-
Name
3,4,5-Trimethoxyphenylboronic acid
- EINECS
- CAS No. 182163-96-8
- Article Data11
- CAS DataBase
- Density 1.21 g/cm3
- Solubility
- Melting Point >230 °C(lit.)
- Formula C9H13BO5
- Boiling Point 370.6 °C at 760 mmHg
- Molecular Weight 212.01
- Flash Point 177.9 °C
- Transport Information
- Appearance beige crystalline powder
- Safety 37/39-26-36
- Risk Codes 36/37/38-22-20/21/22
-
Molecular Structure
-
Hazard Symbols
 Xn,
Xn, Xi
Xi
- Synonyms Boronicacid, (3,4,5-trimethoxyphenyl)- (9CI);Boronicacid, B-(3,4,5-trimethoxyphenyl)-;
- PSA 68.15000
- LogP -0.60780
Synthetic route
-
-
2675-79-8
1-bromo-3,4,5-trimethoxybenzene

-
-
182163-96-8
3,4,5-trimethoxyphenylboronic Acid

| Conditions | Yield |
|---|---|
| Stage #1: 1-bromo-3,4,5-trimethoxybenzene With n-butyllithium In tetrahydrofuran at -78℃; Inert atmosphere; Stage #2: With Trimethyl borate In tetrahydrofuran at -78 - 20℃; Inert atmosphere; | 95% |
| Stage #1: 1-bromo-3,4,5-trimethoxybenzene With n-butyllithium In tetrahydrofuran; hexane at -85℃; for 1h; Stage #2: With Trimethyl borate In tetrahydrofuran at 20℃; for 18h; Further stages.; | 64% |
| Stage #1: 1-bromo-3,4,5-trimethoxybenzene With n-butyllithium In tetrahydrofuran at -78℃; for 0.5h; Stage #2: With Trimethyl borate In tetrahydrofuran at -78℃; for 0.5h; Stage #3: With hydrogenchloride In tetrahydrofuran; water |
-
-
121-43-7
Trimethyl borate

-
-
2675-79-8
1-bromo-3,4,5-trimethoxybenzene

-
-
182163-96-8
3,4,5-trimethoxyphenylboronic Acid

| Conditions | Yield |
|---|---|
| With Mg In tetrahydrofuran Mg (reflux), THF, B(OCH)3, bromobenzene-compound (-10°C) or n-butyllithium, B(OCH3)3, -78°C, THF; | 95% |
-
-
25245-29-8
5-iodo-1,2,3-trimethoxybenzene

-
-
182163-96-8
3,4,5-trimethoxyphenylboronic Acid

| Conditions | Yield |
|---|---|
| Stage #1: 5-iodo-1,2,3-trimethoxybenzene With tert.-butyl lithium In tetrahydrofuran at -78℃; Stage #2: With Trimethyl borate In tetrahydrofuran at -78 - 20℃; Further stages.; | 69% |
-
-
133095-91-7
3,4,5-trimethoxyphenylmagnesium bromide

-
-
182163-96-8
3,4,5-trimethoxyphenylboronic Acid

| Conditions | Yield |
|---|---|
| Stage #1: 3,4,5-trimethoxyphenylmagnesium bromide With Trimethyl borate In tetrahydrofuran at -78 - 20℃; Stage #2: With hydrogenchloride In water | 65% |
-
-
25245-29-8
5-iodo-1,2,3-trimethoxybenzene

-
-
121-43-7
Trimethyl borate

-
-
182163-96-8
3,4,5-trimethoxyphenylboronic Acid

| Conditions | Yield |
|---|---|
| Stage #1: 5-iodo-1,2,3-trimethoxybenzene With tert.-butyl lithium In tetrahydrofuran at -78℃; for 1h; Stage #2: Trimethyl borate In tetrahydrofuran at -78℃; for 1h; | |
| Stage #1: 5-iodo-1,2,3-trimethoxybenzene In tetrahydrofuran at -78℃; for 0.5h; Inert atmosphere; Stage #2: With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 1h; Inert atmosphere; Stage #3: Trimethyl borate In tetrahydrofuran; hexane at 20℃; for 14h; Inert atmosphere; |
-
-
24313-88-0
3,4,5-Trimethoxyaniline

