-
Name
2-Methoxy-5-methylaniline
- EINECS 204-419-1
- CAS No. 120-71-8
- Article Data25
- CAS DataBase
- Density 1.039 g/cm3
- Solubility Slightly soluble in hot water
- Melting Point 50-52 °C(lit.)
- Formula C8H11NO
- Boiling Point 235.398 °C at 760 mmHg
- Molecular Weight 137.181
- Flash Point 102.068 °C
- Transport Information
- Appearance brown to black crystalline chunks
- Safety 53-45
- Risk Codes 45-22
-
Molecular Structure
-
Hazard Symbols
 T,
T,  Xi
Xi
- Synonyms o-Anisidine,5-methyl- (7CI,8CI);1-Amino-2-methoxy-5-methylbenzene;2-Methoxy-5-methylbenzenamine;3-Amino-4-methoxytoluene;4-Methyl-2-aminoanisole;5-Methyl-2-methoxyaniline;5-Methyl-o-anisidine;Cresidine;Krezidin;NSC 406904;NSC 6162;m-Amino-p-cresol methyl ether;p-Cresidine;
- PSA 35.25000
- LogP 2.16700
Synthetic route
-
-
77721-47-2
2-Methoxi-5-methylphenylazid

-
-
120-71-8
Cresidine

| Conditions | Yield |
|---|---|
| With lithium aluminium tetrahydride | 100% |
| With lithium aluminium tetrahydride In diethyl ether |

| Conditions | Yield |
|---|---|
| With palladium on carbon; hydrogen In ethanol at 20℃; under 28443.9 Torr; for 22h; | 99% |
| With ammonium sulfide; ethanol | |
| With hydrogenchloride; tin at 50℃; | |
| With iron; acetic acid |
-
-
29530-48-1
2-Nitro-2'-methoxy-5'-methyl-azobenzol

-
A
-
95-54-5
1,2-diamino-benzene

-
B
-
120-71-8
Cresidine

| Conditions | Yield |
|---|---|
| With lithium perchlorate In methanol; dichloromethane at 20℃; Cyclization; Electrolysis; | A n/a B n/a C 83% |


| Conditions | Yield |
|---|---|
| With O-(4-nitrobenzoyl)hydroxylammonium trifluoromethanesulfonate; iron(II) bromide; silver(I) triflimide In 2,2,2-trifluoroethanol; water at 30℃; for 2h; | 56% |
| With hydroxylamine triflate; iron(II) sulfate In water; acetonitrile at 20℃; for 16h; | 53% |
| Stage #1: p-methylanizole With pyridine; tetra-n-butylammonium tetrakis(pentafluorophenyl)borate; trifluorormethanesulfonic acid In dichloromethane at 25℃; Inert atmosphere; Electrochemical reaction; Stage #2: With piperidine In acetonitrile at 80℃; for 12h; Inert atmosphere; | 13% |
| Multi-step reaction with 2 steps 1: 1.)phenyliodine(III) bis(trifluoroacetate) (PIFA), 2.) Me3SiN3 / 1.) (CF3)2CHOH, r. t. 2: 100 percent / LAH View Scheme |
-
-
29530-48-1
2-Nitro-2'-methoxy-5'-methyl-azobenzol

-
A
-
95-54-5
1,2-diamino-benzene

-
B
-
58380-86-2
2-(2'-methoxy-5'-methylphenyl)-2H-benzotriazole

-
C
-
120-71-8
Cresidine

| Conditions | Yield |
|---|---|
| With sodium hydroxide; lithium perchlorate In tetrahydrofuran; water at 20℃; Cyclization; Electrolysis; | A n/a B 50% C n/a |

-
-
119-10-8
4-methoxy-3-nitrotoluene

-
A
-
120-71-8
Cresidine

-
B
-
6376-14-3
2-methoxy-5-methyl-4-chloroaniline

| Conditions | Yield |
|---|---|
| With hydrogenchloride; tin |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: K2CO3; xylene 2: tin; hydrochloric acid / 50 °C View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: HNO3+H2SO4 2: potassium hydroxide / 110 °C 3: ammonium sulfide; alcohol View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: potassium hydroxide / 110 °C 2: ammonium sulfide; alcohol View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: iodine; iron; chlorine 2: HNO3+H2SO4 3: potassium hydroxide / 110 °C 4: ammonium sulfide; alcohol View Scheme |

