-
Name
N1-(3-Aminophenyl)acetamide
- EINECS 203-021-5
- CAS No. 102-28-3
- Article Data39
- CAS DataBase
- Density 1.203 g/cm3
- Solubility 1-5 g/100 mL at 24 °C in water
- Melting Point 86-88 °C(lit.)
- Formula C8H10N2O
- Boiling Point 388.9 °C at 760 mmHg
- Molecular Weight 150.18
- Flash Point 189 °C
- Transport Information
- Appearance Gray solid.
- Safety 26-36-37/39
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms m-Aminoacetanilide;Acetanilide,3'-amino- (6CI,7CI,8CI);1-Acetylamino-3-aminobenzene;1-Amino-3-(acetylamino)benzene;3-(Acetylamino)aniline;3-Acetamidoaniline;3-Acetylaminobenzeneamine;3-Amino-N-acetylaniline;N-(3-Aminophenyl)acetamide;N-Acetyl-1,3-diaminobenzene;N-Acetyl-m-phenylenediamine;NSC 165576;m-(Acetylamino)aniline;m-Acetamidoaniline;
- PSA 55.12000
- LogP 1.88140
Synthetic route

| Conditions | Yield |
|---|---|
| With iron(III)-acetylacetonate; hydrazine hydrate In methanol at 150℃; Microwave irradiation; Green chemistry; | 99% |

| Conditions | Yield |
|---|---|
| With 1,4-diaza-bicyclo[2.2.2]octane at 65 - 90℃; for 6h; Time; Inert atmosphere; | 98.4% |


| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate; TPGS-750-M In tetrahydrofuran; water at 20℃; for 0.5h; | 98% |
| With aluminum amalgam; water In tetrahydrofuran at 20℃; for 0.5h; | 96% |
| With iron; acetic acid In neat liquid at 50℃; for 0.25h; | 91% |

| Conditions | Yield |
|---|---|
| With chlorosulfonic acid In toluene at 90℃; for 0.5h; Beckmann rearrangement; | 98% |
| With tricarallylic acid at 160℃; for 0.0833333h; Beckmann rearrangement; Inert atmosphere; neat (no solvent); | 90% |

| Conditions | Yield |
|---|---|
| 88.5% |

| Conditions | Yield |
|---|---|
| With dicyclohexyl(2',4',6'-triisopropyl-5-methoxy-3,4,6-trimethyl-[1,1'-biphenyl]-2-yl)phosphine; C50H70NO4PPdS; C50H70NO4PPdS; dicyclohexyl(2',4',6'-triisopropyl-4-methoxy-3,5,6-trimethyl-[1,1'-biphenyl]-2-yl)phosphine; ammonia; sodium t-butanolate In 1,4-dioxane at 80℃; for 24h; Inert atmosphere; | 48% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at -10℃; for 5h; | 34.2% |

| Conditions | Yield |
|---|---|
| With water das Hydrochlorid entsteht; |

| Conditions | Yield |
|---|---|
| With hydrogenchloride das Hydrochlorid entsteht; ueber mehrere Stufen; |

| Conditions | Yield |
|---|---|
| With sodium chloride pH ; |
-
-
64-19-7
acetic acid

-
-
25186-45-2
N-(3-(phenyldiazenyl)phenyl)acetamide

-
A
-
62-53-3
aniline

-
B
-
102-28-3
m-acetamide aniline


| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: HCl / H2O / 8 h / pH = 1,5-3,5 2: 20 percent NaCl-solution / pH View Scheme |


| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: palladium on activated charcoal; hydrogen / methanol / 8 h / 20 °C 2: acetic acid / Reflux View Scheme | |
| Multi-step reaction with 2 steps 1: N-ethyl-N,N-diisopropylamine / dichloromethane / 20 °C / Inert atmosphere 2: palladium 10% on activated carbon; hydrogen / dichloromethane; methanol / 20 °C / 760.05 Torr View Scheme | |
| Multi-step reaction with 2 steps 1: triethylamine / dichloromethane 2: tin(II) chloride dihdyrate / ethanol / 16 h / Reflux View Scheme | |
| Multi-step reaction with 2 steps 1: 0.5 h / 20 °C 2: acetic acid; iron / neat liquid / 0.25 h / 50 °C View Scheme |
-
-
102-28-3
m-acetamide aniline

