-
Name
3,3'-Thiodipropionic acid
- EINECS 203-841-3
- CAS No. 111-17-1
- Article Data28
- CAS DataBase
- Density 1.362 g/cm3
- Solubility 40 g/L (25 °C) in water
- Melting Point 131-134 °C(lit.)
- Formula C6H10O4S
- Boiling Point 409.3 °C at 760 mmHg
- Molecular Weight 178.209
- Flash Point 201.3 °C
- Transport Information
- Appearance White fine crystalline powder
- Safety 26-36-37/39
- Risk Codes 36/37/38-36
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms Propionicacid, 3,3'-thiodi- (6CI,8CI);3,3'-Thiobis(propanoic acid);3,3'-Thiodipropanoic acid;4-Thiaheptanedioic acid;Bis(2-carboxyethyl) sulfide;Diethyl sulfide 2,2'-dicarboxylic acid;NSC 65523;NSC 8166;TDPA;Thiodihydracrylic acid;Thiodipropionic acid;Tyox A;b,b'-Thiodipropionic acid;
- PSA 99.90000
- LogP 0.66900
Synthetic route

| Conditions | Yield |
|---|---|
| Stage #1: acrylic acid With sodium hydroxide In water at 45 - 50℃; for 0.5h; Stage #2: With hydrogen sulfide In water at 45 - 100℃; Concentration; Time; | A n/a B 94.4% |
| Stage #1: acrylic acid With sodium hydroxide In water at 45 - 50℃; for 0.5h; Stage #2: With hydrogen sulfide In water at 45 - 100℃; for 9.46h; Stage #3: With sulfuric acid; iron In water for 2.5h; Concentration; Reagent/catalyst; Inert atmosphere; |

| Conditions | Yield |
|---|---|
| With silica bromide In dichloromethane at 20℃; for 0.166667h; Inert atmosphere; | 94% |

| Conditions | Yield |
|---|---|
| With water for 48h; Rhodococcus rhodochrous AJ270; | 60% |
| With potassium phosphate buffer; Rhodococcus sp. AJ270 at 30℃; for 48h; | 60% |
| With hydrogenchloride |
-
-
57-57-8
β-Propiolactone

-
-
5466-06-8
3-mercaptopropionic acid ethyl ester

-
-
111-17-1
4-thiaheptane-1,7-dioic acid

| Conditions | Yield |
|---|---|
| With sodium hydroxide at 10℃; folgende Verseifung in der Siedehitze; |

| Conditions | Yield |
|---|---|
| With sodium sulfide; water at 200℃; |

| Conditions | Yield |
|---|---|
| With sodium sulfide; water |
-
-
141-76-4
3-iodopropanoic acid

-
A
-
111-17-1
4-thiaheptane-1,7-dioic acid

-
B
-
107-96-0
3-mercaptopropionic acid

| Conditions | Yield |
|---|---|
| With potassium hydrosulfide | |
| With alkali hydrosulfide |
-
-
3680-08-8
3,3'-sulfinyldipropionic acid

-
-
68-11-1
mercaptoacetic acid

-
A
-
111-17-1
4-thiaheptane-1,7-dioic acid

-
B
-
505-73-7
disulfanediyldiacetic acid

| Conditions | Yield |
|---|---|
| at 60℃; Rate constant; |


| Conditions | Yield |
|---|---|
| With potassium hydroxide; hydrogen sulfide at 15 - 25℃; folgende Verseifung mit verduennter Natronlauge bei 65grad; |
-
-
292638-85-8
acrylic acid methyl ester

-
-
107-96-0
3-mercaptopropionic acid

-
-
111-17-1
4-thiaheptane-1,7-dioic acid

| Conditions | Yield |
|---|---|
| With hydrogenchloride |

| Conditions | Yield |
|---|---|
| With aqueous alkali | |
| With sulfuric acid; acetic acid; hydroquinone |
-
-
79-10-7
acrylic acid

-
-
107-96-0
3-mercaptopropionic acid

-
A
-
111-17-1
4-thiaheptane-1,7-dioic acid

-
B
-
39669-77-7
tris-(2-carboxy-ethyl)-sulfonium ; chloride

| Conditions | Yield |
|---|---|
| With hydrogenchloride; water; acetic acid |


| Conditions | Yield |
|---|---|
| With sodium sulfide; water pH 9; |
-
-
57-57-8
β-Propiolactone

