-
Name
2,5-Thiophenedicarboxylic acid
- EINECS 224-284-2
- CAS No. 4282-31-9
- Article Data42
- CAS DataBase
- Density 1.655 g/cm3
- Solubility Very soluble in water.
- Melting Point >300 °C(lit.)
- Formula C6H4O4S
- Boiling Point 444.6 °C at 760 mmHg
- Molecular Weight 172.161
- Flash Point 222.7 °C
- Transport Information
- Appearance white to light yellow crystal powder
- Safety 26-36-37/39
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms 2,5-Dicarboxythiophene;NSC 81789;Thiophene-2,5-dicarboxylic acid;
- PSA 102.84000
- LogP 1.14450
Synthetic route
-
-
18853-40-2
2,5-thiophenedicarbonitrile

-
-
4282-31-9
Thiophene-2,5-dicarboxylic acid

| Conditions | Yield |
|---|---|
| With hydrogenchloride for 6h; Heating; | 79% |

| Conditions | Yield |
|---|---|
| Stage #1: Adipic acid With dmap; thionyl chloride at 78 - 81℃; for 3.5h; Large scale; Stage #2: With sodium hydroxide In water at 70 - 72℃; for 2h; pH=9.3 - 10; Reagent/catalyst; Temperature; pH-value; Large scale; | 75.58% |
-
-
61755-85-9
2,5‐diethyl thiophene‐2,5‐dicarboxylate

-
-
4282-31-9
Thiophene-2,5-dicarboxylic acid

| Conditions | Yield |
|---|---|
| With potassium hydroxide In ethanol for 2h; Heating; | 70% |
| With potassium hydroxide |

| Conditions | Yield |
|---|---|
| With n-butyllithium; carbon dioxide In tetrahydrofuran; water | 60% |
| With diethyl ether; phenyllithium Behandeln des Reaktionsgemisches mit festem Kohlendioxid; |
-
-
67-56-1
methanol

-
-
3141-27-3
2,5-dibromothiophen

-
-
201230-82-2
carbon monoxide

-
A
-
4282-31-9
Thiophene-2,5-dicarboxylic acid

-
B
-
4282-34-2
Dimethyl thiophene-2,5-dicarboxylate

| Conditions | Yield |
|---|---|
| With tert-Amyl alcohol; sodium hydride; cobalt(II) acetate In tetrahydrofuran at 40℃; under 760 Torr; for 54h; Irradiation; | A 17% B 55% |

-
-
3141-27-3
2,5-dibromothiophen

-
-
201230-82-2
carbon monoxide

-
A
-
4282-31-9
Thiophene-2,5-dicarboxylic acid

-
B
-
4282-34-2
Dimethyl thiophene-2,5-dicarboxylate

| Conditions | Yield |
|---|---|
| With methanol; tert-Amyl alcohol; sodium hydride; cobalt(II) acetate In tetrahydrofuran at 40℃; under 760 Torr; for 54h; Irradiation; | A 17% B 55% |

-
-
3141-27-3
2,5-dibromothiophen

-
-
124-38-9
carbon dioxide

-
A
-
7311-63-9
5-bromothiophene-2-carboxylic acid

-
B
-
4282-31-9
Thiophene-2,5-dicarboxylic acid

| Conditions | Yield |
|---|---|
| Stage #1: 2,5-dibromothiophen; carbon dioxide With tert.-butyl lithium In pentane at -70℃; Inert atmosphere; Stage #2: With sulfuric acid; water In pentane at 20℃; Inert atmosphere; | A 34% B 5% |

-
-
188290-36-0
thiophene

-
-
1822-71-5
n-pentylsodium

-
-
14593-46-5
sodium tert-pentoxide

-
-
15719-64-9, 15719-76-3, 97762-63-5
methylammonium carbonate

-
-
4282-31-9
Thiophene-2,5-dicarboxylic acid

| Conditions | Yield |
|---|---|
| aufeinanderfolgender Umsetzung; |

-
-
188290-36-0
thiophene

-
-
4282-31-9
Thiophene-2,5-dicarboxylic acid

| Conditions | Yield |
|---|---|
| With n-heptane; n-pentylsodium; sodium tert-pentoxide anschliessend Behandeln mit festem Kohlendioxid; |

| Conditions | Yield |
|---|---|
| With alkaline permanganate solution |
-
-
13679-74-8
2-Acetyl-5-methylthiophen

