Dayang Chem (Hangzhou) Co.,Ltd.
Dayangchem's R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. DayangChem can provide different quantities
Cas:584-03-2
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquiryAlity Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Xi'an Xszo Chem Co., Ltd.
1. Factory price and high quality must be guaranteed, base on 8 years of production and R&D experience2. Free samples will be provided,ensure specifications and quality are right for customer3. Customers will receive the most professional technical s
Cas:584-03-2
Min.Order:1 Gram
FOB Price: $0.1
Type:Manufacturers
inquiryChemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Cas:584-03-2
Min.Order:5 Kiloliter
FOB Price: $1.2 / 5.0
Type:Manufacturers
inquiryHebei Nengqian Chemical Import and Export Co., LTD
Our Advantage Rich Experience Our products are sold all over Europe,North&South America, Sino-East, Asia and pacific area as well as Africa,we establish long term. Quality service Company cooperates with research institutes. We strictly con
Cas:584-03-2
Min.Order:1 Kilogram
FOB Price: $15.0 / 50.0
Type:Trading Company
inquiryZhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:584-03-2
Min.Order:10 Gram
FOB Price: $146.0 / 176.0
Type:Trading Company
inquiryHebei Sankai Chemical Technology Co., Ltd
1. Product advantages ♦ High purity, all above 98.5%, no impurities after dissolution ♦ We will test each batch to ensure quality ♦ OEM and private brand services designed for free ♦ Various cap colors available ♦ W
Cas:584-03-2
Min.Order:1 Kilogram
FOB Price: $7.0 / 9.0
Type:Trading Company
inquiryShanghai Upbio Tech Co.,Ltd
1.In No Less 10 years exporting experience. you can 100% received goods 2.Lower Price with higher quality 3,Free sample 4,We are sincerely responsible for the "product quality" and "After Service" Upbio is Specializ
Qingdao Beluga Import and Export Co., LTD
1,2-BUTANEDIOL CAS:8584-03-2 Qingdao Belugas Import and Export Co., Ltd. is a scientific and technological company integrating research and development, production and trade of chemical intermediates, specializing in high quality organic intermediat
Cas:584-03-2
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryChangchun Artel lmport and Export trade company
Product Detail Minimum Order Qty. 10 Gram Supply Ability 500 Kilograms/Month Storage store in cool, dry, ventilated place 20℃ Delivery Time 3 business days after payment Payment Term TT,western union,Paypal,
Cas:584-03-2
Min.Order:20 Metric Ton
Negotiable
Type:Trading Company
inquiryHenan Wentao Chemical Product Co., Ltd.
Henan Wentao Chemical Product Co.,Ltd is Located in Zhengzhou High-tech Development Zone with import and export license, We passed ISO 9001:2008 as well, Henan Wentao has developed more than 1000 compounds, which are widely used in the fields of prod
SHANGHAI T&W PHARMACEUTICAL CO., LTD.
A substitute for perfluorooctanoic acid, mainly used as a surfactant, dispersant, additive, etc Appearance:White solid or Colorless liquid Purity:99.3 % We will ship the goods in a timely manner as required We can provide relevant documents acc
Hangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Zibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:584-03-2
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquiryHANGZHOU YUNUO CHEMICAL CO.,LTD
Superior quality, moderate price & quick delivery. Appearance:white powder Storage:Stored in cool, dry and ventilation place; Away from fire and heat Package:25kg/drum, or as per your request. Application:Used as Pharmaceutical Intermediates Tr
Jiangsu Qianyu Molecular Technology Co., LTD.
Our Advantages A. International Top level TechnologyOur company owned biomedicine experts are famous at home and abroad with rich experience in research and development in the field of efficient chiral functional molecules research and development an
Xiamen Jenny Chemical Technology Co., Ltd.
GMP standard, high purity, competitive price, in stock 1. Quick Response: within 6 hours after receiving your email. 2. Quality Guarantee: All products are strictly tested by our QC, confirmed by QA, and approved by a third-party lab in China, USA,
Hangzhou Huarong Pharm Co., Ltd.
Hangzhou Huarong Pharm Co., Ltd.established since 2006 , has been actively developing specialty products for Finished Dosages, APIs, Intermediates, and Fine chemicals markets in North America, Europe, Korea, Japan, Mid-East and all over the World. Hu
TAIZHOU ZHENYU BIOTECHNOLOGY CO., LTD
Zhenyu biotech exported this product to many countries and regions at best price. if you are looking for the material's manufacturer or supplier in china, zhenyu biotech is your best choice. pls contact with us freely for getting detailed
Cas:584-03-2
Min.Order:1 Kilogram
FOB Price: $2.0
Type:Lab/Research institutions
inquiryKAISA GROUP INC
1.Applied in food field.it can improve the immune system and prolong life. 2.Appliedin cosmetic field.it can improve the skin care. 3.Applied in pharmaceutical field.it can treat various dieases. 4.Our product quality assurance will make our customer
Cas:584-03-2
Min.Order:1 Metric Ton
FOB Price: $7.0 / 8.0
Type:Trading Company
inquiryHunan chemfish Pharmaceutical co.,Ltd
Appearance:95%+ Package:R&D,Pilot run Transportation:per client require Port:Express ,Air, Sea
Golden Pharma Co., Limited
GOLDEN PHARMA CO.,LIMITED.is a professional pharmaceutical company,our team have more than 20years expereince in pharmaceutical production and sales. we are a professional technical enterprise specializing in the R & D, production,QA regulation
Kono Chem Co.,Ltd
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:drum and bag Application:for pharma use Transportation:by sea or air Port:Beijing or Guangzhou
Henan Allgreen Chemical Co.,Ltd
high quality Storage:Sealed, dry, microtherm , avoid light and smell. Package:According to the demand of customer Application:Organic synthesis Transportation:by air or by sea
Henan Tianfu Chemical Co., Ltd.
1.Our services:A.Supply sampleB.The packing also can be according the customers` requirmentC.Any inquiries will be replied within 24 hoursD.we provide Commerical Invoice, Packing List, Bill of loading, COA , Health certificate and Origin certificate.
Wuhan Circle Star Chem-medical Technology co.,Ltd.
1,we produce and sell good chemicals around the world.2,our success rate is about 95%. this means, if customer order is accepted, the probability that the customer will obtain the ordered substances, is 95%.3,our staff consists of highly qualified in
Antimex Chemical Limied
Ansciep Chemical is a professional enterprise manufacturing and distributing fine chemicals and speciality chemicals. We have been dedicated to heterocycle compounds and phenyl rings for tens of years. This is our mature product for export. Our quali
Synthetic route
-
-
59765-97-8
4-ethyl-2-methyl-1,3-dioxolane