-
-
182163-96-8
3,4,5-trimethoxyphenylboronic Acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: NaNO2; concd. H2SO4 / H2O / 5 - 10 °C 1.2: 70 percent / aq. CuBr / 0.5 h / Heating 2.1: nBuLi / tetrahydrofuran; hexane / 1 h / -85 °C 2.2: 64 percent / trimethylborate / tetrahydrofuran / 18 h / 20 °C View Scheme | |
| Multi-step reaction with 2 steps 1.1: sodium nitrite; potassium iodide / water; sulfuric acid / 5 h / 50 °C / Inert atmosphere 2.1: tetrahydrofuran / 0.5 h / -78 °C / Inert atmosphere 2.2: 1 h / -78 °C / Inert atmosphere 2.3: 14 h / 20 °C / Inert atmosphere View Scheme |
-
-
1016971-56-4
C27H33B3O12

-
-
182163-96-8
3,4,5-trimethoxyphenylboronic Acid

| Conditions | Yield |
|---|---|
| With water at 25℃; Equilibrium constant; |
-
-
1016971-65-5
C27H33B3O8

-
A
-
301699-39-8
(4-methoxy-3,5-dimethylphenyl)boronic acid

-
B
-
182163-96-8
3,4,5-trimethoxyphenylboronic Acid

| Conditions | Yield |
|---|---|
| With water at 25℃; Equilibrium constant; |
-
-
1016971-67-7
C27H33B3O10

-
A
-
301699-39-8
(4-methoxy-3,5-dimethylphenyl)boronic acid

-
B
-
182163-96-8
3,4,5-trimethoxyphenylboronic Acid

| Conditions | Yield |
|---|---|
| With water at 25℃; Equilibrium constant; |
-
-
1016971-69-9
C39H57B3O8

-
A
-
182163-96-8
3,4,5-trimethoxyphenylboronic Acid

-
B
-
233584-42-4
4-methoxy-3,5-di-tert-butylphenylboronic acid pinacol ester

| Conditions | Yield |
|---|---|
| With water at 25℃; Equilibrium constant; |
-
-
1016971-71-3
C33H45B3O10

-
A
-
182163-96-8
3,4,5-trimethoxyphenylboronic Acid

-
B
-
233584-42-4
4-methoxy-3,5-di-tert-butylphenylboronic acid pinacol ester

| Conditions | Yield |
|---|---|
| With water at 25℃; Equilibrium constant; |
-
-
1016971-54-2
C25H29B3O6

-
A
-
172975-69-8
3,5-dimethylphenyl boronic acid

-
B
-
182163-96-8
3,4,5-trimethoxyphenylboronic Acid

| Conditions | Yield |
|---|---|
| With water at 25℃; Equilibrium constant; |
-
-
2675-79-8
1-bromo-3,4,5-trimethoxybenzene

-
-
182163-96-8
3,4,5-trimethoxyphenylboronic Acid

| Conditions | Yield |
|---|---|
| Stage #1: 1-bromo-3,4,5-trimethoxybenzene With n-butyllithium In tetrahydrofuran at -78℃; Inert atmosphere; Stage #2: With Trimethyl borate In tetrahydrofuran at -78 - 20℃; Inert atmosphere; | 95% |
| Stage #1: 1-bromo-3,4,5-trimethoxybenzene With n-butyllithium In tetrahydrofuran; hexane at -85℃; for 1h; Stage #2: With Trimethyl borate In tetrahydrofuran at 20℃; for 18h; Further stages.; | 64% |
| Stage #1: 1-bromo-3,4,5-trimethoxybenzene With n-butyllithium In tetrahydrofuran at -78℃; for 0.5h; Stage #2: With Trimethyl borate In tetrahydrofuran at -78℃; for 0.5h; Stage #3: With hydrogenchloride In tetrahydrofuran; water |
-
-
121-43-7
Trimethyl borate

