-
Name
4-CYANOBENZAMIDE
- EINECS 221-223-1
- CAS No. 3034-34-2
- Article Data97
- CAS DataBase
- Density 1.24 g/cm3
- Solubility
- Melting Point 227ºC
- Formula C8H6 N2 O
- Boiling Point 350.2 ºC at 760 mmHg
- Molecular Weight 146.148
- Flash Point 165.6 ºC
- Transport Information
- Appearance Beige powder
- Safety Moderately toxic by ingestion and intraperitoneal routes. When heated to decomposition it emits toxic vapors of NOx.
- Risk Codes 20/21/22-36/37/38
-
Molecular Structure
- Hazard Symbols Xn
- Synonyms Benzamide,p-cyano- (6CI,7CI,8CI); 4-Cyanobenzamide; p-Cyanobenzamide
- PSA 66.88000
- LogP 1.35748
Synthetic route
-
-
52707-54-7, 52707-59-2, 64847-77-4
4-cyanobenzaldehyde oxime

-
-
3034-34-2
4-cyanobenzamide

| Conditions | Yield |
|---|---|
| With Cu(II) on nano silica functionalized triazine dendrimer In water at 20℃; for 0.5h; Beckmann Rearrangement; Green chemistry; | 98% |
| With Cu(II)-metformin immobilized ongraphene oxide In water at 100℃; for 0.5h; Beckmann Rearrangement; Green chemistry; | 96% |
| With [Ru(κ(3)-tpy)(κ(1)-P-PPh2Py)Cl2] In toluene for 12h; Beckmann rearrangement; Reflux; | 80% |
| With Cu(II) complex on SiO2-coated Fe3O4 nanoparticles at 80℃; for 1h; Beckmann Rearrangement; Green chemistry; | 80% |

| Conditions | Yield |
|---|---|
| Stage #1: 4-cyanobenzaldehyde With hydroxylamine hydrochloride; sodium hydrogencarbonate In water Stage #2: In water at 100℃; for 10h; | 98% |
| With hydroxylamine hydrochloride; sodium carbonate In water at 80℃; for 48h; | 96% |
| With hydroxylamine hydrochloride; sodium carbonate In water at 80℃; for 22h; Green chemistry; | 95% |

| Conditions | Yield |
|---|---|
| With copper (II) trifluoroacetate hydrate; palladium diacetate In dimethyl sulfoxide at 130℃; for 10h; Sealed tube; Inert atmosphere; | 98% |

| Conditions | Yield |
|---|---|
| With [1,1'-bis(diphenylphosphino)ferrocene]nickel(II) chloride; silver trifluoroacetate; 1-hydroxy-3H-benz[d][1,2]iodoxole-1,3-dione In N,N-dimethyl-formamide at 8℃; for 9h; Reagent/catalyst; Solvent; | 96.2% |
| With iron(III) chloride; 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical; hydroxylamine hydrochloride; iodine; potassium carbonate In 1,2-dichloro-ethane at 90℃; for 32h; | 83% |

| Conditions | Yield |
|---|---|
| Stage #1: terephthalonitrile With Br(1-)*C24H30N4Rh(1+); water In isopropyl alcohol at 25℃; for 0.5h; Inert atmosphere; Stage #2: With potassium tert-butylate In isopropyl alcohol at 25℃; for 12h; Inert atmosphere; | 96% |
| With water In dimethyl sulfoxide at 100℃; Green chemistry; | 96% |
| With sodium tetrahydroborate In ethanol; water at 80℃; for 4h; regioselective reaction; | 94% |

