-
Name
4-Bromo-3,5-dihydroxybenzoic acid
- EINECS 240-606-4
- CAS No. 16534-12-6
- Article Data14
- CAS DataBase
- Density 2.026 g/cm3
- Solubility
- Melting Point 274-276 °C
- Formula C7H5BrO4
- Boiling Point 387.1 °C at 760 mmHg
- Molecular Weight 233.018
- Flash Point 187.9 °C
- Transport Information
- Appearance white to light yellow crystal powder
- Safety 26
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms a-Resorcylic acid, 4-bromo- (8CI);4-Bromo-3,5-resorcylic acid;NSC 151972;4-Bromo-3,5-dihydroxybenzoic acid;
- PSA 77.76000
- LogP 1.55850
Synthetic route
-
-
99-10-5
3,5-Dihydroxybenzoic acid

-
-
16534-12-6
3,5-dihydroxy-4-bromobenzoic acid

| Conditions | Yield |
|---|---|
| With hydrogenchloride; bromine for 2h; Heating; | 100% |
| With hydrogenchloride; bromine In water for 2h; Inert atmosphere; Schlenk technique; Reflux; | 99% |
| With hydrogenchloride; bromine In water for 2h; Reflux; Inert atmosphere; | 99% |
-
-
99-10-5
3,5-Dihydroxybenzoic acid

-
A
-
16534-12-6
3,5-dihydroxy-4-bromobenzoic acid

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide; sodium bromide; tetraethoxy orthosilicate; 3-aminopropyltriethoxysilane In 1,4-dioxane; phosphate buffer for 19h; | A n/a B 91% |

-
-
64-17-5
ethanol

-
-
16534-12-6
3,5-dihydroxy-4-bromobenzoic acid

-
-
350035-53-9
3,5-dihydroxy-4-bromobenzoic acid ethyl ester

| Conditions | Yield |
|---|---|
| With sulfuric acid for 12h; Reflux; | 100% |
| With sulfuric acid for 4h; Reflux; | 97% |
| With sulfuric acid for 24h; Reflux; | 96% |
-
-
16534-12-6
3,5-dihydroxy-4-bromobenzoic acid

-
-
77-78-1
dimethyl sulfate

-
-
26050-64-6
methyl 4-bromo-3,5-dimethoxybenzoate

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone for 4.5h; Reflux; | 100% |
| With potassium carbonate In acetone for 12h; Reflux; | 90% |
| With potassium carbonate In acetone for 4h; Heating; | 76% |
-
-
16534-12-6
3,5-dihydroxy-4-bromobenzoic acid

-
-
107-30-2
chloromethyl methyl ether

-
-
167832-31-7
(methoxy)methyl 3,5-bis(methoxymethoxy)-4-bromobenzoate

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In dichloromethane at 0 - 20℃; for 16h; Inert atmosphere; | 100% |
| With N-ethyl-N,N-diisopropylamine In dichloromethane at 0 - 20℃; | 89% |
| With N-ethyl-N,N-diisopropylamine In dichloromethane at 0 - 20℃; for 48h; |
-
-
67-56-1
methanol

-
-
16534-12-6
3,5-dihydroxy-4-bromobenzoic acid

-
-
34126-16-4
4-bromo-3,5-dihydroxy-benzoic acid methyl ester

| Conditions | Yield |
|---|---|
| With sulfuric acid for 16h; Reflux; | 99% |
| With thionyl chloride for 2h; Heating; | 96% |
| With sulfuric acid for 12h; Heating; | 96% |
-
-
16534-12-6
3,5-dihydroxy-4-bromobenzoic acid

-
-
34126-16-4
4-bromo-3,5-dihydroxy-benzoic acid methyl ester

| Conditions | Yield |
|---|---|
| With thionyl chloride In methanol at 60℃; for 3h; | 99% |
-
-
16534-12-6
3,5-dihydroxy-4-bromobenzoic acid

-
-
75-03-6
ethyl iodide

-
-
149517-92-0
ethyl 4-bromo-3,5-diethoxybenzoate

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 15 - 90℃; for 13h; | 99% |
| With potassium carbonate In N,N-dimethyl-formamide at 20℃; for 18h; | 98% |
| With potassium carbonate In N,N-dimethyl-formamide at 50℃; | 93% |
-
-
16534-12-6
3,5-dihydroxy-4-bromobenzoic acid

-
-
106-95-6
allyl bromide

-
-
355121-57-2
3,5-bis-allyloxy-4-bromo-benzoic acid allyl ester

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 20℃; for 15h; | 97% |
| With K2CO3 In water; N,N-dimethyl-formamide | 97% |
| With potassium carbonate In N,N-dimethyl-formamide at 20℃; for 18h; |
-
-
16534-12-6
3,5-dihydroxy-4-bromobenzoic acid