-
A
-
1918-79-2
2-methylthiophene-5-carboxylic acid

-
B
-
4282-31-9
Thiophene-2,5-dicarboxylic acid

| Conditions | Yield |
|---|---|
| With potassium hydroxide; potassium permanganate Behandeln des Reaktionsprodukts mit wss. Wasserstoffperoxid; |
-
-
13679-74-8
2-Acetyl-5-methylthiophen

-
-
4282-31-9
Thiophene-2,5-dicarboxylic acid

| Conditions | Yield |
|---|---|
| With alkaline permanganate solution | |
| With sodium hydroxide; potassium permanganate Oxidation; |
-
-
28569-48-4
2,5-bis-chloromethyl-thiophene

-
-
4282-31-9
Thiophene-2,5-dicarboxylic acid

| Conditions | Yield |
|---|---|
| With potassium hydroxide; potassium permanganate | |
| With potassium permanganate; acetone |

| Conditions | Yield |
|---|---|
| With sodium ethanolate Abdampfen der mit Wasser versetzten Mischung und Ansaeuern mit Salzsaeure; |
-
-
18761-46-1
1-(5-ethyl-thiophen-2-yl)-ethanone

-
-
4282-31-9
Thiophene-2,5-dicarboxylic acid

| Conditions | Yield |
|---|---|
| With alkaline permanganate solution |
-
-
188290-36-0
thiophene

-
-
124-38-9
carbon dioxide

-
A
-
527-72-0
2-thiophenylcarboxylic acid

-
B
-
4282-31-9
Thiophene-2,5-dicarboxylic acid

| Conditions | Yield |
|---|---|
| With n-butyllithium 1.) methylcyclohexane, THF, from 0 deg C to 25 deg C, 20 h, 2.) methylcyclohexane, THF, ether; Yield given. Multistep reaction. Yields of byproduct given; |


| Conditions | Yield |
|---|---|
| With n-butyllithium 1. THF/-50 - -55 deg C; Yield given. Multistep reaction; | |
| Stage #1: 2,5-dibromothiophen With iodine; magnesium In tetrahydrofuran; 1,4-dioxane at 60℃; for 4h; Inert atmosphere; Stage #2: carbon dioxide In tetrahydrofuran; 1,4-dioxane Solvent; Temperature; |
-
-
527-72-0
2-thiophenylcarboxylic acid

-
-
124-38-9
carbon dioxide

-
A
-
4282-31-9
Thiophene-2,5-dicarboxylic acid

-
B
-
1451-95-2
thiophene-2,3-dicarboxylic acid

| Conditions | Yield |
|---|---|
| With n-butyllithium 1.) THF/hexane, -78 deg C, 30 min, 2.) THF/hexane, -78 -> -10 deg C; Yield given. Multistep reaction. Yields of byproduct given. Title compound not separated from byproducts; |


-
-
4282-31-9
Thiophene-2,5-dicarboxylic acid

| Conditions | Yield |
|---|---|
| With nitric acid |

-
-
4282-31-9
Thiophene-2,5-dicarboxylic acid

| Conditions | Yield |
|---|---|
| With alkaline permanganate solution |

-
-
4282-31-9
Thiophene-2,5-dicarboxylic acid

| Conditions | Yield |
|---|---|
| With alkaline permanganate solution |

-
-
4282-31-9
Thiophene-2,5-dicarboxylic acid

| Conditions | Yield |
|---|---|
| With potassium cyanide Verseifen durch Kochen mit alkoh. Kali; |

| Conditions | Yield |
|---|---|
| With cadmium catalyst unter Kohlendioxid; |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: petroleum ether; aluminium chloride 2: alkaline permanganate solution View Scheme |

| Conditions | Yield |
|---|---|
| With sulfuric acid In water at 60℃; for 2h; Large scale; | 1679 g |
-
-
4282-31-9
Thiophene-2,5-dicarboxylic acid

