-
Name
3-Aminopyridine
- EINECS 207-322-2
- CAS No. 462-08-8
- Article Data198
- CAS DataBase
- Density 1.107 g/cm3
- Solubility It is soluble in water as well as soluble in alcohol, benzene.
- Melting Point 60-63 ºC(lit.)
- Formula C5H6N2
- Boiling Point 251 ºC at 760 mmHg
- Molecular Weight 94.116
- Flash Point 126 ºC
- Transport Information UN 2671 6.1/PG 2
- Appearance white to light yellow crystal powder
- Safety 36/37/39-45-28-26
- Risk Codes 23/24/25-33-36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi,
Xi, T
T
- Synonyms pyridin-3-amine;Amino-3 pyridine;3-Pyridinamine;;3-Amino Pyridine;3-Pyridylamine;Pyridine, 3-amino-;beta-Aminopyridine;
- PSA 38.91000
- LogP 1.24500
Synthetic route

| Conditions | Yield |
|---|---|
| With hydrogen In ethyl acetate at 20℃; under 7600.51 Torr; for 6h; Autoclave; | 99% |
| With 0.2C27H36N2*Pt; hydrogen In tetrahydrofuran at 60℃; under 3000.3 Torr; for 5h; chemoselective reaction; | 99% |
| With hydrogen In ethyl acetate under 760.051 Torr; for 2h; Heating; Flow reactor; Green chemistry; | 99% |

| Conditions | Yield |
|---|---|
| With cyclohexa-1,4-diene; 5%-palladium/activated carbon In methanol at 120℃; for 0.0833333h; Microwave irradiation; | 99% |
| With palladium 10% on activated carbon; hydrazine hydrate In methanol at 120℃; for 0.25h; Microwave irradiation; | 89% |
| With ethanol; palladium Hydrogenation; |

| Conditions | Yield |
|---|---|
| With cyclohexa-1,4-diene; 5%-palladium/activated carbon In methanol at 120℃; for 0.0833333h; Microwave irradiation; | 99% |
| With hydrazine hydrate In ethanol at 70℃; for 4h; chemoselective reaction; | 95% |
| With palladium 10% on activated carbon; hydrazine hydrate In methanol at 120℃; for 0.25h; Microwave irradiation; | 84% |
| With palladium 10% on activated carbon; hydrogen; sodium hydrogencarbonate In methanol at 20℃; under 760.051 Torr; for 2h; Sealed tube; chemoselective reaction; | 100 %Spectr. |

| Conditions | Yield |
|---|---|
| With sodium hypochlorite In water at 0 - 5℃; | 94.1% |
| With sodium hypochlorite at 5 - 10℃; Temperature; | 92.3% |
| With sodium hydroxide; bromine r.t., 30 min.; then 65-75 deg C, 2 h; | 62% |

| Conditions | Yield |
|---|---|
| With titanium tetrachloride; tin(ll) chloride In diethyl ether for 0.5h; Ambient temperature; | 94% |

| Conditions | Yield |
|---|---|
| With copper(ll) sulfate pentahydrate; ammonium hydroxide In PEG1000-DIL; methyl cyclohexane at 60℃; for 6h; | 94% |
| With ammonium hydroxide; potassium phosphate In dimethyl sulfoxide at 80℃; UV-irradiation; | 94% |
| With C46H71Cl3N2Pd; ammonia; sodium t-butanolate In 1,4-dioxane at 100℃; for 16h; Inert atmosphere; Schlenk technique; | 88% |

| Conditions | Yield |
|---|---|
| With iron(III) oxide; sodium hydroxide; copper(l) iodide; ammonia In ethanol; water at 90℃; for 16h; | 94% |
| With [N,N'-bis(5-sulfonatosalicylidene)-1,2-diaminoethane]copper disodium salt; ammonia; sodium hydroxide In water at 120℃; for 12h; sealed tube; | 94% |
| With copper(I) oxide; ammonium hydroxide; C17H14N2O3; potassium hydroxide In ethanol at 60℃; for 24h; Schlenk technique; Inert atmosphere; | 93% |

| Conditions | Yield |
|---|---|
| With sodium hypochlorite; sodium hydroxide In water at 5 - 20℃; Temperature; | 93.8% |
| Multi-step reaction with 2 steps 1: manganese(IV) oxide / ethanol; water / 6 h / 85 °C 2: sodium hydroxide; sodium hypochlorite / 6 h / 90 °C View Scheme |