-
-
100-39-0
benzyl bromide

-
-
158585-09-2
benzyl 3,5-bis(benzyloxy)-4-bromobenzoate

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 25℃; for 4h; | 96% |
| With potassium carbonate In N,N-dimethyl-formamide at 20 - 60℃; Inert atmosphere; | 89% |
| With potassium carbonate In N,N-dimethyl-formamide for 20h; Ambient temperature; | 75% |

-
-
16534-12-6
3,5-dihydroxy-4-bromobenzoic acid

-
-
26050-64-6
methyl 4-bromo-3,5-dimethoxybenzoate

| Conditions | Yield |
|---|---|
| 95% |
-
-
74-87-3
methylene chloride

-
-
16534-12-6
3,5-dihydroxy-4-bromobenzoic acid

-
-
56518-42-4
4-bromo-3,5-dimethoxybenzoic acid

| Conditions | Yield |
|---|---|
| Stage #1: 3,5-dihydroxy-4-bromobenzoic acid With sodium hydride In water at 70℃; for 0.5h; pH=8; Stage #2: methylene chloride With sodium 4-dodecylbenzenesulfonate In methanol; water at 75℃; under 1500.15 Torr; for 1h; Temperature; pH-value; Pressure; Reagent/catalyst; Autoclave; | 94.49% |
-
-
74-96-4
ethyl bromide

-
-
16534-12-6
3,5-dihydroxy-4-bromobenzoic acid

-
-
363166-41-0
4-bromo-3,5-diethoxybenzoic acid

| Conditions | Yield |
|---|---|
| With potassium hydroxide In dimethyl sulfoxide at 20℃; | 92% |
-
-
16534-12-6
3,5-dihydroxy-4-bromobenzoic acid

-
-
26050-64-6
methyl 4-bromo-3,5-dimethoxybenzoate

| Conditions | Yield |
|---|---|
| With potassium carbonate; dimethyl sulfate In acetone for 24h; Reflux; Inert atmosphere; | 92% |
| With potassium carbonate; dimethyl sulfate In acetone Reflux; Inert atmosphere; | 92% |

| Conditions | Yield |
|---|---|
| With tetra-(n-butyl)ammonium iodide; potassium carbonate In N,N-dimethyl-formamide at 50℃; for 72h; | 92% |
| With tetra-(n-butyl)ammonium iodide; potassium carbonate In N,N-dimethyl-formamide at 50℃; for 72h; Inert atmosphere; | 92% |
-
-
3587-60-8
Benzyloxymethyl chloride

-
-
16534-12-6
3,5-dihydroxy-4-bromobenzoic acid

-
-
1450741-52-2
(benzyloxy)methyl 3,5-bis((benzyloxy)methoxy)-4-bromobenzoate

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In dichloromethane at 0 - 20℃; for 18h; Inert atmosphere; Schlenk technique; | 87% |
| With N-ethyl-N,N-diisopropylamine In dichloromethane at 20℃; for 16h; Inert atmosphere; | 87% |
-
-
109-87-5
Dimethoxymethane

-
-
16534-12-6
3,5-dihydroxy-4-bromobenzoic acid

-
-
167832-31-7
(methoxy)methyl 3,5-bis(methoxymethoxy)-4-bromobenzoate

| Conditions | Yield |
|---|---|
| Stage #1: Dimethoxymethane With acetyl chloride; zinc dibromide In dichloromethane at 0℃; for 0.5h; Stage #2: 3,5-dihydroxy-4-bromobenzoic acid With N-ethyl-N,N-diisopropylamine In dichloromethane at 0 - 20℃; for 13h; | 86% |
-
-
16534-12-6
3,5-dihydroxy-4-bromobenzoic acid

-
-
107-30-2
chloromethyl methyl ether

-
-
167830-45-7
4-bromo-3,5-dimethoxymethoxybenzoic acid

| Conditions | Yield |
|---|---|
| Stage #1: 3,5-dihydroxy-4-bromobenzoic acid; chloromethyl methyl ether With N-ethyl-N,N-diisopropylamine In dichloromethane at 0 - 20℃; for 48h; Stage #2: With sodium hydroxide In methanol at 70℃; for 3h; Stage #3: With hydrogenchloride In methanol; water at 0℃; pH=5 - 6; | 60% |
| With N-ethyl-N,N-diisopropylamine In dichloromethane at 0 - 20℃; for 48h; Inert atmosphere; | 60% |
-
-
64-67-5
diethyl sulfate

