-
Name
4-Fluorobenzyl alcohol
- EINECS 207-292-0
- CAS No. 459-56-3
- Article Data239
- CAS DataBase
- Density 1.173 g/cm3
- Solubility Slightly soluble in water.
- Melting Point 23 °C
- Formula C7H7FO
- Boiling Point 202 °C at 760 mmHg
- Molecular Weight 126.13
- Flash Point 94.6 °C
- Transport Information
- Appearance colorless or slightly yellow liquid
- Safety 23-24/25-45-36/37/39-27-26
- Risk Codes 14-34-36/37
-
Molecular Structure
-
Hazard Symbols
 Xi,
Xi, C
C
- Synonyms Benzylalcohol, p-fluoro- (6CI,7CI,8CI);(4-Fluorophenyl)methanol;4-Fluorobenzenemethanol;p-Fluorophenylmethanol;NSC 63347;p-Fluorobenzyl alcohol;
- PSA 20.23000
- LogP 1.31800
Synthetic route

| Conditions | Yield |
|---|---|
| With lithium aluminium tetrahydride In tetrahydrofuran at 20℃; Inert atmosphere; Cooling with ice; | 100% |
| Stage #1: 4-Fluorobenzoic acid With 4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane at 20℃; for 12h; Inert atmosphere; Stage #2: With silica gel In methanol at 50℃; for 3h; | 90% |
| With cobalt(II) tetrafluoroborate hexahydrate; hydrogen; [2-((diphenylphospino)methyl)-2-methyl-1,3-propanediyl]bis[diphenylphosphine] In tetrahydrofuran at 100℃; under 60006 Torr; for 22h; | 84% |

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate In methanol at 20℃; for 1h; | 100% |
| With C24H30Cl2NPRuS2; potassium tert-butylate; hydrogen In dichloromethane; toluene at 80℃; under 15001.5 Torr; for 5h; Autoclave; | 100% |
| With C55H44O2P4Ru; hydrogen In toluene at 80℃; under 38002.6 Torr; for 18h; Glovebox; Autoclave; | 99% |

| Conditions | Yield |
|---|---|
| With cerium(IV) triflate; water at 20℃; for 25h; | 100% |
| With sodium anthraquinone-2-sulfonate; oxygen In water at 30℃; Irradiation; |

| Conditions | Yield |
|---|---|
| With methanol; potassium permanganate at 25℃; chemoselective reaction; | 99% |
| With water In acetonitrile for 0.166667h; Quantum yield; Irradiation; |

| Conditions | Yield |
|---|---|
| With C23H29Cl2N2OPRuS; potassium tert-butylate; hydrogen In 2-methyltetrahydrofuran at 100℃; under 37503.8 Torr; for 16h; Catalytic behavior; Time; Autoclave; | 99% |
| With C27H35Cl2N2OPRuS; potassium tert-butylate; hydrogen In 2-methyltetrahydrofuran at 100℃; under 37503.8 Torr; for 2h; Catalytic behavior; Time; Autoclave; | 99% |
| With C27H34Cl2NO2PRuS; potassium tert-butylate; hydrogen In 2-methyltetrahydrofuran at 100℃; under 37503.8 Torr; for 16h; Catalytic behavior; Time; Autoclave; | 98% |

-
-
459-56-3
4-fluorobenzylic alcohol

| Conditions | Yield |
|---|---|
| With silica gel In ethyl acetate; Petroleum ether | 99% |
| With silica gel In methanol at 60℃; for 3h; Inert atmosphere; | 92% |
| In methanol at 20℃; for 1h; Inert atmosphere; Glovebox; | 92% |
-
-
139058-95-0
4-fluorobenzyl trifluoroacetate

-
A
-
459-56-3
4-fluorobenzylic alcohol

-
B
-
75-89-8
2,2,2-trifluoroethanol

| Conditions | Yield |
|---|---|
| With trans-[(2,6-bis(di-tert-butylphosphinomethyl)pyridine)Fe(H)2(CO)]; hydrogen; sodium methylate In 1,4-dioxane at 40℃; under 18751.9 Torr; for 16h; Glovebox; Inert atmosphere; | A n/a B 97% |
| With trimethylamine-N-oxide; tricarbonyl(η4-1,3-bis(trimethylsilyl)-4,5,6,7-tetrahydro-2H-inden-2-one)iron; hydrogen; triethylamine In toluene at 90℃; under 52505.3 Torr; for 17h; Inert atmosphere; Glovebox; |

-
-
459-56-3
4-fluorobenzylic alcohol

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water Schlenk technique; Inert atmosphere; | 97% |
| With sodium hydroxide In water |
-
-
14629-55-1
(4-fluorobenzyloxy)trimethylsilane

-
-
459-56-3
4-fluorobenzylic alcohol

| Conditions | Yield |
|---|---|
| With 1-butyl-3-methylimidazolium chloride at 20℃; for 0.66h; | 95% |
| With 1,4-diazabicyclo[2.2.2]octane tribromide supported on magnetic Fe3O4 nanoparticles In methanol at 20℃; for 0.0833333h; | 90% |
-
-
42564-51-2
4-fluoro-3-nitrobenzaldehyde

-
-
459-56-3
4-fluorobenzylic alcohol

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate In methanol | 95% |

| Conditions | Yield |
|---|---|
| With iron(III) sulfate; water In toluene at 110℃; for 3h; Ionic liquid; | 93% |
| With water; sodium carbonate | |
| With sodium carbonate for 6h; Heating; Yield given; |
-
-
79606-49-8
4-fluoro-N,N-diphenylbenzamide