-
-
64-17-5
ethanol

-
-
61755-85-9
2,5‐diethyl thiophene‐2,5‐dicarboxylate

| Conditions | Yield |
|---|---|
| With sulfuric acid Reflux; | 100% |
| With sulfuric acid for 120h; Reflux; | 90% |
| With graphene oxide at 100℃; for 24h; | 67% |
| With sulfuric acid at 85℃; for 72h; Inert atmosphere; | 3.2 g |
| for 6h; Reflux; |
-
-
67-56-1
methanol

-
-
4282-31-9
Thiophene-2,5-dicarboxylic acid

-
-
4282-34-2
Dimethyl thiophene-2,5-dicarboxylate

| Conditions | Yield |
|---|---|
| With thionyl chloride at 0 - 20℃; for 3h; Heating / reflux; | 99.9% |
| With thionyl chloride Reflux; | 99% |
| With thionyl chloride Reflux; | 99% |
-
-
280-57-9
1,4-diaza-bicyclo[2.2.2]octane

-
-
4282-31-9
Thiophene-2,5-dicarboxylic acid

-
-
584-03-2
1,2-dihydroxybutane

| Conditions | Yield |
|---|---|
| Stage #1: 1,4-diaza-bicyclo[2.2.2]octane; Thiophene-2,5-dicarboxylic acid; zinc(II) nitrate hexahydrate In N,N-dimethyl-formamide for 1h; Stage #2: 1,2-dihydroxybutane In N,N-dimethyl-formamide at 130℃; for 48h; Sealed tube; | 99% |


| Conditions | Yield |
|---|---|
| With boric acid In toluene at 170 - 240℃; Solvent; Temperature; Inert atmosphere; | 98.7% |
-
-
280-57-9
1,4-diaza-bicyclo[2.2.2]octane

-
-
4282-31-9
Thiophene-2,5-dicarboxylic acid

-
-
107-21-1
ethylene glycol

| Conditions | Yield |
|---|---|
| Stage #1: 1,4-diaza-bicyclo[2.2.2]octane; Thiophene-2,5-dicarboxylic acid; zinc(II) nitrate hexahydrate In N,N-dimethyl-formamide for 1h; Stage #2: ethylene glycol In N,N-dimethyl-formamide at 130℃; for 48h; Sealed tube; | 98% |


| Conditions | Yield |
|---|---|
| In ethanol for 2h; Reflux; | 97.9% |

| Conditions | Yield |
|---|---|
| In toluene for 24h; Reflux; water segregator; | 96% |
-
-
280-57-9
1,4-diaza-bicyclo[2.2.2]octane

-
-
57-55-6
propylene glycol

-
-
4282-31-9
Thiophene-2,5-dicarboxylic acid

| Conditions | Yield |
|---|---|
| Stage #1: 1,4-diaza-bicyclo[2.2.2]octane; Thiophene-2,5-dicarboxylic acid; zinc(II) nitrate hexahydrate In N,N-dimethyl-formamide for 1h; Stage #2: propylene glycol In N,N-dimethyl-formamide at 130℃; for 48h; Sealed tube; | 96% |

-
-
280-57-9
1,4-diaza-bicyclo[2.2.2]octane

-
-
4282-31-9
Thiophene-2,5-dicarboxylic acid

-
-
5343-92-0
1,2-pentanediol

| Conditions | Yield |
|---|---|
| Stage #1: 1,4-diaza-bicyclo[2.2.2]octane; Thiophene-2,5-dicarboxylic acid; zinc(II) nitrate hexahydrate In N,N-dimethyl-formamide for 1h; Stage #2: 1,2-pentanediol In N,N-dimethyl-formamide at 130℃; for 48h; Sealed tube; | 96% |