| Conditions | Yield |
|---|---|
| With tetrafluoroboric acid; tert.-butylnitrite In methanol; water at 0℃; for 1h; Inert atmosphere; | 100% |
| Multi-step reaction with 2 steps 1: tetrahydrofuran / -15 °C 2: tert.-butylnitrite / tetrahydrofuran View Scheme |
-
-
871700-07-1
trifluoromethanesulfonic acid 3-cyclopropyl-1-(2-fluoro-4-iodophenyl)-6,8-dimethyl-2,4,7-trioxo-1,2,3,4,7,8-hexahydro-pyrido[2,3-d]pyrimidin-5-yl ester

-
-
102-28-3
m-acetamide aniline

| Conditions | Yield |
|---|---|
| With 2,6-dimethylpyridine In N,N-dimethyl acetamide at 130℃; for 5h; Product distribution / selectivity; | 99% |
-
-
6629-04-5
ethyl N-cyanoacetylcarbamate

-
-
102-28-3
m-acetamide aniline

-
-
1207441-77-7
(Z)-ethyl 2-(2-(3-acetamidophenyl)hydrazono)-2-cyanoacetylcarbamate

| Conditions | Yield |
|---|---|
| Stage #1: m-acetamide aniline With hydrogenchloride; sodium nitrite In water at 0℃; for 0.5h; Stage #2: ethyl N-cyanoacetylcarbamate With sodium acetate In ethanol; water at 0℃; for 3h; | 99% |

| Conditions | Yield |
|---|---|
| Stage #1: methyl chloroacetate; m-acetamide aniline With sodium carbonate at 90 - 100℃; for 2h; Stage #2: With sodium bromide; zinc at 105 - 110℃; for 2h; Catalytic behavior; Time; | 98.6% |
-
-
102-28-3
m-acetamide aniline

| Conditions | Yield |
|---|---|
| With benzyltrimethylammonium chlorobromate In methanol; dichloromethane for 3h; Ambient temperature; | 98% |
-
-
102-28-3
m-acetamide aniline

-
-
49584-26-1
p-cyanobenzenesulfonyl chloride

-
-
847732-99-4
N-(3-{[(4-Cyanophenyl)sulfonyl]amino}phenyl)acetamide

| Conditions | Yield |
|---|---|
| With sodium acetate In water; isopropyl alcohol at 20℃; for 3.5h; Heating / reflux; | 98% |

| Conditions | Yield |
|---|---|
| With ammonium hydroxide In water at 90 - 98℃; under 7500.75 Torr; for 18h; pH=5 - 7; Temperature; Large scale; | 98% |
-
-
302601-82-7
4-(2-(diethylamino)acetamido)benzoic acid

-
-
102-28-3
m-acetamide aniline

-
-
1357620-21-3
3-acetamido-N-(4-(2-(diethylamino)acetamido)phenyl)benzamide

| Conditions | Yield |
|---|---|
| With dmap; benzotriazol-1-ol; dicyclohexyl-carbodiimide In N,N-dimethyl-formamide at 20℃; for 20h; Inert atmosphere; | 97% |
-
-
102-28-3
m-acetamide aniline

-
-
50617-74-8
N-ethyl-1,3-benzenediamine

| Conditions | Yield |
|---|---|
| With dimethyl sulfide borane In tetrahydrofuran at 60℃; for 24h; Inert atmosphere; | 97% |
-
-
102-28-3
m-acetamide aniline

| Conditions | Yield |
|---|---|
| With tetra-N-butylammonium tribromide In methanol; dichloromethane for 0.166667h; Ambient temperature; | 96% |
-
-
108-77-0
1,3,5-trichloro-2,4,6-triazine

-
-
102-28-3
m-acetamide aniline

-
-
395667-32-0
2-chloro-4,6-bis([3-acetylamino]phenylamino)-triazine

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate In acetone for 3h; Reflux; | 96% |
-
-
88284-48-4
2-(trimethylsilyl)phenyl trifluoromethanesulfonate

