-
Name
4-Chloro-2-nitrobenzoic acid
- EINECS 228-483-5
- CAS No. 6280-88-2
- Article Data18
- CAS DataBase
- Density 1.602 g/cm3
- Solubility water: 0.53 g/100 mL (15 °C)
- Melting Point 141-143 °C(lit.)
- Formula C7H4ClNO4
- Boiling Point 357 °C at 760 mmHg
- Molecular Weight 201.566
- Flash Point 169.7 °C
- Transport Information UN 2811 6.1/PG 3
- Appearance light yellow to light yellow-green crystalline
- Safety 26-36-24/25
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms 2-Nitro-4-chlorobenzoicacid;NSC 6158;
- PSA 83.12000
- LogP 2.46960
Synthetic route
-
-
34662-32-3
4-chloro-2-nitrobenzonitrile

-
-
6280-88-2
4-chloro-2-nitro-benzoic acid

| Conditions | Yield |
|---|---|
| With sulfuric acid | |
| With sulfuric acid | |
| With sulfuric acid | |
| Multi-step reaction with 2 steps 1: sulfuric acid 2: sulfuric acid View Scheme |

| Conditions | Yield |
|---|---|
| With nitric acid at 200℃; | |
| With pyridine; potassium permanganate; water | |
| With potassium permanganate |

| Conditions | Yield |
|---|---|
| With potassium permanganate |
-
-
41994-91-6
4-chloro-2-nitrobenzoic amide

-
-
6280-88-2
4-chloro-2-nitro-benzoic acid

| Conditions | Yield |
|---|---|
| With sulfuric acid |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: durch Austausch von NH2 gegen CN nach Sandmeyer 2: concentrated sulfuric acid View Scheme | |
| Multi-step reaction with 2 steps 2: sulfuric acid View Scheme | |
| Multi-step reaction with 2 steps 1: copper (II)-sulfate; alkali cyanide; water; sulfuric acid; Wasser / Edukt 4: Natriumnitrit 2: aqueous sulfuric acid View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: sulfuric acid; nitric acid 2: KMnO4 View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: SbCl5; chlorine / <40 2: nitric acid / 200 °C View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 2: KMnO4 View Scheme | |
| Multi-step reaction with 2 steps 2: nitric acid / 200 °C View Scheme |

| Conditions | Yield |
|---|---|
| With nitric acid In water at 60℃; for 6h; Cooling with ice; |
-
-
6280-88-2
4-chloro-2-nitro-benzoic acid

-
-
41995-04-4
4-chloro-2-nitro-benzoyl chloride

| Conditions | Yield |
|---|---|
| With thionyl chloride In toluene for 3h; Heating; | 100% |
| With thionyl chloride In DMF (N,N-dimethyl-formamide) at 26 - 65℃; for 5h; | 100% |
| With thionyl chloride at 80℃; Inert atmosphere; | 90% |
-
-
6280-88-2
4-chloro-2-nitro-benzoic acid

-
-
77-78-1
dimethyl sulfate

-
-
42087-80-9
methyl 4-chloro-2-nitrobenzoate

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone for 0.5h; Esterification; Heating; | 100% |
-
-
6280-88-2
4-chloro-2-nitro-benzoic acid

-
-
41994-91-6
4-chloro-2-nitrobenzoic amide

| Conditions | Yield |
|---|---|
| With ammonium hydroxide; thionyl chloride In dichloromethane; ethyl acetate | 99% |
| With ammonium hydroxide; thionyl chloride 1.) PhMe, reflux; Multistep reaction; | |
| Multi-step reaction with 2 steps 1: thionyl chloride / toluene; dimethylformamide / Heating 2: 30 percent aq. NH3 / dioxane / 20 °C View Scheme |
-
-
6280-88-2
4-chloro-2-nitro-benzoic acid

