Dayang Chem (Hangzhou) Co.,Ltd.
As a leading manufacturer and supplier of chemicals in China, DayangChem not only supply popular chemicals, but also DayangChem's R&D center offer custom synthesis services. DayangChem can provide different quantities of custom synthesis ch
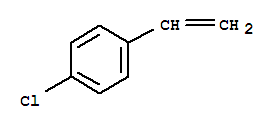
Cas:1073-67-2
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquirySimagchem Corporation
Welcome to Simagchem, your partner in China as a premier supply of bulk specialty chemicals for industry and life science. We introduce experienced quality product and exceptional JIT service with instant market intelligence in China to benefit our
Cas:1073-67-2
Min.Order:1 Metric Ton
Negotiable
Type:Manufacturers
inquiryAlity Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Chemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Cas:1073-67-2
Min.Order:5 Kiloliter
FOB Price: $1.2 / 5.0
Type:Manufacturers
inquiryHenan Tianfu Chemical Co., Ltd.
Our company engages in Sodium Tripolyphosphate (STPP) and Sodium Hexametabphosphate (SHMP) production; development of noble metal catalysts, synthesis of electronic chemical materials and general chemicals Imp&Exp trading business. The company
Cas:1073-67-2
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryZhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:1073-67-2
Min.Order:1 Kilogram
FOB Price: $139.0 / 210.0
Type:Trading Company
inquiryHenan Sinotech Import&Export Corporation
p-Chlorostyrene 4-Chlorostyrene; 1-Chloro-4-ethenyl-benzen; 1-Chloro-4-ethenylbenzene; 1-Chloro-4-vinylbenzene; 4-Chloro-styren; Benzene, 4-chloro-1-ethenyl- ; benzene; 1-Chloro-4-ethenyl-; Parachlorostyren
Qingdao Beluga Import and Export Co., LTD
4-Chlorostyrene CAS:1073-67-2 Qingdao Belugas Import and Export Co., Ltd. is a scientific and technological company integrating research and development, production and trade of chemical intermediates, specializing in high quality organic intermedi
Cas:1073-67-2
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryHenan Wentao Chemical Product Co., Ltd.
Henan Wentao Chemical Product Co.,Ltd is Located in Zhengzhou High-tech Development Zone with import and export license, We passed ISO 9001:2008 as well, Henan Wentao has developed more than 1000 compounds, which are widely used in the fields of prod
Cas:1073-67-2
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryHangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Zibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:1073-67-2
Min.Order:10 Gram
Negotiable
Type:Lab/Research institutions
inquirySiwei Development Group Ltd.
Product name: 4-Chlorostyrene CAS No.:1073-67-2 Molecule Formula:C8H7Cl Molecule Weight:138.59 Purity: 96% Package: 25kg/drum Description:Yellow liquid Manufacture Standards:Enterprise Standard TESTING ITEMS SPECI
Cas:1073-67-2
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryXiamen Jenny Chemical Technology Co., Ltd.
GMP standard, high purity, competitive price, in stock 1. Quick Response: within 6 hours after receiving your email. 2. Quality Guarantee: All products are strictly tested by our QC, confirmed by QA, and approved by a third-party lab in China, USA,
Changchun Artel lmport and Export trade company
My Company Changchun Artel Import and Export trade companyImport and Export Trade Co., Ltd.is located in Changchun, a national historical and cultural city, Jilin Province, China. It covers an area of 22,000 square meters, construction area
Cas:1073-67-2
Min.Order:20 Metric Ton
Negotiable
Type:Trading Company
inquiryShandong Mopai Biotechnology Co., LTD
Shandong Mopai Biotechnology Co., LTD is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemicals. W
KAISA GROUP INC
1.Applied in food field.it can improve the immune system and prolong life. 2.Appliedin cosmetic field.it can improve the skin care. 3.Applied in pharmaceutical field.it can treat various dieases. 4.Our product quality assurance will make our customer
Cas:1073-67-2
Min.Order:1 Metric Ton
FOB Price: $7.0 / 8.0
Type:Trading Company
inquirySuzhou Health Chemicals Co., Ltd.
High quality,stable supply chain.Appearance:white/off-white or light yellow Storage:Store in cool and dry place, keep away from strong light and heat. Package:aluminum bottle,glass bottle,PTFE bottle,cardboard drum Application:This product can be use
Win-Win chemical Co.Ltd
Stock products, own laboratory Package:Grams, Kilograms Application:For R&D Transportation:According to customer request Port:Shanghai
Cas:1073-67-2
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryKono Chem Co.,Ltd
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:drum and bag Application:for pharma use Transportation:by sea or air Port:Beijing or Guangzhou
Hangzhou Dingyan Chem Co., Ltd
R & D enterprises have their own stock in stockAppearance:To be subject to the object Package:Customized Application:pharmaceutical intermediates Transportation:Air Port:Shanghai;Guangzhou
GIHI CHEMICALS CO.,LIMITED
high purity,in stock Package:25kg/drum,or as per customers'demand Application:API,Pharmaceutical intermediates Transportation:air,sea,courier
Wuhan Fortuna Chemical Co.,Ltd
1.High quality : the purity is 99% min . through multiple producing procedures. 2.Competitive price : low price because of our skilled production technolpgy ,save the production cost at most , and give big profit room to our customers 57.Safe and fa
DB BIOTECH CO., LTD
Q1: Are you a manufacturer Answer: Yes, we are factory founded on 2012.Q2: How to contact with us Click "contact supplier" And then send us message the product you interest in, you will get reply within 12 hours.Q3:Which kind of payment terms do you
Shanghai Acmec Biochemical Technology Co., Ltd.
Acmec is a leading manufacturer and supplier of biochemical reagents and life science products. We have over 40,000 items in stock (real-time inventory) and offer discounted prices to registered members of the online store ( www.acmec.com.cn ) Appea
Cas:1073-67-2
Min.Order:1 bottle
Negotiable
Type:Lab/Research institutions
inquiryHenan Allgreen Chemical Co.,Ltd
high quality Storage:Sealed, dry, microtherm , avoid light and smell. Package:According to the demand of customer Application:Organic synthesis Transportation:by air or by sea
Aecochem Corp.
Our clients, like BASF,CHEMO,Brenntag,ASR,Evonik,Merck and etc.Appearance:COA Storage:in stock Application:MSDS/TDS
Hubei Taiho Chemical Co.,LTD
TAIHO Unique Advantages:1.We're factory2.Free samples available3.Commodity inspection can be done4.ISO9001,Kosher certifications5.10 years experiences Storage:Store in cool &dry place Package:aluminium foil bag/fiber can/plastic drum Application:comp
Hunan chemfish Pharmaceutical co.,Ltd
Hunan chemfish Pharmaceutical co.,Ltd.located in Lugu High-tech industral park ,Hunan province . with its own R&D center and more than 10000㎡manufacture plant . Chemfish owns 40 reactors from 1000L to 8000L. With complete auxiliary equipment as
Chemsigma International Co.,Ltd.
bulk productiongoods in stockswe have the best competitive price in the marketmeantime,we are committed to service to our every customer Chemsigma International Co.,Ltd. is a chemical manufacturer, specialize in custom synthesis and organic chemical
Antimex Chemical Limied
Ansciep Chemical is a professional enterprise manufacturing and distributing fine chemicals and speciality chemicals. We have been dedicated to heterocycle compounds and phenyl rings for tens of years. This is our mature product for export. Our quali
Synthetic route

