-
Name
2-Chloropropionic acid
- EINECS 209-952-3
- CAS No. 598-78-7
- Article Data66
- CAS DataBase
- Density 1.285 g/cm3
- Solubility soluble in water
- Melting Point -12--14 °C
- Formula C3H5ClO2
- Boiling Point 188.983 °C at 760 mmHg
- Molecular Weight 108.525
- Flash Point 68.095 °C
- Transport Information UN 2511 8/PG 3
- Appearance colourless liquid
- Safety 23-26-28A-36/37/39-45
- Risk Codes 21/22-35
-
Molecular Structure
-
Hazard Symbols
 C
C
- Synonyms Propionicacid, 2-chloro- (7CI,8CI);Propionic acid, a-chloro- (4CI);(RS)-2-Chloropropanoic acid;(RS)-2-Chloropropionic acid;(RS)-a-Chloropropionic acid;2-Chloropropanoic acid;2-Chloropropionic acid;DL-2-Chloropropionic acid;NSC173;NSC 401806;a-Chloropropionic acid;
- PSA 37.30000
- LogP 0.69830
Synthetic route
-
-
26910-40-7
1-chloroethyl phenyl sulfoxide

-
-
124-38-9
carbon dioxide

-
-
598-78-7
(R,S)-2-chloropropionic acid

| Conditions | Yield |
|---|---|
| Stage #1: 1-chloroethyl phenyl sulfoxide With cyclohexylmagnesiumchloride In tetrahydrofuran; toluene at -78℃; for 0.166667h; Inert atmosphere; Stage #2: carbon dioxide In tetrahydrofuran; toluene at -78℃; for 3h; | 100% |

| Conditions | Yield |
|---|---|
| With 4H3N*4H(1+)*CuMo6O18(OH)6(4-); water; oxygen; sodium carbonate at 50℃; under 760.051 Torr; for 12h; | 99% |
| With C4H11FeMo6NO24(3-)*3C16H36N(1+); water; oxygen; sodium carbonate at 50℃; under 760.051 Torr; for 8h; Green chemistry; | 97% |
| With sodium chlorite; sodium dihydrogenphosphate In water; tert-butyl alcohol for 1.5h; |
-
-
802294-64-0
propionic acid

-
-
598-78-7
(R,S)-2-chloropropionic acid

| Conditions | Yield |
|---|---|
| With sulfuric acid; chlorine; propionic acid anhydride at 130℃; under 760.051 Torr; for 3h; Time; | 97.22% |
| Stage #1: propionic acid With phosphorus trichloride at 160℃; Stage #2: With trichloroisocyanuric acid at 160℃; | 76% |
| Chlorierung; |

| Conditions | Yield |
|---|---|
| With phenylsilane; C12H23N2O2P In tetrahydrofuran at 23℃; for 2h; | 91% |

| Conditions | Yield |
|---|---|
| With potassium phosphate; carbon dioxide; CrH6Mo6O24(3-)*3H3N*3H(1+) In dimethyl sulfoxide at 80℃; under 750.075 Torr; for 24h; Green chemistry; | 87% |

| Conditions | Yield |
|---|---|
| With pyridinium polyhydrogen fluoride; potassium chloride; sodium nitrite for 72h; Ambient temperature; | 74% |
| With nitric acid folgendes Erwaermen mit konz.HCl; |
-
-
802294-64-0
propionic acid

-
A
-
75-99-0
2,2-Dichloropropionic acid

-
B
-
598-78-7
(R,S)-2-chloropropionic acid

| Conditions | Yield |
|---|---|
| With chlorine; pyrographite; propionic acid anhydride at 130℃; under 760.051 Torr; for 2.5h; | A 10.47% B 68.23% |
-
-
148586-45-2
2-Chloro-2-(toluene-4-sulfinyl)-propionic acid

-
-
598-78-7
(R,S)-2-chloropropionic acid

| Conditions | Yield |
|---|---|
| With ethylmagnesium bromide In tetrahydrofuran at -78℃; for 0.166667h; | 67% |
| With ethylmagnesium bromide; water; ammonium chloride 1.) THF, -78 deg C, 10 min; Yield given. Multistep reaction; |
-
-
802294-64-0
propionic acid