-
-
2675-79-8
1-bromo-3,4,5-trimethoxybenzene

-
-
182163-96-8
3,4,5-trimethoxyphenylboronic Acid

| Conditions | Yield |
|---|---|
| With Mg In tetrahydrofuran Mg (reflux), THF, B(OCH)3, bromobenzene-compound (-10°C) or n-butyllithium, B(OCH3)3, -78°C, THF; | 95% |
-
-
25245-29-8
5-iodo-1,2,3-trimethoxybenzene

-
-
182163-96-8
3,4,5-trimethoxyphenylboronic Acid

| Conditions | Yield |
|---|---|
| Stage #1: 5-iodo-1,2,3-trimethoxybenzene With tert.-butyl lithium In tetrahydrofuran at -78℃; Stage #2: With Trimethyl borate In tetrahydrofuran at -78 - 20℃; Further stages.; | 69% |
-
-
133095-91-7
3,4,5-trimethoxyphenylmagnesium bromide

-
-
182163-96-8
3,4,5-trimethoxyphenylboronic Acid

| Conditions | Yield |
|---|---|
| Stage #1: 3,4,5-trimethoxyphenylmagnesium bromide With Trimethyl borate In tetrahydrofuran at -78 - 20℃; Stage #2: With hydrogenchloride In water | 65% |
-
-
25245-29-8
5-iodo-1,2,3-trimethoxybenzene

-
-
121-43-7
Trimethyl borate

-
-
182163-96-8
3,4,5-trimethoxyphenylboronic Acid

| Conditions | Yield |
|---|---|
| Stage #1: 5-iodo-1,2,3-trimethoxybenzene With tert.-butyl lithium In tetrahydrofuran at -78℃; for 1h; Stage #2: Trimethyl borate In tetrahydrofuran at -78℃; for 1h; | |
| Stage #1: 5-iodo-1,2,3-trimethoxybenzene In tetrahydrofuran at -78℃; for 0.5h; Inert atmosphere; Stage #2: With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 1h; Inert atmosphere; Stage #3: Trimethyl borate In tetrahydrofuran; hexane at 20℃; for 14h; Inert atmosphere; |
-
-
24313-88-0
3,4,5-Trimethoxyaniline

-
-
182163-96-8
3,4,5-trimethoxyphenylboronic Acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: NaNO2; concd. H2SO4 / H2O / 5 - 10 °C 1.2: 70 percent / aq. CuBr / 0.5 h / Heating 2.1: nBuLi / tetrahydrofuran; hexane / 1 h / -85 °C 2.2: 64 percent / trimethylborate / tetrahydrofuran / 18 h / 20 °C View Scheme | |
| Multi-step reaction with 2 steps 1.1: sodium nitrite; potassium iodide / water; sulfuric acid / 5 h / 50 °C / Inert atmosphere 2.1: tetrahydrofuran / 0.5 h / -78 °C / Inert atmosphere 2.2: 1 h / -78 °C / Inert atmosphere 2.3: 14 h / 20 °C / Inert atmosphere View Scheme |
-
-
1016971-56-4
C27H33B3O12

-
-
182163-96-8
3,4,5-trimethoxyphenylboronic Acid

| Conditions | Yield |
|---|---|
| With water at 25℃; Equilibrium constant; |
-
-
1016971-65-5
C27H33B3O8

-
A
-
301699-39-8
(4-methoxy-3,5-dimethylphenyl)boronic acid

-
B
-
182163-96-8
3,4,5-trimethoxyphenylboronic Acid

| Conditions | Yield |
|---|---|
| With water at 25℃; Equilibrium constant; |
-
-
1016971-67-7
C27H33B3O10

-
A
-
301699-39-8
(4-methoxy-3,5-dimethylphenyl)boronic acid

-
B
-
182163-96-8
3,4,5-trimethoxyphenylboronic Acid

| Conditions | Yield |
|---|---|
| With water at 25℃; Equilibrium constant; |
-
-
1016971-69-9
C39H57B3O8

