Dayang Chem (Hangzhou) Co.,Ltd.
DayangChem exported this product to many countries and regions at best price. If you are looking for the material's manufacturer or supplier in China, DayangChem is your best choice. Pls contact with us freely for getting detailed product spe
Cas:593-60-2
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquiryAlity Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Chemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Hebei yanxi chemical co.,LTD.
Hebei yanxi chemical co. LTD. has expanded a compositive entity from initially only as a small manufacturer. The company dedicated to the development, production and marketing of chemicals. After many years of efforts, we have established stable
Cas:593-60-2
Min.Order:1 Metric Ton
FOB Price: $1.0 / 2.0
Type:Trading Company
inquiryHenan Tianfu Chemical Co., Ltd.
Our company was built in 2009 with an ISO certificate.In the past 5 years, we have grown up as a famous fine chemicals supplier in China and we had established stable business relationships with Samsung,LG,Merck,Thermo Fisher Scientific and so on.Our
Cas:593-60-2
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryHenan Wentao Chemical Product Co., Ltd.
We are leading fine chemicals supplier in China with ISO certificate, Our main business covers the fields below: 1.Noble Metal Catalysts (Pt.Pd...) 2.Organic Phosphine Ligands (Tert-butyl-phosphine.Cyclohexyl-phosphine...) 3.OLED
Cas:593-60-2
Min.Order:1 Gram
FOB Price: $2.0
Type:Lab/Research institutions
inquiryHangzhou Keyingchem Co.,Ltd
Hangzhou KeyingChem Co., Ltd. exported this product to many countries and regions at best price. If you are looking for the material’s manufacturer or supplier in China, KeyingChem is your best choice. Pls contact with us freely for getting det
Hangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Zibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:593-60-2
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquiryHANGZHOU YUNUO CHEMICAL CO.,LTD
Superior quality, moderate price & quick delivery. Appearance:colourless liquid Storage:Stored in cool, dry and ventilation place; Away from fire and heat Package:25kg/drum, or as per your request. Application:Used as Pharmaceutical Intermediat
Zibo Dorne chemical technology co. LTD
Product Details Grade: pharmaceutical grade Purity:99%+ ProductionCapacity: 1000 Kilogram/Month Scope of use: For scientific research only(The product must be used legally) Our Advantage 1. Best quality with competitive price. 2. Quick shipping,
Siwei Development Group Ltd.
Product name: Vinylbromide Bromoethylene CAS No.:593-60-2 Molecule Formula:C2H3Br Molecule Weight:106.95 Purity: 98.0% Package: 200kg/drum Description:Colorless liquid Manufacture Standards:Enterprise Standard TEST
Cas:593-60-2
Min.Order:1 Kilogram
Negotiable
Type:Trading Company
inquiryHunan chemfish Pharmaceutical co.,Ltd
593-60-2 Appearance:95%+ Package:R&D,Pilot run Application:593-60-2 Transportation:per client require Port:Express ,Air, Sea
SAGECHEM LIMITED
SAGECHEM is a chemical R&D, manufacturing and distribution company in China since 2009, including pharmaceutical intermediates, agrochemical, dyestuff intermediates, organosilicone, API and etc. We also offer a full range of services in custom synthe
Zhuozhou Wenxi import and Export Co., Ltd
Product Description Description & Specification Category Pharmaceutical Raw Materials, Fine Chemicals, Bulk drug Standard Medical standard
Cas:593-60-2
Min.Order:1 Kilogram
FOB Price: $112.0
Type:Trading Company
inquiryAntimex Chemical Limied
Ansciep Chemical is a professional enterprise manufacturing and distributing fine chemicals and speciality chemicals. We have been dedicated to heterocycle compounds and phenyl rings for tens of years. This is our mature product for export. Our quali
Hubei Langyou International Trading Co., Ltd
CAS No.593-60-2 Vinyl bromide Application:CAS No.593-60-2 Vinyl bromide
Hunan Longxianng Runhui Trading Co.,Ltd
CAS No.593-60-2 Vinyl bromideAppearance:Powder Storage:2-8°C Package:1KG -200KG Drum with pallet Application:Pharma intermediates Transportation:By express (Door to door) such as FEDEX, DHL, EMS for small amount. By air(airport to airport) or by se
Cas:593-60-2
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryHunan Russell Chemicals Technology Co.,Ltd
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:Foil bag; Drum; Plastic bottle Application:Pharma;Industry;Agricultural Transportation:by sea or air Port:any port in China
Yurui(Shanghai)Chemical Co.,Ltd
How May We Serve You We can Supply HPLC\HMMR\MS report for you ,pls feel free to contact us! Capability on chemical synthesis ● Shanghai High-Tech Enterprises ● Strong R&D Team from USA, Japan, Korea ● 65000M2+ Factory, 300+ reac
Shanghai united Scientific Co.,Ltd.
United Scientific Company Located in Shanghai of China , is a competitive player in the global specialty and fine chemical market. Fenghua has both the expertise and flexibility to produce a wide range of chemicals. Focusing on developing the innovat
Shanghai AngewChem Co., Ltd.
Shanghai AngewChemCo., Ltd. is an innovative enterprise on fine chemicals and pharmaceuticals. Based on Shanghai R&D center and Hunan chemical manufacturing plant, we offer chemical research, process development, and large-scale production. Complete
changchun tuocai technology co.,ltld
CAS No.593-60-2 Vinyl bromideAppearance:Powder Storage:2-8°C Package:1KG -200KG Drum with pallet Application:Pharma intermediates Transportation:by sea or by air or by courier
Hebei Minshang Biotechnology Co., Ltd
CAS No.593-60-2 Vinyl bromide Application:Pharma intermediates
Shanghai Yanchu chemicals Co., Ltd
low price and good quality Application:It is an important raw material and intermediate used in Organic Synthesis, Pharmaceuticals, Agrochemicals and D
Hangzhou Fenjun Chemical Co Ltd.
Hangzhou Keying Chem Co., Ltd. Is a comprehensive enterprise, dedicated to the development, production and marketing of chemicals. As a technology innovative and service professional enterprise, Our company mainly engages in global pharmaceutical, ch
Synthetic route