-
-
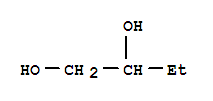
584-03-2
1,2-dihydroxybutane

| Conditions | Yield |
|---|---|
| With water under 76.0051 - 760.051 Torr; Reflux; | 98.5% |
-
-
4437-85-8, 90970-72-2, 90971-04-3, 127128-77-2
1,2-butylene carbonate

-
A
-
67-56-1
methanol

-
B
-
584-03-2
1,2-dihydroxybutane

| Conditions | Yield |
|---|---|
| With (bis[(2-diisopropylphosphino)ethyl]amine)Mn(CO)2Br; hydrogen; sodium t-butanolate In tetrahydrofuran at 120℃; under 22502.3 Torr; for 26h; Schlenk technique; Glovebox; Autoclave; | A 74% B 97% |
| With carbonylhydrido(tetrahydroborato)[bis(2-diphenylphosphinoethyl)-amino]ruthenium(II); potassium carbonate In isopropyl alcohol at 140℃; Glovebox; | A 94 %Chromat. B 94% |
| With [bis({2‐[bis(propan‐2‐yl)phosphanyl]ethyl})amine](bromo)(carbonyl)(hydride)iron(II); potassium tert-butylate; isopropyl alcohol In tetrahydrofuran at 140℃; for 18h; Catalytic behavior; Mechanism; Concentration; Solvent; Temperature; Reagent/catalyst; Time; Inert atmosphere; Schlenk technique; Green chemistry; | A 83 %Chromat. B 72% |

-
-
584-03-2
1,2-dihydroxybutane

| Conditions | Yield |
|---|---|
| With silica gel; triethylamine In diethyl ether; Petroleum ether Substitution; Detrifluoroacetylation; | 96% |

| Conditions | Yield |
|---|---|
| With water In N,N-dimethyl-formamide at 110℃; for 20h; | 92% |
| With water at 90℃; under 11251.1 Torr; | 92.8% |
| With water at 90℃; under 11251.1 Torr; | 92.7% |
-
-
4437-85-8, 90970-72-2, 90971-04-3, 127128-77-2
1,2-butylene carbonate

-
-
584-03-2
1,2-dihydroxybutane

| Conditions | Yield |
|---|---|
| With potassium tert-butylate; hydrogen; C16H18BrCoINO2 In dibutyl ether at 160℃; under 45004.5 Torr; for 20h; Sealed tube; Autoclave; | 92% |
| With ammonium bromide; ethylenediamine at 80℃; for 5h; Microwave irradiation; | > 95 %Spectr. |
-
-
600-15-7, 565-70-8
2-Hydroxybutanoic acid