-
-
66937-55-1
N-cyclohexylglycine t-butyl ester

| Conditions | Yield |
|---|---|
| With triethylamine; HATU In N,N-dimethyl-formamide for 12h; | 98% |
-
-
6280-88-2
4-chloro-2-nitro-benzoic acid

-
-
41513-04-6
2-chloro-5-bromonitrobenzene

| Conditions | Yield |
|---|---|
| With trichloroisocyanuric acid; bromine In tetrachloromethane at 10 - 100℃; for 18h; Photolysis; | 96% |
| With copper(l) iodide; oxygen; copper(I) bromide In dimethyl sulfoxide at 160℃; under 760.051 Torr; for 30h; Schlenk technique; Sealed tube; | 81% |
| With 2.9-dimethyl-1,10-phenanthroline; oxygen; copper diacetate; silver sulfate; sodium bromide In dimethyl sulfoxide at 160℃; under 760.051 Torr; for 20h; Schlenk technique; | 64% |
-
-
119072-55-8, 7188-38-7
tert-butylisonitrile

-
-
6280-88-2
4-chloro-2-nitro-benzoic acid

-
-
104-88-1
4-chlorobenzaldehyde

-
-
120-20-7
2-(3,4-dimethoxyphenyl)-ethylamine

| Conditions | Yield |
|---|---|
| In methanol at 20℃; for 24h; | 95% |

-
-
61-54-1
tryptamine

-
-
119072-55-8, 7188-38-7
tert-butylisonitrile

-
-
6280-88-2
4-chloro-2-nitro-benzoic acid

-
-
104-88-1
4-chlorobenzaldehyde

| Conditions | Yield |
|---|---|
| In methanol at 20℃; for 24h; | 95% |

-
-
6280-88-2
4-chloro-2-nitro-benzoic acid

-
-
64-04-0
phenethylamine

-
-
284487-93-0
4-chloro-2-nitro-N-phenethylbenzamide

| Conditions | Yield |
|---|---|
| With dmap; benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In dichloromethane; N,N-dimethyl-formamide at 60℃; for 4h; | 94% |
| With pyridine; 2-(1H-9-azabenzotriazol-1-yl)-1,1,3,3-tetramethyluroniumhexafluorophosphate In N,N-dimethyl-formamide for 1h; |
-
-
6280-88-2
4-chloro-2-nitro-benzoic acid

-
-
68-12-2, 33513-42-7
N,N-dimethyl-formamide

-
-
2953-58-4
N,N-dimethyl-4-chloro-2-nitrobenzamide

| Conditions | Yield |
|---|---|
| With trichlorophosphate at 120℃; for 1h; Sealed tube; | 94% |
| With trichlorophosphate at 120℃; for 1h; | 94% |
| With dipotassium peroxodisulfate; (p-cymene)ruthenium(II) chloride; 4,5-bis(diphenylphosphino)-9,9-dimethylxanthene at 160℃; for 24h; | 37% |
| With potassium thiosulfate; [RhCl2(p-cymene)]2; 4,5-bis(diphenylphosphino)-9,9-dimethylxanthene at 160℃; for 24h; Inert atmosphere; Sealed tube; | 37% |

| Conditions | Yield |
|---|---|
| With dmap; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In dichloromethane at 20℃; | 93% |
-
-
13669-42-6
3-quinolylcarboxaldehyde

-
-
119072-55-8, 7188-38-7
tert-butylisonitrile

-
-
6280-88-2
4-chloro-2-nitro-benzoic acid

-
-
120-20-7
2-(3,4-dimethoxyphenyl)-ethylamine

| Conditions | Yield |
|---|---|
| In methanol at 20℃; for 24h; | 93% |

-
-
6280-88-2
4-chloro-2-nitro-benzoic acid

-
-
22996-18-5
(4-chloro-2-nitrophenyl)methanol

| Conditions | Yield |
|---|---|
| With dimethylsulfide borane complex In tetrahydrofuran at 80℃; for 3h; Inert atmosphere; | 91% |
| With dimethylsulfide borane complex In tetrahydrofuran at 80℃; for 3h; Inert atmosphere; | 52% |
| With boron dimethyl-trifluoro sulphide In tetrahydrofuran at 0 - 60℃; Inert atmosphere; | |
| With borane-THF In tetrahydrofuran at 0 - 20℃; for 72h; |

