-
Name
2,4-Difluorophenylboronic acid
- EINECS -0
- CAS No. 144025-03-6
- Article Data8
- CAS DataBase
- Density 1.35 g/cm3
- Solubility
- Melting Point 247-250 ºC
- Formula C6H5BF2O2
- Boiling Point 251 ºC at 760 mmHg
- Molecular Weight 157.912
- Flash Point 105.6 ºC
- Transport Information
- Appearance off-white to light beige or yellow powder
- Safety 37/39-26-36
- Risk Codes 36/37/38-22
-
Molecular Structure
-
Hazard Symbols
 Xn,
Xn, Xi
Xi
- Synonyms 2,4-Difluorobenzeneboronic acid;2,4-Difluorobenzene boronic acid;
- PSA 40.46000
- LogP -0.35540
Synthetic route
-
-
121-43-7
Trimethyl borate

-
-
144025-04-7
(2,4-difluorophenyl)magnesium bromide

-
-
144025-03-6
2,4-difluorophenylboronic acid

| Conditions | Yield |
|---|---|
| With sulfuric acid In tetrahydrofuran; water | 84.3% |
| With sulfuric acid In tetrahydrofuran; water | 84.3% |
| In tetrahydrofuran | 74.8% |
| In tetrahydrofuran | 74.8% |
| Stage #1: Trimethyl borate; (2,4-difluorophenyl)magnesium bromide In tetrahydrofuran at -60℃; for 5h; Stage #2: With hydrogenchloride In tetrahydrofuran; water at 0℃; pH=1; Inert atmosphere; | 40% |
-
-
348-57-2
2,4-difluorobromobenzene

-
-
121-43-7
Trimethyl borate

-
-
144025-03-6
2,4-difluorophenylboronic acid

| Conditions | Yield |
|---|---|
| With n-butyllithium In diethyl ether; hexane n-BuLi (2.5 M, hexane) was added to C6F2BrH3 in Et2O at -70 °C, 1.5 equiv. of cold soln. of B(OMe)3 was added in Et2O, stirring at -60 °C for 1 h, mixt. was allowed to warm to 15 °C within 1 h; poured into 5 % HCl, org. layer was sepd., aq. layer was twice extd. with ether, extracts were combined, dried with MgSO4, evapn., solid was stored over water at 15 °C for 1 month; | 77% |
-
-
871300-65-1
C6H3BF5(1-)

-
-
144025-03-6
2,4-difluorophenylboronic acid

| Conditions | Yield |
|---|---|
| In phosphate buffer pH=6.9 - 7.0; Kinetics; |
-
-
512198-16-2
tris(2,4-difluorophenyl)boroxine

-
-
7732-18-5
water

-
-
144025-03-6
2,4-difluorophenylboronic acid

| Conditions | Yield |
|---|---|
| In acetonitrile | |
| In diethyl ether |
-
-
109-86-4
2-methoxy-ethanol

-
-
144025-03-6
2,4-difluorophenylboronic acid

| Conditions | Yield |
|---|---|
| With sodium carbonate In water; acetylacetone |

-
-
144025-03-6
2,4-difluorophenylboronic acid

| Conditions | Yield |
|---|---|
| With hydrogenchloride In water | 1.15 g |
-
-
348-57-2
2,4-difluorobromobenzene

-
-
688-71-1
tri-n-propyl borate

-
-
7732-18-5
water

-
-
144025-03-6
2,4-difluorophenylboronic acid

| Conditions | Yield |
|---|---|
| Stage #1: 2,4-difluorobromobenzene With magnesium Stage #2: tri-n-propyl borate Stage #3: water Acidic conditions; |

-
-
144025-03-6
2,4-difluorophenylboronic acid

-
-
618-51-9
3-Iodobenzoic acid

| Conditions | Yield |
|---|---|
| Stage #1: 3-Iodobenzoic acid With benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; N-ethyl-N,N-diisopropylamine; Wang resin In dichloromethane at 20℃; for 22h; Stage #2: 2,4-difluorophenylboronic acid With tris(dibenzylideneacetone)dipalladium (0); potassium carbonate In N,N-dimethyl-formamide at 20℃; Stage #3: With trifluoroacetic acid In dichloromethane at 20℃; for 13h; | 100% |
-
-
109-04-6
2-bromo-pyridine