-
-
584-03-2
1,2-dihydroxybutane

| Conditions | Yield |
|---|---|
| With hydrogen In water at 119.84℃; under 7500.75 - 60006 Torr; for 2h; Catalytic behavior; Reagent/catalyst; Autoclave; Sealed tube; | 92% |

| Conditions | Yield |
|---|---|
| Stage #1: 1-butylene With Oxone; potassium chloride In water; acetone at 30℃; for 12h; Large scale; Stage #2: With water; potassium hydroxide at 25 - 40℃; for 4h; pH=12; Large scale; | 88% |
| With dihydrogen peroxide; tungsten(VI) oxide; acetic acid at 70℃; | |
| With formic acid; dihydrogen peroxide at -17℃; beim anschliessenden Erwaermen mit wss. Natronlauge; |
-
-
106-88-7
ethyloxirane

-
-
124-38-9
carbon dioxide

-
-
62-53-3
aniline

-
A
-
101835-17-0
5-ethyl-3-phenyl-1,3-oxazolidine-2-one

-
B
-
584-03-2
1,2-dihydroxybutane

| Conditions | Yield |
|---|---|
| With C24H25N4O3(1+)*I(1-); 1,8-diazabicyclo[5.4.0]undec-7-ene at 110℃; under 3750.38 Torr; for 25h; Autoclave; | A 80% B n/a |


| Conditions | Yield |
|---|---|
| With hydrogen In 1,4-dioxane at 159.84℃; under 60006 Torr; for 24h; Autoclave; | A 12% B 79% |
| With hydrogen In 1,4-dioxane at 139.84℃; under 60006 Torr; for 4h; Autoclave; |
-
-
167354-12-3
(2R)-1-(benzyloxy)-2-butanol

-
-
584-03-2
1,2-dihydroxybutane

| Conditions | Yield |
|---|---|
| With palladium 10% on activated carbon; hydrogen In ethanol | 79% |
-
-
62214-39-5
(2S)-but-3-ene-1,2-diol

-
-
584-03-2
1,2-dihydroxybutane

| Conditions | Yield |
|---|---|
| In palladium-carbon; ethanol | 68% |
-
-
64244-53-7
<(2-tetrahydropyranoyloxy)methyl>oxirane

-
-
75-16-1
methylmagnesium bromide

-
-
584-03-2
1,2-dihydroxybutane

| Conditions | Yield |
|---|---|
| Stage #1: <(2-tetrahydropyranoyloxy)methyl>oxirane; methylmagnesium bromide With copper(I) bromide In tetrahydrofuran at -10℃; for 1h; Inert atmosphere; Stage #2: With toluene-4-sulfonic acid In methanol at 20℃; | 64% |
-
-
56-81-5
glycerol

-
A
-
57-55-6
propylene glycol

-
B
-
110-63-4
Butane-1,4-diol

-
C
-
513-85-9
2.3-butanediol

-
D
-
584-03-2
1,2-dihydroxybutane

-
E
-
1825-14-5, 3950-21-8, 36402-52-5, 42075-32-1, 72345-23-4, 625-69-4
pentane-1,3-diol

| Conditions | Yield |
|---|---|
| With sodium hydroxide; hydrogen at 212℃; under 51716.2 Torr; Product distribution / selectivity; | A 60% B n/a C n/a D n/a E n/a |

-
-
124-38-9
carbon dioxide

-
-
201230-82-2
carbon monoxide

-
A
-
67-56-1
methanol

-
B
-
71-23-8
propan-1-ol

-
C
-
57-55-6
propylene glycol

-
D
-
626-95-9
1,4-Pentanediol

-
E
-
111-29-5
1 ,5-pentanediol

-
F
-
110-63-4
Butane-1,4-diol

-
G
-
60-29-7
diethyl ether

-
H
-
34557-54-5
methane

-
I
-
64-17-5
ethanol

-
J
-
6032-29-7
(+/-)-2-pentanol

-
K
-
18826-95-4, 107-88-0
1.3-butanediol

-
L
-
5343-92-0
1,2-pentanediol

-
M
-
74-84-0
ethane

-
N
-
74-98-6
propane

-
O
-
115-10-6
Dimethyl ether

-
P
-
540-67-0
ethyl methyl ether

-
Q
-
78-83-1
2-methyl-propan-1-ol

-
R
-
584-02-1
2-pentanol

-
S
-
71-41-0
pentan-1-ol

-
T
-
3174-67-2
1,3-pentanediol

-
U
-
79-20-9
acetic acid methyl ester

-
V
-
67-63-0
isopropyl alcohol

-
W
-
78-92-2, 15892-23-6
iso-butanol

-
X
-
75-65-0
tert-butyl alcohol

-
Y
-
71-36-3
butan-1-ol

-
Z
-
584-03-2
1,2-dihydroxybutane

| Conditions | Yield |
|---|---|
| With hydrogen; Cu/Co/Al at 260 - 320℃; under 45004.5 - 75007.5 Torr; Conversion of starting material; | A 57.5% B n/a C n/a D n/a E n/a F n/a G n/a H n/a I 28.5% J n/a K n/a L n/a M n/a N n/a O n/a P n/a Q n/a R n/a S n/a T n/a U n/a V n/a W n/a X n/a Y n/a Z n/a |
| With hydrogen; Cu/ZnO/Al2O3 at 285 - 300℃; under 45004.5 - 67506.8 Torr; Conversion of starting material; | A 57.5% B n/a C n/a D n/a E n/a F n/a G n/a H n/a I 28.5% J n/a K n/a L n/a M n/a N n/a O n/a P n/a Q n/a R n/a S n/a T n/a U n/a V n/a W n/a X n/a Y n/a Z n/a |