-
-
7732-18-5
water

-
A
-
111-17-1
4-thiaheptane-1,7-dioic acid

-
B
-
107-96-0
3-mercaptopropionic acid

-
-
7664-93-9
sulfuric acid

-
-
64-19-7
acetic acid

-
-
123-31-9
hydroquinone

-
-
79-10-7
acrylic acid

-
-
107-96-0
3-mercaptopropionic acid

-
-
111-17-1
4-thiaheptane-1,7-dioic acid


| Conditions | Yield |
|---|---|
| in neutraler oder schwach mineralsaurer wss. Loesung; |

-
-
110-89-4
piperidine

-
-
7732-18-5
water

-
-
123-31-9
hydroquinone

-
-
79-10-7
acrylic acid

-
-
107-96-0
3-mercaptopropionic acid

-
A
-
111-17-1
4-thiaheptane-1,7-dioic acid

-
B
-
52662-37-0
(+/-)-2-methyl-3-thia-adipic acid

-
-
7647-01-0
hydrogenchloride

-
-
64-19-7
acetic acid

-
-
123-31-9
hydroquinone

-
-
79-10-7
acrylic acid

-
-
107-96-0
3-mercaptopropionic acid

-
A
-
111-17-1
4-thiaheptane-1,7-dioic acid

-
-
7647-01-0
hydrogenchloride

-
-
64-19-7
acetic acid

-
-
107-96-0
3-mercaptopropionic acid

-
A
-
111-17-1
4-thiaheptane-1,7-dioic acid


-
-
111-17-1
4-thiaheptane-1,7-dioic acid

| Conditions | Yield |
|---|---|
| With sodium sulfide |

-
-
111-17-1
4-thiaheptane-1,7-dioic acid

| Conditions | Yield |
|---|---|
| With water |
-
-
3680-08-8
3,3'-sulfinyldipropionic acid

-
-
10035-10-6, 12258-64-9
hydrogen bromide

-
-
64-19-7
acetic acid

-
-
111-17-1
4-thiaheptane-1,7-dioic acid

-
-
7647-01-0
hydrogenchloride

-
-
13532-18-8
methyl 3-(methylthio)propionate

-
A
-
111-17-1
4-thiaheptane-1,7-dioic acid

-
B
-
75-18-3
dimethylsulfide

-
-
7647-01-0
hydrogenchloride

-
-
13327-56-5
3-(methylthio)propanoic acid ethyl ester

-
A
-
111-17-1
4-thiaheptane-1,7-dioic acid

-
B
-
75-18-3
dimethylsulfide

-
-
13532-18-8
methyl 3-(methylthio)propionate

-
-
7664-93-9
sulfuric acid

-
A
-
111-17-1
4-thiaheptane-1,7-dioic acid

-
B
-
75-18-3
dimethylsulfide

-
-
7647-01-0
hydrogenchloride

-
-
646-01-5
3-(methylsulfanyl)propionic acid

-
A
-
111-17-1
4-thiaheptane-1,7-dioic acid

-
B
-
74-93-1
methylthiol

-
C
-
75-18-3
dimethylsulfide


| Conditions | Yield |
|---|---|
| With dihydrogen peroxide In neat (no solvent) at 20℃; for 0.0333333h; Reagent/catalyst; | 99% |
| With dihydrogen peroxide at 20℃; for 0.0166667h; Reagent/catalyst; | 98% |
| With dihydrogen peroxide In neat (no solvent) at 20℃; for 0.0166667h; Reagent/catalyst; chemoselective reaction; | 98% |
-
-
111-17-1
4-thiaheptane-1,7-dioic acid

-
-
138500-88-6
(4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl)methanamine

| Conditions | Yield |
|---|---|
| With 1-hydroxy-pyrrolidine-2,5-dione; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride for 36h; | 98% |
-
-
111-17-1
4-thiaheptane-1,7-dioic acid