-
-
144025-03-6
2,4-difluorophenylboronic acid

-
-
391604-55-0
2-(2,4-difluorophenyl)pyridine

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); sodium carbonate In tetrahydrofuran at 70℃; for 24h; Suzuki coupling; | 100% |
| With tetrakis(triphenylphosphine) palladium(0); sodium carbonate In tetrahydrofuran; water at 70℃; Inert atmosphere; | 98% |
| With sodium carbonate; tetrakis(triphenylphosphine) palladium(0) In tetrahydrofuran; water at 70℃; for 24h; | 97% |
-
-
648904-46-5
trifluoromethanesulfonic acid 6-methoxy-1-[4-(2-piperidin-1-ylethoxy)phenoxy]naphthalen-2-yl ester

-
-
144025-03-6
2,4-difluorophenylboronic acid

| Conditions | Yield |
|---|---|
| Stage #1: trifluoromethanesulfonic acid 6-methoxy-1-[4-(2-piperidin-1-ylethoxy)phenoxy]naphthalen-2-yl ester; 2,4-difluorophenylboronic acid With cesium fluoride; tricyclohexylphosphine; palladium diacetate In acetonitrile at 90℃; for 0.166667h; Suzuki Coupling; Stage #2: With hydrogenchloride In diethyl ether | 100% |
-
-
540-38-5
4-Iodophenol

-
-
144025-03-6
2,4-difluorophenylboronic acid

-
-
59089-68-8
4-(2,4-difluorophenyl)-phenol

| Conditions | Yield |
|---|---|
| With potassium carbonate; palladium(0)bis(tricyclohexylphosphine) In water; N,N-dimethyl-formamide Inert atmosphere; Reflux; | 100% |
-
-
1380281-66-2
(6-Bromopyridin-2-yl)-[5-(2-tert-butyl-pyrimidin-4-yl)-2-methylphenyl]amine

-
-
144025-03-6
2,4-difluorophenylboronic acid

-
-
1380281-67-3
[5-(2-tert-Butyl-pyrimidin-4-yl)-2-methylphenyl]-[6-(2,4-difluorophenyl)-pyridin-2-yl]amine

| Conditions | Yield |
|---|---|
| With sodium carbonate; tetrakis(triphenylphosphine) palladium(0) In ethanol; water at 75℃; for 3h; Suzuki Coupling; | 100% |
-
-
144025-03-6
2,4-difluorophenylboronic acid

-
-
1380281-57-1
2-bromo-6-(2,4-difluorophenyl)pyridine

| Conditions | Yield |
|---|---|
| With sodium carbonate; tetrakis(triphenylphosphine) palladium(0) In ethanol; water at 75℃; for 2.5h; Product distribution / selectivity; Suzuki Coupling; | 100% |

-
-
144025-03-6
2,4-difluorophenylboronic acid

| Conditions | Yield |
|---|---|
| With palladium diacetate; sodium carbonate In water at 20℃; for 3h; Suzuki Coupling; Inert atmosphere; | 100% |
-
-
586-78-7
para-nitrophenyl bromide

-
-
144025-03-6
2,4-difluorophenylboronic acid

-
-
1178888-74-8
2,4-difluoro-4′-nitrobiphenyl

| Conditions | Yield |
|---|---|
| With 1,1',1'',1'''-benzene-1,2,4,5-tetrayltetrakis(methylene)tetrakis-(piperidin-4-ol); palladium diacetate; potassium carbonate In ethanol; water at 20℃; Suzuki-Miyaura Coupling; | 100% |
-
-
1015792-46-7
5-ethyl-4-iodo-2(5H)-furanone

-
-
144025-03-6
2,4-difluorophenylboronic acid

| Conditions | Yield |
|---|---|
| With 1,1',1'',1'''-benzene-1,2,4,5-tetrayltetrakis(methylene)tetrakis-(piperidin-4-ol); palladium diacetate; potassium carbonate In ethanol; water at 20℃; for 0.25h; Suzuki-Miyaura Coupling; | 100% |

-
-
144025-03-6
2,4-difluorophenylboronic acid

| Conditions | Yield |
|---|---|
| With palladium diacetate; sodium carbonate; triphenylphosphine In 1,4-dioxane; water Suzuki-Miyaura Coupling; Inert atmosphere; Reflux; | 100% |
-
-
89-55-4
5-bromosalicyclic acid