| Conditions | Yield |
|---|---|
| With [N,N'-bis(5-sulfonatosalicylidene)-1,2-diaminoethane]copper disodium salt; ammonia; sodium hydroxide In water at 120℃; for 12h; sealed tube; | 93% |
| Stage #1: 3-Bromopyridine With copper(l) iodide; potassium carbonate; L-proline In dimethyl sulfoxide Inert atmosphere; Stage #2: With ammonium hydroxide In dimethyl sulfoxide at 90℃; for 12h; Inert atmosphere; | 93% |
| With oxalic acid hydrazide; ammonium hydroxide; tetrabutylammomium bromide; potassium carbonate; 2,5-hexanedione; copper(II) oxide In water at 90℃; for 1.33333h; | 90% |

| Conditions | Yield |
|---|---|
| Stage #1: N-(pyridin-3-yl)acetamide With Schwartz's reagent In tetrahydrofuran at 20℃; for 0.05h; Inert atmosphere; Stage #2: With water In tetrahydrofuran Inert atmosphere; | 89% |

| Conditions | Yield |
|---|---|
| With hydrogen In water; toluene at 50℃; under 4500.45 Torr; for 5h; Reagent/catalyst; | A 11.5% B 88.5% |
| With 1% platinum on charcoal; hydrogen; sodium carbonate In water; toluene at 50℃; under 4500.45 Torr; for 5h; Catalytic behavior; Reagent/catalyst; | |
| With hydrogen; NE-01M02; sodium carbonate In water; toluene at 50℃; under 4500.45 Torr; for 5h; Catalytic behavior; Reagent/catalyst; | |
| With platinum on activated charcoal; hydrogen; sodium carbonate In water; toluene at 50℃; under 4500.45 Torr; for 5h; Reagent/catalyst; |
-
-
10296-29-4
3-azidopyridine

-
-
462-08-8
pyridin-3-ylamine

| Conditions | Yield |
|---|---|
| With iron In water at 20℃; Inert atmosphere; | 88% |
-
-
63458-55-9
N-monochloronicotinamide

-
-
462-08-8
pyridin-3-ylamine

| Conditions | Yield |
|---|---|
| Stage #1: N-monochloronicotinamide In water at 90℃; for 1.5h; Stage #2: With hydrogenchloride In water at 15 - 50℃; Product distribution / selectivity; | 86% |

-
-
462-08-8
pyridin-3-ylamine

| Conditions | Yield |
|---|---|
| With ammonium sulfate; bis(1,5-cyclooctadiene)nickel (0); sodium t-butanolate at 100 - 110℃; for 12h; | 85% |
-
-
170839-31-3
benzyl N-(pyridin-3-yl)carbamate

-
-
462-08-8
pyridin-3-ylamine

| Conditions | Yield |
|---|---|
| With methanol; sodium tetrahydroborate; nickel(II) chloride hexahydrate at 20℃; for 0.25h; chemoselective reaction; | 78% |
| With hydrogenchloride; water |

| Conditions | Yield |
|---|---|
| With copper(ll) sulfate pentahydrate; potassium phosphate tribasic heptahydrate; water; Sucrose In water at 90℃; for 15h; Catalytic behavior; Green chemistry; | 75% |
-
-
1462-86-8
3-amino-pyridine-2-carboxylic acid

-
-
87-13-8
diethyl 2-ethoxymethylenemalonate

-
A
-
462-08-8
pyridin-3-ylamine

-
B
-
14029-71-1
2-(pyridin-3-yl)aminomethylene-malonic acid diethyl ester

| Conditions | Yield |
|---|---|
| at 140 - 150℃; for 12h; | A n/a B 63% |


-
-
462-08-8
pyridin-3-ylamine

| Conditions | Yield |
|---|---|
| With methanesulfonic acid; palladium on activated charcoal In ethanol Heating; | 63% |
-
-
25604-13-1
succinic acid 3-pyridylmonoamide

-
-
462-08-8
pyridin-3-ylamine

| Conditions | Yield |
|---|---|
| With ammonium peroxydisulfate; silver nitrate; trifluoroacetic acid In water at 70℃; for 6h; | 60% |
-
-
18364-47-1
N-methyl-N-(3-pyridyl)amine