-
A
-
182163-96-8
3,4,5-trimethoxyphenylboronic Acid

-
B
-
233584-42-4
4-methoxy-3,5-di-tert-butylphenylboronic acid pinacol ester

| Conditions | Yield |
|---|---|
| With water at 25℃; Equilibrium constant; |
-
-
1016971-71-3
C33H45B3O10

-
A
-
182163-96-8
3,4,5-trimethoxyphenylboronic Acid

-
B
-
233584-42-4
4-methoxy-3,5-di-tert-butylphenylboronic acid pinacol ester

| Conditions | Yield |
|---|---|
| With water at 25℃; Equilibrium constant; |
-
-
1016971-54-2
C25H29B3O6

-
A
-
172975-69-8
3,5-dimethylphenyl boronic acid

-
B
-
182163-96-8
3,4,5-trimethoxyphenylboronic Acid

| Conditions | Yield |
|---|---|
| With water at 25℃; Equilibrium constant; |
-
-
1016971-55-3
C26H31B3O9

-
A
-
172975-69-8
3,5-dimethylphenyl boronic acid

-
B
-
182163-96-8
3,4,5-trimethoxyphenylboronic Acid

| Conditions | Yield |
|---|---|
| With water at 25℃; Equilibrium constant; |
-
-
5419-55-6
Triisopropyl borate

-
-
2675-79-8
1-bromo-3,4,5-trimethoxybenzene

-
-
7732-18-5
water

-
-
182163-96-8
3,4,5-trimethoxyphenylboronic Acid

| Conditions | Yield |
|---|---|
| Stage #1: 1-bromo-3,4,5-trimethoxybenzene With n-butyllithium In tetrahydrofuran at -78℃; Inert atmosphere; Stage #2: Triisopropyl borate In tetrahydrofuran at -78 - 0℃; Inert atmosphere; Stage #3: water With hydrogenchloride In tetrahydrofuran Inert atmosphere; |

-
-
182163-96-8
3,4,5-trimethoxyphenylboronic Acid

-
-
570402-53-8
1-[2-(4-methoxy-phenyl)-ethyl]-3,4-bis-trifluoromethanesulfonyloxy-1H-pyrrole-2,5-dicarboxylic acid dimethyl ester

-
-
220231-03-8
dimethyl 3,4-bis(3,4,5-trimethoxyphenyl)-1-<2-(4-methoxyphenyl)ethyl>pyrrole-2,5-dicarboxylate

| Conditions | Yield |
|---|---|
| With sodium carbonate; tetrakis(triphenylphosphine) palladium(0) In tetrahydrofuran for 20h; Suzuki cross-coupling; Heating; | 100% |

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); caesium carbonate In ethanol; N,N-dimethyl-formamide at 120℃; for 0.333333h; Suzuki coupling; Microwave irradiation; | 100% |
-
-
1184926-29-1
2-bromo-5-(2-trimethylsilanyl-ethoxymethyl)-5H-pyrrolo[2,3-b]pyrazine

-
-
1334675-00-1
(R)-2-(3-cyano-azetidin-1-yl)-1-methyl-2-oxo-ethyl-ammonium trifluoroacetate

-
-
182163-96-8
3,4,5-trimethoxyphenylboronic Acid

-
-
1334675-29-4
2-(3,4,5-trimethoxyphenyl)-5-(2-trimethylsilanylethoxymethyl)-5H-pyrrolo[2,3-b]pyrazine-7-carboxylic acid [(R)-2-(3-cyanoazetidin-1-yl)-1-methyl-2-oxoethyl]amide

| Conditions | Yield |
|---|---|
| Stage #1: 2-bromo-5-(2-trimethylsilanyl-ethoxymethyl)-5H-pyrrolo[2,3-b]pyrazine With Vilsmeier reagent In chloroform at 20℃; for 8h; Reflux; Inert atmosphere; Stage #2: With sodium hydrogencarbonate In chloroform Stage #3: (R)-2-(3-cyano-azetidin-1-yl)-1-methyl-2-oxo-ethyl-ammonium trifluoroacetate; 3,4,5-trimethoxyphenylboronic Acid | 100% |