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 0 - 20℃; | 100% |
-
-
856678-58-5
7-iodo-benzofuran-5-sulfonyl chloride

-
-
120-71-8
Cresidine

-
-
856678-59-6
7-iodo-N-(2-methoxy-5-methylphenyl)-1-benzofuran-5-sulfonamide

| Conditions | Yield |
|---|---|
| With pyridine In dichloromethane at 20℃; | 99% |
-
-
120-71-8
Cresidine

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 20℃; for 0.166667h; | 99% |

| Conditions | Yield |
|---|---|
| Stage #1: C18H17N3O10S3 With hydrogenchloride; sodium nitrite In water at 0 - 5℃; for 1.5h; Stage #2: Cresidine With sodium hydroxide In water; acetone at 0 - 25℃; pH=6; | 96% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; pyridine for 3h; Heating; | 89% |
| In tetrahydrofuran | 60% |
-
-
32315-10-9
bis(trichloromethyl) carbonate

-
-
40216-82-8, 60080-69-5
methyl (S)-2,5-diaminopentanoate dihydrochloride

-
-
120-71-8
Cresidine

| Conditions | Yield |
|---|---|
| Stage #1: bis(trichloromethyl) carbonate; Cresidine With triethylamine In tetrahydrofuran at 0 - 20℃; for 2.5h; Stage #2: methyl (S)-2,5-diaminopentanoate dihydrochloride With triethylamine In tetrahydrofuran at 65℃; for 12h; | 89% |

-
-
271261-75-7
2,4-dioctyloxy-5-nitrobenzoyl chloride

-
-
120-71-8
Cresidine

-
-
271261-76-8
3-(2,4-dioctyloxy-5-nitrobenzoylamino)-4-methoxytoluene

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 0 - 20℃; Substitution; | 88.7% |
-
-
41459-62-5
diisopropyl (chloroethynyl)phosphonate

-
-
120-71-8
Cresidine

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetonitrile for 14h; Reflux; | 88% |

| Conditions | Yield |
|---|---|
| With formic acid In methanol for 10h; Inert atmosphere; Reflux; | 88% |
-
-
94205-62-6
2-(4'-aminophenyl)-4-quinolinecarboxylic acid

-
-
120-71-8
Cresidine

| Conditions | Yield |
|---|---|
| Stage #1: 2-(4'-aminophenyl)-4-quinolinecarboxylic acid With hydrogenchloride; sodium nitrite at 2℃; for 0.5h; Stage #2: Cresidine With hydrogenchloride; sodium acetate at 10 - 20℃; for 16h; pH=4.5; | 87% |
-
-
120-71-8
Cresidine

-
-
475474-43-2
2-(4'-aminophenyl)-6-bromo-4-quinolinecarboxylic acid

| Conditions | Yield |
|---|---|
| Stage #1: 2-(4'-aminophenyl)-6-bromo-4-quinolinecarboxylic acid With hydrogenchloride; sodium nitrite at 2℃; for 0.5h; Stage #2: Cresidine With hydrogenchloride; sodium acetate at 10 - 20℃; pH=4.5; | 87% |
-
-
120-71-8
Cresidine

-
-
873980-68-8
4-bromo-2-methoxy-5-methylaniline

| Conditions | Yield |
|---|---|
| With N-Bromosuccinimide In N,N-dimethyl-formamide at 0 - 20℃; for 18h; | 86% |
| With bromine In dichloromethane at 0 - 23℃; for 16h; | 51% |
| With bromine In dichloromethane at 0 - 20℃; | 50% |
| With N-Bromosuccinimide In N,N-dimethyl-formamide at 20℃; for 16h; |
-
-
108-94-1
cyclohexanone

-
-
134390-45-7
3-phenyl-2,2-dihydroxy-3-oxopropionic acid ethyl ester

-
-
120-71-8
Cresidine

| Conditions | Yield |
|---|---|
| With benzoic acid In toluene at 65℃; for 36h; | 85% |


| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 20℃; for 8h; Cooling with ice; | 85% |

| Conditions | Yield |
|---|---|
| In 2-methoxy-ethanol for 6h; Heating; | 85% |
-
-
120-71-8
Cresidine