-
-
462-08-8
pyridin-3-ylamine

| Conditions | Yield |
|---|---|
| With piperidine; dichloro(dimethylglyoxime)(dimethylglyoximato)cobalt(III); (4s,6s)-2,4,5,6-tetra(9H-carbazol-9-yl)isophthalonitrile In acetonitrile at -78℃; for 72h; Reagent/catalyst; Sealed tube; Inert atmosphere; Irradiation; | 60% |
-
-
56700-70-0
3-(tert-butoxycarbonylamino)pyridine

-
-
462-08-8
pyridin-3-ylamine

| Conditions | Yield |
|---|---|
| With silica gel In dichloromethane for 0.05h; Irradiation; | 56% |
-
-
4122-56-9
methyl o-formylbenzoate

-
-
157562-24-8
3-aminopicolinic acid trimethylsilyl ester

-
A
-
462-08-8
pyridin-3-ylamine

| Conditions | Yield |
|---|---|
| In xylene at 140 - 145℃; for 18h; | A 49% B 50% |

-
-
1462-86-8
3-amino-pyridine-2-carboxylic acid

-
-
611-10-9
2-ethoxycarbonyl-1-cyclopentanone

-
A
-
462-08-8
pyridin-3-ylamine

-
B
-
61319-84-4
ethyl 2-(3-pyridylamino)-1-cyclopentene-1-carboxylate

| Conditions | Yield |
|---|---|
| at 150℃; for 20h; | A n/a B 50% C 13% D 36% |

-
-
157562-25-9
3-(3-oxobutylamido)picolinic acid

-
A
-
462-08-8
pyridin-3-ylamine

-
B
-
39642-60-9
N,N′-bis(m-pyridyl)urea

| Conditions | Yield |
|---|---|
| In 1,3,5-trimethyl-benzene at 160℃; for 15h; | A 47% B 47% |

| Conditions | Yield |
|---|---|
| With ammonium peroxydisulfate; silver nitrate; trifluoroacetic acid In water at 70℃; for 6h; | A 40% B 45% |

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate; 5%-palladium/activated carbon; hydrazine hydrate; lithium hydroxide In 1,4-dioxane at 170℃; for 16h; Molecular sieve; Inert atmosphere; | 45% |

| Conditions | Yield |
|---|---|
| With copper(l) iodide; cesium acetate In dimethyl sulfoxide at 90℃; for 24h; Inert atmosphere; | A 17% B 42% |


| Conditions | Yield |
|---|---|
| Stage #1: pyridin-3-ylamine With sulfuric acid at 20℃; Cooling with ice; Stage #2: With sodium nitrite In water Reflux; | 100% |
| With sulfuric acid Diazotization; | |
| With sulfuric acid; water; sodium nitrite at 0℃; Erwaermen des Reaktionsgemisches auf 50grad; | |
| With sulfuric acid; water; sodium nitrite at 0℃; Erwaermen des Reaktionsgemisches auf 80grad.; |
-
-
462-08-8
pyridin-3-ylamine

-
-
116-11-0
2-Methoxypropene

-
-
32405-72-4
3-Pyridinyl(1-methylethylidene)amine

| Conditions | Yield |
|---|---|
| With pyridinium p-toluenesulfonate; triethylamine In chloroform at 100℃; for 9h; | 100% |
-
-
462-08-8
pyridin-3-ylamine

-
-
42166-50-7
3-hydrazinopyridine

| Conditions | Yield |
|---|---|
| Stage #1: pyridin-3-ylamine With hydrogenchloride; sodium nitrite In water at 0 - 5℃; for 0.583333h; Stage #2: With hydrogenchloride; tin(ll) chloride In water at 0 - 10℃; for 1.5h; | 100% |
| Stage #1: pyridin-3-ylamine With hydrogenchloride; sodium nitrite In water at 5℃; for 0.75h; Stage #2: With hydrogenchloride; tin(ll) chloride In water at 0 - 10℃; for 1.5h; Stage #3: With sodium hydroxide In water at 0 - 10℃; Product distribution / selectivity; | 83% |
| Stage #1: pyridin-3-ylamine With hydrogenchloride; sodium nitrite In water for 0.75h; Stage #2: With tin(ll) chloride In water at 5℃; for 1.5h; Stage #3: With sodium hydroxide In water | 83% |
-
-
462-08-8
pyridin-3-ylamine

| Conditions | Yield |
|---|---|
| With fluorosilicic acid In methanol | 100% |

| Conditions | Yield |
|---|---|
| at 200℃; for 0.05h; | 100% |
-
-
462-08-8
pyridin-3-ylamine