-
-
182163-96-8
3,4,5-trimethoxyphenylboronic Acid

-
-
583-53-9
1,2-dibromobenzene

| Conditions | Yield |
|---|---|
| With palladium diacetate; potassium carbonate In ethanol; N,N-dimethyl acetamide at 20℃; for 3h; Suzuki Coupling; | 100% |
-
-
610-94-6
2-bromobenzoic acid methyl ester

-
-
182163-96-8
3,4,5-trimethoxyphenylboronic Acid

-
-
928847-09-0
3',4',5'-trimethoxybiphenyl-2-carboxylic acid methyl ester

| Conditions | Yield |
|---|---|
| With sodium carbonate; tetrakis(triphenylphosphine) palladium(0) In water; toluene at 100℃; | 99% |
| With sodium carbonate; tetrakis(triphenylphosphine) palladium(0) In toluene at 100℃; Suzuki reaction; | 45% |
-
-
1033610-26-2
C15H12INO3

-
-
182163-96-8
3,4,5-trimethoxyphenylboronic Acid

-
-
1033609-97-0
6-methoxy-2-(4-methoxyphenyl)-3-(3,4,5-trimethoxyphenyl)furo[2,3-b]pyridine

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate; tetrakis(triphenylphosphine) palladium(0) In 1,4-dioxane; water for 0.05h; Suzuki Coupling; Heating / reflux; | 99% |
-
-
1055900-77-0
3-chloro-2-(4-methylphenyl)-5-phenylfuran

-
-
182163-96-8
3,4,5-trimethoxyphenylboronic Acid

-
-
1055900-91-8
C26H24O4

| Conditions | Yield |
|---|---|
| With potassium fluoride; tris(dibenzylideneacetone)dipalladium (0); tri-tert-butyl phosphine at 120℃; for 10h; Suzuki -Miyaura coupling; | 99% |
-
-
182163-96-8
3,4,5-trimethoxyphenylboronic Acid

-
-
513-42-8
3-hydroxy-2-methyl-1-propene

| Conditions | Yield |
|---|---|
| With palladium diacetate; caesium carbonate; 4,5-bis(diphenylphos4,5-bis(diphenylphosphino)-9,9-dimethylxanthenephino)-9,9-dimethylxanthene at 100℃; for 12h; Temperature; Time; Suzuki-Miyaura Coupling; | 99% |
| With palladium diacetate; caesium carbonate; 4,5-bis(diphenylphos4,5-bis(diphenylphosphino)-9,9-dimethylxanthenephino)-9,9-dimethylxanthene at 120 - 140℃; Microwave irradiation; |
-
-
1309980-37-7
diethyl 1-[(4-methylbenzenesulfonyl)oxy]ethene-alpha-phosphonate

-
-
182163-96-8
3,4,5-trimethoxyphenylboronic Acid

| Conditions | Yield |
|---|---|
| With dicyclohexyl-(2',6'-dimethoxybiphenyl-2-yl)-phosphane; palladium diacetate; caesium carbonate In toluene at 60℃; Suzuki Coupling; Schlenk technique; Inert atmosphere; | 99% |
-
-
182163-96-8
3,4,5-trimethoxyphenylboronic Acid

-
-
25245-29-8
5-iodo-1,2,3-trimethoxybenzene

| Conditions | Yield |
|---|---|
| With N-iodo-succinimide; potassium acetate In acetonitrile at 50℃; for 4h; | 99% |
-
-
586-78-7
para-nitrophenyl bromide

-
-
182163-96-8
3,4,5-trimethoxyphenylboronic Acid

| Conditions | Yield |
|---|---|
| With tetra-butylammonium acetate; Pd EnCat-30TM In ethanol at 120℃; for 0.166667h; Suzuki cross-coupling; microwave irradiation; | 98% |
-
-
580-13-2
2-bromonaphthalene