| Conditions | Yield |
|---|---|
| With N-n-butyl-N-methylpiperidine bis(trifluoromethanesulfonyl)imide salt; sodium hydroxide at 160℃; for 5h; | 99% |
| With copper In quinoline | |
| With ferulic acid decarboxylase from Saccharomyces cerevisiae In aq. phosphate buffer; dimethyl sulfoxide at 25℃; pH=7.4; Kinetics; Enzymatic reaction; |

| Conditions | Yield |
|---|---|
| Stage #1: para-Chlorobenzyl alcohol With triethylphosphine hydrobromide at 100℃; for 0.166667h; Microwave irradiation; Stage #2: formaldehyd With potassium carbonate In water at 100℃; for 0.0833333h; Microwave irradiation; | 97% |

| Conditions | Yield |
|---|---|
| With hydrogen In toluene at 20℃; under 22801.5 Torr; for 12h; Autoclave; chemoselective reaction; | 96% |
| With piperazine; NiO doped titania In methanol at 20℃; for 3h; Inert atmosphere; Sealed tube; UV-irradiation; | 95% |
| With bis(1,5-cyclooctadiene)diiridium(I) dichloride; ethanol; 1,2-bis-(diphenylphosphino)ethane In tetrahydrofuran at 70℃; for 22h; Inert atmosphere; stereoselective reaction; | 91% |
-
-
23135-16-2
1,2-dibromo-1-(4-chlorophenyl)ethane

