-
Name
2-Methylindoline
- EINECS 229-971-0
- CAS No. 6872-06-6
- Article Data89
- CAS DataBase
- Density 0.989 g/cm3
- Solubility negligible
- Melting Point -51 °C
- Formula C9H11N
- Boiling Point 232.6 °C at 760 mmHg
- Molecular Weight 133.193
- Flash Point 93.3 °C
- Transport Information
- Appearance clear yellow to brown liquid
- Safety 26-36/37/39-60-57
- Risk Codes 36/37/38-52/53-22
-
Molecular Structure
-
Hazard Symbols
 Xn,
Xn,  Xi
Xi
- Synonyms Indoline,2-methyl- (6CI,7CI,8CI);2,3-Dihydro-2-methyl-1H-indole;2,3-Dihydro-2-methylindole;2-Methyl-1H-indoline;2-Methyl-2,3-dihydro-1H-indole;2-Methyl-2,3-dihydroindole;NSC 65598;a-Methyldihydroindole;
- PSA 12.03000
- LogP 2.18110
Synthetic route

| Conditions | Yield |
|---|---|
| With tetrahydroxydiboron; palladium on activated carbon In 2,2,2-trifluoroethanol; water at 40℃; for 24h; Schlenk technique; | 99% |
| With zinc borohydride In diethyl ether for 48h; Ambient temperature; | 94% |
| With formic acid; C29H32ClIrNO; sodium formate In water at 30℃; for 16h; pH=4.5; | 92% |
-
-
32704-22-6
2-allyl-phenylamine

-
-
6872-06-6
2-methyl indoline

| Conditions | Yield |
|---|---|
| With C48H66N2O2Zr(2-); trityl tetrakis(pentafluorophenyl)borate In chlorobenzene at 140℃; for 48h; Glovebox; Inert atmosphere; | 94% |
| With C25H37BN4O2Zr(2+) In benzene-d6 at 20℃; for 72h; Reagent/catalyst; Inert atmosphere; stereoselective reaction; | 90% |
| With (pentamethylcyclopentadienyl)2LaCH(TMS)2 In pentane for 24h; Ambient temperature; |
-
-
1534360-76-3
2-(prop-2-yn-1-yl)aniline

-
-
6872-06-6
2-methyl indoline

| Conditions | Yield |
|---|---|
| With C33H63N15Zr3 In toluene at 140℃; for 48h; Glovebox; Inert atmosphere; | 93% |
-
-
149312-00-5, 18982-43-9
1ξ-(2-chloro-phenyl)-2-nitro-propene

-
-
6872-06-6
2-methyl indoline

| Conditions | Yield |
|---|---|
| With potassium carbonate; copper(l) chloride In water at 110℃; under 760.051 Torr; for 6h; Reagent/catalyst; Temperature; | 92% |
-
-
131880-76-7
1-(2-methyl-2,3-dihydro-indol-1-yl)-ethanone

-
A
-
54813-73-9
1-ethyl-2-methylindoline

-
B
-
6872-06-6
2-methyl indoline

| Conditions | Yield |
|---|---|
| With triethyl borane; Triethoxysilane; sodium hydroxide In hexane at 80℃; for 6h; Reagent/catalyst; Solvent; Inert atmosphere; Sealed tube; | A 10% B 90% |
| With Triethoxysilane; sodium triethylborohydride In hexane at 80℃; for 6h; Schlenk technique; Glovebox; Sealed tube; Inert atmosphere; | A 90% B n/a |
| With Triethoxysilane; sodium triethylborohydride In hexane at 80℃; for 6h; Inert atmosphere; Schlenk technique; High pressure; Sealed tube; | A 90% B n/a |
| With sodium triethylborohydride In tetrahydrofuran at 80℃; for 12h; Inert atmosphere; Schlenk technique; Glovebox; Sealed tube; | A n/a B 72% |
| With sodium triethylborohydride In tetrahydrofuran at 80℃; for 6h; Inert atmosphere; Schlenk technique; High pressure; Sealed tube; | A n/a B 72% |
-
-
15804-71-4
2-bromo-β-methyl-β-nitrostyrene

-
-
6872-06-6
2-methyl indoline

| Conditions | Yield |
|---|---|
| With copper(l) chloride; sodium hydroxide In water at 120℃; under 760.051 Torr; for 4h; Reagent/catalyst; Temperature; | 90% |