-
A
-
590-92-1
3-Bromopropionic acid

-
B
-
107-94-8
chloropropionic acid

-
C
-
598-72-1
2-Bromopropionic acid

-
D
-
598-78-7
(R,S)-2-chloropropionic acid

| Conditions | Yield |
|---|---|
| With hydrogenchloride; oxygen; potassium bromide; sodium nitrite In chloroform; water at 40℃; under 760.051 Torr; for 18h; Sealed tube; Irradiation; | A 65% B n/a C 23% D n/a |

-
-
1617-17-0
2-chlorpropionitrile

-
A
-
27816-36-0
2-Chloropropionamide

-
B
-
598-78-7
(R,S)-2-chloropropionic acid

| Conditions | Yield |
|---|---|
| at 30℃; for 1.5h; Rhodococcus sp. (SP 361), pH 7; | A 8% B 43% |
-
-
849585-22-4
LACTIC ACID

-
-
598-78-7
(R,S)-2-chloropropionic acid

| Conditions | Yield |
|---|---|
| With phosphorus pentachloride Behandeln des α-Chlorpropionsaeurechlorids mit Wasser; |

| Conditions | Yield |
|---|---|
| With hydrogenchloride at 140℃; |

| Conditions | Yield |
|---|---|
| With phosphorus at 105 - 110℃; | |
| With phosphonic Acid at 100 - 120℃; |

| Conditions | Yield |
|---|---|
| With nitric acid; (R,S)-2-chloropropionic acid unter Durchleiten von Luft bei 90grad; |

| Conditions | Yield |
|---|---|
| With chlorine Irradiation.im UV-Licht; | |
| With hydrogenchloride Electrolysis.an einer Platinanode; | |
| With hydrogenchloride Electrolysis.an Platinanode; |
-
-
802294-64-0
propionic acid

-
A
-
107-94-8
chloropropionic acid

-
B
-
565-64-0
2,3-dichloropropanoic acid

-
C
-
598-78-7
(R,S)-2-chloropropionic acid

| Conditions | Yield |
|---|---|
| beim Chlorieren in der fluessigen Phase unter Belichtung; |

-
-
802294-64-0
propionic acid

-
A
-
5961-88-6
cyclic anhydride of β-sulfopropionic acid

-
B
-
598-78-7
(R,S)-2-chloropropionic acid

| Conditions | Yield |
|---|---|
| With sulfuryl dichloride Irradiation.Gluehlampenlicht; |

| Conditions | Yield |
|---|---|
| With hydrogenchloride at 20 - 40℃; |
-
-
16714-77-5
1,3-dichlorobutan-2-one

-
-
598-78-7
(R,S)-2-chloropropionic acid

| Conditions | Yield |
|---|---|
| With sodium hypobromide |

| Conditions | Yield |
|---|---|
| With phosphorus pentachloride Verseifen des entstandenen α-Chlor-propionitrils durch Erhitzen mit konz.Salzsaeure; |

| Conditions | Yield |
|---|---|
| at 90 - 120℃; beim Chlorieren; |
-
-
802294-64-0
propionic acid

-
-
7719-12-2, 52843-90-0
phosphorus trichloride

-
-
598-78-7
(R,S)-2-chloropropionic acid

| Conditions | Yield |
|---|---|
| at 90 - 120℃; beim Chlorieren; |

| Conditions | Yield |
|---|---|
| at 90 - 120℃; beim Chlorieren; |


| Conditions | Yield |
|---|---|
| at 105 - 110℃; beim Chlorieren; |

| Conditions | Yield |
|---|---|
| at 90 - 120℃; beim Chlorieren; |
-
-
7647-01-0
hydrogenchloride