-
-
124-38-9
carbon dioxide

-
-
201230-82-2
carbon monoxide

-
A
-
67-56-1
methanol

-
B
-
71-23-8
propan-1-ol

-
C
-
57-55-6
propylene glycol

-
D
-
626-95-9
1,4-Pentanediol

-
E
-
111-29-5
1 ,5-pentanediol

-
F
-
110-63-4
Butane-1,4-diol

-
G
-
60-29-7
diethyl ether

-
H
-
64-17-5
ethanol

-
I
-
6032-29-7
(+/-)-2-pentanol

-
J
-
18826-95-4, 107-88-0
1.3-butanediol

-
K
-
5343-92-0
1,2-pentanediol

-
L
-
115-10-6
Dimethyl ether

-
M
-
540-67-0
ethyl methyl ether

-
N
-
78-83-1
2-methyl-propan-1-ol

-
O
-
584-02-1
2-pentanol

-
P
-
71-41-0
pentan-1-ol

-
Q
-
3174-67-2
1,3-pentanediol

-
R
-
79-20-9
acetic acid methyl ester

-
S
-
67-63-0
isopropyl alcohol

-
T
-
78-92-2, 15892-23-6
iso-butanol

-
U
-
75-65-0
tert-butyl alcohol

-
V
-
71-36-3
butan-1-ol

-
W
-
584-03-2
1,2-dihydroxybutane

| Conditions | Yield |
|---|---|
| With hydrogen; Zn/Cr/K at 350 - 420℃; under 75007.5 - 135014 Torr; Conversion of starting material; | A 57.5% B n/a C n/a D n/a E n/a F n/a G n/a H 28.5% I n/a J n/a K n/a L n/a M n/a N n/a O n/a P n/a Q n/a R n/a S n/a T n/a U n/a V n/a W n/a |

-
-
56-81-5
glycerol

-
A
-
57-55-6
propylene glycol

-
B
-
107-21-1
ethylene glycol

-
C
-
513-85-9
2.3-butanediol

-
D
-
584-03-2
1,2-dihydroxybutane

| Conditions | Yield |
|---|---|
| With sodium hydroxide; hydrogen; HC-1 catalyst at 193 - 216℃; under 82745.9 Torr; Product distribution / selectivity; | A 56% B 4.37% C n/a D n/a |


-
A
-
57-55-6
propylene glycol

-
B
-
50-70-4
D-sorbitol

-
C
-
107-21-1
ethylene glycol

-
D
-
584-03-2
1,2-dihydroxybutane

| Conditions | Yield |
|---|---|
| With phosphotungstic acid; ruthenium-carbon composite; hydrogen In water at 250℃; under 45004.5 Torr; for 0.833333h; Autoclave; | A 5.5% B 12.4% C 53.1% D 6.2% |

-
-
13814-27-2
1,2-butanediol diacetate

-
A
-
55378-41-1
1-hydroxy-2-acetoxybutane

-
B
-
584-03-2
1,2-dihydroxybutane

| Conditions | Yield |
|---|---|
| 1,3-disubstituted tetraalkyldistannoxane (X = Y = Cl) In methanol; chloroform for 120h; Ambient temperature; | A 53% B 22% |
-
-
56-81-5
glycerol

-
A
-
57-55-6
propylene glycol

-
B
-
513-85-9
2.3-butanediol

-
C
-
584-03-2
1,2-dihydroxybutane

-
D
-
1825-14-5, 3950-21-8, 36402-52-5, 42075-32-1, 72345-23-4, 625-69-4
pentane-1,3-diol

| Conditions | Yield |
|---|---|
| With sodium hydroxide; hydrogen at 209 - 213℃; under 51716.2 - 82745.9 Torr; Product distribution / selectivity; | A 51% B n/a C n/a D n/a |