-
-
6872-06-6
2-methyl indoline

| Conditions | Yield |
|---|---|
| With hydrazine | 80% |

| Conditions | Yield |
|---|---|
| Zn(2+) montmorillonite for 0.05h; Cyclization; 3-Aza-Cope rearrangement; microwave irradiation; | 80% |
| Multi-step reaction with 2 steps 1: BF3/Et2O / xylene / 72 h / Heating 2: Cp'2LaCH(TMS)2 (Cp'= η5Me5C5) / pentane / 24 h / Ambient temperature View Scheme |
-
-
2058-74-4
1-methyl-1H-indole-2,3-dione

-
-
51315-70-9
2-methyl-1-(toluene-4-sulfonyl)-2,3-dihydro-1H-indole

-
-
6872-06-6
2-methyl indoline

| Conditions | Yield |
|---|---|
| Stage #1: 1-methyl-1H-indole-2,3-dione With sodium amalgam In N,N-dimethyl-formamide at 20℃; for 4h; Stage #2: 2-methyl-1-(toluene-4-sulfonyl)-2,3-dihydro-1H-indole In N,N-dimethyl-formamide at 110℃; for 18h; | 79% |

| Conditions | Yield |
|---|---|
| With hydrogen; platinum on activated charcoal In acetic acid at 100℃; under 45600 Torr; for 2h; | A n/a B 70% |
| With hydrogen; platinum on activated charcoal In acetic acid at 100℃; under 45600 Torr; for 2h; Product distribution; other catalyst, other solvent, other temperature, other pressure; | A n/a B 70% |
| With hydrogen; cobalt(II) acetate for 3h; | A 17% B 55% |
| With hydrogen In decalin at 180℃; under 37503.8 Torr; for 16h; Autoclave; |
-
-
61610-64-8
2-(ortho-bromophenyl)-1-methylethylamine

-
-
6872-06-6
2-methyl indoline

| Conditions | Yield |
|---|---|
| With 1,1'-bis-(diphenylphosphino)ferrocene; palladium 10% on activated carbon; sodium t-butanolate In 1,3,5-trimethyl-benzene at 140℃; for 24h; Inert atmosphere; | 70% |

-
-
6872-06-6
2-methyl indoline

| Conditions | Yield |
|---|---|
| With C52H62FeN5O4S2(1-)*C8H20N(1+) In N,N-dimethyl-formamide at 115℃; for 24h; Molecular sieve; | 57% |
| With tetrabutylammonium tricarbonylnitrosylferrate In 1,2-dichloro-ethane; N,N-dimethyl-formamide at 120℃; for 1h; Microwave irradiation; Inert atmosphere; |

| Conditions | Yield |
|---|---|
| With bis{rhodium[3,3'-(1,3-phenylene)bis(2,2-dimethylpropanoic acid)]} In toluene at 120℃; for 16h; Inert atmosphere; | A 30% B 30% |


-
-
6872-06-6
2-methyl indoline

| Conditions | Yield |
|---|---|
| With water inactive 2-methyl-indoline; |

-
-
6872-06-6
2-methyl indoline
-
-
81777-39-1
tert-Butyl-[1-(2-methyl-2,3-dihydro-indol-1-yl)-meth-(E)-ylidene]-amine

-
-
6872-06-6
2-methyl indoline

| Conditions | Yield |
|---|---|
| With potassium hydroxide In dimethyl sulfoxide for 3.5h; heating; |
-
-
77435-11-1
ethyl 1-benzyl-2-methyl-1H-indole-3-carboxylate

-
A
-
53855-47-3
ethyl 2-methyl-1H-indole-3-carboxylate

-
B
-
77435-28-0
3-hydroxymethyl-2-methylindoline

-
C
-
6872-06-6
2-methyl indoline

| Conditions | Yield |
|---|---|
| With ammonia; sodium In diethyl ether for 2h; Heating; | A n/a B 245 mg C 207 mg |


| Conditions | Yield |
|---|---|
| at 190℃; under 220652 Torr; Hydrogenation; |


| Conditions | Yield |
|---|---|
| bei der elektrolytischen Reduktion in saurer Loesung; |


-
-
95-20-5
2-methyl-1H-indole

-
-
10034-85-2
hydrogen iodide

-
A
-
1821-39-2
2-propylaniline

-
B
-
6872-06-6
2-methyl indoline


| Conditions | Yield |
|---|---|
| In benzene for 0.833333h; Irradiation; | |
| With air In benzene for 0.833333h; Irradiation; | |
| In acetonitrile for 1h; Irradiation; | |
| With 2-(1,3-bis(3'-hexylimidazol-2'-ylidene)phenylene)bis(dimethylamido)iodo zirconium (IV) In (2)H8-toluene at 160℃; for 192h; Inert atmosphere; Sealed tube; Glovebox; |
-
-
32704-22-6
2-allyl-phenylamine