| Conditions | Yield |
|---|---|
| With potassium hydroxide In ethanol at 40℃; for 1h; | 70% |
| With potassium hydroxide In ethanol Heating; | 56% |
| With potassium hydroxide at 60℃; |

| Conditions | Yield |
|---|---|
| With N,N-dimethyl-formamide; potassium bromide; trichlorophosphate In acetonitrile at 20℃; Reagent/catalyst; Sonication; | 60% |

-
-
60-29-7
diethyl ether

-
-
78-74-0
1,1,2-tribromoethane

-
A
-
108-86-1
bromobenzene

-
B
-
593-60-2
Vinyl bromide

-
C
-
92-52-4
biphenyl

-
D
-
71-43-2
benzene

| Conditions | Yield |
|---|---|
| Zersetzen des Reaktionsproduktes mit Wasser; |

-
-
557-91-5
1,1-Dibromoethane

-
-
64-17-5
ethanol

-
-
141-52-6
sodium ethanolate

-
A
-
593-60-2
Vinyl bromide

-
B
-
74-86-2
acetylene


| Conditions | Yield |
|---|---|
| With diethyl ether; phenylmagnesium bromide und Zers. des Reaktionsproduktes mit Wasser; |
-
-
856929-61-8
1,4-dibenzyl-2,2,5,5-tetramethyl-piperazine

-
-
106-93-4
ethylene dibromide

-
A
-
593-60-2
Vinyl bromide


| Conditions | Yield |
|---|---|
| at 120 - 125℃; |

| Conditions | Yield |
|---|---|
| With hydrogen bromide; copper(I) bromide at 60℃; | |
| With pyridine-base; hydrogen bromide; cadmium(II) bromide at 215 - 230℃; | |
| With BaBr2-charcoal; hydrogen bromide at 200℃; |

| Conditions | Yield |
|---|---|
| With mercury bromide; hydrogen bromide at 200℃; |
-
-
110-52-1
1,4-dibromo-butane