| Conditions | Yield |
|---|---|
| With thionyl chloride for 6h; | 90% |
| With thionyl chloride at 0 - 50℃; for 24h; Inert atmosphere; | 67% |
| With sulfuric acid Reflux; |
-
-
1121-60-4
pyridine-2-carbaldehyde

-
-
119072-55-8, 7188-38-7
tert-butylisonitrile

-
-
6280-88-2
4-chloro-2-nitro-benzoic acid

-
-
54149-39-2
O-(2-Methoxy-ethyl)-hydroxylamine

| Conditions | Yield |
|---|---|
| In methanol at 20℃; for 24h; | 90% |

-
-
61-54-1
tryptamine

-
-
6280-88-2
4-chloro-2-nitro-benzoic acid

-
-
885463-77-4
4-chloro-N-[2-(1H-indol-3-yl)ethyl]-2-nitrobenzamide

| Conditions | Yield |
|---|---|
| Stage #1: 4-chloro-2-nitro-benzoic acid With benzotriazol-1-ol; dicyclohexyl-carbodiimide In N,N-dimethyl-formamide at 20℃; Stage #2: tryptamine In N,N-dimethyl-formamide at 20℃; for 12h; | 89% |
-
-
119072-55-8, 7188-38-7
tert-butylisonitrile

-
-
6280-88-2
4-chloro-2-nitro-benzoic acid

-
-
123-11-5
4-methoxy-benzaldehyde

-
-
22483-09-6
2,2-dimethoxyethylamine

| Conditions | Yield |
|---|---|
| In methanol at 20℃; for 24h; | 89% |


| Conditions | Yield |
|---|---|
| With copper(l) iodide; oxygen In dimethyl sulfoxide at 160℃; under 760.051 Torr; for 30h; Reagent/catalyst; Solvent; Schlenk technique; Sealed tube; | 87% |
| With 2.9-dimethyl-1,10-phenanthroline; oxygen; copper (I) acetate; silver sulfate; sodium iodide In dimethyl sulfoxide at 160℃; for 24h; Schlenk technique; | 72% |
| With 2.9-dimethyl-1,10-phenanthroline; oxygen; copper diacetate; silver sulfate; sodium iodide In dimethyl sulfoxide at 160℃; under 760.051 Torr; for 20h; Schlenk technique; | 72% |
-
-
6280-88-2
4-chloro-2-nitro-benzoic acid

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine; N-[(dimethylamino)-3-oxo-1H-1,2,3-triazolo[4,5-b]pyridin-1-yl-methylene]-N-methylmethanaminium hexafluorophosphate In N,N-dimethyl-formamide at 0 - 30℃; for 16h; | 87% |
-
-
119072-55-8, 7188-38-7
tert-butylisonitrile

-
-
6280-88-2
4-chloro-2-nitro-benzoic acid

-
-
123-11-5
4-methoxy-benzaldehyde

-
-
120-20-7
2-(3,4-dimethoxyphenyl)-ethylamine

| Conditions | Yield |
|---|---|
| In methanol at 20℃; for 24h; | 87% |

-
-
1075-47-4
4-methylphenylglyoxal

-
-
931-53-3
Cyclohexyl isocyanide

-
-
6280-88-2
4-chloro-2-nitro-benzoic acid

-
-
5003-71-4
3-bromopropylamine hydrochloride

| Conditions | Yield |
|---|---|
| Stage #1: 4-methylphenylglyoxal; Cyclohexyl isocyanide; 4-chloro-2-nitro-benzoic acid; 3-bromopropylamine hydrochloride With sodium hydroxide In methanol at 20℃; for 24h; Stage #2: With caesium carbonate In methanol at 20℃; for 1h; | 87% |

