-
Name
2-Methylindene
- EINECS
- CAS No. 2177-47-1
- Article Data54
- CAS DataBase
- Density 1.003 g/cm3
- Solubility
- Melting Point 80°C
- Formula C10H10
- Boiling Point 205.567 °C at 760 mmHg
- Molecular Weight 130.189
- Flash Point 70.76 °C
- Transport Information
- Appearance transparent to yellow liquid
- Safety
- Risk Codes
-
Molecular Structure
- Hazard Symbols
- Synonyms 2-Methylindene;2-Methyl-1H-indene;Indene,2-methyl- (7CI,8CI);
- PSA 0.00000
- LogP 2.64600
Synthetic route
-
-
17496-14-9
2,3-dihydro-2-methyl-1H-inden-1-one

-
-
2177-47-1
2-Methylindene

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid; 2,2-Dimethyl-1,3-propanediol In toluene for 24h; Reflux; | 92% |
| With oxalic acid | |
| Multi-step reaction with 2 steps 1: NaBH4 / tetrahydrofuran; methanol / 4 h / 25 °C 2: p-toluenesulfonic acid*H2O / benzene / 1 h / Heating View Scheme |
-
-
68846-65-1
2-methylene-2,3-dihydro-1H-indene

-
-
2177-47-1
2-Methylindene

| Conditions | Yield |
|---|---|
| With hydrogen bromide In acetonitrile for 1h; Heating; | 90% |
-
-
10485-09-3
2-bromoindene

-
A
-
2177-47-1
2-Methylindene

-
B
-
70063-93-3
1,2-dimethylindene

-
C
-
95-13-6
1-indene

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine)palladium dichloride In benzene at 80℃; for 2h; Yields of byproduct given; | A 88% B n/a C n/a |

-
-
105991-56-8
2-Trimethylsilanylmethyl-indan-2-ol

-
-
2177-47-1
2-Methylindene

| Conditions | Yield |
|---|---|
| With potassium hydride In tetrahydrofuran Ambient temperature; | 86% |
-
-
507261-02-1
1,1-dibromo-3-(2-bromophenyl)-2-methylpropene

-
-
2177-47-1
2-Methylindene

| Conditions | Yield |
|---|---|
| With n-butyllithium In tetrahydrofuran; hexane at -90℃; for 0.5h; | 85% |
| Stage #1: 1,1-dibromo-3-(2-bromophenyl)-2-methylpropene With n-butyllithium In tetrahydrofuran; hexane at -90℃; for 0.5h; Stage #2: With methanol; ammonium chloride In tetrahydrofuran; hexane | 85% |
-
-
89121-39-1
1-allyl-2-(chloromethyl)benzene

-
A
-
2177-47-1
2-Methylindene

-
B
-
68846-65-1
2-methylene-2,3-dihydro-1H-indene

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); triethylamine In acetonitrile for 1h; Heating; | A 1% B 82% |
| With tetrakis(triphenylphosphine) palladium(0); triethylamine In acetonitrile for 1h; Product distribution; Heating; other o-alkenyl- and o-alkynylbenzyl halides, other time, other reagents; | A 1% B 82% |
-
-
120906-16-3
1-allyl-2-(bromomethyl)benzene

-
A
-
2177-47-1
2-Methylindene

-
B
-
68846-65-1
2-methylene-2,3-dihydro-1H-indene

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); triethylamine In acetonitrile for 0.5h; Heating; | A 1% B 82% |
-
-
33223-84-6
2-hydroxy-2-methylindan

-
-
2177-47-1
2-Methylindene

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In benzene for 4h; Dehydration; Heating; | 69% |
| With phosphoric acid at 150℃; | 37% |
| With phosphorus pentoxide; benzene |
-
-
120906-17-4
1-allyl-2-(iodomethyl)benzene

-
A
-
2177-47-1
2-Methylindene

-
B
-
68846-65-1
2-methylene-2,3-dihydro-1H-indene

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); triethylamine In acetonitrile for 0.5h; Heating; | A 18% B 64% |

-
A
-
2177-47-1
2-Methylindene

-
B
-
68846-65-1
2-methylene-2,3-dihydro-1H-indene

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); triethylamine In acetonitrile for 1h; Heating; | A 1% B 60% |

| Conditions | Yield |
|---|---|
| With bis(triphenylphosphine)nickel(II) diiodide; tricyclohexylphosphine In tetrahydrofuran at 70℃; Kumada Cross-Coupling; Inert atmosphere; chemoselective reaction; | 57% |