-
A
-
95-20-5
2-methyl-1H-indole

-
B
-
635-46-1
1,2,3,4-tetrahydroisoquinoline

-
C
-
65826-91-7
1-(2-aminophenyl)-2-propanol

-
D
-
6872-06-6
2-methyl indoline

| Conditions | Yield |
|---|---|
| With water; β‐cyclodextrin for 1h; Irradiation; |


| Conditions | Yield |
|---|---|
| In benzene for 0.833333h; Irradiation; |


| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: ZnCl2 / xylene / 5.5 h / 145 °C 2.1: selenenyl bromide resin; SnCl4 / CH2Cl2 / 0.5 h / -20 °C 2.2: n-Bu3SnH; AIBN / toluene / 2 h / 90 °C View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: dimethylformamide / 12 h / 20 °C 2.1: ZnCl2 / xylene / 5.5 h / 145 °C 3.1: selenenyl bromide resin; SnCl4 / CH2Cl2 / 0.5 h / -20 °C 3.2: n-Bu3SnH; AIBN / toluene / 2 h / 90 °C View Scheme |


-
-
6872-06-6
2-methyl indoline

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 92 percent / t-BuLi / tetrahydrofuran / 1.) -20 deg C 2: 80 percent / N2H4 View Scheme |
-
-
17697-12-0
benzeneseleninic anhydride

-
-
6872-06-6
2-methyl indoline

-
-
85677-01-6
2-methyl-3-(phenylselanyl)-1H-indole

| Conditions | Yield |
|---|---|
| In tetrahydrofuran for 1h; | 100% |
| In tetrahydrofuran at 0℃; for 2h; | 96% |
-
-
124-63-0
methanesulfonyl chloride

-
-
6872-06-6
2-methyl indoline

-
-
66929-70-2
1H-indole,2,3-dihydro-2-methyl-1-(methylsulfonyl)

| Conditions | Yield |
|---|---|
| In pyridine at 20℃; for 20h; | 100% |
| With pyridine at 20℃; for 2h; Inert atmosphere; | 30% |
2-Methylindoline Chemical Properties
Product Name: 2-Methylindoline (CAS NO.6872-06-6)
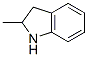
MF: C9H11N
MW: 133.190340 g/mol
Flash Point: 93.3 °C
Melting Point: -51 °C
EINECS: 229-971-0
Water Solubility: negligible
IUPAC Name: 2-methyl-2,3-dihydro-1H-indole
Density: 1.023 g/mL at 25 °C(lit.)
Refractive index: n20/D 1.569(lit.)
Enthalpy of Vaporization: 46.93 kJ/mol
Boiling Point: 232.6 °C at 760 mmHg
Vapour Pressure: 0.0585 mmHg at 25°C
Product Categories: Indoles and derivatives; pharmacetical; Indoline & Oxindole; Heterocyclic Compounds; Building Blocks; Heterocyclic Building Blocks; Indoles
2-Methylindoline Toxicity Data With Reference
| 1. | ipr-mus LD50:360 mg/kg | FATOAO Farmakologiya i Toksikologiya (Moscow). 29 (1966),721. |
2-Methylindoline Consensus Reports
Reported in EPA TSCA Inventory.
2-Methylindoline Safety Profile
A poison by intraperitoneal route. When heated to decomposition it emits toxic vapors of NOx.
Safety Information of 2-Methylindoline (CAS NO.6872-06-6):
Hazard Codes : Xi ,Xn
,Xn
Risk Statements: 36/37/38-52/53-22
36/37/38: Irritating to eyes, respiratory system and skin
52/53: Harmful to aquatic organisms, may cause long-term adverse effects in the aquatic environment
22: Harmful if swallowed
Safety Statements: 26-36/37/39-60-57
26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice
36/37/39: Wear suitable protective clothing, gloves and eye/face protection 60: This material and/or its container must be disposed of as hazardous waste
57: Use appropriate container to avoid environmental contamination
WGK Germany: 3
RTECS: NM1926350
Hazard Note : Irritant
2-Methylindoline Specification
2-Methylindoline , its CAS NO. is 6872-06-6, the synonyms are R,S)-2-Methyl-2,3-dihydro-1H-indole ; 1H-Indole, 2,3-dihydro-2-methyl- ; 1H-Indole,2,3-dihydro-2-methyl- ; 2,3-dihydro-2-methyl-1h-indol ; 2,3-Dihydro-2-methyl-1H-indole ; 2-Methyl-2,3-dihydroindole ; 2-methyl-indolin ; alpha-Methyldihydroindole .
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View