-
-
106-98-9
1-butylene

-
A
-
86943-35-3
(+/-)-2-hydroxybutanal

-
B
-
123-38-6
propionaldehyde

-
C
-
5077-67-8
1-Hydroxy-2-butanone

-
D
-
584-03-2
1,2-dihydroxybutane

| Conditions | Yield |
|---|---|
| With air; dihydrogen peroxide for 0.5h; Product distribution; Kinetics; Further Variations:; reaction time; Oxidation; UV-irradiation; | A 7% B 50.6% C 24.2% D 18.2% |


| Conditions | Yield |
|---|---|
| With Rh/C; hydrogen; ortho-tungstic acid; copper(II) sulfate In water at 245℃; under 45004.5 Torr; for 0.5h; Autoclave; | A 7.4% B 50% C 6.7% |
| With phosphotungstic acid; platinum on activated charcoal; hydrogen In water at 250℃; under 45004.5 Torr; for 0.833333h; Autoclave; | A 12.8% B 23.2% C 6% |
| With hydrogen In water at 219.84℃; under 31503.2 - 48754.9 Torr; for 8h; Autoclave; |

-
-
56-81-5
glycerol

-
A
-
67-56-1
methanol

-
B
-
849585-22-4
LACTIC ACID

-
C
-
57-55-6
propylene glycol

-
D
-
64-17-5
ethanol

-
E
-
107-21-1
ethylene glycol

-
F
-
513-85-9
2.3-butanediol

-
G
-
584-03-2
1,2-dihydroxybutane

-
H
-
1825-14-5, 3950-21-8, 36402-52-5, 42075-32-1, 72345-23-4, 625-69-4
pentane-1,3-diol

| Conditions | Yield |
|---|---|
| With sodium hydroxide; hydrogen; solid catalyst similar to the “G” catalyst disclosed in U.S. Pat. No. 6,479,713 or the “HC-1” catalyst available from Sud Chemie (Louisville, Ky.) In water at 154 - 229℃; under 62059.4 - 82745.9 Torr; Product distribution / selectivity; | A 0.6% B 0.7% C 47.3% D 0.1% E 2.1% F n/a G n/a H n/a |

-
-
50-70-4
D-sorbitol

-
A
-
106-69-4
hexane-1,2,6-triol

-
B
-
140946-33-4
1,4,5-hexanetriol

-
C
-
20221-50-5, 24211-51-6, 122923-46-0, 5581-21-5
1,2,5,6-hexanetetrol

-
D
-
584-03-2
1,2-dihydroxybutane

| Conditions | Yield |
|---|---|
| With propylene glycol; hydrogen; copper In water at 210℃; under 93089.1 Torr; Concentration; | A 5.51% B 26.54% C 46.12% D 10.26% |

-
-
56-81-5
glycerol

-
A
-
57-55-6
propylene glycol

-
B
-
513-85-9
2.3-butanediol

-
C
-
584-03-2
1,2-dihydroxybutane

| Conditions | Yield |
|---|---|
| With sodium hydroxide; hydrogen at 202℃; under 82745.9 Torr; Product distribution / selectivity; | A 43% B n/a C n/a |
| With sodium hydroxide; hydrogen; solid catalyst similar to the "G" catalyst disclosed in US 6,479,713 or the "HC-1" catalyst available from Sd Chemie (Louisville, KY) at 154 - 229℃; under 62059.4 - 82745.9 Torr; Product distribution / selectivity; | A 33.2% B n/a C n/a |
| With sodium hydroxide; hydrogen at 196℃; under 82745.9 Torr; Product distribution / selectivity; | A 27% B n/a C n/a |
| With sodium hydroxide; hydrogen; solid catalyst similar to the “G” catalyst disclosed in U.S. Pat. No. 6,479,713 or the “HC-1” catalyst available from Sud Chemie (Louisville, Ky.) In water at 150 - 210℃; under 51716.2 Torr; Product distribution / selectivity; | A 10% B n/a C n/a |
| With sodium hydroxide; hydrogen; Süd Chemie HC-1 catalyst In water at 159 - 212℃; under 96435 Torr; Product distribution / selectivity; |


-
A
-
57-55-6
propylene glycol

-
B
-
2319-57-5, 2418-52-2, 6968-16-7, 7493-90-5, 7541-59-5
1,2,3,4-butanetetrol

-
C
-
107-21-1
ethylene glycol

-
D
-
584-03-2
1,2-dihydroxybutane

| Conditions | Yield |
|---|---|
| With ruthenium-carbon composite; hydrogen In water at 215℃; under 39003.9 Torr; for 1.5h; Catalytic behavior; Reagent/catalyst; Autoclave; | A 9.6% B 9% C 40.6% D 7.8% |