-
-
107-18-6
allyl alcohol

-
-
39557-51-2
diallyl 3,3'-thiodipropanoate

| Conditions | Yield |
|---|---|
| toluene-4-sulfonic acid In toluene Heating; | 93% |
| With toluene-4-sulfonic acid In toluene for 8h; Heating / reflux; | 20 g |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In ethanol at 80℃; for 2h; | 92% |


| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In acetonitrile at 40℃; for 4h; Temperature; Reagent/catalyst; Solvent; Sonication; Inert atmosphere; | 89.03% |
| With dmap; dicyclohexyl-carbodiimide In acetonitrile at 40℃; for 4h; Temperature; Reagent/catalyst; Solvent; Inert atmosphere; | 89.03% |
-
-
111-17-1
4-thiaheptane-1,7-dioic acid

-
-
66-71-7
1,10-Phenanthroline

-
-
7732-18-5
water

-
-
6046-93-1
copper(II) acetate monohydrate

| Conditions | Yield |
|---|---|
| Stage #1: 4-thiaheptane-1,7-dioic acid; water; copper(II) acetate monohydrate at 70℃; for 1h; Stage #2: 1,10-Phenanthroline In ethanol at 70℃; for 2h; | 86% |


| Conditions | Yield |
|---|---|
| With sulfuric acid Heating; | 85% |
| With sulfuric acid |
-
-
141-86-6
2,6-Diaminopyridine

-
-
111-17-1
4-thiaheptane-1,7-dioic acid

-
-
1120337-12-3
1,3,5,13,15,17-hexaaza-6,12,18,24-tetraoxo-9,21-dithia-2,3,4:14,15,16-dipyridine cyclotetracosane

| Conditions | Yield |
|---|---|
| With hydrogenchloride In ethanol; water at 85℃; | 85% |
-
-
111-17-1
4-thiaheptane-1,7-dioic acid

-
-
36653-82-4
1-Hexadecanol

-
-
3287-12-5
dihexadecyl 3,3'-thiodipropionate

| Conditions | Yield |
|---|---|
| With Novozym 435 at 80℃; under 600.048 Torr; for 4h; | 84% |

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide In methanol at 40℃; for 1h; chemoselective reaction; | 82% |
| With potassium permanganate | |
| With water; bromine | |
| With dihydrogen peroxide; acetic acid | |
| With Oxone In tetrahydrofuran; water at 20℃; for 3h; |
-
-
111-17-1
4-thiaheptane-1,7-dioic acid

-
-
91-76-9
benzoguanamine

-
-
1198294-59-5
1,11(2,6)-ditriazina-2,10,12,20-tetraaza-3,9,13,19-tetraoxo-6,16-dithiacyclocosa-phan-1(4),11(4)-diphenyl

| Conditions | Yield |
|---|---|
| With hydrogenchloride In ethanol; water Heating; | 82% |

| Conditions | Yield |
|---|---|
| Stage #1: guanidine carbonate; 4-thiaheptane-1,7-dioic acid; neodymium(III) nitrate hexahydrate In water pH=6.55; Stage #2: With nitric acid In water pH=4 - 4.5; | 80.5% |

-
-
111-17-1
4-thiaheptane-1,7-dioic acid

-
-
18733-39-6
β,β'-thiodipropionic acid dichloroanhydride

| Conditions | Yield |
|---|---|
| With thionyl chloride In benzene Heating; | 80% |
| With thionyl chloride In chloroform; N,N-dimethyl-formamide at 50℃; for 0.166667h; | 80% |
| With thionyl chloride |

| Conditions | Yield |
|---|---|
| With sulfuric acid Heating; | 80% |
| With sulfuric acid for 4h; Reflux; |

| Conditions | Yield |
|---|---|
| With sulfuric acid Heating; | 79% |
| With sulfuric acid | |
| With toluene-4-sulfonic acid; benzene unter Entfernen des entstehenden Wassers; |
-
-
111-17-1
4-thiaheptane-1,7-dioic acid

-
-
79-19-6
thiosemicarbazide

-
-
303065-25-0
5,5'-(thiobis(ethane-2,1-diyl))bis(1,3,4-thiadiazol-2-amine)

| Conditions | Yield |
|---|---|
| With trichlorophosphate at 90℃; for 3h; | 79% |
| With trichlorophosphate at 90℃; for 3h; | 60% |
-
-
366-18-7
[2,2]bipyridinyl