-
-
931-53-3
Cyclohexyl isocyanide

-
-
6280-88-2
4-chloro-2-nitro-benzoic acid

-
-
5003-71-4
3-bromopropylamine hydrochloride

-
-
1076-95-5
p-methoxyphenylglyoxal

| Conditions | Yield |
|---|---|
| Stage #1: Cyclohexyl isocyanide; 4-chloro-2-nitro-benzoic acid; 3-bromopropylamine hydrochloride; p-methoxyphenylglyoxal With sodium hydroxide In methanol at 20℃; for 24h; Stage #2: With caesium carbonate In methanol at 20℃; for 1h; | 86% |

-
-
119072-55-8, 7188-38-7
tert-butylisonitrile

-
-
6280-88-2
4-chloro-2-nitro-benzoic acid

-
-
123-11-5
4-methoxy-benzaldehyde

-
-
54149-39-2
O-(2-Methoxy-ethyl)-hydroxylamine

| Conditions | Yield |
|---|---|
| In methanol at 20℃; for 24h; | 84% |


| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane at 20℃; Steglich Esterification; | 83% |
-
-
534-22-5
2-methylfuran

-
-
6280-88-2
4-chloro-2-nitro-benzoic acid

-
-
1045704-63-9
2-methyl-5-(4-chloro-2-nitrophenyl)-furan

| Conditions | Yield |
|---|---|
| With palladium(II) trifluoroacetate; triphenylphosphine; silver carbonate In 1,2-dimethoxyethane; dimethyl sulfoxide at 120℃; for 24h; Inert atmosphere; Glovebox; | 83% |
-
-
6280-88-2
4-chloro-2-nitro-benzoic acid

-
-
84518-77-4
ethyl 1,2,3,4-tetrahydro-β-carboline-3-carboxylate

-
-
130472-81-0
2-(4-Chloro-2-nitro-benzoyl)-2,3,4,9-tetrahydro-1H-β-carboline-3-carboxylic acid ethyl ester

| Conditions | Yield |
|---|---|
| With dicyclohexyl-carbodiimide In dichloromethane for 3h; Ambient temperature; | 82% |
-
-
6280-88-2
4-chloro-2-nitro-benzoic acid

-
-
56978-50-8
4,4'-dichloro-2,2'-dinitrobiphenyl

| Conditions | Yield |
|---|---|
| With copper(l) iodide In dimethyl sulfoxide at 140℃; for 20h; Catalytic behavior; Solvent; Reagent/catalyst; Inert atmosphere; | 82% |
| With triphenylphosphine; silver carbonate; palladium dichloride In dimethyl sulfoxide at 130℃; for 6h; Inert atmosphere; | 77% |

| Conditions | Yield |
|---|---|
| With 1,10-Phenanthroline; caesium carbonate; copper(l) chloride; silver(l) oxide In N,N-dimethyl-formamide at 110℃; for 23h; Inert atmosphere; | 81% |
| With 1,10-Phenanthroline; di-tert-butyl peroxide; oxygen; potassium carbonate; copper(I) bromide In toluene at 140℃; for 24.25h; Molecular sieve; Schlenk technique; Sealed tube; regioselective reaction; | 78% |
| With bis(tricyclohexylphosphine)nickel(II) dichloride; N,N′-bis(2,6-diisopropylphenyl)imidazol-2-ylidene hydrochloride; silver carbonate; p-benzoquinone at 170℃; for 24h; Sealed tube; | 72% |
-
-
6280-88-2
4-chloro-2-nitro-benzoic acid