-
-
1073-67-2
4-vinylbenzyl chloride

| Conditions | Yield |
|---|---|
| With indium; cobalt(II) chloride hexahydrate In methanol at 20℃; for 1h; | 95% |
| With gallium; bis(cyclopentadienyl)titanium dichloride In tetrahydrofuran at 20℃; for 0.166667h; Inert atmosphere; Ultrasonic irradiation; chemoselective reaction; | 95% |
| With indium; niobium pentachloride In tetrahydrofuran at 20℃; for 0.166667h; Sonication; chemoselective reaction; | 95% |

-
-
1073-67-2
4-vinylbenzyl chloride

| Conditions | Yield |
|---|---|
| With molybdenum hexacarbonyl In toluene for 1h; Reflux; chemoselective reaction; | 95% |

| Conditions | Yield |
|---|---|
| With dichloro bis(acetonitrile) palladium(II) In N,N-dimethyl-formamide at 20℃; for 0.166667h; | 94% |
-
-
77295-59-1
1-chloro-4-(2,2-dibromovinyl)benzene

-
-
1073-67-2
4-vinylbenzyl chloride

| Conditions | Yield |
|---|---|
| With samarium In methanol at 45℃; for 6h; | 94% |
-
-
61078-14-6
1-methyl-2-(methylsulfonyl)-1H-benzo[d]imidazole

-
-
104-88-1
4-chlorobenzaldehyde

-
-
1073-67-2
4-vinylbenzyl chloride

| Conditions | Yield |
|---|---|
| With potassium tert-butylate In N,N-dimethyl-formamide at 20℃; for 1h; Inert atmosphere; | 93% |
| With potassium tert-butylate In N,N-dimethyl-formamide at 20℃; for 1h; Inert atmosphere; | 93% |

| Conditions | Yield |
|---|---|
| With sodium iodide; tin(ll) chloride In ethanol for 0.0666667h; Reflux; Green chemistry; | 92% |
| With carbon monoxide In tetrahydrofuran; water at 110℃; under 6840.46 Torr; for 2h; Autoclave; chemoselective reaction; | 85% |
| With iododioxobis(triphenylphosphine)rhenium(V) In toluene for 0.583333h; Reflux; chemoselective reaction; | 78% |

| Conditions | Yield |
|---|---|
| With Hoveyda-Grubbs catalyst second generation; di-μ-bromobis-(tritert-butylphosphine)dipalladium(I) In tetrahydrofuran at 60℃; under 7500.75 Torr; for 16h; Autoclave; | 92% |
-
-
104-88-1
4-chlorobenzaldehyde

-
-
18107-18-1
diazomethyl-trimethyl-silane

-
-
1073-67-2
4-vinylbenzyl chloride

| Conditions | Yield |
|---|---|
| With Rh(I)[PPh2CH2CH2Si(OEt)3]3Cl; triphenylphosphine; isopropyl alcohol In tetrahydrofuran at 25℃; for 4h; Reagent/catalyst; chemoselective reaction; | 87% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In methanol; water at 100℃; for 10h; Hiyama Coupling; Sealed tube; | 86% |
-
-
3020-28-8
(iodomethyl)triphenylphosphonium iodide

-
-
104-88-1
4-chlorobenzaldehyde

-
-
1073-67-2
4-vinylbenzyl chloride

| Conditions | Yield |
|---|---|
| With dibutyl telluride In tetrahydrofuran at 80℃; for 22h; | 85% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide; palladium diacetate In water at 120℃; for 0.166667h; Hiyama reaction; microwave irradiation; | 84% |
-
-
1779-49-3
Methyltriphenylphosphonium bromide

-
-
104-88-1
4-chlorobenzaldehyde

-
-
1073-67-2
4-vinylbenzyl chloride

| Conditions | Yield |
|---|---|
| With 1,8-diazabicyclo[5.4.0]undec-7-ene In dichloromethane for 5h; Wittig reaction; Heating; | 82% |
| Stage #1: Methyltriphenylphosphonium bromide With 1,8-diazabicyclo[5.4.0]undec-7-ene In dichloromethane for 0.5h; Reflux; Stage #2: 4-chlorobenzaldehyde In dichloromethane for 5h; Reflux; | 82% |
| Stage #1: Methyltriphenylphosphonium bromide With n-butyllithium In tetrahydrofuran for 0.25h; Stage #2: 4-chlorobenzaldehyde In tetrahydrofuran Wittig olefination; Further stages.; | 73% |