-
-
56-81-5
glycerol

-
A
-
849585-22-4
LACTIC ACID

-
B
-
57-55-6
propylene glycol

-
C
-
64-17-5
ethanol

-
D
-
107-21-1
ethylene glycol

-
E
-
513-85-9
2.3-butanediol

-
F
-
584-03-2
1,2-dihydroxybutane

-
G
-
1825-14-5, 3950-21-8, 36402-52-5, 42075-32-1, 72345-23-4, 625-69-4
pentane-1,3-diol

| Conditions | Yield |
|---|---|
| With sodium hydroxide; hydrogen; solid catalyst similar to the “G” catalyst disclosed in U.S. Pat. No. 6,479,713 or the “HC-1” catalyst available from Sud Chemie (Louisville, Ky.) In water at 178 - 198℃; under 62059.4 Torr; Product distribution / selectivity; | A 1% B 38% C 0.1% D 1.6% E n/a F n/a G n/a |


| Conditions | Yield |
|---|---|
| With hydrogen; 5%-palladium/activated carbon In ethanol at 20℃; under 2625.26 Torr; for 16h; | 37% |
-
-
50-99-7
D-glucose

-
A
-
57-55-6
propylene glycol

-
B
-
107-21-1
ethylene glycol

-
C
-
584-03-2
1,2-dihydroxybutane

| Conditions | Yield |
|---|---|
| With palladium on activated charcoal; water; zinc at 250℃; for 0.5h; Temperature; | A 33.3% B 7.1% C 7.6% |
| With 5% active carbon-supported ruthenium; hydrogen; ortho-tungstic acid; sodium hydroxide In water; glycerol at 200℃; under 37503.8 Torr; for 4h; |

-
-
56-81-5
glycerol

-
A
-
67-56-1
methanol

-
B
-
849585-22-4
LACTIC ACID

-
C
-
57-55-6
propylene glycol

-
D
-
107-21-1
ethylene glycol

-
E
-
513-85-9
2.3-butanediol

-
F
-
584-03-2
1,2-dihydroxybutane

-
G
-
1825-14-5, 3950-21-8, 36402-52-5, 42075-32-1, 72345-23-4, 625-69-4
pentane-1,3-diol

| Conditions | Yield |
|---|---|
| With sodium hydroxide; hydrogen; solid catalyst similar to the “G” catalyst disclosed in U.S. Pat. No. 6,479,713 or the “HC-1” catalyst available from Sud Chemie (Louisville, Ky.) In water at 185 - 205℃; under 82745.9 Torr; Product distribution / selectivity; | A 0.7% B 0.5% C 33.2% D 1.5% E n/a F n/a G n/a |

-
-
56-81-5
glycerol

-
A
-
849585-22-4
LACTIC ACID

-
B
-
57-55-6
propylene glycol

-
C
-
107-21-1
ethylene glycol

-
D
-
513-85-9
2.3-butanediol

-
E
-
584-03-2
1,2-dihydroxybutane

-
F
-
1825-14-5, 3950-21-8, 36402-52-5, 42075-32-1, 72345-23-4, 625-69-4
pentane-1,3-diol

| Conditions | Yield |
|---|---|
| With sodium hydroxide; hydrogen at 177 - 228℃; under 62059.4 - 82745.9 Torr; Product distribution / selectivity; | A 0.5% B 33.2% C 1.5% D n/a E n/a F n/a |


| Conditions | Yield |
|---|---|
| With triethylamine In toluene Cyclization; | 99% |

| Conditions | Yield |
|---|---|
| With ruthenium(III) acetate at 20℃; for 8h; | 99% |

-
-
584-03-2
1,2-dihydroxybutane

-
-
943433-23-6
[Ti(N-phenylsalicylideneimine(-H))2(OCH2CH(C2H5)O)]

| Conditions | Yield |
|---|---|
| In benzene byproducts: (CH3)2CHOH; under unhydrous atm., soln. of ligand added to soln. of Ti compd. (3.06:3.01), refluxed, monitored by isopropanol liberated; concd., crystd., elem. anal.; | 99% |
-
-
124-38-9
carbon dioxide

-
-
584-03-2
1,2-dihydroxybutane

-
-
4437-85-8, 90970-72-2, 90971-04-3, 127128-77-2
1,2-butylene carbonate

| Conditions | Yield |
|---|---|
| With 2-Cyanopyridine; cerium(IV) oxide at 139.84℃; under 37503.8 Torr; for 1h; Autoclave; | 99% |
| With 1,10-Phenanthroline; calcium carbide; zinc trifluoromethanesulfonate In 1-methyl-pyrrolidin-2-one at 180℃; under 37503.8 Torr; for 24h; Autoclave; Glovebox; Sealed tube; | 82% |
| Stage #1: carbon dioxide With 1-butyl-3-methyl-1H-imidazol-3-iumhydrogencarbonate at 25℃; under 760.051 Torr; for 6h; Stage #2: 1,2-dihydroxybutane at 25℃; under 760.051 Torr; for 12h; | 40.4% |
| With 1,8-diazabicyclo[5.4.0]undec-7-ene; 2-methyl-but-3-yn-2-ol In N,N-dimethyl-formamide at 120℃; under 22502.3 Torr; for 10h; Autoclave; | 99 %Chromat. |
| With tetraethylammonium iodide In acetonitrile at 20℃; under 760.051 Torr; Electrolysis; | 47.6 %Chromat. |
-
-
616-38-6
carbonic acid dimethyl ester