-
-
802294-64-0
propionic acid

-
A
-
107-94-8
chloropropionic acid

-
B
-
598-78-7
(R,S)-2-chloropropionic acid

| Conditions | Yield |
|---|---|
| Electrolysis; |

-
-
7782-50-5
chlorine

-
-
802294-64-0
propionic acid

-
A
-
107-94-8
chloropropionic acid

-
B
-
598-78-7
(R,S)-2-chloropropionic acid

| Conditions | Yield |
|---|---|
| im UV-Licht(λ:350-400nm); |

-
-
56-23-5
tetrachloromethane

-
-
7782-50-5
chlorine

-
-
802294-64-0
propionic acid

-
A
-
107-94-8
chloropropionic acid

-
B
-
598-78-7
(R,S)-2-chloropropionic acid

| Conditions | Yield |
|---|---|
| im UV-Licht(λ:350-400nm); |

-
-
40843-73-0
4-(2',4'-dichlorophenoxy)-phenol

-
-
598-78-7
(R,S)-2-chloropropionic acid

-
-
40843-25-2
diclofop

| Conditions | Yield |
|---|---|
| With sodium hydroxide; phosphoric acid In 5,5-dimethyl-1,3-cyclohexadiene; water | 99.5% |

-
-
598-78-7
(R,S)-2-chloropropionic acid

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane at 0 - 20℃; for 12h; Reagent/catalyst; Temperature; Inert atmosphere; diastereoselective reaction; | 99% |
-
-
7689-03-4
camptothecin

-
-
598-78-7
(R,S)-2-chloropropionic acid

-
-
1112310-87-8
camptothecin-20-O-2-chloropropionate

| Conditions | Yield |
|---|---|
| With dmap; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In dichloromethane at 20℃; for 2h; | 98.1% |
-
-
598-78-7
(R,S)-2-chloropropionic acid

-
-
142-73-4
iminodiacetic acid

-
-
22149-55-9
α-alanine-N-diacetic acid

| Conditions | Yield |
|---|---|
| at 120℃; Temperature; | 97% |

| Conditions | Yield |
|---|---|
| With thionyl chloride In hexane for 3h; Heating; | 95% |
| With pyridine; phosgene at 55℃; for 1h; Reagent/catalyst; Temperature; | 95.4% |
| With thionyl chloride In N,N-dimethyl-formamide at 85℃; for 2h; | 85% |

| Conditions | Yield |
|---|---|
| With potassium chloride In tetrachloromethane at 72℃; for 4h; Kinetics; Rate constant; Thermodynamic data; activation energy, other temperatures (55, 63, 80 deg C); | 95% |
| With sulfuric acid | 85% |
| With hydrogenchloride |
-
-
95-53-4
o-toluidine

-
-
598-78-7
(R,S)-2-chloropropionic acid

-
-
19281-31-3
N-(2-methylphenyl)-2-chloropropanamide

| Conditions | Yield |
|---|---|
| Stage #1: (R,S)-2-chloropropionic acid With 2-chloro-4,6-dimethoxy-1 ,3,5-triazine In N,N-dimethyl-formamide for 0.25h; Stage #2: o-toluidine With 4-methyl-morpholine In water; N,N-dimethyl-formamide at 0 - 10℃; for 6.25h; | 95% |
-
-
121065-74-5
tert-butyl 2-hydroxymethylacrylate

-
-
598-78-7
(R,S)-2-chloropropionic acid

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane at 0 - 20℃; for 1h; | 93.4% |
| With dmap; dicyclohexyl-carbodiimide |
-
-
26473-47-2
2-methyl-3-sulfanylpropanoic acid

-
-
598-78-7
(R,S)-2-chloropropionic acid

-
-
106014-16-8
2,5-dimethyl-3-thia-adipic acid

| Conditions | Yield |
|---|---|
| Stage #1: 2-methyl-3-sulfanylpropanoic acid With sodium hydroxide In water at 20℃; for 0.166667h; Stage #2: (R,S)-2-chloropropionic acid In water for 2.5h; | 93% |
-
-
51958-58-8
chloromethyl trimethyl benzene

-
-
15336-78-4
N,N'-bis(glycidyl)-ethyleneurea

-
-
598-78-7
(R,S)-2-chloropropionic acid

-
-
422507-82-2
N,N'-bis(2-hydroxy-3-acryloyloxy-propyl)-ethyleneurea

| Conditions | Yield |
|---|---|
| With triethylamine | 92% |

-
-
933717-56-7
N1-(3-trifluoromethoxy-phenyl)-ethane-1,2-diamine

-
-
598-78-7
(R,S)-2-chloropropionic acid

-
-
1206600-53-4
2-chloro-N-[2-(3-trifluoromethoxy-phenylamino)-ethyl]-propionamide

| Conditions | Yield |
|---|---|
| With dicyclohexyl-carbodiimide In dichloromethane at -30℃; | 92% |
-
-
529-34-0
3,4-dihydronaphthalene-1(2H)-one