| Conditions | Yield |
|---|---|
| With 10H-phenothiazine; bis(μ-chloro)bis{(2-(1-(hydroxyimino)methyl)phenyl-C1,N)palladium(II)}; potassium acetate In N,N-dimethyl acetamide at 105℃; under 775.743 Torr; Heck-Mizoroki reaction; Inert atmosphere; | 82% |
| With sodium acetate; palladium diacetate; tris-(o-tolyl)phosphine In N,N-dimethyl-formamide at 90℃; under 7600.51 Torr; Heck reaction; Inert atmosphere; Autoclave; | |
| With N-Methyldicyclohexylamine; palladium diacetate; tetra-(n-butyl)ammonium iodide; tri tert-butylphosphoniumtetrafluoroborate In methanol; toluene at 130℃; under 7500.75 Torr; for 0.333333h; Heck Reaction; |

| Conditions | Yield |
|---|---|
| With caesium carbonate; triphenylphosphine; palladium dichloride In tetrahydrofuran; water at 85℃; for 4h; Suzuki-Miyaura reaction; | 81% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetonitrile at 20℃; Wittig Olefination; | 81% |
-
-
104586-14-3
[2-(4-Chloro-phenyl)-ethyl]-trimethyl-stannane

-
-
1073-67-2
4-vinylbenzyl chloride

| Conditions | Yield |
|---|---|
| With tris(4-methylphenyl)methylium tetrafluoroborate In 1,2-dichloro-ethane at 20℃; | 79% |

| Conditions | Yield |
|---|---|
| With potassium fluoride; 4 A molecular sieve; tetrabutyl-ammonium chloride; bis(dibenzylideneacetone)-palladium(0) In toluene for 24h; Ambient temperature; | 79% |

| Conditions | Yield |
|---|---|
| With sodium hydride In mineral oil at 135℃; for 10h; Inert atmosphere; Schlenk technique; | 79% |

| Conditions | Yield |
|---|---|
| With palladium diacetate; C22H27P; triethylamine In 1,4-dioxane; Hexadecane at 120℃; under 15001.5 Torr; for 20h; Heck Reaction; Schlenk technique; Inert atmosphere; | 79% |

| Conditions | Yield |
|---|---|
| at 140℃; for 4h; Ionic liquid; | 70% |
| With copper In quinoline at 185 - 195℃; Decarboxylation; | 50% |
| With quinoline; copper | |
| With hydrogen bromide; acetic acid at 100℃; Erwaermen des Reaktionsprodukts mit wss. Na2CO3-Loesung auf dem Dampfbad; |

-
A
-
1073-67-2
4-vinylbenzyl chloride

| Conditions | Yield |
|---|---|
| at 260℃; under 1 Torr; | A 70% B n/a |

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetonitrile at 20℃; Wittig Olefination; | 69% |

| Conditions | Yield |
|---|---|
| With ammonium chloride In tetrahydrofuran; nitrogen | 67.1% |
| With ammonium chloride In tetrahydrofuran; nitrogen | 67.1% |
| With ammonium chloride In dibutyl ether; nitrogen | 55.4% |

-
-
88691-45-6, 132536-25-5, 88691-39-8
(E)-[2-(4-chlorophenyl)ethenyl]trimethylsilane

-
-
123-62-6
propionic acid anhydride

-
A
-
1073-67-2
4-vinylbenzyl chloride

| Conditions | Yield |
|---|---|
| With bis(1,5-cyclooctadiene)diiridium(I) dichloride In toluene at 120℃; for 24h; Inert atmosphere; | A n/a B 67% |

-
-
29805-11-6
1-(4-chlorophenyl)prop-2-yn-1-ol

-
-
65-85-0
benzoic acid

-
A
-
1073-67-2
4-vinylbenzyl chloride

-
B
-
1241573-14-7
1-(4-chlorophenyl)-2-oxopropyl benzoate

| Conditions | Yield |
|---|---|
| With RuCl2(η-C6H6)(TPPMS) In water at 100℃; for 6h; Inert atmosphere; | A 6% B 65% |


| Conditions | Yield |
|---|---|
| With ammonium chloride In tetrahydrofuran; nitrogen | 64.8% |