-
-
102-28-3
m-acetamide aniline

-
-
19619-91-1
N-(3-(phenylamino)phenyl)acetamide

| Conditions | Yield |
|---|---|
| With cesium fluoride In acetonitrile at 20℃; for 10h; | 95% |

| Conditions | Yield |
|---|---|
| With sodium carbonate; sodium bromide at 10 - 100℃; | 94.89% |
| With sodium carbonate; sodium bromide at 100℃; | 94.89% |
| With sodium carbonate; sodium bromide at 100℃; | 94.89% |
-
-
871700-56-0
1-(2-fluoro-4-iodophenyl)-3-(4-methoxybenzyl)-6,8-dimethyl-2,4,7-trioxo-1,2,3,4,7,8-hexahydropyrido[2,3-d]pyrimidin-5-yl trifluoromethanesulfonate

-
-
102-28-3
m-acetamide aniline

| Conditions | Yield |
|---|---|
| With 2,6-dimethylpyridine In N,N-dimethyl acetamide at 130℃; for 2h; | 94.6% |
-
-
2927-71-1
2,4-dichloro-5-fluoropyrimidine

-
-
102-28-3
m-acetamide aniline

-
-
1166389-12-3
N-(3-(2-chloro-5-fluoropyrimidin-4-ylamino)phenyl)acetamide

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In isopropyl alcohol at 80℃; for 16h; | 94% |
| With N-ethyl-N,N-diisopropylamine In isopropyl alcohol at 80℃; for 16h; | 94% |

| Conditions | Yield |
|---|---|
| In water at 100℃; for 3h; | 93.8% |

| Conditions | Yield |
|---|---|
| With triethylamine In 1,2-dichloro-ethane at 50 - 70℃; | 93.7% |
-
-
871700-32-2
3-cyclopropyl-1-(2-fluoro-4-iodophenyl)-6,8-dimethyl-2,4,7-trioxo-1,2,3,4,7,8-hexahydropyrido[2,3-d]pyrimidin-5-yl 4-methylbenzenesulfonate

-
-
102-28-3
m-acetamide aniline

| Conditions | Yield |
|---|---|
| With 2,6-dimethylpyridine In N,N-dimethyl acetamide at 130℃; for 4h; Product distribution / selectivity; | 93% |

| Conditions | Yield |
|---|---|
| In isopropyl alcohol at 110℃; Sealed tube; Microwave irradiation; | 92% |
-
-
2831-66-5
2,4-dichloro-1,3,5-triazine

-
-
102-28-3
m-acetamide aniline

-
-
1211876-46-8
N-(3-(4-chloro-1,3,5-triazin-2-ylamino)phenyl)acetamide

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In 1,2-dimethoxyethane at 20℃; for 16h; Cooling with ice; | 91.7% |
-
-
153493-48-2
5,6-dichlorofurazano<3,4-b>pyrazine

-
-
102-28-3
m-acetamide aniline

| Conditions | Yield |
|---|---|
| With N,N-diethylaniline In acetonitrile Substitution; Heating; | 91% |

| Conditions | Yield |
|---|---|
| With [(9,9-Me2-4,5-(PPh2)2-xanthene]Pd(η3-allyl)OTf In 1,4-dioxane at 100℃; for 24h; | 91% |
-
-
50-00-0
formaldehyd

-
-
108-73-6
3,5-dihydroxyphenol

-
-
102-28-3
m-acetamide aniline

-
-
1353370-43-0
C36H36N6O6

| Conditions | Yield |
|---|---|
| With acetic acid In ethanol; water at 20℃; for 12h; | 91% |

-
-
82671-02-1
2,6-dichloro-5-fluoro-pyridine-3-carbonitrile

-
-
102-28-3
m-acetamide aniline

-
-
1374215-08-3
C14H10ClFN4O

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In 1-methyl-pyrrolidin-2-one at 20 - 100℃; for 3h; | 91% |
-
-
17763-67-6
Phenyl triflate