-
-
111-17-1
4-thiaheptane-1,7-dioic acid

-
-
7732-18-5
water

-
-
6046-93-1
copper(II) acetate monohydrate

-
-
142576-33-8, 1574389-81-3
{[Cu(μ3-tdp)(2,2'-bipyridine)]·H2O}n

| Conditions | Yield |
|---|---|
| Stage #1: 4-thiaheptane-1,7-dioic acid; water; copper(II) acetate monohydrate at 70℃; for 1h; Stage #2: [2,2]bipyridinyl In ethanol at 70℃; for 2h; | 79% |


| Conditions | Yield |
|---|---|
| Stage #1: guanidine carbonate; 4-thiaheptane-1,7-dioic acid; praseodymium(III) nitrate hexahydrate In water pH=6.55; Stage #2: With nitric acid In water pH=4 - 4.5; | 78.1% |


| Conditions | Yield |
|---|---|
| With sulfuric acid Heating; | 78% |
-
-
111-17-1
4-thiaheptane-1,7-dioic acid

| Conditions | Yield |
|---|---|
| With triphenylphosphine; diethylazodicarboxylate In tetrahydrofuran at 0 - 20℃; for 18h; | 78% |

| Conditions | Yield |
|---|---|
| With potassium hydroxide In methanol; water | 77% |


| Conditions | Yield |
|---|---|
| Stage #1: guanidine carbonate; 4-thiaheptane-1,7-dioic acid; samarium(III) nitrate hexahydrate In water pH=6.55; Stage #2: With nitric acid In water pH=4 - 4.5; | 76.7% |


| Conditions | Yield |
|---|---|
| With sulfuric acid Heating; | 76% |
| With Novozym 435 at 80℃; under 600.048 Torr; for 24h; |

| Conditions | Yield |
|---|---|
| With sulfuric acid Heating; | 76% |

| Conditions | Yield |
|---|---|
| With dmap; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In dichloromethane; N,N-dimethyl-formamide at 20℃; for 48h; Inert atmosphere; | 76% |
-
-
111-17-1
4-thiaheptane-1,7-dioic acid

-
-
109-76-2
Trimethylenediamine

-
-
1192661-22-5
1,9,13,21-tetraaza-5,17-dithiacyclotetracosane-2,8,14,20-tetraone

| Conditions | Yield |
|---|---|
| With hydrogenchloride In ethanol; water at 85℃; for 14h; pH=3 - 4; | 75% |

| Conditions | Yield |
|---|---|
| With Na2CO3 In water byproducts: CO2; under aerobic condns.; to aq. soln. of thiodipropionic acid, added Na2CO3; mixt. stirred, then soln. mixed with aq. ZnCl2; soln. stirred for 10 min; acetone added; left overnight at room temp.; crystals filtered off, washed with acetone and Et2O; dried in air; elem. anal.; | 75% |