-
-
2177-47-1
2-Methylindene

| Conditions | Yield |
|---|---|
| With [1,3-bis(2,6-diisopropylphenyl)imidazol-2-ylidene]gold bis(trifluoromethanesulfonyl)imidate; water In tetrahydrofuran at 20℃; for 2h; Inert atmosphere; Schlenk technique; | 53% |
-
-
17496-14-9
2,3-dihydro-2-methyl-1H-inden-1-one

-
-
33223-84-6
2-hydroxy-2-methylindan

-
-
7732-18-5
water

-
-
2177-47-1
2-Methylindene

| Conditions | Yield |
|---|---|
| With MeMgBr; toluene-4-sulfonic acid; hydroquinone In toluene | 48% |

-
-
849799-68-4
1-ethynyl-2-(prop-1-yn-1-yl)benzene

-
-
2177-47-1
2-Methylindene

| Conditions | Yield |
|---|---|
| With [1,3-bis(2,6-diisopropylphenyl)imidazol-2-ylidene]gold bis(trifluoromethanesulfonyl)imidate; water In tetrahydrofuran at 20℃; for 2h; Inert atmosphere; Schlenk technique; | 42% |

| Conditions | Yield |
|---|---|
| With magnesium In diethyl ether; dichloromethane; water; toluene | 30% |


-
A
-
2177-47-1
2-Methylindene

-
B
-
68846-65-1
2-methylene-2,3-dihydro-1H-indene

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); triethylamine In acetonitrile for 120h; Heating; | A 23% B 26% |
-
-
693-04-9
butyl magnesium bromide

-
A
-
2177-47-1
2-Methylindene

-
B
-
229031-59-8
3-butyl-2-methyl-1H-indene

| Conditions | Yield |
|---|---|
| 1,3-bis[(diphenylphosphino)propane]dichloronickel(II) In tetrahydrofuran; toluene at 55℃; for 2h; | A n/a B 24% |


-
A
-
2177-47-1
2-Methylindene

-
B
-
229031-59-8
3-butyl-2-methyl-1H-indene

| Conditions | Yield |
|---|---|
| With butyl magnesium bromide; 1,3-bis[(diphenylphosphino)propane]dichloronickel(II) In tetrahydrofuran; toluene at 55℃; for 2h; | A n/a B 24% |

| Conditions | Yield |
|---|---|
| at 140℃; | |
| at 140℃; |
-
-
54553-50-3, 54553-51-4, 58540-44-6, 88229-97-4, 88229-98-5
(+/-)-cis-1-Acetoxy-2-methyl-indan

-
-
2177-47-1
2-Methylindene

| Conditions | Yield |
|---|---|
| at 350 - 650℃; Pyrolysis; |
-
-
54553-50-3, 54553-51-4, 58540-44-6, 88229-97-4, 88229-98-5
(+/-)-trans-1-Acetoxy-2-methyl-indan

-
-
2177-47-1
2-Methylindene

| Conditions | Yield |
|---|---|
| at 350 - 600℃; Pyrolysis; |

| Conditions | Yield |
|---|---|
| With sodium acetate; acetic acid (electrolysis); |

-
-
2177-47-1
2-Methylindene

| Conditions | Yield |
|---|---|
| at 200℃; |
-
-
119-64-2
tetralin

-
-
124-18-5
decane

-
A
-
292638-84-7
styrene

-
B
-
91-20-3
naphthalene

-
C
-
447-53-0
1,2-Dihydronaphthalene

-
D
-
2177-47-1
2-Methylindene

-
E
-
767-60-2
3-Methylindene

-
F
-
95-13-6
1-indene

| Conditions | Yield |
|---|---|
| at 730℃; Product distribution; Mechanism; other temperature; | A 4.4 % Chromat. B 14 % Chromat. C 35.7 % Chromat. D 1.2 % Chromat. E 4.3 % Chromat. F 7.1 % Chromat. |

-
-
496-11-7
INDANE

-
A
-
91-20-3
naphthalene

-
B
-
447-53-0
1,2-Dihydronaphthalene

-
C
-
2177-47-1
2-Methylindene

-
D
-
767-60-2
3-Methylindene

-
E
-
108-88-3
toluene

-
F
-
95-13-6
1-indene

| Conditions | Yield |
|---|---|
| at 800℃; Product distribution; Mechanism; | A 2.5 % Chromat. B 1.1 % Chromat. C 2.6 % Chromat. D 5.7 % Chromat. E 4.7 % Chromat. F 77.2 % Chromat. |