-
-
598-78-7
(R,S)-2-chloropropionic acid

-
-
250652-65-4
2-[(2-chloropropanoyl)oxy]tetralone
-
-
56-35-9
bis(tri-n-butyltin)oxide

-
-
598-78-7
(R,S)-2-chloropropionic acid

-
-
33550-23-1
Tri-n-Butylzinn-2-chlorpropionat

| Conditions | Yield |
|---|---|
| byproducts: H2O; | 91% |
| byproducts: H2O; | 91% |
-
-
134469-07-1
Benzimidazol-2-thiol

-
-
598-78-7
(R,S)-2-chloropropionic acid

-
-
21547-70-6
2-(2-benzimidazolylthio)propionic acid

| Conditions | Yield |
|---|---|
| With potassium hydroxide In ethanol for 6h; Reflux; | 91% |

-
-
598-78-7
(R,S)-2-chloropropionic acid

| Conditions | Yield |
|---|---|
| With sodium hydroxide; sodium hydrogencarbonate Heating; | 90% |
-
-
37052-78-1
2-Mercapto-5-methoxybenzimidazole

-
-
598-78-7
(R,S)-2-chloropropionic acid

-
-
919467-09-7
2-(5-methoxy-1(3)H-benzimidazol-2-ylmercapto)-propionic acid

| Conditions | Yield |
|---|---|
| With potassium hydroxide In ethanol for 6h; Reflux; | 90% |

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane at 20℃; for 48h; | 90% |
-
-
6325-91-3
5-nitro-2-mercaptobenzimidazole

-
-
598-78-7
(R,S)-2-chloropropionic acid

-
-
114381-56-5
2-(5-nitro-1(3)H-benzimidazol-2-ylmercapto)-propionic acid

| Conditions | Yield |
|---|---|
| With potassium hydroxide In ethanol for 6h; Reflux; | 89% |
-
-
598-78-7
(R,S)-2-chloropropionic acid

-
-
25369-78-2
5-chloro-1H-benzoimidazole-2-thiol

-
-
6963-77-5
2-(5-chloro-1(3)H-benzimidazol-2-ylmercapto)-propionic acid

| Conditions | Yield |
|---|---|
| With potassium hydroxide In ethanol for 6h; Reflux; | 89% |
-
-
1122-41-4
2,4-dichlorobenzenethiol

-
-
598-78-7
(R,S)-2-chloropropionic acid

-
-
119424-79-2
2-(2,4-dichlorophenylsulfanyl)propionic acid

| Conditions | Yield |
|---|---|
| With sodium thiophenolate; sodium hydroxide for 5h; | 88.8% |
-
-
67572-54-7
3-(3,4-diossimetilenefenil)-4-ammino-5-mercapto-4H-1,2,4-triazolo

-
-
598-78-7
(R,S)-2-chloropropionic acid

| Conditions | Yield |
|---|---|
| With sodium hydroxide; sodium hydrogencarbonate Heating; | 88% |
-
-
4528-64-7
4,4-diphenyl-2-cyclohexen-1-one