| Conditions | Yield |
|---|---|
| With triphenylphosphine; diethylazodicarboxylate In tetrahydrofuran at 0 - 20℃; for 18h; | 74% |
3,3'-Thiodipropionic acid Chemical Properties
Molecular Structure of BIS(2-CARBOXYETHYL) SULFIDE(111-17-1):
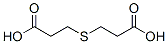
IUPAC Name:3-(2-carboxyethylsulfanyl)propanoic acid
Molecular Formula:C6H10O4S
Molecular Weight:178.206200 g/mol
Appearance:white crystalline powder
Melting Point:131-134 °C(lit.)
Density:1.362 g/cm3
Flash Point:201.3 °C
Enthalpy of Vaporization:72.57 kJ/mol
Boiling Point:409.3 °C at 760 mmHg
Vapour Pressure:7.61E-08 mmHg at 25 °C
Water Solubility:1.974e+005 mg/L at 25 °C
BRN:1210299
EINECS:203-841-3
Synonyms of BIS(2-CARBOXYETHYL) SULFIDE(111-17-1):
3,3'-THIODIPROPIONIC ACID;3,3-THIODIPROPIONIC ACID;3,3'-THIODIPROPIONIC ACID, POLYMER-BOUND;DIETHYL SULFIDE 2,2'-DICARBOXYLIC ACID;BIS(2-CARBOXYETHYL) SULFIDE;BETA,BETA'-THIODIPROPIONIC ACID;RARECHEM AL BO 0314;3,3'-Thiodipropionic acid
Categories of BIS(2-CARBOXYETHYL) SULFIDE(111-17-1):
Others;Supported Reagents;Supported Synthesis
3,3'-Thiodipropionic acid Uses
3,3'-Thiodipropionic acid Toxicity Data With Reference
| 1. | skn-rbt 500 mg/24H MLD | 28ZPAK Sbornik Vysledku Toxixologickeho Vysetreni Latek A Pripravku Marhold, J.V.,Institut Pro Vychovu Vedoucicn Pracovniku Chemickeho Prumyclu Praha,Czechoslovakia.: 1972,171. | ||
| 2. | eye-rbt 20 mg/24H MOD | 28ZPAK Sbornik Vysledku Toxixologickeho Vysetreni Latek A Pripravku Marhold, J.V.,Institut Pro Vychovu Vedoucicn Pracovniku Chemickeho Prumyclu Praha,Czechoslovakia.: 1972,171. | ||
| 3. | orl-rat LD50:3980 mg/kg | 28ZPAK Sbornik Vysledku Toxixologickeho Vysetreni Latek A Pripravku Marhold, J.V.,Institut Pro Vychovu Vedoucicn Pracovniku Chemickeho Prumyclu Praha,Czechoslovakia.: 1972,171. | ||
| 4. | ipr-rat LD50:500 mg/kg | AFREAW Advances in Food Research. 3 (1951),197. | ||
| 5. | orl-mus LD50:2000 mg/kg | AFREAW Advances in Food Research. 3 (1951),197. | ||
| 6. | ipr-mus LD50:250 mg/kg | AFREAW Advances in Food Research. 3 (1951),197. | ||
| 7. | ivn-mus LD50:175 mg/kg | AFREAW Advances in Food Research. 3 (1951),197. |
3,3'-Thiodipropionic acid Consensus Reports
3,3'-Thiodipropionic acid Safety Profile
Hazard Codes:Xi

Risk Statements:36/37/38-36
36/37/38:Irritating to eyes, respiratory system and skin
36:Irritating to the eyes
Safety Statements:26-36-37/39
26:In case of contact with eyes, rinse immediately with plenty of water and seek medical advice
36:Wear suitable protective clothing
37/39:Wear suitable gloves and eye/face protection
WGK Germany:2
RTECS:UF7990000
HS Code:29309070
A poison by intraperitoneal and intravenous routes. Moderately toxic by ingestion. A skin and eye irritant. When heated to decomposition it emits toxic fumes of SOx. See also SULFIDES.
3,3'-Thiodipropionic acid Specification
Store in a cool, dry place. Store in a tightly closed container.
Related Products
- 3,10-Diaminotricyclo(5.2.1.0(sup 2,6))decane
- 3,10-Dinitrophenanthrene
- 3-((10-ETHYL-11-(p-HYDROXYPHENYL)DIBENZ-(B,F)OXEPIN-3-YL)OXY)-1,2-PROPANEDIOL HYDRATE (4:1)
- 3-(1,1,2,2-Tetrafluoroethoxy)aniline
- 3-(1,1,2,2-Tetrafluoroethoxy)benzaldehyde
- 3-(1,1,2,2-Tetrafluoroethoxy)bromobenzene
- 3-(1,1,2,2-Tetrafluoroethoxy)toluene
- 3-[1,1'-Biphenyl]-4-yl-1,2,3,4-tetrahydro-1-naphthol
- 3,11-Dichloro-6,11-dihydro-6-methyldibenzo[c,f][1,2]thiazepine 5,5-dioxide
- 3-[(1,1-Dimethyl-2-hydroxyethyl)amino]-2-hydroxypropanesulfonicacid
- 1111732-94-5
- 111174-59-5
- 1111-74-6
- 111174-62-0
- 1111-78-0
- 111-18-2
- 11118-25-5
- 11118-57-3
- 1111-88-2
- 1111-89-3
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View