-
-
29817-09-2, 22664-13-7
1-methylene-2-phenylcyclopropane

-
A
-
7555-67-1
1-phenylmethylenecyclopropane

-
B
-
2177-47-1
2-Methylindene

-
C
-
68846-65-1
2-methylene-2,3-dihydro-1H-indene

| Conditions | Yield |
|---|---|
| In gas at 150.7 - 280.8℃; Kinetics; Thermodynamic data; also further temperatures in the presence of O2; Ea, ΔH(excit.), ΔS(excit.); |


| Conditions | Yield |
|---|---|
| In cyclohexane at 25℃; Quantum yield; Irradiation; | |
| In hexane Quantum yield; Ambient temperature; Irradiation; |

| Conditions | Yield |
|---|---|
| at 42℃; for 0.0166667h; Irradiation; | |
| at 42℃; Product distribution; Irradiation; effect of excitation wavelength; various reaction times; |

| Conditions | Yield |
|---|---|
| at 42℃; for 0.0166667h; Product distribution; Irradiation; initial sample temperatures from -15 deg C to 25 deg C; also with varying pressures of n-butane; |


| Conditions | Yield |
|---|---|
| at 42℃; for 0.0166667h; Irradiation; | |
| at 42℃; Product distribution; Irradiation; effect of excitation wavelength; various reaction times; |
-
-
2177-47-1
2-Methylindene

-
-
56423-64-4
(2-methylindenyl)lithium

| Conditions | Yield |
|---|---|
| With n-butyllithium In hexane at 20℃; for 5h; | 98.4% |
| With n-butyllithium In hexane | 85% |

| Conditions | Yield |
|---|---|
| Stage #1: 2-Methylindene With n-butyllithium In tetrahydrofuran; hexane at 20℃; for 3h; Inert atmosphere; Cooling with ice; Stage #2: 1-iodo-butane In tetrahydrofuran; hexane for 2h; Cooling with ice; | 97% |
-
-
2177-47-1
2-Methylindene

-
-
81060-09-5
1-tert-butoxy-6-iodohexane

| Conditions | Yield |
|---|---|
| With tetra-(n-butyl)ammonium iodide; potassium hydroxide In toluene at 25℃; for 24h; | 97% |

| Conditions | Yield |
|---|---|
| With hydrogen; platinum(IV) oxide In tetrahydrofuran under 1034.3 - 1551.4 Torr; | 95% |
| With C7H14N3(1+)*Cl(1-); sodium triethylborohydride; cobalt(II) chloride In tetrahydrofuran at 60℃; under 37503.8 Torr; for 16h; Autoclave; | 95% |
| With hydrogen; palladium on activated charcoal In hexane for 5h; Catalytic hydrogenation; | 91% |
-
-
2177-47-1
2-Methylindene

-
-
110452-27-2
2,2'-dimethyl-1,1'-biindenyl

| Conditions | Yield |
|---|---|
| With n-butyllithium; copper dichloride 1.) ether, -30 deg C, 30 min, 2.) ether, -30 deg C, 30 min; | 95% |

| Conditions | Yield |
|---|---|
| Stage #1: diiodomethane With diethylzinc; trifluoroacetic acid In hexane; dichloromethane for 0.333333h; cooling; Stage #2: 2-Methylindene In hexane; dichloromethane at 20℃; for 3h; | 94% |
-
-
2177-47-1
2-Methylindene

-
-
1079-66-9
chloro-diphenylphosphine

-
-
718638-63-2
3-(diphenylphosphino)-2-methylindene

| Conditions | Yield |
|---|---|
| Stage #1: 2-Methylindene With n-butyllithium In diethyl ether at -78 - 20℃; Stage #2: chloro-diphenylphosphine In diethyl ether at -78 - 20℃; | 94% |
-
-
2177-47-1
2-Methylindene

-
-
1126942-56-0
7-(bromomethyl)-2-methyl-1H-indene

-
-
1126942-68-4
2-methyl-1-[(2-methyl-1H-inden-7-yl)methyl]-1H-indene

| Conditions | Yield |
|---|---|
| Stage #1: 2-Methylindene With n-butyllithium In diethyl ether; hexane at 0 - 20℃; for 2h; Stage #2: 7-(bromomethyl)-2-methyl-1H-indene In tetrahydrofuran; diethyl ether; hexane at -80 - 20℃; for 12h; | 92% |

| Conditions | Yield |
|---|---|
| Stage #1: bis(pinacol)diborane With potassium ethoxide In N,N-dimethyl acetamide at 20℃; for 1h; Inert atmosphere; Sealed tube; Stage #2: bromobenzene; 2-Methylindene With (1,2-dimethoxyethane)dichloronickel(II) In N,N-dimethyl acetamide at 0 - 20℃; for 24h; Inert atmosphere; Sealed tube; | 91% |