-
-
14604-48-9
vinyl cation

-
A
-
74-96-4
ethyl bromide

-
B
-
593-60-2
Vinyl bromide

-
C
-
74-85-1
ethene

-
D
-
74-86-2
acetylene

| Conditions | Yield |
|---|---|
| With hydrogen at 100℃; under 60 - 600 Torr; Product distribution; Kinetics; Thermodynamic data; labelled with tritium; -ΔH; |

-
-
74-96-4
ethyl bromide

-
A
-
593-60-2
Vinyl bromide

-
B
-
557-91-5
1,1-Dibromoethane

-
C
-
74-84-0
ethane

-
D
-
74-85-1
ethene

-
E
-
106-97-8
n-butane

| Conditions | Yield |
|---|---|
| at 24.9℃; Quantum yield; Irradiation; dependence of the product quantum yields on ethyl bromide pressure, photolysios time and additives (as N2, CF4, NO).; |


| Conditions | Yield |
|---|---|
| copper and nickel chlorides at 170℃; Equilibrium constant; Thermodynamic data; 200 - 250 deg C, ΔH(excit.), ΔS(excit.); |

| Conditions | Yield |
|---|---|
| With sulphur hexafluoride In gaseous matrix Rate constant; Thermodynamic data; Irradiation; effect of laser fluence, effect of partial pressure of SF6, energy distribution, activation energy; |


| Conditions | Yield |
|---|---|
| With bromine at 23.9 - 258.9℃; Kinetics; Irradiation; carrier gas, He, the role of resonance stabilization; |
-
-
359-63-7
N,N-bis(trifluoromethyl)hydroxylamine

-
-
106-93-4
ethylene dibromide

-
A
-
593-60-2
Vinyl bromide

-
B
-
6141-70-4
1,2-bisethane

| Conditions | Yield |
|---|---|
| With sodium hydride 1.) Et2O, -78 deg C, 0.5 h, 2.) diglyme, 50 deg C, 1 h; Yield given. Multistep reaction. Yields of byproduct given; |

-
-
57718-12-4
(6-iodohex-1-yn-1-yl)benzene

-
-
106-95-6
allyl bromide

-
A
-
593-60-2
Vinyl bromide

-
B
-
57718-21-5
(1-cyclopentylidene-3-butenyl)benzene

| Conditions | Yield |
|---|---|
| With tert.-butyl lithium 1.) pentane, Et2O, from -78 deg C to RT, 1 h, 2.) pentane, Et2O, -78 deg C; Yield given. Multistep reaction. Yields of byproduct given; |

-
-
563-79-1
2,3-Dimethyl-2-butene

-
-
4222-23-5
3-bromo-3-methyldiazirine

-
A
-
593-60-2
Vinyl bromide

-
B
-
96335-77-2
1-Bromo-1,2,2,3,3-pentamethyl-cyclopropane

| Conditions | Yield |
|---|---|
| In n-heptane Product distribution; Quantum yield; Mechanism; Irradiation; energy data: ΔH; | |
| In n-heptane at 80℃; Product distribution; Mechanism; Thermodynamic data; ΔH; |


| Conditions | Yield |
|---|---|
| With hydrogen bromide at -261.2℃; for 5.03333h; Product distribution; Irradiation; |


| Conditions | Yield |
|---|---|
| With bromine In methanol at 25℃; Mechanism; deuterium kinetic isotope effect; also deuterated ethylenes; other solvent; |
-
-
34557-54-5
methane