-
-
190773-57-0
N,N’,N’’,N’’’-tetrakis(2-pyridylmethyl)-1,6-diaminohexane

-
-
4282-31-9
Thiophene-2,5-dicarboxylic acid

-
-
6147-53-1
cobalt(II) diacetate tetrahydrate

| Conditions | Yield |
|---|---|
| Stage #1: N,N’,N’’,N’’’-tetrakis(2-pyridylmethyl)-1,6-diaminohexane; cobalt(II) diacetate tetrahydrate In methanol at 20℃; Stage #2: Thiophene-2,5-dicarboxylic acid In methanol at 20℃; | 96% |

-
-
4282-31-9
Thiophene-2,5-dicarboxylic acid

| Conditions | Yield |
|---|---|
| In toluene C4H2S(COOH)2 added to soln. of W2((CH3)3CCOO)4 in toluene; soln. stirredfor 7-10 d at rt; precipitate filtered off; detd. by elem. anal.; | 95% |
| In toluene under Ar atm. to soln. (W2(O2C(t)Bu)4) in toluene 2,5-Th(COOH)2 was added and stirred at room temp. for 5-10 days; ppt. was filtered, washed with toluene and hexane, and dried in vacuo; elem. anal.; | 95% |

| Conditions | Yield |
|---|---|
| With acetic acid In ethanol at 20℃; for 120 - 168h; pH=4 - 5; | 94% |


| Conditions | Yield |
|---|---|
| Stage #1: 1,4-diaza-bicyclo[2.2.2]octane; Thiophene-2,5-dicarboxylic acid; zinc(II) nitrate hexahydrate In N,N-dimethyl-formamide for 1h; Stage #2: glycerol In N,N-dimethyl-formamide at 130℃; for 48h; Sealed tube; | 94% |

-
-
4282-31-9
Thiophene-2,5-dicarboxylic acid

-
-
95-84-1
3-amino-4-hydroxytoluene

-
-
3723-19-1
5,5'-dimethyl-2,2'-thiophene-2,5-diyl-bis-benzooxazole

| Conditions | Yield |
|---|---|
| With boric acid; tin(ll) chloride In 1,2-dichloro-benzene at 220℃; for 24h; Inert atmosphere; Dean-Stark; | 94% |

| Conditions | Yield |
|---|---|
| In toluene for 24h; Reflux; water segregator; | 93% |
-
-
4282-31-9
Thiophene-2,5-dicarboxylic acid

-
-
20989-17-7
(2S)-2-phenylglycinol

-
-
1259933-47-5
(+)-2,5-bis[4'-(S)-phenyloxazolin-2'-yl]thiophene

| Conditions | Yield |
|---|---|
| In toluene for 24h; Reflux; water segregator; | 93% |
-
-
4282-31-9
Thiophene-2,5-dicarboxylic acid

-
-
1117-97-1
N,0-dimethylhydroxylamine

-
-
821797-87-9
N2,N5-dimethoxy-N2,N5-dimethylthiophene-2,5-dicarboxamide

| Conditions | Yield |
|---|---|
| Stage #1: Thiophene-2,5-dicarboxylic acid; N,0-dimethylhydroxylamine In toluene at 0℃; for 0.166667h; Stage #2: With phosphorus trichloride In toluene at 20 - 60℃; for 0.5h; | 92% |
-
-
4282-31-9
Thiophene-2,5-dicarboxylic acid

-
-
1349082-74-1
2-(5-{6-[5-(pyrazin-2-yl)-1H-1,2,4-triazol-3-yl]-pyridin-2-yl}-1H-1,2,4-triazol-3-yl)pyrazine