-
-
144025-03-6
2,4-difluorophenylboronic acid

-
-
22494-42-4
2',4'-difluoro-4-hydroxybiphenyl-3-carboxylic acid

| Conditions | Yield |
|---|---|
| With 2-[5-(4-methylphenyl)isoxazol-3-yl]-5-(5-phenylisoxazol-3-yl)-1,3,4-oxadiazole·2PdCl2; potassium carbonate In water; N,N-dimethyl-formamide at 100℃; for 0.0166667h; Suzuki Coupling; | 99% |
| With potassium carbonate; palladium dichloride In water; N,N-dimethyl-formamide at 75℃; Temperature; Solvent; Reagent/catalyst; Sonication; Cooling; | 98.37% |
| With C11H8Cl4N2O3Pd; potassium carbonate In methanol; water; N,N-dimethyl-formamide at 100℃; for 0.0833333h; Suzuki Coupling; | 97% |
-
-
623-12-1
4-chloromethoxybenzene

-
-
144025-03-6
2,4-difluorophenylboronic acid

-
-
90101-30-7
2,4-difluoro-4'-methoxybiphenyl

| Conditions | Yield |
|---|---|
| With dicyclohexyl-(2',6'-dimethoxybiphenyl-2-yl)-phosphane; potassium phosphate; palladium diacetate In toluene at 80℃; for 16h; Suzuki-Miyaura coupling; | 99% |
| With potassium carbonate; 2-(dicyclohexylphosphino)-2'-methylbiphenyl; palladium on activated charcoal In N,N-dimethyl acetamide; water at 100℃; for 2h; Suzuki-Miyaura cross-coupling; | 64% |
-
-
1144042-97-6
C29H29N3O4S

-
-
144025-03-6
2,4-difluorophenylboronic acid

-
A
-
1144043-13-9
2-(2,4-difluorophenylthio)-N-isopropylbenzamide

-
B
-
1144042-90-9
(+)-(S)-2-(benzyloxycarbonylamino)-1-(2,4-difluorophenyl)-3-(1H-indol-3-yl)propan-1-one

| Conditions | Yield |
|---|---|
| With copper(I) 2-hydroxy-3-methylbenzoate; oxygen In N,N-dimethyl-formamide at 23℃; for 3h; optical yield given as %ee; | A 99% B 97% |

-
-
56634-50-5
4-bromo-5H-furan-2-one

-
-
144025-03-6
2,4-difluorophenylboronic acid

| Conditions | Yield |
|---|---|
| With bis-triphenylphosphine-palladium(II) chloride; potassium fluoride In tetrahydrofuran; water for 5h; Inert atmosphere; Reflux; | 99% |
-
-
1201683-05-7
C17H18BrNS2

-
-
144025-03-6
2,4-difluorophenylboronic acid

-
-
1341232-96-9
2-(2,4-difluorophenyl)-4-(5-hexylthieno[3,2-b]thien-2-yl)pyridine

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); sodium carbonate In tetrahydrofuran; water at 70℃; for 0.333333h; Suzuki coupling; Inert atmosphere; Microwave irradiation; | 99% |
-
-
3899-91-0
4-methoxyphenyl p-toluenesulfonate

-
-
144025-03-6
2,4-difluorophenylboronic acid

-
-
90101-30-7
2,4-difluoro-4'-methoxybiphenyl

| Conditions | Yield |
|---|---|
| With potassium phosphate; nickelocene; tricyclohexylphosphine In tetrahydrofuran at 20℃; for 8h; Reagent/catalyst; Glovebox; Inert atmosphere; | 99% |
-
-
7385-85-5
naphthalen-2-yl tosylate

-
-
144025-03-6
2,4-difluorophenylboronic acid

| Conditions | Yield |
|---|---|
| With potassium phosphate; bis(tricyclohexylphosphine)nickel(II) bromide In tetrahydrofuran; water at 20℃; Reagent/catalyst; Glovebox; | 99% |
-
-
144025-03-6
2,4-difluorophenylboronic acid

| Conditions | Yield |
|---|---|
| With potassium phosphate; bis(tricyclohexylphosphine)nickel(II) bromide In tetrahydrofuran; water at 20℃; Glovebox; | 99% |
-
-
98027-84-0
2,6-dichloro-4-iodopyridine

-
-
144025-03-6
2,4-difluorophenylboronic acid

| Conditions | Yield |
|---|---|
| With (1,1'-bis(diphenylphosphino)ferrocene)palladium(II) dichloride; sodium hydrogencarbonate In 1,2-dimethoxyethane; water at 90℃; for 1h; Inert atmosphere; | 98.5% |
-
-
99-90-1
para-bromoacetophenone