-
-
75-63-8
Bromotrifluoromethane

-
A
-
74-83-9
methyl bromide

-
B
-
593-60-2
Vinyl bromide

-
C
-
373-52-4
bromofluoromethane

-
D
-
75-46-7
trifluoromethan

-
E
-
75-38-7
Vinylidene fluoride

-
F
-
74-95-3
1,1-dibromomethane

| Conditions | Yield |
|---|---|
| Mn exchaged zeolite ZSM-5 at 599.85℃; Product distribution; other catalysts, temp.; |


| Conditions | Yield |
|---|---|
| Kinetics; Bromwasserstoffabspaltung; |

| Conditions | Yield |
|---|---|
| at 120 - 140℃; |
-
-
7732-18-5
water

-
-
10035-10-6, 12258-64-9
hydrogen bromide

-
-
74-86-2
acetylene

-
A
-
593-60-2
Vinyl bromide

-
B
-
557-91-5
1,1-Dibromoethane

| Conditions | Yield |
|---|---|
| at 100℃; under 80 - 240 Torr; Kinetics; |


| Conditions | Yield |
|---|---|
| at 200℃; |

-
-
107-21-1
ethylene glycol

-
-
106-93-4
ethylene dibromide

-
A
-
593-60-2
Vinyl bromide

-
B
-
762-49-2
2-fluoroethyl bromide

| Conditions | Yield |
|---|---|
| at 150℃; |


| Conditions | Yield |
|---|---|
| With (2R,3R)-2,3-(2,5,8,11-tetrathiadodecamethylene)-1,4-dioxaspiro<4.5>decane; nickel dichloride In diethyl ether at -10 - 0℃; for 24h; Product distribution; var. of ligand, temp., ratio, time; asymmetric induction; | 100% |
| all-(S) 4-Me2N-cis-decaline[3,2-b](1-Ph2P-ferrocene)*PdCl2 In diethyl ether at 0℃; for 24h; | 96% |

| Conditions | Yield |
|---|---|
| Stage #1: Vinyl bromide With magnesium In tetrahydrofuran at 20℃; for 2h; Stage #2: (R)-Citronellal In tetrahydrofuran at 20℃; for 3h; Further stages.; | 100% |
-
-
593-60-2
Vinyl bromide

-
-
132563-81-6
1-(2,4-difluorophenyl)-2-(tetrahydropyranyloxy)propan-1-one

-
-
126945-39-9
(3S,4R)-3-(2,4-Difluorophenyl)-4-(2-tetrahydropyranyloxy)-1-penten-3-ol

| Conditions | Yield |
|---|---|
| With sodium chloride; ammonium chloride; magnesium In tetrahydrofuran; acetone | 100% |
-
-
26654-39-7
4,4-dimethyl-1,3-oxazolidin-2-one

-
-
593-60-2
Vinyl bromide

-
-
929554-28-9
4,4-dimethyl-3-vinyloxazolidin-2-one

| Conditions | Yield |
|---|---|
| With copper(l) iodide; potassium carbonate; N,N`-dimethylethylenediamine In toluene at 113℃; for 16h; | 100% |
-
-
593-60-2
Vinyl bromide

-
-
389079-71-4
N-(2-(cyclohex-1-en-1-yl)ethyl)-4-methylbenzenesulfonamide

-
-
1180843-91-7
C17H23NO2S

| Conditions | Yield |
|---|---|
| With copper(l) iodide; potassium carbonate; N,N`-dimethylethylenediamine In tetrahydrofuran at 80℃; Inert atmosphere; | 100% |

| Conditions | Yield |
|---|---|
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; triethylamine; triphenylphosphine In tetrahydrofuran at 20 - 50℃; Sonogashira Cross-Coupling; Inert atmosphere; | 100% |
| With copper(l) iodide; tetrakis(triphenylphosphine) palladium(0) In tetrahydrofuran at 24℃; for 4h; Inert atmosphere; Alkaline conditions; | 96% |
| Stage #1: Vinyl bromide With copper(l) iodide; tetrakis(triphenylphosphine) palladium(0); diethylamine In tetrahydrofuran at 20℃; for 0.0833333h; Sonogashira Cross-Coupling; Inert atmosphere; Stage #2: 4-bromo-1-ethynylbenzene In tetrahydrofuran at 20℃; Inert atmosphere; | 79.1% |

| Conditions | Yield |
|---|---|
| With aluminium bromide at -50℃; for 1h; | 100% |
-
-
593-60-2
Vinyl bromide