-
-
182163-96-8
3,4,5-trimethoxyphenylboronic Acid

| Conditions | Yield |
|---|---|
| With [1,3-bis(2,6-diisopropylphenyl)imidazol-2-ylidene](3-chloropyridyl)palladium(ll) dichloride; potassium carbonate at 110℃; for 0.166667h; Suzuki-Miyaura coupling; Microwave irradiation; Neat (no solvent); | 98% |
| With tetra-butylammonium acetate; Pd EnCat-30TM In ethanol at 120℃; for 0.166667h; Suzuki cross-coupling; microwave irradiation; |

| Conditions | Yield |
|---|---|
| With potassium phosphate; tetrakis(triphenylphosphine) palladium(0) In 1,4-dioxane; water at 90℃; Suzuki-Miyaura Coupling; Inert atmosphere; | 98% |
-
-
49669-13-8
1-(6-bromopyridin-2-yl)-ethanone

-
-
182163-96-8
3,4,5-trimethoxyphenylboronic Acid

| Conditions | Yield |
|---|---|
| With potassium phosphate; palladium diacetate; triphenylphosphine In 1,4-dioxane; water at 110℃; Suzuki-Miyaura Coupling; Sealed tube; Inert atmosphere; | 98% |

| Conditions | Yield |
|---|---|
| Stage #1: 2-iodolselenophene; 3,4,5-trimethoxyphenylboronic Acid With palladium diacetate In 1,2-dichloro-ethane at 20℃; for 0.25h; Inert atmosphere; Stage #2: With potassium carbonate In water; 1,2-dichloro-ethane at 90℃; for 1h; | 98% |

| Conditions | Yield |
|---|---|
| Stage #1: 3,4,5-trimethoxyphenylboronic Acid With sodium azide; copper(II) sulfate In methanol Stage #2: C18H20O3 With sodium L-ascorbate In methanol; water at 20℃; | 97% |

-
-
182163-96-8
3,4,5-trimethoxyphenylboronic Acid

| Conditions | Yield |
|---|---|
| With NHC-Pd(II)-Im; potassium hydroxide In ethanol at 20℃; for 24h; Temperature; Suzuki-Miyaura Coupling; Schlenk technique; | 97% |
-
-
182163-96-8
3,4,5-trimethoxyphenylboronic Acid

-
-
566915-55-7
3-benzyloxy-3'-bromo-4-methoxy-biphenyl

-
-
847063-38-1
3-benzyloxy-4,3'',4'',5''-tetramethoxy-[1,1',3',1'']terphenyl

| Conditions | Yield |
|---|---|
| Stage #1: 3-benzyloxy-3'-bromo-4-methoxy-biphenyl With tetrakis(triphenylphosphine) palladium(0) In toluene at 20℃; for 0.166667h; Stage #2: 3,4,5-trimethoxyphenylboronic Acid With sodium carbonate In ethanol; toluene for 3h; Suzuki coupling; Heating; | 96% |
| With sodium carbonate; tetrakis(triphenylphosphine) palladium(0) In ethanol; toluene Suzuki cross-coupling reaction; |
-
-
946007-93-8
(3R,4S,5R)-5-(6-bromo-7-methoxy-benzo[1,3]dioxol-5-yl)-3,4-dimethyl-dihydro-furan-2-one

-
-
182163-96-8
3,4,5-trimethoxyphenylboronic Acid

-
-
139068-10-3
eupomatilone 4

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate; tetrakis(triphenylphosphine) palladium(0) In 1,2-dimethoxyethane; water for 18h; Suzuki coupling; Heating; | 96% |
-
-
1144503-16-1
dimethyl N-benzyl-3,4-bis(trifluoromethanesulfonyloxy)pyrrole-2,5-dicarboxylate

-
-
182163-96-8
3,4,5-trimethoxyphenylboronic Acid

-
-
1144503-24-1
dimethyl N-benzyl-3,4-bis(3,4,5-trimethoxyphenyl)pyrrole-2,5-dicarboxylate