-
-
874298-19-8
4-(3-pyridyl)ureido-phenylboronic acid pinacol ester

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; toluene at 20℃; for 2.5h; | 100% |
| In tetrahydrofuran at 20℃; for 6h; | 94.3% |
| In tetrahydrofuran at 20℃; for 6h; | 94.3% |
| In tetrahydrofuran; toluene at 20℃; for 16h; | 93% |
| In 1,2-dimethoxyethane |
-
-
462-08-8
pyridin-3-ylamine

-
-
17823-69-7
2-cyano-3,3-bis(methylthio)acrylamide

-
-
1456732-10-7
2-cyano-3-(methylthio)-3-(pyridin-3-ylamino)acrylamide

| Conditions | Yield |
|---|---|
| In ethanol at 75℃; for 18h; | 100% |
-
-
462-08-8
pyridin-3-ylamine

-
-
24801-88-5
3-(triethoxypropyl) isocyanate

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; for 48h; | 100% |

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; | 100% |
-
-
462-08-8
pyridin-3-ylamine

-
-
1238056-80-8
pentafluorophenyl indirubin-5-carboxylate

-
-
1238056-92-2
5-[N-(pyridin-3-yl)aminocarbonyl]indirubin

| Conditions | Yield |
|---|---|
| With dmap In 1,4-dioxane Inert atmosphere; Reflux; | 99.9% |

| Conditions | Yield |
|---|---|
| In acetonitrile at 100℃; for 0.833333h; Microwave irradiation; | 99.4% |

| Conditions | Yield |
|---|---|
| With triethylamine In tetrahydrofuran; diethyl ether for 1h; Ambient temperature; | 99% |
| 96% | |
| With pyridine; dmap In dichloromethane at 0℃; for 2h; Temperature; Concentration; | 95.5% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at -20℃; for 0.25h; | 99% |
-
-
462-08-8
pyridin-3-ylamine

-
-
623-00-7
4-bromobenzenecarbonitrile

-
-
189100-30-9
4-(pyridin-3-ylamino)benzonitrile

| Conditions | Yield |
|---|---|
| With sodium phenoxide; 4,5-bis(diphenylphosphino)-9,9-dimethylxanthene; tris(dibenzylideneacetone)dipalladium (0) In 1,4-dioxane at 170℃; for 2h; microwave irradiation; | 99% |
-
-
462-08-8
pyridin-3-ylamine

-
-
196314-83-7
1-(isocyanatoethyl)-1,2-closo-dodecaborane

-
-
196314-53-1
1-((3'-pyridinylcarbamido)ethyl)-o-carborane

| Conditions | Yield |
|---|---|
| In acetonitrile absence of air and moisture; equimolar amts., stirring at room temp. up to 24 h; solvent removal, recrystn. or chromy. (not specified); | 99% |

| Conditions | Yield |
|---|---|
| With 3,5-Lutidine In dichloromethane at 25℃; | 99% |
-
-
462-08-8
pyridin-3-ylamine

-
-
1311568-99-6
1-{3-[(tert-butoxycarbonyl)amino]propyl}-2-(trimethoxymethyl)-1H-benzimidazole-6-carboxylic acid

-
-
1311569-35-3
tert-butyl {3-[6-(pyridin-3-ylcarbamoyl)-2-(trimethoxymethyl)-1H-benzimidazol-1-yl]propyl}carbamate

| Conditions | Yield |
|---|---|
| Stage #1: 1-{3-[(tert-butoxycarbonyl)amino]propyl}-2-(trimethoxymethyl)-1H-benzimidazole-6-carboxylic acid With benzotriazol-1-yloxyl-tris-(pyrrolidino)-phosphonium hexafluorophosphate In N,N-dimethyl-formamide for 0.166667h; Stage #2: pyridin-3-ylamine With triethylamine In N,N-dimethyl-formamide at 45℃; for 20h; | 99% |
-
-
462-08-8
pyridin-3-ylamine