-
-
102-28-3
m-acetamide aniline

-
-
19619-91-1
N-(3-(phenylamino)phenyl)acetamide

| Conditions | Yield |
|---|---|
| With C47H70BrO4PPdSi; sodium t-butanolate In tetrahydrofuran at 20℃; Schlenk technique; Inert atmosphere; Sealed tube; | 91% |
| With C46H27F24FeNiP2(1+)*CF3O3S(1-); triethylamine In 2-methyltetrahydrofuran at 100℃; for 16h; Schlenk technique; Inert atmosphere; Glovebox; Sealed tube; | 25 %Chromat. |

-
-
102-28-3
m-acetamide aniline

-
-
363135-51-7
N-(3-Formylquinolin-7-yl)acetamide

| Conditions | Yield |
|---|---|
| Stage #1: 2-iminiomethylvinamidinium trichloride monohydrate; m-acetamide aniline With piperidine In butan-1-ol at 80℃; for 2h; Stage #2: With acetic acid In water; butan-1-ol Stage #3: With sodium hydroxide In water at 20℃; for 1h; | 90% |
3'-Aminoacetanilide Chemical Properties
Product Name:N1-(3-Aminophenyl)acetamide
The MF of N1-(3-Aminophenyl)acetamide(102-28-3) is C8H10N2O.
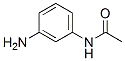
The MW of N1-(3-Aminophenyl)acetamide(102-28-3) is 150.18.
Synonyms: N-ACETYL-M-PHENYLENEDIAMINE;N-ACETYL-1,3-PHENYLENEDIAMINE;N1-(3-AMINOPHENYL)ACETAMIDE;3’-amino-acetanilid;3-acetylaminoaniline
EINECS: 203-021-5
Product Categories: Intermediates of Dyes and Pigments;AMINEPRIMARY;Aromatic Carboxylic Acids, Amides, Anilides, Anhydrides & Salts
Mol File: 102-28-3.mol
mp:86-88 °C(lit.)
Surface Tension of it is 53 dyne/cm and the density is 1.203 g/cm3
Flash Point: 189 °C
Enthalpy of Vaporization: 63.82 kJ/mol
Boiling Point: 388.9 °C at 760 mmHg
Water Solubility: 1-5 g/100 mL at 24 oC
CAS DataBase Reference:102-28-3(CAS DataBase Reference)
EPA Substance Registry:System 102-28-3(EPA Substance)
3'-Aminoacetanilide Uses
The N1-(3-Aminophenyl)acetamide(102-28-3) is used primarily as a reaction intermediate for dyes and disperse dyes
3'-Aminoacetanilide Toxicity Data With Reference
| 1. | eye-rbt 500 mg/24H SEV | 28ZPAK Sbornik Vysledku Toxixologickeho Vysetreni Latek A Pripravku Marhold, J.V.,Institut Pro Vychovu Vedoucicn Pracovniku Chemickeho Prumyclu Praha,Czechoslovakia.: 1972,129. | ||
| 2. | mmo-sat 333 µg/plate | EMMUEG Environmental and Molecular Mutagenesis. 11 (Suppl 12)(1988),1. | ||
| 3. | ivn-mus LD50:320 mg/kg | CSLNX* U.S. Army Armament Research & Development Command, Chemical Systems Laboratory, NIOSH Exchange Chemicals. (Aberdeen Proving Ground, MD 21010) NX#02899 . |
3'-Aminoacetanilide Consensus Reports
3'-Aminoacetanilide Safety Profile
Poison by intravenous route. Moderately toxic by ingestion. A severe eye irritant. Mutation data reported. When heated to decomposition it emits toxic fumes of NOx.
Hazard Codes: Xi
Xi
Risk Statements:36/37/38
R36/37/38:Irritating to eyes, respiratory system and skin ;
Safety Statements:26-36-37/39
S26:In case of contact with eyes, rinse immediately with plenty of water and seek medical advice ;
S36:Wear suitable protective clothing ;
S37/39:Wear suitable gloves and eye/face protection ;
WGK Germany:3
RTECS:AD8050000
3'-Aminoacetanilide Specification
It is Incompatible with Strong oxidizing agents, acids, acetic anhydride, acid chlorides, carbon dioxide.Hazardous Decomposition Products is Nitrogen oxides, carbon monoxide, carbon dioxide.
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View