-
-
144025-03-6
2,4-difluorophenylboronic acid

-
-
53591-79-0
1-(2',4'difluoro[1,1']biphenyl-4-yl)ethanone

| Conditions | Yield |
|---|---|
| With C20H12N2O8PdS2(2-)*2Na(1+); sodium hydroxide In water at 100℃; for 6h; | 98% |
| With C40H56Cl2FeN2O4P2; sodium carbonate In water; acetone at 20℃; for 4h; Suzuki coupling; | 92% |
| With [Pd(N-(3-chloro-2-quinoxalinyl)-N'-(2,6-diisopropylphenyl)imidazolium)(PPh3)Cl2]; potassium carbonate In water at 70℃; for 3h; Catalytic behavior; Suzuki-Miyaura Coupling; | 55% |
| With cis,cis,cis-1,2,3,4-tetrakis(diphenylphosphinomethyl)cyclope; potassium carbonate; bis(η3-allyl-μ-chloropalladium(II)) In xylene at 130℃; for 20h; Suzuki cross-coupling reaction; | |
| With (R,R)-N,N'-bis[(2,4-dimethoxyphenyl)methyl]-1,2-cyclohexanediamine PdCl2 complex; potassium carbonate In N,N-dimethyl-formamide at 120℃; for 24h; Reactivity; Reagent/catalyst; Suzuki coupling; Aerobic conditions; | 99 %Spectr. |
-
-
1246612-19-0
6-bromo-N-phenyl-N-(pyridin-2-yl)pyridin-2-amine

-
-
144025-03-6
2,4-difluorophenylboronic acid

-
-
1246612-18-9
F2C6H3C5H3NN(C6H5)C5H4N

| Conditions | Yield |
|---|---|
| With palladium diacetate; potassium carbonate; triphenylphosphine In 1,2-dimethoxyethane; water for 6h; Inert atmosphere; Reflux; | 98% |
-
-
102-79-4
n-butyldiethanolamine

-
-
144025-03-6
2,4-difluorophenylboronic acid

| Conditions | Yield |
|---|---|
| With magnesium sulfate In acetone at 35℃; for 1h; | 98% |
-
-
634-47-9
2-chloro-4-methylquinoline

-
-
144025-03-6
2,4-difluorophenylboronic acid

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); sodium carbonate In ethanol; water; toluene at 110℃; for 16h; Suzuki-Miyaura Coupling; Inert atmosphere; | 98% |
| Suzuki Coupling; Inert atmosphere; Schlenk technique; |

| Conditions | Yield |
|---|---|
| With bis-triphenylphosphine-palladium(II) chloride; potassium phosphate In 1,4-dioxane at 50℃; for 8h; Inert atmosphere; regioselective reaction; | 98% |

| Conditions | Yield |
|---|---|
| With C35H40N4O9(2+)*2Cl(1-); palladium diacetate; potassium hydroxide In ethanol at 100℃; for 1.5h; Suzuki Coupling; Green chemistry; | 98% |
-
-
119-30-2
2-hydroxy-5-iodobenzoic acid

-
-
144025-03-6
2,4-difluorophenylboronic acid

-
-
22494-42-4
2',4'-difluoro-4-hydroxybiphenyl-3-carboxylic acid

| Conditions | Yield |
|---|---|
| With C32H26Cl2N8O4Pd2; potassium carbonate In methanol; water for 0.0833333h; Suzuki-Miyaura Coupling; Reflux; | 98% |
| With choline chloride; palladium diacetate; sodium carbonate; glycerol at 60℃; for 5h; Suzuki-Miyaura Coupling; | 52% |

-
-
144025-03-6
2,4-difluorophenylboronic acid

| Conditions | Yield |
|---|---|
| With (1,1'-bis(diphenylphosphino)ferrocene)palladium(II) dichloride; sodium carbonate In ethanol; water; N,N-dimethyl-formamide; toluene at 86℃; for 2.66667h; Suzuki Coupling; Inert atmosphere; | 97% |
-
-
174265-12-4
2-bromo-5-chlorobenzaldehyde

-
-
144025-03-6
2,4-difluorophenylboronic acid

| Conditions | Yield |
|---|---|
| With potassium phosphate monohydrate; C66H84O10P2Pd In water; toluene at 45℃; for 3h; Reagent/catalyst; Solvent; Suzuki-Miyaura Coupling; Inert atmosphere; | 97% |
| With potassium phosphate monohydrate; C50H64NO6PPdS In water at 45℃; for 3h; Suzuki-Miyaura Coupling; Inert atmosphere; Sealed tube; |
-
-
585-76-2
m-bromobenzoic acid

-
-
144025-03-6
2,4-difluorophenylboronic acid

| Conditions | Yield |
|---|---|
| With 4-(di-tert-butylphosphino)ethyltrimethylammonium chloride; sodium carbonate; sodium tetrachloropalladate(II) In water at 20℃; Suzuki reaction; | 96% |
-
-
106-47-8
4-chloro-aniline