-
-
16534-12-6
3,5-dihydroxy-4-bromobenzoic acid

-
-
149517-92-0
ethyl 4-bromo-3,5-diethoxybenzoate

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone for 8h; Esterification; etherification; Heating; | 51% |
-
-
16534-12-6
3,5-dihydroxy-4-bromobenzoic acid

-
-
100-46-9
benzylamine

-
-
675850-37-0
N-Benzyl-4-bromo-3,5-dihydroxy-benzamide

| Conditions | Yield |
|---|---|
| 39% | |
| 39% |
-
-
16534-12-6
3,5-dihydroxy-4-bromobenzoic acid

-
-
876170-40-0
methyl 4-bromo-3-hydroxy-5-methoxybenzoate

| Conditions | Yield |
|---|---|
| With potassium carbonate; methyl iodide In N,N-dimethyl-formamide at 20℃; for 60h; | 13.1% |
-
-
16534-12-6
3,5-dihydroxy-4-bromobenzoic acid

-
-
149-91-7
3,4,5-trihydroxybenzoic acid

| Conditions | Yield |
|---|---|
| With potassium hydroxide beim Schmelzen; |
-
-
16534-12-6
3,5-dihydroxy-4-bromobenzoic acid

-
-
860757-51-3
4-bromo-3,5-dihydroxy-2,6-dinitro-benzoic acid

| Conditions | Yield |
|---|---|
| With nitric acid |
-
-
16534-12-6
3,5-dihydroxy-4-bromobenzoic acid

-
-
77-78-1
dimethyl sulfate

-
-
56518-42-4
4-bromo-3,5-dimethoxybenzoic acid

| Conditions | Yield |
|---|---|
| With sodium hydroxide |

| Conditions | Yield |
|---|---|
| beim Schmelzen; |
-
-
16534-12-6
3,5-dihydroxy-4-bromobenzoic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 10 steps 1.1: 64 percent / conc. H2SO4 / 18 h / Heating 2.1: 97 percent / PPh3; diisopropyl azodicarboxylate / tetrahydrofuran / 18 h / 20 °C 3.1: 90 percent / diisobutylaluminium hydride / hexane; tetrahydrofuran / 2 h / 20 °C 4.1: 87 percent / imidazole / CH2Cl2 / 1 h / 20 °C 5.1: trimethylborate; n-BuLi / hexane; tetrahydrofuran / 2 h / -78 - 20 °C 5.2: 81 percent / H2O 6.1: 92 percent / aq. Na2CO3; Ba(OH)2*H2O / Pd(PPh3)4 / 1,2-dimethoxy-ethane; H2O / 19 h / Heating 7.1: 92 percent / benzylidenebis(tricyclohexylphosphane)dichlororuthenium / CH2Cl2 / 48 h / 20 °C 8.1: 98 percent / Bu4NF*3H2O / tetrahydrofuran / 0.75 h / 20 °C 9.1: 87 percent / PPh3; diisopropyl azodicarboxylate / tetrahydrofuran / 18 h / 20 °C 10.1: 84 percent / LiOH / tetrahydrofuran; methanol; H2O / 4 h / 40 °C View Scheme |
-
-
16534-12-6
3,5-dihydroxy-4-bromobenzoic acid

-
-
934990-38-2
2-[2,6-bis(pent-4-enyloxy)phenyl]-6-[2,6-bis(pent-4-enyloxy)-4-(tert-butyldimethylsilyloxymethyl)phenyl]pyridine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1.1: 64 percent / conc. H2SO4 / 18 h / Heating 2.1: 97 percent / PPh3; diisopropyl azodicarboxylate / tetrahydrofuran / 18 h / 20 °C 3.1: 90 percent / diisobutylaluminium hydride / hexane; tetrahydrofuran / 2 h / 20 °C 4.1: 87 percent / imidazole / CH2Cl2 / 1 h / 20 °C 5.1: trimethylborate; n-BuLi / hexane; tetrahydrofuran / 2 h / -78 - 20 °C 5.2: 81 percent / H2O 6.1: 92 percent / aq. Na2CO3; Ba(OH)2*H2O / Pd(PPh3)4 / 1,2-dimethoxy-ethane; H2O / 19 h / Heating View Scheme |
-
-
16534-12-6
3,5-dihydroxy-4-bromobenzoic acid