-
-
1073-67-2
4-vinylbenzyl chloride

-
-
23135-16-2
1,2-dibromo-1-(4-chlorophenyl)ethane

| Conditions | Yield |
|---|---|
| With bromine In chloroform at 0℃; Inert atmosphere; | 100% |
| With Oxone; ammonium bromide In water; acetonitrile for 13h; Reflux; | 96% |
| With acetic acid; lithium bromide at 25℃; for 2h; Green chemistry; | 95% |

| Conditions | Yield |
|---|---|
| With N-Bromosuccinimide; water In acetonitrile at 20℃; | 100% |
| With trifluoroacetic acid; potassium bromide In water; dimethyl sulfoxide at 50℃; for 1.6h; Reagent/catalyst; Electrolysis; | 94% |
| With (2S,3S)-3-hydroxy-2-(4-methylphenylsulfonamido)butanoic acid; water; N-bromoacetamide In tert-butyl alcohol at 20℃; for 0.5h; regioselective reaction; | 84% |

| Conditions | Yield |
|---|---|
| With 1,1,1-trifluoro-2-propanone; dihydrogen peroxide | 100% |
| With tert.-butylhydroperoxide; 2C13H10N3O2(1-)*MoO2(2+) In methanol; dichloromethane for 1h; Catalytic behavior; Reagent/catalyst; | 100% |
| With tert.-butylhydroperoxide; cis-[MoO2(N'-(3-ethoxy-2-hydroxybenzylidene)-2-methoxybenzohydrazide-2H)(OH2)] In methanol; dichloromethane for 1h; Catalytic behavior; Reflux; | 100% |
-
-
1073-67-2
4-vinylbenzyl chloride

-
-
20433-10-7
5-(4-methoxyphenyl)-2-phenyl-2H-tetrazole

-
-
75745-49-2
5-(4-chlorophenyl)-3-(4-methoxyphenyl)-1-phenyl-4,5-dihydro-1H-pyrazole

| Conditions | Yield |
|---|---|
| In benzene for 2h; UV-irradiation; | 100% |
-
-
1073-67-2
4-vinylbenzyl chloride

| Conditions | Yield |
|---|---|
| bis(ethylene)rhodium(I) chloride dimer In toluene byproducts: 4-chlorophenylethane; under Ar; to a soln. of the Rh-complex in toluene was added the arylethene, reaction with the oxaazaborolidine for 4 h; distn.; elem. anal.; | 100% |
-
-
1073-67-2
4-vinylbenzyl chloride

| Conditions | Yield |
|---|---|
| With meso-tetraphenylporphyrin iron(III) chloride; cesium fluoride; zinc In N,N-dimethyl-formamide at 60℃; for 2h; Inert atmosphere; diastereoselective reaction; | 100% |

| Conditions | Yield |
|---|---|
| In dichloromethane at -40 - 20℃; Inert atmosphere; | 100% |


| Conditions | Yield |
|---|---|
| With hydrogen In ethanol at 60℃; under 3000.3 Torr; for 5h; | 99% |
| With methanol; palladium diacetate; 4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane In dichloromethane at 25℃; for 12h; Sealed tube; Inert atmosphere; chemoselective reaction; | 99% |
| With triethylsilane; 1% Pd on activated carbon In water at 20℃; for 2h; Reagent/catalyst; Green chemistry; chemoselective reaction; | 99% |
-
-
1073-67-2
4-vinylbenzyl chloride

-
-
55962-05-5
[N-(p-tolylsulfonyl)imino]phenyliodinane

-
-
97401-93-9
N-(p-toluenesulfonyl)-2-(4-chlorophenyl)aziridine

| Conditions | Yield |
|---|---|
| With [CuI(tris[(tetramethylguanidino)-phenyl]amine)][PF6] In acetonitrile for 2h; Inert atmosphere; Molecular sieve; | 99% |
| With copper-loaded dopamine-grafted Fe3O4 nanoparticles In acetonitrile at 70℃; for 2h; Inert atmosphere; Microwave irradiation; Green chemistry; | 96% |
| With [PhB{CH2P(p-CF3Ph)2}3]Cu(PPh3) In dichloromethane at 20℃; for 5h; Inert atmosphere; Schlenk technique; | 95% |
-
-
1073-67-2
4-vinylbenzyl chloride

-
-
6773-29-1
dimethyl diazomalonate

-
-
127604-92-6, 132145-65-4
dimethyl 2-(4-chlorophenyl)cyclopropane-1,1-dicarboxylate

| Conditions | Yield |
|---|---|
| Stage #1: 4-vinylbenzyl chloride With bis[rhodium(α,α,α',α'-tetramethyl-1,3-benzenedipropionic acid)] In dichloromethane at 0℃; for 0.0833333h; Inert atmosphere; Stage #2: dimethyl diazomalonate In dichloromethane at 0 - 23℃; Inert atmosphere; | 99% |
| Stage #1: 4-vinylbenzyl chloride; dimethyl diazomalonate In dichloromethane at 0 - 22℃; Inert atmosphere; Stage #2: With tetrakis(hydroxymethyl)phosphonium hydroxide In benzene at 60℃; for 12h; | 99% |
| With bis{rhodium[3,3'-(1,3-phenylene)bis(2,2-dimethylpropanoic acid)]} In dichloromethane at 20℃; for 2h; Inert atmosphere; | 90% |

