of the product : Wash mouth out with water, and get medical aid immediately.
2,4'-Dichloroacetophenone (CAS NO.937-20-2) should avoid the condition like incompatible materials, dust generation. It is not compatible with strong oxidizing agents. And also prevent it to broken down into hazardous decomposition products: hydrogen chloride, carbon monoxide, carbon dioxide. However, its hazardous polymerization has not been reported. Store in a tightly closed container. In addition, 2,4'-Dichloroacetophenone (CAS NO.937-20-2) should be stored in a cool, dry, well-ventilated area away from incompatible substances.
Related Products
- 20,22-DIHYDRODIGITOXIN
- 20,29,30-Trinorlupane,(17alpha)-
- 20-ETHYL-6-β,8-DIHYDROXY-1-α-METHOXY-4-METHYLHETERATISAN-14-ONE
- 20-Ethylprostaglandin F2-alpha
- 20-Isopropylcholanthrene
- 20-METHYLCHOLANTHREN-15-ONE
- 20-METHYLCHOLANTHRENE PICRATE
- 20-METHYLCHOLANTHRENE-TRINITRO-BENZENE
- 20(S)-Ginsenoside C-K
- 2,10-DIFLUOROBENZO(rst)PENTAPHENE
- 937207-71-1
- 93720-89-9
- 93-72-1
- 937265-83-3
- 93729-77-2
- 937-30-4
- 937369-38-5
- 937369-39-6
- 937370-61-1
- 937370-62-2
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View