-
-
144025-03-6
2,4-difluorophenylboronic acid

| Conditions | Yield |
|---|---|
| With dicyclohexyl-(2',6'-dimethoxybiphenyl-2-yl)-phosphane; potassium phosphate; palladium diacetate In toluene at 80℃; for 10h; Suzuki-Miyaura coupling; | 96% |
2,4-Difluorophenylboronic acid Specification
The 2,4-Difluorophenylboronic acid, with the CAS registry number 144025-03-6, is also known as Boronic acid, B-(2,4-difluorophenyl)-. It belongs to the product categories of Blocks; BoronicAcids; Fluoro Compounds; Boric Acid; Substituted Boronic Acids; Fluorobenzene; Boronic Acid; Aryl; Halogenated; Organoborons; B (Classes of Boron Compounds); Boronic Acids; Boronic Acids; Boronic Acids and Derivatives. This chemical's molecular formula is C6H5BF2O2 and molecular weight is 157.91. What's more, both its IUPAC name and systematic name are the same which is called (2,4-Difluorophenyl)boronic acid.
Physical properties about 2,4-Difluorophenylboronic acid are: (1)ACD/LogP: 1.73; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 1.73; (4)ACD/LogD (pH 7.4): 1.69; (5)ACD/BCF (pH 5.5): 12.15; (6)ACD/BCF (pH 7.4): 11.2; (7)ACD/KOC (pH 5.5): 207.87; (8)ACD/KOC (pH 7.4): 191.61; (9)#H bond acceptors: 2; (10)#H bond donors: 2; (11)#Freely Rotating Bonds: 3; (12)Polar Surface Area: 18.46 Å2; (13)Index of Refraction: 1.485; (14)Molar Refractivity: 33.51 cm3; (15)Molar Volume: 116.7 cm3; (16)Surface Tension: 36.1 dyne/cm; (17)Density: 1.35 g/cm3; (18)Flash Point: 105.6 °C; (19)Enthalpy of Vaporization: 51.6 kJ/mol; (20)Boiling Point: 251 °C at 760 mmHg; (21) Vapour Pressure: 0.011 mmHg at 25 °C.
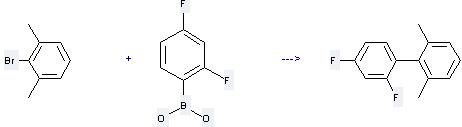
Uses of 2,4-Difluorophenylboronic acid: it is used to produce other chemicals. For example, it can react with 2-Bromo-1,3-dimethyl-benzene to get 2,4-Difluoro-2',6'-dimethyl-biphenyl. The reaction occurs with reagents K2CO3, cis,cis,cis-1,2,3,4-Tetrakis(diphenylphosphinomethyl)cyclopentane and solvent xylene at temperature of 130 °C. The reaction time is 20 hours. The yield is 60 %.
When you are dealing with this chemical, you should be very careful. This chemical is inflammation to the skin, eyes and respiratory system or other mucous membranes. If swallowed, it's harmful to health. Therefore, you should wear suitable protective clothing, gloves and eye/face protection. And in case of contacting with eyes, you should rinse immediately with plenty of water and seek medical advice.
You can still convert the following datas into molecular structure:
(1) SMILES: Fc1cc(F)ccc1B(O)O
(2) InChI: InChI=1S/C6H5BF2O2/c8-4-1-2-5(7(10)11)6(9)3-4/h1-3,10-11H
(3) InChIKey: QQLRSCZSKQTFGY-UHFFFAOYSA-N
Related Products
- 20,22-DIHYDRODIGITOXIN
- 20,29,30-Trinorlupane,(17alpha)-
- 20-ETHYL-6-β,8-DIHYDROXY-1-α-METHOXY-4-METHYLHETERATISAN-14-ONE
- 20-Ethylprostaglandin F2-alpha
- 20-Isopropylcholanthrene
- 20-METHYLCHOLANTHREN-15-ONE
- 20-METHYLCHOLANTHRENE PICRATE
- 20-METHYLCHOLANTHRENE-TRINITRO-BENZENE
- 20(S)-Ginsenoside C-K
- 2,10-DIFLUOROBENZO(rst)PENTAPHENE
- 14402-50-7
- 14402-67-6
- 144026-79-9
- 14402-88-1
- 14403-45-3
- 144034-80-0
- 144036-17-9
- 14404-25-2
- 144043-17-4
- 144043-20-9
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View