| Conditions | Yield |
|---|---|
| With water; 3-chloro-benzenecarboperoxoic acid at 30℃; for 2h; | 99% |
| With dihydrogen peroxide; ethyl acetate; lipase [PSLG6] In water at 60℃; for 0.0833333h; microwave irradiation; | 90% |
| With osmium(VIII) oxide; 4-methylmorpholine N-oxide In water; acetone at 0 - 20℃; for 18h; Inert atmosphere; | 90% |

| Conditions | Yield |
|---|---|
| With sodium periodate; C53H44As2N2O3Ru In water; ethyl acetate; acetonitrile at 25℃; for 0.5h; | 99% |
| With sodium periodate; C22H23ClIN2Os(1+)*F6P(1-) In water; tert-butyl alcohol at 60℃; for 1h; Schlenk technique; Inert atmosphere; | 99% |
| With sodium periodate; C18H15ClFN2Ru(1+)*Cl(1-) In water; tert-butyl alcohol at 60℃; for 1h; Catalytic behavior; Schlenk technique; Inert atmosphere; | 99% |
-
-
1073-67-2
4-vinylbenzyl chloride

-
-
1122-91-4
4-bromo-benzaldehyde

-
-
52792-18-4
(E)-4-(4-chlorostyryl)benzaldehyde

| Conditions | Yield |
|---|---|
| With sodium acetate In N,N-dimethyl-formamide for 3h; Heck Reaction; Schlenk technique; Inert atmosphere; Reflux; | 99% |
| With C28H34ClN3O5Pd; sodium carbonate In 1-methyl-pyrrolidin-2-one at 120℃; for 20h; Heck Reaction; | 95% |
| With sodium acetate; tetraphenylphosphonium bromide; palladium dichloride In N,N-dimethyl-formamide at 140℃; for 1.5h; Arylation; Heck reaction; | 99 % Chromat. |
| With palladium diacetate; triethylamine; tris-(o-tolyl)phosphine In N,N-dimethyl acetamide at 140℃; Heck Reaction; |
-
-
1073-67-2
4-vinylbenzyl chloride

| Conditions | Yield |
|---|---|
| With bis(1,5-cyclooctadiene)rhodium(I) tetrafluoroborate; hydrogen; iodine; 2,2'-bis-(diphenylphosphino)-1,1'-binaphthyl at 20℃; under 30003 Torr; for 6h; | 99% |
| With polymethylhydrosiloxane; iodine In chloroform at 20℃; for 0.333333h; | 95% |
| With phosphoric acid; iodine Inert atmosphere; Schlenk technique; Green chemistry; | 84% |
| With triethylsilane; tetrafluoroboric acid; iodine; copper(II) oxide In dichloromethane at -30℃; for 3h; | 61% |

| Conditions | Yield |
|---|---|
| Stage #1: 4-vinylbenzyl chloride; 4-cyanobenzaldehyde With phenylborondichloride In dichloromethane at 25℃; for 12h; Stage #2: With water | 99% |
-
-
1073-67-2
4-vinylbenzyl chloride

-
-
25015-63-8
4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane

| Conditions | Yield |
|---|---|
| With bis(1,5-cyclooctadiene)diiridium(I) dichloride; 1,4-di(diphenylphosphino)-butane In tetrahydrofuran at 20℃; for 24h; | 99% |
| With bis(diphenylphosphino)butane; bis(1,5-cyclooctadiene)diiridium(I) dichloride In tetrahydrofuran under N2; pinacol borane was reacted with ligand in THF at room temp. for 24 h in the presence of Ir-complex and bis(diphenylphosphino)butane; chromy.; | 99% |
| With sodium triethylborohydride; scandium tris(trifluoromethanesulfonate) In tetrahydrofuran; toluene at 100℃; for 24h; Microwave irradiation; Inert atmosphere; regioselective reaction; | 96% |
-
-
1073-67-2
4-vinylbenzyl chloride

-
-
35059-50-8
t-butyl diazoacetate

| Conditions | Yield |
|---|---|
| With C67H51IrN2O2 In tetrahydrofuran at -78℃; for 24.1667h; optical yield given as %ee; enantioselective reaction; | 99% |
| chiral phenyliridium(III)-salen complex In tetrahydrofuran at -78 - 20℃; | 99 % Spectr. |
-
-
1073-67-2
4-vinylbenzyl chloride