-
-
3034-34-2
4-cyanobenzamide

| Conditions | Yield |
|---|---|
| With ammonium carbonate In dimethyl sulfoxide at 25℃; for 6h; | 85% |

| Conditions | Yield |
|---|---|
| With tris-(dibenzylideneacetone)dipalladium(0); tris(2-morpholinophenyl)phosphine; potassium carbonate In water; tert-butyl alcohol at 85℃; for 10h; Schlenk technique; Inert atmosphere; | 82% |

| Conditions | Yield |
|---|---|
| With dmap; 1,1'-bis-(diphenylphosphino)ferrocene; nickel(II) chloride hexahydrate; zinc In acetonitrile at 80℃; for 6h; Schlenk technique; Inert atmosphere; Sealed tube; | 82% |

| Conditions | Yield |
|---|---|
| With hydroxylamine hydrochloride; 2,4,6-triphenylpyrylium tetrafluoroborate; oxygen; ammonium bromide In acetonitrile at 40℃; under 750.075 Torr; for 24h; Molecular sieve; Irradiation; | 82% |

| Conditions | Yield |
|---|---|
| With dmap; 1,1'-bis-(diphenylphosphino)ferrocene; nickel(II) chloride hexahydrate; zinc In acetonitrile at 80℃; for 6h; Schlenk technique; Inert atmosphere; Sealed tube; | 80% |
| With dmap; 1,1'-bis-(diphenylphosphino)ferrocene; nickel(II) chloride hexahydrate; zinc In acetonitrile at 80℃; for 6h; Inert atmosphere; Sealed tube; | 80% |

| Conditions | Yield |
|---|---|
| Stage #1: 4-cyanobenzaldehyde With ammonia at -33℃; for 1h; Inert atmosphere; Stage #2: With potassium permanganate at -33℃; for 1h; Inert atmosphere; Reflux; Stage #3: With sodium sulfite | A 78% B 20% |

| Conditions | Yield |
|---|---|
| Stage #1: 4-cyanobenzaldehyde With ammonia at -33℃; for 1h; Inert atmosphere; Stage #2: With potassium permanganate for 1h; Inert atmosphere; Reflux; | A n/a B 78% |

| Conditions | Yield |
|---|---|
| With ammonium chloride; N-ethyl-N,N-diisopropylamine; 2-(1-oxypyridin-2-yl)-1,1,3,3-tetramethylisothiouronium tetrafluoroborate In N,N-dimethyl-formamide at 20℃; for 0.5h; | 77% |
| With tris[2-phenylpyridinato-C2,N]iridium(III); dipotassium hydrogenphosphate; borane-ammonia complex; di-tert-butyl dicarbonate; magnesium chloride In acetonitrile at 20℃; for 36h; Schlenk technique; Irradiation; Green chemistry; | 58% |
| Multi-step reaction with 2 steps 1: (COCl)2, DMF / CH2Cl2 / Heating 2: conc. NH4OH / ethyl acetate / 0.17 h View Scheme |

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 20℃; for 1h; Cooling with ice; | 76% |

-
-
3034-34-2
4-cyanobenzamide

| Conditions | Yield |
|---|---|
| With lithium hydroxide monohydrate In water at 20℃; for 2h; | 76% |

| Conditions | Yield |
|---|---|
| With water; palladium diacetate; acetic acid; scandium tris(trifluoromethanesulfonate) at 30℃; for 24h; | A 65% B 18% |
| With chitosan-supported ruthenium catalyst (ChRu) In water at 120℃; for 1h; Microwave irradiation; Sealed tube; Overall yield = 7 %; | |
| With nitrile hydratase from Rhodococcus rhodochrous J1 W72Y mutant In aq. phosphate buffer at 35℃; for 4h; pH=7.4; Reagent/catalyst; Enzymatic reaction; regioselective reaction; |

| Conditions | Yield |
|---|---|
| With (1,3-(dicyclohexylphosphino)propane )Pd(ethylene); ammonia; potassium carbonate In dimethyl sulfoxide at 110℃; under 450 Torr; for 24h; Schlenk technique; Inert atmosphere; | 60% |
| With ammonia; palladium diacetate; catacxium A In 1,4-dioxane at 130℃; under 3000.3 Torr; for 20h; Autoclave; | 32 %Chromat. |
| With 1,1'-bis-(diphenylphosphino)ferrocene; ammonia; palladium diacetate In 1,4-dioxane at 130℃; under 1500.15 Torr; for 20h; | 30 %Chromat. |
-
-
69386-99-8, 18705-39-0
terephthalaldehyde oxime