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); sodium carbonate In tetrahydrofuran; water at 20℃; Suzuki-Miyaura coupling; Reflux; Inert atmosphere; | 96% |

-
-
182163-96-8
3,4,5-trimethoxyphenylboronic Acid

| Conditions | Yield |
|---|---|
| With dichloro(1,1'-bis(diphenylphosphanyl)ferrocene)palladium(II)*CH2Cl2; sodium carbonate In 1,2-dimethoxyethane; water at 105℃; for 2h; Suzuki coupling; Microwave irradiation; Inert atmosphere; | 96% |
-
-
5751-20-2
2-Methylthiouracil

-
-
182163-96-8
3,4,5-trimethoxyphenylboronic Acid

-
-
1388177-39-6
2-(methylsulfanyl)-4-(3,4,5-trimethoxyphenyl)pyrimidine

| Conditions | Yield |
|---|---|
| Stage #1: 2-Methylthiouracil With triethylamine; bromo-tris(1-pyrrolidinyl)phosphonium hexafluorophosphate In 1,4-dioxane at 20℃; for 2h; Inert atmosphere; Sealed tube; Stage #2: 3,4,5-trimethoxyphenylboronic Acid With bis-triphenylphosphine-palladium(II) chloride; sodium carbonate In 1,4-dioxane; water at 100℃; for 4h; Suzuki coupling; Inert atmosphere; Sealed tube; chemoselective reaction; | 96% |
-
-
1388177-38-5
4-(benzo[d][1,3]dioxol-5-yl)-2-(methylsulfanyl)pyrimidine

-
-
182163-96-8
3,4,5-trimethoxyphenylboronic Acid

-
-
1388177-59-0
4-(benzo[d][1,3]dioxol-5-yl)-2-(3,4,5-trimethoxyphenyl)pyrimidine

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); copper(I) thiophene-2-carboxylate In tetrahydrofuran at 100℃; for 1h; Liebeskind-Srogl reaction; Inert atmosphere; Microwave irradiation; regioselective reaction; | 96% |

-
-
182163-96-8
3,4,5-trimethoxyphenylboronic Acid

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); potassium carbonate In 1,4-dioxane; water at 95℃; for 16h; Suzuki Coupling; Inert atmosphere; | 96% |

-
-
182163-96-8
3,4,5-trimethoxyphenylboronic Acid

| Conditions | Yield |
|---|---|
| Stage #1: 3-bromo-1-tert-butyloxycarbonyl-5-methoxy-2-indolyl diphenyl phosphate; 3,4,5-trimethoxyphenylboronic Acid With palladium diacetate; ruphos In toluene at 20℃; for 0.0833333h; Suzuki Coupling; Inert atmosphere; Stage #2: With potassium phosphate In water; toluene at 60℃; for 20h; Suzuki Coupling; Inert atmosphere; regioselective reaction; | 96% |
-
-
119072-55-8, 7188-38-7
tert-butylisonitrile

-
-
182163-96-8
3,4,5-trimethoxyphenylboronic Acid

-
-
118-41-2
Eudesmic acid

| Conditions | Yield |
|---|---|
| Stage #1: tert-butylisonitrile; 3,4,5-trimethoxyphenylboronic Acid With copper diacetate; palladium diacetate In N,N-dimethyl-formamide at 100℃; for 24h; Molecular sieve; Sealed tube; Stage #2: With water In N,N-dimethyl-formamide Molecular sieve; Sealed tube; | 96% |
-
-
93683-65-9
6-chloro-3-nitropicolinonitrile