-
-
108-98-5
thiophenol

-
-
4548-56-5
4-chloro-2-nitrophenyl phenyl sulfide

| Conditions | Yield |
|---|---|
| With copper(l) iodide; 1,10-Phenanthroline; oxygen; potassium carbonate In dimethyl sulfoxide at 140℃; for 24h; Molecular sieve; | 81% |
-
-
7304-32-7
2-fluoro-5-nitrobenzoic acid

-
-
6280-88-2
4-chloro-2-nitro-benzoic acid

-
-
1393456-33-1
C12H6ClFN2O4

| Conditions | Yield |
|---|---|
| With dichloro bis(acetonitrile) palladium(II); silver carbonate; tricyclohexylphosphine In 1,2-dimethoxyethane; dimethyl sulfoxide at 120℃; for 24h; Inert atmosphere; | 80% |
-
-
119072-55-8, 7188-38-7
tert-butylisonitrile

-
-
6280-88-2
4-chloro-2-nitro-benzoic acid

-
-
5680-79-5
glycine ethyl ester hydrochloride

-
-
59259-01-7
2-<2-(1,3-dioxolanyl)>benzaldehyde

-
-
1426688-04-1
4-(2-(tert-butyl)-3-oxo-2,3-dihydroisoquinolin-4-yl)-8-chloro-3,4-dihydro-1H-benzo[e][1,4]diazepine-2,5-dione

| Conditions | Yield |
|---|---|
| Stage #1: tert-butylisonitrile; 4-chloro-2-nitro-benzoic acid; glycine ethyl ester hydrochloride; 2-<2-(1,3-dioxolanyl)>benzaldehyde With sodium carbonate In methanol at 20℃; for 12h; Ugi Condensation; Inert atmosphere; Stage #2: With perchloric acid In water; acetonitrile at 20℃; for 8h; Inert atmosphere; Stage #3: With acetic acid; zinc In water; acetonitrile at 60℃; for 8h; Inert atmosphere; | 80% |

4-Chloro-2-Nitrobenzoic Acid Chemical Properties
Molecular Weight: 201.56
EINECS: 228-483-5
Melting point: 141-143 °C(lit.)
Density: 1.5
Flash point: >100°C
Appearance: Slightly yellow powder.
Solubility in water: Very soluble
4-Chloro-2-nitrobenzoic acid's(6280-88-2) Synonyms: 2-Nitro-4-chlorobenzoic acid;4-CHLORO-2-NITROBENZOIC ACID;Benzoic acid, 4-chloro-2-nitro-;4-Chloro-2-nitrobenzoic acid, 97+%;4,2-CNBA;4-Chloro-2-Nitrobenzoic;4-CHLORO-2-NITROBENZOIC ACID 98+%
4-Chloro-2-nitrobenzoic acid's(6280-88-2) Structural formula:
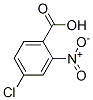
4-Chloro-2-Nitrobenzoic Acid Uses
4-Chloro-2-Nitrobenzoic Acid Safety Profile
Hazard Codes Xi
Risk Statements 36/37/38
Safety Statements 26-36-24/25
RIDADR UN 2811 6.1/PG 3
WGK Germany 3
Hazard Note Irritant
HS Code 29163900
4-Chloro-2-Nitrobenzoic Acid Specification
【Disposal Code】4
【Incompatibilities】Oxidizing agents, strong bases.
【Stability】Stable at room temperature in closed containers under normal storage and handling conditions.
【Decomposition】Hydrogen chloride, nitrogen oxides, carbon monoxide, irritating and toxic fumes and gases, carbon dioxide, nitrogen.
Transport Information for 4-Chloro-2-nitrobenzoic acid(6280-88-2)
【HS Code】2916 39 00
Related Products
- 4-Chloro-2-Nitrobenzoic Acid
- 6280-89-3
- 628-09-1
- 6280-96-2
- 6280-98-4
- 6281-06-7
- 628-11-5
- 62811-75-0
- 6281-23-8
- 628-12-6
- 62813-01-8
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View