-
-
100-55-0
3-hydroxymethylpyridin

-
-
78675-94-2
N-(pyridin-3-ylmethyl)pyridin-3-amine

| Conditions | Yield |
|---|---|
| With potassium hydroxide at 130℃; for 25h; Schlenk technique; Inert atmosphere; | 99% |

| Conditions | Yield |
|---|---|
| With potassium hydroxide at 130℃; for 25h; Schlenk technique; Inert atmosphere; | 99% |
| With potassium tert-butylate; benzonitrile In 1,4-dioxane at 120℃; for 15h; Inert atmosphere; Glovebox; Sealed tube; | 82% |
| With samarium diiodide; potassium tert-butylate In tetrahydrofuran; toluene at 140℃; for 1h; Microwave irradiation; Inert atmosphere; | 78% |
| With [(NH-C3H5)Tr(NHP(iPr)2)2CoCl2]; potassium tert-butylate In toluene at 80℃; for 24h; | 61% |
-
-
462-08-8
pyridin-3-ylamine

-
-
73831-13-7
4-cyano-3-methylbenzoic acid

| Conditions | Yield |
|---|---|
| Stage #1: 4-cyano-3-methylbenzoic acid With oxalyl dichloride; N,N-dimethyl-formamide In tetrahydrofuran at 20 - 66℃; for 3h; Stage #2: pyridin-3-ylamine With pyridine at 20℃; for 16h; | 99% |

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine; HATU In N,N-dimethyl-formamide at 20℃; for 24h; | 99% |

| Conditions | Yield |
|---|---|
| With [COD(Pd-AlPhos)2]; 1,8-diazabicyclo[5.4.0]undec-7-ene In tert-butyl methyl ether at 20℃; for 3h; Reagent/catalyst; Inert atmosphere; Sealed tube; Schlenk technique; | 99% |
-
-
462-08-8
pyridin-3-ylamine

-
-
55162-34-0
1-bromo-4-(2-chloroethoxy)benzene

| Conditions | Yield |
|---|---|
| With [COD(Pd-AlPhos)2]; 1,8-diazabicyclo[5.4.0]undec-7-ene In tert-butyl methyl ether at 20℃; for 3h; Inert atmosphere; Sealed tube; Schlenk technique; | 99% |
-
-
462-08-8
pyridin-3-ylamine


| Conditions | Yield |
|---|---|
| Stage #1: 4-(4-(4-(4-(((6aS)-5-((allyloxy)carbonyl)-2-methoxy-12-oxo-6-((tetrahydro-2H-pyran-2-yl)oxy)-5,6,6a,7,8,9,10,12-octahydrobenzo[e]pyrido[1,2-a][1,4] diazepin-3-yl)oxy)butanamido)-1-methyl-1H-pyrrole-2- carboxamido)phenyl)-1-methyl-1H-pyrrole-2-carboxylic acid With triethylamine; N-[(dimethylamino)-3-oxo-1H-1,2,3-triazolo[4,5-b]pyridin-1-yl-methylene]-N-methylmethanaminium hexafluorophosphate In dichloromethane at 20℃; for 0.25h; Stage #2: pyridin-3-ylamine In dichloromethane at 20℃; for 16h; | 99% |
-
-
462-08-8
pyridin-3-ylamine

-
-
87-13-8
diethyl 2-ethoxymethylenemalonate

-
-
14029-71-1
2-(pyridin-3-yl)aminomethylene-malonic acid diethyl ester

| Conditions | Yield |
|---|---|
| at 150℃; | 98% |
| In ethanol Reflux; | 97% |
| In ethanol for 24h; Reflux; | 96% |
-
-
462-08-8
pyridin-3-ylamine

-
-
1943-83-5
2-chloroethyl isothiocyanate

-
-
13908-58-2
1-(2-chloro-ethyl)-3-pyridin-3-yl-urea

| Conditions | Yield |
|---|---|
| In toluene at 0 - 20℃; for 5.5h; | 98% |
| In toluene at 0 - 20℃; for 5.5h; | 98% |
| In tetrahydrofuran for 1.5h; Ambient temperature; | 81% |
-
-
462-08-8
pyridin-3-ylamine