-
-
73448-13-2
tert-butyldimethylsilyl hex-5-yn-1-yl ether

-
-
138059-76-4
8-<(tert-butyldimethylsilyl)oxy>-1-octen-3-yne

| Conditions | Yield |
|---|---|
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; diethylamine for 2h; Ambient temperature; | 99% |

| Conditions | Yield |
|---|---|
| for 1h; Ambient temperature; in vacuo; | 99% |
-
-
593-60-2
Vinyl bromide

-
-
768-60-5
4-methoxyphenylacetylen

-
-
55088-86-3
1-(but-3-en-1-yn-1-yl)-4-methoxybenzene

| Conditions | Yield |
|---|---|
| With copper(l) iodide; tetrakis(triphenylphosphine) palladium(0); diethylamine In tetrahydrofuran at 0 - 20℃; for 19h; Inert atmosphere; | 99% |
| With copper(l) iodide; tetrakis(triphenylphosphine) palladium(0); diethylamine In tetrahydrofuran at 0 - 20℃; Inert atmosphere; | 99% |
| Stage #1: Vinyl bromide With copper(l) iodide; tetrakis(triphenylphosphine) palladium(0); diethylamine In tetrahydrofuran at 20℃; for 0.0833333h; Sonogashira Cross-Coupling; Inert atmosphere; Stage #2: 4-methoxyphenylacetylen In tetrahydrofuran at 20℃; Inert atmosphere; | 92.5% |

| Conditions | Yield |
|---|---|
| Stage #1: Vinyl bromide With tert.-butyl lithium In diethyl ether at -78℃; for 0.25h; Stage #2: C21H17NO4 In tetrahydrofuran; diethyl ether at 0℃; for 0.166667h; Further stages.; | 99% |
-
-
593-60-2
Vinyl bromide

-
-
654085-09-3
[(4'-(4-t-butylphenyl)-2,2':6',2''-terpyridine)Rh(Br)]

-
-
654085-21-9
Rh(III)Br2(ethenyl)(4'-(4-tertbutylphenyl)-2,2':6',2''-terpyridine)

| Conditions | Yield |
|---|---|
| In ethanol Ar; adding 100 equiv. of bromoethene to a mixt. of Rh complex with ethanol at room temp.; pouring the mixt. into pentane after 5 min, decantation, washing the solid with pentane, drying under reduced pressure; | 99% |

| Conditions | Yield |
|---|---|
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; diisopropylamine In tetrahydrofuran at 0 - 20℃; Inert atmosphere; | 99% |
| With copper(l) iodide; tetrakis(triphenylphosphine) palladium(0); diethylamine In tetrahydrofuran at 20℃; Inert atmosphere; Schlenk technique; | 95% |
-
-
593-60-2
Vinyl bromide

-
-
766-97-2
4-n-methylphenylacetylene

-
-
30011-66-6
1-(but-3-en-1-yn-1-yl)-4-methylbenzene

| Conditions | Yield |
|---|---|
| With copper(l) iodide; tetrakis(triphenylphosphine) palladium(0); diethylamine In tetrahydrofuran at 0 - 20℃; for 19h; Inert atmosphere; | 99% |
| With copper(l) iodide; tetrakis(triphenylphosphine) palladium(0); triethylamine In tetrahydrofuran at 20℃; Sealed tube; Inert atmosphere; | 95% |
| With copper(l) iodide; tetrakis(triphenylphosphine) palladium(0); diethylamine In tetrahydrofuran at 20℃; Inert atmosphere; Schlenk technique; | 84% |

| Conditions | Yield |
|---|---|
| With copper(l) iodide; tetrakis(triphenylphosphine) palladium(0); diethylamine In tetrahydrofuran at 0 - 20℃; Sonogashira Cross-Coupling; Inert atmosphere; Schlenk technique; | 99% |
| With copper(l) iodide; tetrakis(triphenylphosphine) palladium(0); diethylamine at 0 - 20℃; for 6.5h; | 98% |