-
-
7732-18-5
water

| Conditions | Yield |
|---|---|
| With 3-ethyl-1-methyl-1H-imidazol-3-ium bromide In acetonitrile at 160℃; for 72h; Reagent/catalyst; Autoclave; | 92% |


| Conditions | Yield |
|---|---|
| In toluene for 24h; Reflux; water segregator; | 91% |
-
-
4282-31-9
Thiophene-2,5-dicarboxylic acid

| Conditions | Yield |
|---|---|
| In toluene C4H2S(COOH)2 added to soln. of Mo2((CH3)3CCOO)4 in toluene; soln. stirred for 7-10 d at rt; precipitate filtered off; detd. by elem. anal., MALDI; | 90% |
| In toluene under Ar atm. to soln. (Mo2(O2C(t)Bu)4) in toluene 2,5-Th(COOH)2 was added and stirred at room temp. for 5-10 days; ppt. was filtered; elem. anal.; | 90% |

| Conditions | Yield |
|---|---|
| With NH3*H2O In water High Pressure; mixt. of MnCl2*4H2O, thiohene-2,5-dicarboxylic acid, H2O adjusted to pH 7.4 by aq. soln. of NH3*H2O, placed in autoclave, heated at 110°Cfor 3 d, slow cooled to room temp.; ppt. filtered, washed with H2O; elem. anal.; | 90% |


| Conditions | Yield |
|---|---|
| With NH3*H2O In water High Pressure; mixt. of CoCl2*6H2O, thiohene-2,5-dicarboxylic acid, H2O adjusted to pH 7.4 by aq. soln. of NH3*H2O, placed in autoclave, heated at 110°Cfor 3 d, slow cooled to room temp.; ppt. filtered, washed with H2O; elem. anal.; | 90% |

-
-
4282-31-9
Thiophene-2,5-dicarboxylic acid

-
-
68-12-2, 33513-42-7
N,N-dimethyl-formamide

| Conditions | Yield |
|---|---|
| at 120℃; for 72h; | 90% |


| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide at 135℃; for 24h; | 90% |

-
-
4282-31-9
Thiophene-2,5-dicarboxylic acid

-
-
6147-53-1
cobalt(II) diacetate tetrahydrate

-
-
263713-26-4
N,N’-(1,4-phenylenebis(methylene))bis(1-(pyridin-2-yl)-N-(pyridin-2-ylmethyl)methanamine)

| Conditions | Yield |
|---|---|
| Stage #1: cobalt(II) diacetate tetrahydrate; N,N’-(1,4-phenylenebis(methylene))bis(1-(pyridin-2-yl)-N-(pyridin-2-ylmethyl)methanamine) In methanol at 20℃; Stage #2: Thiophene-2,5-dicarboxylic acid In methanol at 20℃; | 90% |

-
-
4282-31-9
Thiophene-2,5-dicarboxylic acid

-
-
95-55-6
2-amino-phenol

-
-
2866-43-5
2,2'-thiophene-2,5-diylbis(benzoxazole)

| Conditions | Yield |
|---|---|
| With boric acid; tin(ll) chloride In 1,2-dichloro-benzene at 220℃; for 24h; Inert atmosphere; Dean-Stark; | 89.23% |
| Stage #1: Thiophene-2,5-dicarboxylic acid; 2-amino-phenol at 150℃; for 2h; Inert atmosphere; Stage #2: With ammonia In water at 20℃; pH=~ 10 - 11; Cooling with cold water; | 5.5% |
| With boric acid In 1-methyl-pyrrolidin-2-one; toluene at 20 - 185℃; for 13h; |
-
-
4282-31-9
Thiophene-2,5-dicarboxylic acid

-
-
112245-13-3
(S)-tert-leucinol

| Conditions | Yield |
|---|---|
| In toluene for 24h; Reflux; water segregator; | 89% |

| Conditions | Yield |
|---|---|
| at 120℃; for 120h; High pressure; | A 89% B n/a |

-
-
64-18-6
formic acid

-
-
4282-31-9
Thiophene-2,5-dicarboxylic acid

-
-
10026-11-6
zirconium(IV) chloride

-
-
68-12-2, 33513-42-7
N,N-dimethyl-formamide

| Conditions | Yield |
|---|---|
| In 1-methyl-pyrrolidin-2-one at 120℃; for 48h; Sonication; | 89% |