-
-
591-50-4
iodobenzene

-
-
1657-49-4, 1657-50-7, 4714-23-2, 130059-92-6, 130059-96-0
1-chloro-4-styrylbenzene

| Conditions | Yield |
|---|---|
| With tetrabutyl ammonium fluoride; palladium diacetate; potassium carbonate at 70℃; for 36h; Heck reaction; air; | 99% |
| With triethylamine In water; acetonitrile at 60℃; for 2h; Heck Reaction; | 98% |
| With potassium hydroxide In water; N,N-dimethyl-formamide at 90℃; for 2h; | 96% |

| Conditions | Yield |
|---|---|
| With tris-(dibenzylideneacetone)dipalladium(0); 2,5-di(2,4,6-trimethylphenyl)-1,2,5-thiadiazolidine-1-oxide; triethylamine In N,N-dimethyl-formamide at 200℃; for 0.166667h; Heck reaction; Microwave irradiation; | 99% |
| With tris-(dibenzylideneacetone)dipalladium(0); 2,5-di(2,4,6-trimethylphenyl)-1,2,5-thiadiazolidine-1-oxide; triethylamine In N,N-dimethyl-formamide at 200℃; for 0.166667h; Mizoroki-Heck reaction; Microwave irradiation; | 99% |
| With potassium phosphate tribasic trihydrate In N,N-dimethyl-formamide at 120℃; for 4h; Heck Reaction; | 99% |

| Conditions | Yield |
|---|---|
| With tris-(dibenzylideneacetone)dipalladium(0); 2,5-di(2,4,6-trimethylphenyl)-1,2,5-thiadiazolidine-1-oxide; tetrabutylammomium bromide; sodium acetate In 1-methyl-pyrrolidin-2-one at 200℃; for 0.5h; Heck reaction; Microwave irradiation; | 99% |
| With 2,5-di(2,4,6-trimethylphenyl)-1,2,5-thiadiazolidine-1-oxide; tetrabutylammomium bromide; sodium acetate; bis(dibenzylideneacetone)-palladium(0) In 1-methyl-pyrrolidin-2-one at 200℃; for 0.5h; Mizoroki-Heck reaction; Microwave irradiation; | 99% |
| With potassium phosphate; C34H46Br4N8Pd2 In N,N-dimethyl-formamide at 110℃; for 4h; Heck Reaction; | 80% |
| With tetrabutylammomium bromide; palladium diacetate; potassium carbonate; 1-[phenyl(piperidin-1-yl)methyl]naphthalen-2-ol In N,N-dimethyl acetamide at 140℃; for 40h; Heck Reaction; Inert atmosphere; |
-
-
623-73-4
diazoacetic acid ethyl ester

-
-
1073-67-2
4-vinylbenzyl chloride

-
-
395683-39-3
ethyl (1R,2S)-2-(4-chlorophenyl)cyclopropane-1-carboxylate

| Conditions | Yield |
|---|---|
| With C20H21ClN3ORh; silver trifluoromethanesulfonate In dichloromethane at 0℃; Inert atmosphere; diastereoselective reaction; | 99% |
-
-
919299-02-8
3-diazo-3,6-dihydro-2H-pyran-2-one

-
-
1073-67-2
4-vinylbenzyl chloride

-
-
1161824-01-6
C13H11ClO2

| Conditions | Yield |
|---|---|
| Stage #1: 3-diazo-3,6-dihydro-2H-pyran-2-one; 4-vinylbenzyl chloride In dichloromethane at -78℃; for 0.166667h; Molecular sieve; Inert atmosphere; Stage #2: With C67H51IrN2O2 In dichloromethane at -78℃; for 72h; Molecular sieve; Inert atmosphere; optical yield given as %ee; enantioselective reaction; | 99% |
-
-
1073-67-2
4-vinylbenzyl chloride

-
-
98-80-6
phenylboronic acid

-
-
1657-50-7
(E)-1-(4-chlorophenyl)-2-phenylethene

| Conditions | Yield |
|---|---|
| With C38H36Cl2Fe2N2Pd2; oxygen In N,N-dimethyl-formamide at 50℃; for 3h; Heck reaction; | 99% |
| With palladium diacetate; acetic anhydride; 2,3-dicyano-5,6-dichloro-p-benzoquinone In acetic acid at 90℃; for 27h; Heck reaction; | 99% |
| With [2,2]bipyridinyl; methanesulfonic acid; palladium(II) trifluoroacetate In N,N-dimethyl-formamide at 100℃; for 24h; Schlenk technique; | 95% |
-
-
1073-67-2
4-vinylbenzyl chloride