| Conditions | Yield |
|---|---|
| With copper(l) iodide; tetrakis(triphenylphosphine) palladium(0); diethylamine In tetrahydrofuran at 20℃; Inert atmosphere; Schlenk technique; | 99% |
| With copper(l) iodide; tetrakis(triphenylphosphine) palladium(0); diethylamine In tetrahydrofuran at 0 - 20℃; Inert atmosphere; | 88% |
| With copper(l) iodide; tetrakis(triphenylphosphine) palladium(0); triethylamine at 0 - 20℃; Sonogashira Cross-Coupling; Schlenk technique; Inert atmosphere; | 76% |
| With copper(l) iodide; tetrakis(triphenylphosphine) palladium(0); triethylamine In tetrahydrofuran at 0 - 25℃; Inert atmosphere; | 45.9% |

| Conditions | Yield |
|---|---|
| With copper(l) iodide; tetrakis(triphenylphosphine) palladium(0); diethylamine In tetrahydrofuran at 20℃; Inert atmosphere; Schlenk technique; | 99% |
| With copper(l) iodide; tetrakis(triphenylphosphine) palladium(0) In tetrahydrofuran; diethylamine at 0 - 20℃; Inert atmosphere; | 69% |
| With copper(l) iodide; tetrakis(triphenylphosphine) palladium(0); diethylamine In tetrahydrofuran at 0 - 20℃; for 4h; Inert atmosphere; Schlenk technique; | 29% |
| With copper(l) iodide; tetrakis(triphenylphosphine) palladium(0); diethylamine In tetrahydrofuran at 0 - 20℃; for 4h; Glovebox; |

| Conditions | Yield |
|---|---|
| Stage #1: Vinyl bromide With copper(l) iodide; tetrakis(triphenylphosphine) palladium(0); diethylamine In tetrahydrofuran at 20℃; for 0.0833333h; Sonogashira Cross-Coupling; Inert atmosphere; Stage #2: 4-trifluoromethylphenylacetylene In tetrahydrofuran at 20℃; Inert atmosphere; | 99% |
| With copper(l) iodide; tetrakis(triphenylphosphine) palladium(0); diethylamine In tetrahydrofuran at 0 - 20℃; for 4h; Inert atmosphere; Schlenk technique; | 89% |
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; triethylamine In tetrahydrofuran at 20℃; Sealed tube; Inert atmosphere; | |
| With copper(l) iodide; tetrakis(triphenylphosphine) palladium(0); diethylamine at 20℃; Sonogashira Cross-Coupling; Inert atmosphere; |

| Conditions | Yield |
|---|---|
| With copper(l) iodide; tetrakis(triphenylphosphine) palladium(0); diethylamine In tetrahydrofuran at 0 - 20℃; Sonogashira Cross-Coupling; Inert atmosphere; Schlenk technique; | 99% |

| Conditions | Yield |
|---|---|
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; triethylamine; triphenylphosphine In tetrahydrofuran at 50℃; for 2h; Sonogashira Cross-Coupling; | 98% |
| With copper(l) iodide; tetrakis(triphenylphosphine) palladium(0); diethylamine In tetrahydrofuran at 0 - 20℃; Inert atmosphere; | 90% |
| With copper(l) iodide; tetrakis(triphenylphosphine) palladium(0); diethylamine In tetrahydrofuran at 0 - 20℃; Sonogashira Cross-Coupling; Inert atmosphere; Schlenk technique; | 90% |
-
-
593-60-2
Vinyl bromide

-
-
127643-19-0
3-[1,3]Dioxolan-2-ylmethyl-4-methyl-pentanal

-
-
127600-26-4
5-[1,3]Dioxolan-2-ylmethyl-6-methyl-hept-1-en-3-ol

| Conditions | Yield |
|---|---|
| With iodine; magnesium In tetrahydrofuran | 98% |
-
-
593-60-2
Vinyl bromide

-
-
83060-72-4
3-amino-5-phenylthiophene-2-carbonitrile

| Conditions | Yield |
|---|---|
| With NaOCH3 In ethanol; chloroform | 98% |
-
-
593-60-2
Vinyl bromide