-
-
64-18-6
formic acid

-
-
4282-31-9
Thiophene-2,5-dicarboxylic acid

-
-
10026-11-6
zirconium(IV) chloride

-
-
68-12-2, 33513-42-7
N,N-dimethyl-formamide

| Conditions | Yield |
|---|---|
| In 1-methyl-pyrrolidin-2-one at 80℃; for 48h; | 89% |

2,5-Thiophenedicarboxylic acid Chemical Properties
Empirical Formula: C6H4O4S
Molecular Weight: 172.1586
Index of Refraction: 1.662
Molar Refractivity: 38.49 cm3
Molar Volume: 103.9 cm3
Surface Tension: 83.9 dyne/cm
Density: 1.655 g/cm3
Flash Point: 222.7 °C
Enthalpy of Vaporization: 74.03 kJ/mol
Boiling Point: 444.6 °C at 760 mmHg
Vapour Pressure: 1.1E-08 mmHg at 25 °C
Melting point: >300 °C(lit.)
Appearance: White to light yellow crystal powder
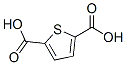
Structure of 2,5-Thiophenedicarboxylic acid (CAS NO.4282-31-9):
Product Category of 2,5-Thiophenedicarboxylic acid (CAS NO.4282-31-9): Thiophenes;Thiophens;Functional Materials;Reagents for Conducting Polymer Research;Thiophene Derivatives (for Conduting Polymer Research);Thiophen;Heterocyclic Building Blocks
2,5-Thiophenedicarboxylic acid Safety Profile
Hazard Codes:  Xi
Xi
Risk Statements: 36/37/38
R36/37/38:Irritating to eyes, respiratory system and skin.
Safety Statements: 26-36-37/39
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S36:Wear suitable protective clothing.
S37/39:Wear suitable gloves and eye/face protection.
2,5-Thiophenedicarboxylic acid Specification
2,5-Thiophenedicarboxylic acid , its cas register number is 4282-31-9. It also can be called Thiophene-2,5-dicarboxylic acid ; and NSC 81789 . It is hazardous, so the first aid measures and others should be known. Such as: When on the skin: first, should flush skin with plenty of water immediately for at least 15 minutes while removing contaminated clothing. Secondly, get medical aid. Or in the eyes: Flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids. Then get medical aid soon. While, it's inhaled: Remove from exposure and move to fresh air immediately. Give artificial respiration while not breathing. When breathing is difficult, give oxygen. And as soon as to get medical aid. Then you have the ingesting of the product: Wash mouth out with water, and get medical aid immediately. Notes to physician: Treat supportively and symptomatically.
In addition, 2,5-Thiophenedicarboxylic acid (CAS NO.4282-31-9) is not compatible with strong oxidizing agents, and you must not take it with incompatible materials. And also prevent it to broken down into hazardous decomposition products: Carbon monoxide, carbon dioxide, sulfur oxides (SOx), including sulfur oxide and sulfur dioxide.
Related Products
- 20,22-DIHYDRODIGITOXIN
- 20,29,30-Trinorlupane,(17alpha)-
- 20-ETHYL-6-β,8-DIHYDROXY-1-α-METHOXY-4-METHYLHETERATISAN-14-ONE
- 20-Ethylprostaglandin F2-alpha
- 20-Isopropylcholanthrene
- 20-METHYLCHOLANTHREN-15-ONE
- 20-METHYLCHOLANTHRENE PICRATE
- 20-METHYLCHOLANTHRENE-TRINITRO-BENZENE
- 20(S)-Ginsenoside C-K
- 2,10-DIFLUOROBENZO(rst)PENTAPHENE
- 4282-33-1
- 4282-34-2
- 42823-46-1
- 42823-72-3
- 4282-40-0
- 4282-42-2
- 4282-44-4
- 4282-45-5
- 4282-46-6
- 4282-47-7
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View