-
-
584-03-2
1,2-dihydroxybutane

-
-
4437-85-8, 90970-72-2, 90971-04-3, 127128-77-2
1,2-butylene carbonate

| Conditions | Yield |
|---|---|
| In neat (no solvent) at 110℃; for 24h; Temperature; Green chemistry; | 99% |
| With zinc(II) trifluoroacetate hydrate; 1,3-bis(imidazol-1-ylmethyl)benzene for 9h; Reagent/catalyst; Reflux; | |
| With tetrabutylammomium bromide at 180℃; under 8250.83 Torr; for 0.05h; Flow reactor; | |
| With sodium methylate In methanol at 64 - 90℃; |
-
-
280-57-9
1,4-diaza-bicyclo[2.2.2]octane

-
-
4282-31-9
Thiophene-2,5-dicarboxylic acid

-
-
584-03-2
1,2-dihydroxybutane

| Conditions | Yield |
|---|---|
| Stage #1: 1,4-diaza-bicyclo[2.2.2]octane; Thiophene-2,5-dicarboxylic acid; zinc(II) nitrate hexahydrate In N,N-dimethyl-formamide for 1h; Stage #2: 1,2-dihydroxybutane In N,N-dimethyl-formamide at 130℃; for 48h; Sealed tube; | 99% |


| Conditions | Yield |
|---|---|
| In benzene refluxing (10 h); recrystn. (benzene/CH2Cl2); elem. anal.; | 98.95% |

| Conditions | Yield |
|---|---|
| With iron(III) chloride hexahydrate at 20℃; for 2h; | 98% |

| Conditions | Yield |
|---|---|
| With C29H44IrN5P2; potassium tert-butylate In diethylene glycol dimethyl ether at 130℃; for 20h; Solvent; Reagent/catalyst; Temperature; Inert atmosphere; Glovebox; | 98% |
-
-
201230-82-2
carbon monoxide

-
-
584-03-2
1,2-dihydroxybutane

-
-
4437-85-8, 90970-72-2, 90971-04-3, 127128-77-2
1,2-butylene carbonate

| Conditions | Yield |
|---|---|
| With N-chloro-succinimide; (neocuproine)Pd(OAc)2; sodium acetate In acetonitrile at 55℃; under 760.051 Torr; for 24h; Molecular sieve; | 97% |
| With air; potassium iodide; palladium(II) iodide In ISOPROPYLAMIDE at 100℃; under 15201 Torr; for 15h; Autoclave; | 94% |
| With oxygen; potassium iodide; palladium(II) iodide In N,N-dimethyl acetamide at 100℃; under 15201 Torr; for 15h; | 94% |
| With triethylamine; copper dichloride 1.) THF, 30 kg/cm2, 80 deg C, 4h, 2.) room temp, 18 h, 1 atm; Yield given. Multistep reaction; | |
| With oxygen; sodium carbonate; copper dichloride In acetonitrile at 100℃; under 22502.3 Torr; for 3h; Autoclave; |
-
-
58479-61-1
tert-butylchlorodiphenylsilane

-
-
584-03-2
1,2-dihydroxybutane

| Conditions | Yield |
|---|---|
| With 4-methylpyridine-1-oxide In dichloromethane at 20℃; Molecular sieve; regioselective reaction; | 97% |
-
-
124-38-9
carbon dioxide

-
-
115-19-5
2-methyl-but-3-yn-2-ol

-
-
584-03-2
1,2-dihydroxybutane

-
A
-
4437-85-8, 90970-72-2, 90971-04-3, 127128-77-2
1,2-butylene carbonate

-
B
-
115-22-0
3-Hydroxy-3-methyl-2-butanone

| Conditions | Yield |
|---|---|
| With 1,8-diazabicyclo[5.4.0]undec-7-ene; zinc(II) chloride In acetonitrile at 80℃; under 7500.75 Torr; for 24h; Autoclave; Sealed tube; chemoselective reaction; | A 97% B 65 %Chromat. |
| Stage #1: carbon dioxide; 2-methyl-but-3-yn-2-ol With C15H18N2O2 In acetonitrile at 25℃; under 760.051 Torr; for 24h; Inert atmosphere; Schlenk technique; Stage #2: 1,2-dihydroxybutane With 1-methyl-2,3,4,6,7,8-hexahydro-1H-pyrimido[1,2-a]pyrimidine In acetonitrile at 25℃; for 24h; Inert atmosphere; Schlenk technique; | A 89 %Spectr. B n/a |