-
-
2177-47-1
2-Methylindene

-
-
1126942-58-2
chloro(dimethyl)(2-methyl-1H-inden-7-yl)silane

-
-
1126942-86-6
dimethyl(2-methyl-1H-inden-1-yl)(2-methyl-1H-inden-7-yl)silane

| Conditions | Yield |
|---|---|
| Stage #1: 2-Methylindene With n-butyllithium In diethyl ether; hexane at 0 - 20℃; for 12h; Stage #2: With copper(l) cyanide In diethyl ether; hexane at -50 - -30℃; for 1h; Stage #3: chloro(dimethyl)(2-methyl-1H-inden-7-yl)silane In diethyl ether; hexane at -80 - 20℃; for 12h; | 88% |
-
-
616-47-7
1-methyl-1H-imidazole

-
-
2177-47-1
2-Methylindene

-
-
75-78-5
dimethylsilicon dichloride

-
-
143232-13-7
bis(2-methyl-1-indenyl)-dimethylsilane

| Conditions | Yield |
|---|---|
| With n-butyllithium In tetrahydrofuran; toluene | 87% |

-
-
2177-47-1
2-Methylindene

| Conditions | Yield |
|---|---|
| With tris(pentafluorophenyl)borate at 20℃; for 3h; Glovebox; | 87% |

| Conditions | Yield |
|---|---|
| Stage #1: 2-bromoindene With iodine; magnesium In tetrahydrofuran at 80℃; for 3h; Inert atmosphere; Stage #2: dimethylsilicon dichloride In tetrahydrofuran; hexane at -78℃; for 18h; Stage #3: 2-Methylindene With 1,3-dimethyl-2-imidazolidinone; n-butyllithium In tetrahydrofuran; hexane at -78 - 20℃; for 5h; Inert atmosphere; | 87% |


| Conditions | Yield |
|---|---|
| Stage #1: 2-Methylindene With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 2h; Metallation; Stage #2: dimethylmonochlorosilane In tetrahydrofuran; hexane at 20℃; for 12h; silylation; Further stages.; | 86% |

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide; Ru(2,2':6',2''-terpyridine)(2,6-pyridinedicarboxylate) In tert-Amyl alcohol at 20℃; for 12h; Conversion of starting material; | 86% |
| With dihydrogen peroxide; ruthenium(2,2',6':2''-terpyridine)(2,6-pyridinedicarboxylate) In tert-Amyl alcohol at 20℃; for 12h; | 86 % Chromat. |
-
-
2177-47-1
2-Methylindene

| Conditions | Yield |
|---|---|
| Stage #1: 2-Methylindene With n-butyllithium In diethyl ether; hexane at 20℃; Inert atmosphere; Stage #2: chloro(1,3-dimethyl-1H-inden-2-yl)dimethylsilane In diethyl ether; hexane at -80 - 20℃; Inert atmosphere; | 86% |

| Conditions | Yield |
|---|---|
| Stage #1: 2-Methylindene With n-butyllithium In diethyl ether; hexane at 0℃; Stage #2: ethyl bromide In diethyl ether at 20℃; | 85% |
-
-
2177-47-1
2-Methylindene

-
-
75-78-5
dimethylsilicon dichloride

-
-
143232-13-7
bis(2-methyl-1-indenyl)-dimethylsilane

| Conditions | Yield |
|---|---|
| Stage #1: 2-Methylindene With n-butyllithium In tetrahydrofuran; toluene at 20 - 80℃; for 1h; Stage #2: dimethylsilicon dichloride In tetrahydrofuran; toluene at 20 - 60℃; | 84% |
| Stage #1: 2-Methylindene With n-butyllithium In tetrahydrofuran; toluene at 80℃; for 1h; Stage #2: dimethylsilicon dichloride In tetrahydrofuran; toluene at 40 - 60℃; | 84% |
| With n-butyllithium 1) n-hexane, THF, reflux, 1 h , 2) r. t., 15 h; Multistep reaction; | |
| With n-butyllithium In tetrahydrofuran; hexane; water | |
| With n-butyllithium In tetrahydrofuran; water; toluene | 8.16 g (84%) |
-
-
2177-47-1
2-Methylindene