-
A
-
623-26-7
terephthalonitrile

-
B
-
3034-34-2
4-cyanobenzamide

| Conditions | Yield |
|---|---|
| With nickel(II) chloride dihydrate In acetonitrile at 80℃; Molecular sieve; Inert atmosphere; | A 58% B 42% |

| Conditions | Yield |
|---|---|
| With palladium diacetate; ammonium bicarbonate; dicyclohexyl-carbodiimide; triphenylphosphine In acetonitrile at 100℃; Inert atmosphere; | 55% |

| Conditions | Yield |
|---|---|
| With sodium azide; iodine; sodium hydrogencarbonate In water at 100℃; for 2h; | 53% |

| Conditions | Yield |
|---|---|
| 96% | |
| With sodium nitrite In sulfuric acid | 95% |
| 94% | |
| With acetic anhydride; acetic acid; sodium nitrite In water | 92% |
| With sulfuric acid; acetic acid; sodium nitrite In water | 86% |
-
-
3034-34-2
4-cyanobenzamide

-
-
86065-51-2
1-(4-cyanophenyl)urea

| Conditions | Yield |
|---|---|
| With [bis(acetoxy)iodo]benzene; ammonium carbamate In 2,2,2-trifluoroethanol at 0 - 20℃; for 23.5h; Reagent/catalyst; Solvent; Hofmann Rearrangement; Inert atmosphere; | 95% |

| Conditions | Yield |
|---|---|
| With sodium hydride In N,N-dimethyl-formamide at 0 - 20℃; Inert atmosphere; | 95% |

| Conditions | Yield |
|---|---|
| With ammonia; hydrogen In toluene at 120℃; under 22502.3 Torr; for 16h; Autoclave; | 91% |
| With formic acid; palladium 10% on activated carbon; triethylamine In tetrahydrofuran at 40℃; for 3h; | 98 %Chromat. |

| Conditions | Yield |
|---|---|
| With dichloro(pentamethylcyclopentadienyl)rhodium (III) dimer; silver carbonate In acetonitrile at 115℃; for 10h; Inert atmosphere; | 90% |

| Conditions | Yield |
|---|---|
| With iron(II) chloride tetrahydrate; N-methyl-N-trimethylsilyl-2,2,2-trifluoroacetamide In tetrahydrofuran at 70℃; for 2h; | 90% |
| With diethyl chlorophosphate at 120℃; for 0.25h; Neat (no solvent); | 86% |
| Stage #1: 4-cyanobenzamide With N-methylbenzamide; phenylsilane; C28H18ClMnN2O2; potassium tert-butylate In tetrahydrofuran at 50℃; Inert atmosphere; Glovebox; Sealed tube; Stage #2: With sodium hydroxide In tetrahydrofuran for 2h; Inert atmosphere; Glovebox; Sealed tube; Stage #3: Glovebox; Inert atmosphere; | 82% |
| at 315℃; for 1h; Temperature; | 72% |
| With N-methyl-N-trimethylsilyl-2,2,2-trifluoroacetamide; zinc trifluoromethanesulfonate In tetrahydrofuran at 70℃; for 24h; chemoselective reaction; |
-
-
3034-34-2
4-cyanobenzamide

-
-
54050-86-1
4-Amidinobenzamide

| Conditions | Yield |
|---|---|
| With sodium amide In dimethyl sulfoxide Reagent/catalyst; Solvent; Sealed tube; | 85% |
| With hydrogenchloride; ethanol; benzene Behandeln des Reaktionsprodukts mit aethanol. Ammoniak; |
-
-
2432-51-1
ethylthioacetic acid methyl ester