| Conditions | Yield |
|---|---|
| Stage #1: 1,4-diaza-bicyclo[2.2.2]octane; zinc(II) nitrate hexahydrate; isophthalic acid In N,N-dimethyl-formamide for 1h; Stage #2: 1,2-dihydroxybutane In N,N-dimethyl-formamide at 130℃; for 48h; | 96% |


| Conditions | Yield |
|---|---|
| With phosphorus pentoxide; silica gel at 20℃; | 95% |
| With dmap; triethylamine In dichloromethane at 20℃; for 16h; Cooling with ice; | 86% |
| With sulfuric acid for 0.25h; Heating; | 73% |
| With pyridine |

| Conditions | Yield |
|---|---|
| With C39H31BMnNO2P2; potassium hydride In toluene at 150℃; for 36h; | 95% |
| With cesiumhydroxide monohydrate In toluene at 150℃; for 24h; Inert atmosphere; Sealed tube; | 94% |
| With 1,10-Phenanthroline; cesiumhydroxide monohydrate; nickel dibromide In toluene at 150℃; for 24h; Inert atmosphere; Sealed tube; | 93% |

| Conditions | Yield |
|---|---|
| toluene-4-sulfonic acid In water; toluene at 135℃; for 4h; Heating / reflux; with Dean-Stark tube; | 94.3% |
-
-
57-13-6
urea

-
-
584-03-2
1,2-dihydroxybutane

-
-
4437-85-8, 90970-72-2, 90971-04-3, 127128-77-2
1,2-butylene carbonate

| Conditions | Yield |
|---|---|
| With 1-hexadecyl-3-methylimidazolium chloride; zinc(II) chloride In neat (no solvent) at 160℃; under 112.511 Torr; for 3h; Green chemistry; | 94.3% |
-
-
584-03-2
1,2-dihydroxybutane

| Conditions | Yield |
|---|---|
| With water-d2; phospho(enol)pyruvic acid mono potassium salt; adenosine 5'-triphosphate disodium salt; barium(II) chloride; magnesium chloride; glycerol kinase from S. cerevisiae or from E. coli and pyruvate kinase In various solvent(s) for 72h; Ambient temperature; | 94% |

| Conditions | Yield |
|---|---|
| With cesiumhydroxide monohydrate In toluene at 150℃; for 24h; Inert atmosphere; Sealed tube; | 94% |
| With 1,10-Phenanthroline; cesiumhydroxide monohydrate; nickel dibromide In toluene at 150℃; for 24h; Inert atmosphere; Sealed tube; | 92% |
| With trimethylamine-N-oxide; tricarbonyl(η4-1,3-bis(trimethylsilyl)-4,5,6,7-tetrahydro-2H-inden-2-one)iron In toluene at 150℃; for 24h; Green chemistry; | 78% |

| Conditions | Yield |
|---|---|
| With Amberlyst15 In cyclohexane Reflux; | 94% |
-
-
22664-18-2, 60322-66-9, 60362-79-0, 60362-80-3, 99881-01-3, 108266-76-8, 108266-77-9
5-exo-methylbicyclo-[2.2.1]heptan-2-exo-ol

-
-
584-03-2
1,2-dihydroxybutane

| Conditions | Yield |
|---|---|
| With naphthalene-1,5-disulfonate In benzene at 80℃; for 3h; | 92.9% |
-
-
75-36-5
acetyl chloride

-
-
584-03-2
1,2-dihydroxybutane

-
A
-
13814-27-2
1,2-butanediol diacetate

-
B
-
24469-20-3
(+/-)-2-hydroxybutyl acetate

| Conditions | Yield |
|---|---|
| With 2,4,6-trimethyl-pyridine In dichloromethane at -78℃; Yields of byproduct given; | A n/a B 92% |
| With 2,4,6-trimethyl-pyridine In dichloromethane at -78℃; Yield given. Yields of byproduct given; |

-
-
584-03-2
1,2-dihydroxybutane

-
-
600-15-7, 565-70-8
2-Hydroxybutanoic acid

| Conditions | Yield |
|---|---|
| With C24H33IrN4O3; water; sodium carbonate for 18h; Reflux; | 92% |
| at 35℃; for 12h; Product distribution; production by resting cells of sp. strain TB-42; optimization of culture medium for preparation of this strain; effect of CaCO3; effect of aeration; pH 7.0; effect of temperature; |
-
-
98-59-9
p-toluenesulfonyl chloride

-
-
584-03-2
1,2-dihydroxybutane

-
-
185215-29-6
1,2-Bis(4-toluenesulfonyloxy)butane

| Conditions | Yield |
|---|---|
| With trimethylamine hydrochloride; triethylamine In acetonitrile at 0 - 5℃; for 1h; | 92% |
| With N,N,N',N'-tetramethylhexamethylenediamine In acetonitrile at 0 - 5℃; for 1h; Tosylation; | 91% |
Related products
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View