-
-
598-78-7
(R,S)-2-chloropropionic acid

-
-
295777-97-8
4,4-diphenyl-6-(2-chloropropionyloxy)-cyclohex-2-en-1-one

| Conditions | Yield |
|---|---|
| Stage #1: (R,S)-2-chloropropionic acid With manganese triacetate In benzene for 2.5h; Substitution; Heating; Stage #2: 4,4-diphenyl-2-cyclohexen-1-one In benzene for 20h; Oxidation; Heating; | 88% |
2-Chloropropionic acid Consensus Reports
Reported in EPA TSCA Inventory.
2-Chloropropionic acid Standards and Recommendations
ACGIH TLV: TWA 0.1 ppm (skin) 85INA8 Documentation of the Threshold Limit Values and Biological Exposure Indices 5 (1986),136(89).
DOT Classification: 8; Label: Corrosive
2-Chloropropionic acid Specification
The Propanoic acid,2-chloro-, with the CAS registry number 598-78-7, is also known as alpha-Chloropropionic acid. It belongs to the product categories of Organic acids; C1 to C5; Carbonyl Compounds; Carboxylic Acids. Its EINECS number is 209-952-3. This chemical's molecular formula is C3H5ClO2 and molecular weight is 108.52. What's more, its systematic name is 2-chloropropanoic acid. It is a neurotoxin. It is stable at common pressure and temperature, and it should be sealed and stored in a cool, ventilated and dry place. It is mainly used to manufacture pesticide herbicides.
Physical properties of Propanoic acid,2-chloro- are: (1)ACD/LogP: 0.30; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): -2.22; (4)ACD/LogD (pH 7.4): -3.37; (5)ACD/BCF (pH 5.5): 1; (6)ACD/BCF (pH 7.4): 1; (7)ACD/KOC (pH 5.5): 1; (8)ACD/KOC (pH 7.4): 1; (9)#H bond acceptors: 2; (10)#H bond donors: 1; (11)#Freely Rotating Bonds: 1; (12)Polar Surface Area: 26.3 Å2; (13)Index of Refraction: 1.441; (14)Molar Refractivity: 22.31 cm3; (15)Molar Volume: 84.4 cm3; (16)Polarizability: 8.84×10-24cm3; (17)Surface Tension: 37.6 dyne/cm; (18)Density: 1.285 g/cm3; (19)Flash Point: 68.1 °C; (20)Enthalpy of Vaporization: 46.87 kJ/mol; (21)Boiling Point: 189 °C at 760 mmHg; (22)Vapour Pressure: 0.26 mmHg at 25°C.
Preparation of Propanoic acid,2-chloro-: this chemical can be prepared by alanine at the ambient temperature. This reaction will need reagents pyridinium polyhydrogen fluoride, KCl, NaNO2. The yield is about 74%.

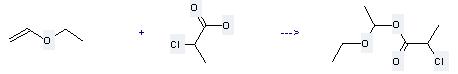
Uses of Propanoic acid,2-chloro-: it can be used to produce 2-chloro-propionic acid 1-ethoxy-ethyl ester at the temperature of 5 - 10 °C. It will need reagent cupric bromide and solvent diethyl ether with the reaction time of 3 hours. The yield is about 75%.
When you are using this chemical, please be cautious about it as the following:
It is harmful in contact with skin and if swallowed. It can cause severe burns. You should not breathe gas/fumes/vapour/spray. After contact with skin, you must wash immediately with plenty of water. In case of contact with eyes, you should rinse immediately with plenty of water and seek medical advice. When using it, you need wear suitable protective clothing, gloves and eye/face protection. In case of accident or if you feel unwell, you must seek medical advice immediately (show the label where possible).
You can still convert the following datas into molecular structure:
(1)SMILES: ClC(C(=O)O)C
(2)Std. InChI: InChI=1S/C3H5ClO2/c1-2(4)3(5)6/h2H,1H3,(H,5,6)
(3)Std. InChIKey: GAWAYYRQGQZKCR-UHFFFAOYSA-N
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| guinea pig | LD50 | skin | 126mg/kg (126mg/kg) | "Documentation of the Threshold Limit Values and Biological Exposure Indices," 5th ed., Cincinnati, OH, American Conference of Governmental Industrial Hygienists, Inc., 1986Vol. 5, Pg. 136(89), 1986. | |
| mouse | LD50 | oral | 400mg/kg (400mg/kg) | BEHAVIORAL: CONVULSIONS OR EFFECT ON SEIZURE THRESHOLD KIDNEY, URETER, AND BLADDER: HEMATURIA SKIN AND APPENDAGES (SKIN): HAIR: OTHER | National Technical Information Service. Vol. OTS0555355. |
| rat | LD50 | oral | 800mg/kg (800mg/kg) | BEHAVIORAL: CONVULSIONS OR EFFECT ON SEIZURE THRESHOLD KIDNEY, URETER, AND BLADDER: HEMATURIA SKIN AND APPENDAGES (SKIN): HAIR: OTHER | National Technical Information Service. Vol. OTS0555355. |
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View