-
-
312753-94-9
2-methyl-2,3-dihydro-1H-inden-2-amine

| Conditions | Yield |
|---|---|
| With benzene-1,3-dicarbonitrile; 1,2,4-triphenylbenzene; ammonia In water; acetonitrile at 20℃; Irradiation; | 83% |
| Stage #1: 2-Methylindene With bis{rhodium[3,3'-(1,3-phenylene)bis(2,2-dimethylpropanoic acid)]}; O-(2,4-dinitrophenyl)hydroxylamine In 2,2,2-trifluoroethanol at 0 - 5℃; for 5h; Stage #2: With palladium on activated charcoal; hydrogen In 2,2,2-trifluoroethanol at 25℃; for 15h; | 60% |
| Multi-step reaction with 2 steps 1: bis{rhodium[3,3'-(1,3-phenylene)bis(2,2-dimethylpropanoic acid)]}; O-(2,4-dinitrophenyl)hydroxylamine / 2,2,2-trifluoroethanol / 2.5 h / 0 - 5 °C / Inert atmosphere 2: palladium 10% on activated carbon; hydrogen / methanol / 15 h / 760.05 Torr View Scheme |

| Conditions | Yield |
|---|---|
| Stage #1: 2-Methylindene With sodium hydride In tetrahydrofuran at 0 - 20℃; for 0.5h; Stage #2: Diethyl carbonate In tetrahydrofuran for 7h; Reflux; | 83% |

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In chloroform-d1 at 20℃; for 5h; Friedel-Crafts Alkylation; Inert atmosphere; stereoselective reaction; | 83% |
-
-
686-69-1
diethylchlorophosphine

-
-
2177-47-1
2-Methylindene

-
-
1461-22-9
tributyltin chloride

-
-
247027-70-9
1-diethylphosphanyl-1-tributylstannyl-2-methylindene

| Conditions | Yield |
|---|---|
| With n-BuLi In diethyl ether; hexane byproducts: LiCl; (anaerobic conds.); addn. of n-BuLi in hexane at 0°C to 2-methylindene in Et2O, stirring (room temp., 1 h), addn. of ClPEt2 at 0°C, stirring (r.t., ca. 1 h), addn. of n-BuLi at 0°C, stirring (1 h), addn. of Bu3SnCl, stirring (1.5 h); removal of volatiles (vac.), addn. of hexane, filtration, removal of solvent; | 80% |

-
-
38274-14-5
2,2'-bis-(bromomethyl)-1,1'-biphenyl

-
-
2177-47-1
2-Methylindene

| Conditions | Yield |
|---|---|
| Stage #1: 2-Methylindene With n-butyllithium In tetrahydrofuran at 0 - 20℃; for 3h; Inert atmosphere; Stage #2: 2,2'-bis-(bromomethyl)-1,1'-biphenyl In tetrahydrofuran at -40 - 20℃; Inert atmosphere; Schlenk technique; | 75% |
2-Methylindene Chemical Properties
Molecular Structure of 2-Methylindene (CAS No.2177-47-1):
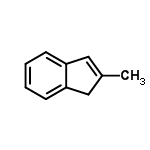
Molecular Formula: C10H10
Molecular Weight: 130.1864
CAS No: 2177-47-1
Index of Refraction: 1.572
Molar Refractivity: 42.73 cm3
Molar Volume: 129.8 cm3
Surface Tension: 35.3 dyne/cm
Density: 1.002 g/cm3
Flash Point: 70.8 °C
Enthalpy of Vaporization: 42.38 kJ/mol
Boiling Point: 205.6 °C at 760 mmHg
Vapour Pressure: 0.356 mmHg at 25°C
InChI: InChI=1/C10H10/c1-8-6-9-4-2-3-5-10(9)7-8/h2-6H,7H2,1H3
InChIKey: YSAXEHWHSLANOM-UHFFFAOYAW
Std. InChI: InChI=1S/C10H10/c1-8-6-9-4-2-3-5-10(9)7-8/h2-6H,7H2,1H3
Std. InChIKey: YSAXEHWHSLANOM-UHFFFAOYSA-N
IUPAC Name: 2-Methyl-1H-indene
Product Categories: Indanone Indene;Arenes;Building Blocks;Organic Building Blocks
2-Methylindene Safety Profile
WGK Germany: 3
2-Methylindene Specification
2-Methylindene (CAS No.2177-47-1), its synonyms are 1H-indene, 2-methyl- ; 2-Methyl-1H-indene .
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View