-
-
182163-96-8
3,4,5-trimethoxyphenylboronic Acid

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); potassium carbonate In 1,4-dioxane; water at 95℃; Suzuki Coupling; Inert atmosphere; | 96% |
3,4,5-Trimethoxyphenylboronic acid Chemical Properties
Molecular structure of 3,4,5-Trimethoxyphenylboronic acid (CAS NO.182163-96-8):
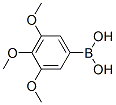
IUPAC Name: (3,4,5-Trimethoxyphenyl)boronic acid
Molecular Formula: C9H13BO5
Molecular Weight: 212.01 g/mol
Density: 1.21 g/cm3
Melting Point: >230 °C(lit.)
Boiling Point: 370.6 °C at 760 mmHg
Flash Point: 177.9 °C
Index of Refraction: 1.513
Molar Refractivity: 52.38 cm3
Molar Volume: 174 cm3
Surface Tension: 43.6 dyne/cm
Enthalpy of Vaporization: 65.15 kJ/mol
Vapour Pressure: 3.79E-06 mmHg at 25 °C
Storage temp.: refrigerator (+4 °C)
H-Bond Donor: 2
H-Bond Acceptor: 5
Rotatable Bond Count: 4
Exact Mass: 212.085604
MonoIsotopic Mass: 212.085604
Topological Polar Surface Area: 68.2
Heavy Atom Count: 15
Canonical SMILES: B(C1=CC(=C(C(=C1)OC)OC)OC)(O)O
InChI: InChI=1S/C9H13BO5/c1-13-7-4-6(10(11)12)5-8(14-2)9(7)15-3/h4-5,11-12H,1-3H3
InChIKey: RULQUTYJXDLRFL-UHFFFAOYSA-N
Product Categories: Substituted Boronic Acids; Alkoxy; Aryl; Organoborons; Boronic acid; Boronic acid series; Boronic Acids; Boronic Acids and Derivatives; Boronic Acids
3,4,5-Trimethoxyphenylboronic acid Uses
3,4,5-Trimethoxyphenylboronic acid (CAS NO.182163-96-8) is commonly used as fine chemical material and pharmaceutical intermediate.
3,4,5-Trimethoxyphenylboronic acid Safety Profile
Hazard Codes:  Xn,
Xn,  Xi
Xi
Risk Statements: 36/37/38-22-20/21/22
R36/37/38:Irritating to eyes, respiratory system and skin.
R22:Harmful if swallowed.
R20/21/22:Harmful by inhalation, in contact with skin and if swallowed.
Safety Statements: 37/39-26-36
S37/39:Wear suitable gloves and eye/face protection.
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S36:Wear suitable protective clothing.
WGK Germany: 3
Hazard Note: Irritant
3,4,5-Trimethoxyphenylboronic acid Specification
3,4,5-Trimethoxyphenylboronic acid (CAS NO.182163-96-8) is also named as Boronicacid, B-(3,4,5-trimethoxyphenyl)- ; Boronicacid, (3,4,5-trimethoxyphenyl)- (9CI) ; 3,4,5-Trimethoxybenzeneboronic acid . 3,4,5-Trimethoxyphenylboronic acid (CAS NO.182163-96-8) is beige crystalline powder.
Related Products
- 3,10-Diaminotricyclo(5.2.1.0(sup 2,6))decane
- 3,10-Dinitrophenanthrene
- 3-((10-ETHYL-11-(p-HYDROXYPHENYL)DIBENZ-(B,F)OXEPIN-3-YL)OXY)-1,2-PROPANEDIOL HYDRATE (4:1)
- 3-(1,1,2,2-Tetrafluoroethoxy)aniline
- 3-(1,1,2,2-Tetrafluoroethoxy)benzaldehyde
- 3-(1,1,2,2-Tetrafluoroethoxy)bromobenzene
- 3-(1,1,2,2-Tetrafluoroethoxy)toluene
- 3-[1,1'-Biphenyl]-4-yl-1,2,3,4-tetrahydro-1-naphthol
- 3,11-Dichloro-6,11-dihydro-6-methyldibenzo[c,f][1,2]thiazepine 5,5-dioxide
- 3-[(1,1-Dimethyl-2-hydroxyethyl)amino]-2-hydroxypropanesulfonicacid
- 182165-34-0
- 182169-63-7
- 18217-00-0
- 18218-04-7
- 182181-19-7
- 182181-38-0
- 182187-39-9
- 182198-35-2
- 182198-89-6
- 1822-00-0
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View