-
-
1567325-02-3
pent-4-ynyl 4-(trifluoromethyl)benzoate

| Conditions | Yield |
|---|---|
| With copper(l) iodide; tetrakis(triphenylphosphine) palladium(0); diethylamine In tetrahydrofuran at 0 - 20℃; Sonogashira Cross-Coupling; Inert atmosphere; Schlenk technique; | 98% |

| Conditions | Yield |
|---|---|
| With bromine In dichloromethane at 0 - 22℃; for 2h; Inert atmosphere; | 97% |
| With bromine In chloroform at 20℃; for 12h; Bromination; | 87% |
| With bromine |
-
-
593-60-2
Vinyl bromide

-
-
71996-27-5
2-(phenylselenenyl)-2-cyclopentenone

| Conditions | Yield |
|---|---|
| 97% |

-
-
593-60-2
Vinyl bromide

-
-
228579-91-7
(2S)-di-tert-butyl 5-methoxypyrrolidine-1,2-dicarboxylate

-
-
1017802-73-1
L-N-(tert-butyloxycarbonyl)-5-vinylproline tert-butyl ester

| Conditions | Yield |
|---|---|
| Stage #1: Vinyl bromide With iodine; magnesium In tetrahydrofuran for 0.5h; Stage #2: With copper(I) bromide dimethylsulfide complex In tetrahydrofuran at -40℃; for 0.75h; Stage #3: (2S)-di-tert-butyl 5-methoxypyrrolidine-1,2-dicarboxylate With boron trifluoride diethyl etherate In tetrahydrofuran at -78 - 20℃; for 4.5h; | 97% |
-
-
593-60-2
Vinyl bromide

-
-
12151-13-2
cycloocta-1,5-dienebis(triphenylphosphine)nickel(0)

-
B
-
5259-72-3, 10060-40-9, 111-78-4
1,5-dicyclooctadiene

| Conditions | Yield |
|---|---|
| In hexane addn. of Ni-complex to soln. of vinyl bromide in hexane (stirring, -50-(-70)°C), after 30 min warming to -10°C, stirring 30 min; filtration, washing (cold hexane), drying (vac., -10°C); | A 97% B n/a |

-
-
593-60-2
Vinyl bromide

-
-
69786-76-1
Li(Cr(CO)3(η6-C6H4OMe-2))

-
-
164577-77-9, 52456-48-1, 147127-36-4
η6-[(2-methoxy)vinylbenzene]tricarbonylchromium(0)

| Conditions | Yield |
|---|---|
| tetrakis(triphenylphosphine) palladium(0) In tetrahydrofuran react. with 1 equiv. CuBr*SMe2 in THF at -78°C, then with 1 equiv. CH2=CHBr and Pd(PPh3)4, -78°C to room temp.; | 97% |


| Conditions | Yield |
|---|---|
| Stage #1: Vinyl bromide With copper(l) iodide; triethylamine; palladium dichloride In tetrahydrofuran at 20℃; for 0.0833333h; Schlenk technique; Inert atmosphere; Stage #2: 4-Phenyl-1-butyne In tetrahydrofuran at 20℃; Schlenk technique; Inert atmosphere; | 97% |
| With copper(l) iodide; tetrakis(triphenylphosphine) palladium(0) In tetrahydrofuran at 24℃; for 13h; Inert atmosphere; Alkaline conditions; | 96% |
| With copper(l) iodide; tetrakis(triphenylphosphine) palladium(0); diethylamine In tetrahydrofuran at 24℃; for 14h; Inert atmosphere; | 95% |

| Conditions | Yield |
|---|---|
| Stage #1: C29H56O2Si2 With 9-bora-bicyclo[3.3.1]nonane In tetrahydrofuran at 0 - 20℃; for 7h; Stage #2: Vinyl bromide With tetrakis(triphenylphosphine) palladium(0); water; sodium hydroxide In tetrahydrofuran at 20℃; for 1.5h; optical yield given as %de; | A 97% B n/a |

Related products
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View

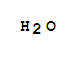














 T;
T;  F+
F+