-
-
3034-34-2
4-cyanobenzamide

-
-
1181384-68-8
N-butyryl-4-cyanobenzamide

| Conditions | Yield |
|---|---|
| Stage #1: 4-cyanobenzamide With N-Bromosuccinimide; iron(II) chloride In acetonitrile at 20℃; for 0.0166667h; Stage #2: ethylthioacetic acid methyl ester In acetonitrile at 45℃; for 20h; | 85% |

| Conditions | Yield |
|---|---|
| With dmap In dichloromethane at 0 - 20℃; Inert atmosphere; | 85% |
| With dmap In acetonitrile at 20℃; for 15h; Inert atmosphere; Sealed tube; | 85% |
| With dmap In dichloromethane at 0 - 23℃; Inert atmosphere; | 84.5% |
-
-
3034-34-2
4-cyanobenzamide

| Conditions | Yield |
|---|---|
| With tetra-N-butylammonium tribromide; 1,8-diazabicyclo[5.4.0]undec-7-ene In dichloromethane for 1h; Ambient temperature; | 83% |

| Conditions | Yield |
|---|---|
| Stage #1: Cyclohexyl iodide; 4-cyanobenzamide With copper(l) iodide; lithium tert-butoxide In N,N-dimethyl-formamide; acetonitrile at 20℃; for 0.0833333h; Inert atmosphere; Stage #2: In N,N-dimethyl-formamide; acetonitrile at 20℃; for 24h; Inert atmosphere; Irradiation; | 83% |

| Conditions | Yield |
|---|---|
| With water; sodium hydroxide In ethanol at 90℃; for 17h; | 82% |
| With potassium tert-butylate In tert-butyl alcohol at 20℃; for 12h; Solvent; Inert atmosphere; | 78% |

| Conditions | Yield |
|---|---|
| Stage #1: 4-cyanobenzamide; Hexafluoroacetone With pyridine In benzene at 20℃; for 0.5h; Stage #2: With thionyl chloride In benzene at 20℃; for 2h; | 82% |
4-CYANOBENZAMIDE Chemical Properties
IUPAC Name: 4-Cyanobenzamide
Synonyms: 4-Cyanobenamide ; 4-Cyano-benzamid;benzamide, 4-cyano- ; Cyanobenzamide ; P-Cyano-benzamid ; P-Cyano benzamide ; 4-Cyanobenzamide ; Benzamide, 4-cyano- (9ci)
Product Categories: AMIDE;Aromatic Carboxylic Acids, Amides, Anilides, Anhydrides & Salts;Aromatic Building Blocks
CAS NO: 3034-34-2
Molecular Formula: C8H6N2O
Molecular Weight: 146.15
Molecular Structure: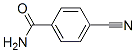
EINECS: 221-223-1 Mol
File: 3034-34-2.mol
Index of Refraction: 1.592
Surface Tension: 59.9 dyne/cm
Density: 1.24 g/cm3
Flash Point: 165.6 °C
Melting point: 227 °C
Enthalpy of Vaporization: 59.48 kJ/mol
Boiling Point: 350.2 °C at 760 mmHg
Vapour Pressure: 4.46E-05 mmHg at 25°C
Appearance: 4-Cyanobenzamide (CAS NO.3034-34-2) is beige powder.
4-CYANOBENZAMIDE Toxicity Data With Reference
| 1. | ipr-rat LD50:609 mg/kg | APFRAD Annales Pharmaceutiques Francaises. 48 (1990),23. | ||
| 2. | orl-mus LD :>1620 mg/kg | APFRAD Annales Pharmaceutiques Francaises. 41 (1983),391. | ||
| 3. | ipr-mus LD50:783 mg/kg | APFRAD Annales Pharmaceutiques Francaises. 48 (1990),23. |
4-CYANOBENZAMIDE Safety Profile
Risk Statements 20/21/22-36/37/38
R20/21/22: 4-Cyanobenzamide (CAS NO.3034-34-2) is harmful by inhalation, in contact with skin and if swallowed.
R36/37/38:Irritating to eyes, respiratory system and skin.
Safety Statements 26-37/39
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S37/39: When 4-Cyanobenzamide (CAS NO.3034-34-2) is used ,wear suitable gloves and eye/face protection.
RIDADR 3439
RTECS CV2484500
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View