-
-
934991-07-8
2-[2,6-bis(hex-5-enyloxy)phenyl]-6-[2,6-bis(hex-5-enyloxy)-4-(2-tetrahydropyranyloxymethyl)phenyl]pyridine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1.1: 64 percent / conc. H2SO4 / 18 h / Heating 2.1: 80 percent / K2CO3; KI / dimethylformamide / 16 h / 70 °C 3.1: 87 percent / diisobutylaluminium hydride / hexane; tetrahydrofuran / 3 h / 20 °C 4.1: 88 percent / p-TsOH / CH2Cl2 / 16 h / 20 °C 5.1: trimethylborate; n-BuLi / hexane; tetrahydrofuran / 2 h / -78 - 20 °C 5.2: 60 percent / H2O 6.1: 84 percent / aq. Na2CO3; Ba(OH)2*H2O / Pd(PPh3)4 / 1,2-dimethoxy-ethane; H2O / 19 h / Heating View Scheme |
4-Bromo-3,5-dihydroxybenzoic acid Specification
The Benzoicacid, 4-bromo-3,5-dihydroxy-, with the CAS registry number 16534-12-6, is also known as 4-Bromo-3,5-resorcylic acid. It belongs to the product categories of Aromatic Carboxylic Acids, Amides, Anilides, Anhydrides & Salts; Organic acids; Alcohols; Monomers; Polymer Science. Its EINECS number is 240-606-4. This chemical's molecular formula is C7H5BrO4 and molecular weight is 233.02. What's more, its systematic name is 4-bromo-3,5-dihydroxybenzoic acid. It is stable at common pressure and temperature, and it should be sealed and stored in a ventilated and dry place. Moreover, it should be protected from oxides and light.
Physical properties of Benzoicacid, 4-bromo-3,5-dihydroxy- are: (1)ACD/LogP: 2.52; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 0.53; (4)ACD/LogD (pH 7.4): -0.64; (5)ACD/BCF (pH 5.5): 1; (6)ACD/BCF (pH 7.4): 1; (7)ACD/KOC (pH 5.5): 5.73; (8)ACD/KOC (pH 7.4): 1; (9)#H bond acceptors: 4; (10)#H bond donors: 3; (11)#Freely Rotating Bonds: 3; (12)Polar Surface Area: 44.76 Å2; (13)Index of Refraction: 1.703; (14)Molar Refractivity: 44.63 cm3; (15)Molar Volume: 115 cm3; (16)Polarizability: 17.69×10-24cm3; (17)Surface Tension: 85.7 dyne/cm; (18)Density: 2.026 g/cm3; (19)Flash Point: 187.9 °C; (20)Enthalpy of Vaporization: 67.09 kJ/mol; (21)Boiling Point: 387.1 °C at 760 mmHg; (22)Vapour Pressure: 1.1E-06 mmHg at 25°C.
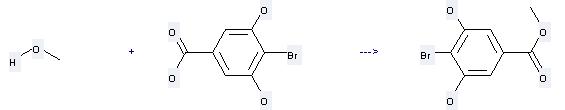
Uses of Benzoicacid, 4-bromo-3,5-dihydroxy-: it can be used to produce 4-Brom-3,5-dihydroxybenzoesaeure-methylester by heating. It will need reagent SOCl2 with the reaction time of 2 hours. The yield is about 96%.
When you are using this chemical, please be cautious about it as the following:
It is irritating to eyes, respiratory system and skin. In case of contact with eyes, you should rinse immediately with plenty of water and seek medical advice.
You can still convert the following datas into molecular structure:
(1)InChI: InChI=1S/C7H5BrO4/c8-6-4(9)1-3(7(11)12)2-5(6)10/h1-2,9-10H,(H,11,12)
(2)InChIKey: NUTRHYYFCDEALP-UHFFFAOYSA-N
(3)Canonical SMILES: C1=C(C=C(C(=C1O)Br)O)C(=O)O
Related Products
- 4-Bromo-3-(hydroxymethyl)-1-methyl-1H-pyrazole
- 4-Bromo-3-(trifluoromethyl)-1H-pyrazole
- 4-Bromo-3-(trifluoromethyl)benzaldehyde
- 4-Bromo-3-(trifluoromethyl)benzoic acid
- 4-Bromo-3-(trifluoromethyl)benzoic acid
- 4-Bromo-3-(trifluoromethyl)phenol
- 4-Bromo-3,3,4,4-tetrafluorobut-1-ene
- 4-Bromo-3,3,4,4-tetrafluorobutan-1-ol
- 4-Bromo-3,4,5-trifluoro-1,1-biphenyl
- 4-Bromo-3',4'-dimethoxybenzophenone
- 16534-24-0
- 16535-89-0
- 16536-30-4
- 1653-64-1
- 165377-44-6
- 165383-72-2
- 165385-93-3
- 16544-67-5
- 165450-17-9
- 1654-52-0
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View