-
-
91-01-0
1,1-Diphenylmethanol

-
-
1104997-38-7
(E)-(3-(4-chlorophenyl)prop-2-ene-1,1-diyl)dibenzene

| Conditions | Yield |
|---|---|
| With trifluorormethanesulfonic acid In ethylene dibromide at 60℃; Air atmosphere; stereoselective reaction; | 99% |
| With N-octadecyl-N-(4-sulfobutyl)pyrrolidinium trifluoromethanesulfonate In dichloromethane at 80℃; for 12h; | 89% |
| With iron(III) chloride hexahydrate; toluene-4-sulfonic acid In dichloromethane at 45℃; for 24h; stereospecific reaction; | 80% |
-
-
1073-67-2
4-vinylbenzyl chloride

-
-
103-19-5
di(p-tolyl) disulfide

-
-
1313220-39-1
(1-(4-chlorophenyl)ethane-1,2-diyl)bis(p-tolylsulfane)

| Conditions | Yield |
|---|---|
| With iodine In water at 20℃; for 12h; | 99% |
-
-
1073-67-2
4-vinylbenzyl chloride

-
-
3240-34-4
[bis(acetoxy)iodo]benzene

-
-
1657-49-4, 1657-50-7, 4714-23-2, 130059-92-6, 130059-96-0
1-chloro-4-styrylbenzene

| Conditions | Yield |
|---|---|
| With palladium diacetate; potassium carbonate In PEG-400 at 60℃; for 18h; Heck type coupling reaction; under air; | 99% |
-
-
1073-67-2
4-vinylbenzyl chloride

-
-
1359763-70-4
3-methyl-1,3-diphenyl-4-(4-vinylphenyl)butan-1-one

| Conditions | Yield |
|---|---|
| With 1,4-bis(dicyclohexylphosphino)butane di-trifluoroborate; palladium diacetate; sodium t-butanolate In toluene at 110℃; for 14h; Inert atmosphere; | 99% |
-
-
1073-67-2
4-vinylbenzyl chloride

-
-
99-91-2
para-chloroacetophenone

-
-
63483-67-0
1-{4-[(E)-2-(4-chlorophenyl)ethenyl]phenyl}ethanone

| Conditions | Yield |
|---|---|
| With tetrakis[μ-1-[(3-methoxyphenyl)methyl]-2-phenyl-3-[2-oxo-2-[(2-phenolato-kO)aminokN]ethyl]-1H-imidazoliumato-kC4]tetrapalladium; tetrabutylammomium bromide; sodium acetate at 140℃; for 2h; Heck Reaction; | 99% |
| With tetrabutylammomium bromide; sodium acetate; C84H60N12O8Pd4 at 140℃; for 2h; Heck Reaction; Inert atmosphere; Schlenk technique; | 90% |
| With tetrabutylammomium bromide; palladium diacetate; silica gel; potassium carbonate for 1h; Neat (no solvent); | 77% |
| With [PdBr2(PPh3)2]; potassium carbonate In N,N-dimethyl-formamide at 20 - 100℃; for 24h; Heck Reaction; Inert atmosphere; stereoselective reaction; | 38% |
-
-
1073-67-2
4-vinylbenzyl chloride

-
-
70957-05-0
1-(N,N‑dimethylcarbamoyl)indole

-
-
1454704-55-2
(E)-2-(4-chlorostyryl)-N,N-dimethyl-1H-indole-1-carboxamide

| Conditions | Yield |
|---|---|
| With tris(acetonitrile)(η5-pentamethylcyclopentadienyl)rhodium(III) hexafluoroantimonate; copper(II) acetate monohydrate In tetrahydrofuran at 100℃; for 24h; Heck Reaction; Inert atmosphere; stereoselective reaction; | 99% |
| With silver hexafluoroantimonate; [ruthenium(II)(η6-1-methyl-4-isopropyl-benzene)(chloride)(μ-chloride)]2; copper(II) acetate monohydrate In 1,4-dioxane at 100℃; for 24h; Schlenk technique; Inert atmosphere; regioselective reaction; | 72% |
| With silver hexafluoroantimonate; [RhCl2(p-cymene)]2; oxygen; copper diacetate; sodium acetate In 1,2-dichloro-ethane at 20 - 100℃; under 760.051 Torr; for 15.25h; Sealed tube; Inert atmosphere; | 65% |
Related products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View
 Xi,
Xi, F
F