-
-
50597-88-1
2-iodo-4-methylanisole

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid; sodium iodide In water; acetonitrile at 10 - 25℃; for 2h; | 82% |
| With toluene-4-sulfonic acid; sodium iodide In water; acetonitrile at 10 - 20℃; for 2h; Inert atmosphere; | 82% |
| With sulfuric acid Diazotization.Behandlung der Diazoniumsalz-Loesung mit KI, zuletzt auf dem Dampfbad; | |
| With hydrogenchloride; potassium iodide; sodium nitrite Multistep reaction; |
-
-
438249-85-5
4-chloro-2-(2-pyrazinyl)-6-(trifluoromethyl)pyrimidine

-
-
120-71-8
Cresidine

-
-
438249-23-1
4-(2-Methoxy-5-methylanilino)-2-(2-pyrazinyl)-6-trifluoromethylpyrimidine

| Conditions | Yield |
|---|---|
| 81% |
-
-
874210-52-3
4-dodecyloxy-2-methoxy-5-nitro-benzoyl chloride

-
-
120-71-8
Cresidine

-
-
874210-63-6
4-dodecyloxy-2-methoxy-N-(2-methoxy-5-methyl-phenyl)-5-nitro-benzamide

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 20℃; for 18h; | 77.1% |
2-Methoxy-5-methylaniline Chemical Properties
5-METHYL-o-ANISIDINE(120-71-8) is also named 2-Amino-4-methylphenolmethylether;2-Amino-p-cresol methyl ether;2-Amino-p-cresolmethylether;2-methoxy-5-methyl-benzenamin;2-Methoxy-5-methylbenzenamine;2-methoxy-5-methyl-Benzenamine;3-amino-para-cresol,methylether;4-Methyl-2-aminoanisole.
Melting Point: 50-52 oC(lit.)
Boiling Point: 235 oC(lit.)
Vapor Density: 4.7 (vs air)
Flash Point >230 oF
Water Solubility: Slightly soluble in hot water.
Index of Refraction: 1.549
Molar Refractivity: 41.99 cm3
Molar Volume: 131.9 cm3
Surface Tension: 37.8 dyne/cm
Enthalpy of Vaporization: 47.22 kJ/mol
Vapour Pressure: 0.0502 mmHg at 25 oC
The chemical structure of 5-METHYL-o-ANISIDINE(120-71-8) is
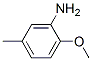 .
.2-Methoxy-5-methylaniline Uses
2-Methoxy-5-methylaniline Toxicity Data With Reference
| 1. | skn-rbt 500 mg/24H MLD | 85JCAE Prehled Prumyslove Toxikologie; Organicke Latky Marhold, J.,Prague, Czechoslovakia.: Avicenum,1986,721. | ||
| 2. | eye-rbt 100 mg/24H MOD | 85JCAE Prehled Prumyslove Toxikologie; Organicke Latky Marhold, J.,Prague, Czechoslovakia.: Avicenum,1986,721. | ||
| 3. | mmo-sat 62,500 ng/plate | ENMUDM Environmental Mutagenesis. 7 (Suppl 5)(1985),1. | ||
| 4. | mma-sat 3330 ng/plate | ENMUDM Environmental Mutagenesis. 7 (Suppl 5)(1985),1. | ||
| 5. | mma-esc 2 mg/plate | ENMUDM Environmental Mutagenesis. 7 (Suppl 5)(1985),1. | ||
| 6. | otr-rat:emb 31 µg/plate | JJATDK JAT, Journal of Applied Toxicology. 1 (1981),190. | ||
| 7. | orl-rat LD50:1450 mg/kg | HURC** Huntingdon Research Center Reports .(Brooklandville, MD.: )1972. |
2-Methoxy-5-methylaniline Consensus Reports
2-Methoxy-5-methylaniline Safety Profile
Hazard Codes:
 T,
T, Xi
Xi Risk Statements: 45-22
45: May cause cancer
22: Harmful if swallowed
Safety Statements: 53-45
53: Avoid exposure - obtain special instruction before use
45: In case of accident or if you feel unwell, seek medical advice immediately (show label where possible)
WGK Germany: 3
RTECS: BZ6720000
F: 9
F9: Keep under nitrogen.
Hazardous Substances Data: 120-71-8(Hazardous Substances Data).
2-Methoxy-5-methylaniline Standards and Recommendations
Related Products
- 2-Methoxy-5-methylaniline
- 1207181-35-8
- 1207181-57-4
- 1207181-58-5
- 1207181-59-6
- 1207181-61-0
- 1207181-62-1
- 1207181-63-2
- 12071-83-9
- 120722-04-5
- 1207253-06-2
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View