-
-
79-04-9
chloroacetyl chloride

-
-
78205-18-2
2-chloro-N-(pyridine-3-yl)acetamide

| Conditions | Yield |
|---|---|
| In acetone at 0℃; | 98% |
| With triethylamine In dichloromethane at 0 - 20℃; | 83% |
| With sodium hydroxide In water at 20℃; | 48% |
3-Aminopyridine Consensus Reports
Reported in EPA TSCA Inventory.
3-Aminopyridine Standards and Recommendations
DOT Classification: 6.1; Label: Poison
3-Aminopyridine Specification
The 3-Aminopyridine, with the CAS registry number 462-08-8, is also known as 3-Pyridinamine. It belongs to the product categories of Fine Chemical & Intermediates; Pyridine; Organics; Pyridine Series; Pyridines Derivates. Its EINECS registry number is 207-322-2. This chemical's molecular formula is C5H6N2 and molecular weight is 94.11. What's more, both its IUPAC name and systematic name are the same which is called Pyridin-3-amine. 3-Aminopyridine is an aminopyridine. This chemical can be prepared by nicotinamide.
Physical properties about 3-Aminopyridine are: (1)# of Rule of 5 Violations: 0; (2)#H bond acceptors: 2; (3)#H bond donors: 2; (4)#Freely Rotating Bonds: 1; (5)Polar Surface Area: 38.91 Å2; (6)Index of Refraction: 1.587; (7)Molar Refractivity: 28.58 cm3; (8)Molar Volume: 84.9 cm3; (9)Surface Tension: 51.2 dyne/cm; (10)Density: 1.107 g/cm3; (11)Flash Point: 126 °C; (12)Enthalpy of Vaporization: 48.83 kJ/mol; (13)Boiling Point: 251 °C at 760 mmHg; (14)Vapour Pressure: 0.021 mmHg at 25 °C.
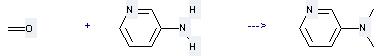
Uses of 3-Aminopyridine: (1) it is used for intermediates of medicine and dye and it also can be used as analytical reagent; (2) it is used to produce other chemicals. For example, it can react with Formaldehyde to get Dimethyl-pyridin-3-yl-amine. The reaction occurs with reagents H2SO4 and zinc-powder.
When you are dealing with this chemical, you should be very careful. This chemical is toxic by inhalation, in contact with skin and if swallowed. It is irritating to eyes, respiratory system and skin. Therefore, you could take off immediately all contaminated clothing. And you must wear suitable protective clothing, gloves and eye/face protection. In case of contact with eyes, you should rinse immediately with plenty of water and seek medical advice.
You can still convert the following datas into molecular structure:
(1) SMILES: Nc1cccnc1
(2) InChI: InChI=1S/C5H6N2/c6-5-2-1-3-7-4-5/h1-4H,6H2
(3) InChIKey: CUYKNJBYIJFRCU-UHFFFAOYSA-N
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| bird - wild | LD50 | oral | 13300ug/kg (13.3mg/kg) | Archives of Environmental Contamination and Toxicology. Vol. 12, Pg. 355, 1983. | |
| mouse | LD50 | intracrebral | 4mg/kg (4mg/kg) | BEHAVIORAL: CONVULSIONS OR EFFECT ON SEIZURE THRESHOLD | Australian Journal of Experimental Biology and Medical Science. Vol. 36, Pg. 365, 1958. |
| mouse | LD50 | intraperitoneal | 28mg/kg (28mg/kg) | Journal of Medicinal Chemistry. Vol. 8, Pg. 296, 1965. | |
| mouse | LD50 | intravenous | 24mg/kg (24mg/kg) | Annales Pharmaceutiques Francaises. Vol. 26, Pg. 345, 1968. | |
| mouse | LD50 | subcutaneous | 30mg/kg (30mg/kg) | Naunyn-Schmiedeberg's Archiv fuer Experimentelle Pathologie und Pharmakologie. Vol. 226, Pg. 163, 1955. | |
| quail | LD50 | oral | 178mg/kg (178mg/kg) | Archives of Environmental Contamination and Toxicology. Vol. 12, Pg. 355, 1983. |
Related Products
- 3-Aminopyridine
- 3-Aminopyridine hydrochloride
- 3-Aminopyridine-2-carboxaldehyde-thiosemicarbazone
- 3-Aminopyridine-2-carboxamide
- 3-Aminopyridine-4-boronic acid pinacol ester
- 3-Aminopyridine-4-carboxylic acid methyl ester
- 462100-06-7
- 462-10-2
- 462102-80-3
- 4621-04-9
- 4621-66-3
- 462-20-4
- 46227-74-1
- 462-34-0
- 46235-50-1
- 4623-55-6
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View