-
A
-
459-56-3
4-fluorobenzylic alcohol

-
B
-
122-39-4
diphenylamine

| Conditions | Yield |
|---|---|
| With potassium phosphate; C29H55FeNOP2; hydrogen In tetrahydrofuran at 130℃; under 37503.8 Torr; for 3h; Catalytic behavior; | A n/a B 93% |
-
-
459-57-4
4-fluorobenzaldehyde

-
A
-
459-56-3
4-fluorobenzylic alcohol

-
B
-
932724-63-5
tris(4-fluorobenzyl)amine

-
C
-
140-75-0
para-fluorobenzylamine

-
D
-
100-46-9
benzylamine

-
E
-
134227-41-1
1-(4-fluorophenyl)-N-[(4-fluorophenyl)methyl]methanamine

| Conditions | Yield |
|---|---|
| With ammonia; hydrogen; Ni/C catalyst In water at 120℃; under 28502.9 - 33753.4 Torr; for 17.5h; | A 1.2% B 1.4% C 90.7% D 0.4% E 1.8% |


| Conditions | Yield |
|---|---|
| With C33H29FeMnN2O3P; hydrogen; potassium carbonate In ethanol at 90℃; under 37503.8 Torr; for 16h; Catalytic behavior; Solvent; Pressure; Autoclave; | 90% |
| With [(Rc,Sp)-N-2-picoIynyI-1-(-2-bis(4-methoxy-3,5-dimethlphenyl)phosphino)ferrocenylethylamine]-kN1-kN2-kP-tricarbonylmanganese(I) bromide; hydrogen; potassium carbonate In ethanol at 90℃; Solvent; Reagent/catalyst; Temperature; Autoclave; | 90% |
| With potassium tert-butylate; C39H41FeMnN2O5P(1+)*Br(1-); 1-Methylnaphthalene In ethanol at 100℃; for 22h; Temperature; Inert atmosphere; Schlenk technique; Darkness; | 77% |
| Multi-step reaction with 2 steps 1: lithium tert-butoxide / tetrahydrofuran / 0 - 20 °C 2: sodium hydroxide / water / 1 h / 20 °C View Scheme | |
| With C41H46FeMnN3O5P(1+)*Br(1-); hydrogen; potassium carbonate In ethanol at 70℃; under 15001.5 Torr; for 5h; Catalytic behavior; Reagent/catalyst; Temperature; | 43.3 %Spectr. |

| Conditions | Yield |
|---|---|
| With water In dimethyl sulfoxide at 50℃; for 24h; Schlenk technique; | 87% |
| With water; dimethyl sulfoxide at 50℃; for 24h; Schlenk technique; Sealed tube; | 87% |
| With water; potassium carbonate |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water at 25℃; for 3.5h; Cannizzaro Reaction; | A n/a B 86% |
| With potassium hydroxide | |
| With barium dihydroxide; formaldehyd at 100 - 110℃; for 0.2h; | A 88 % Chromat. B 11 % Chromat. |
| Stage #1: 4-fluorobenzaldehyde In neat (no solvent) for 0.0333333h; Green chemistry; Stage #2: With 1,4-diaza-bicyclo[2.2.2]octane In neat (no solvent) at 40℃; for 0.00416667h; Reagent/catalyst; Cannizzaro Reaction; Microwave irradiation; Green chemistry; |

-
-
459-56-3
4-fluorobenzylic alcohol

| Conditions | Yield |
|---|---|
| With methanol; sodium triflate; sodium hydrogencarbonate; 1-(chloromethyl)-4-fluoro-1,4-diazoniabicyclo[2.2.2]octane bis(hexafluorophosphate); silver(l) oxide In acetone at 65℃; for 4h; Sealed vial; Inert atmosphere; | 80% |
| With copper(II) bis(trifluoromethanesulfonate); tetrabutylammonium triphenyldifluorosilicate In acetonitrile at 23 - 60℃; for 3h; Sealed tube; regiospecific reaction; | 60% |
-
-
210422-66-5
4-fluorobenzaldehyde p-toluenesulfonylhydrazone

-
-
459-56-3
4-fluorobenzylic alcohol

| Conditions | Yield |
|---|---|
| With water; potassium carbonate at 130℃; for 0.166667h; Microwave irradiation; | 80% |

| Conditions | Yield |
|---|---|
| With methyldiphenylsilane; palladium on activated charcoal In water; acetonitrile at 80℃; under 7500.75 Torr; for 9h; | 80% |

-
-
459-56-3
4-fluorobenzylic alcohol

| Conditions | Yield |
|---|---|
| With methanol; sodium tetrahydroborate; nickel(II) chloride hexahydrate at 20℃; for 2h; | 80% |
-
-
18539-44-1
(±)-2-[(4-fluorobenzyl)oxy]tetrahydropyran

-
-
459-56-3
4-fluorobenzylic alcohol

| Conditions | Yield |
|---|---|
| With 1,4-diazabicyclo[2.2.2]octane tribromide supported on magnetic Fe3O4 nanoparticles In methanol at 20℃; | 80% |
-
-
20671-52-7
lithium pyrrolide

-
-
459-57-4
4-fluorobenzaldehyde

-
A
-
459-56-3
4-fluorobenzylic alcohol

-
B
-
13169-73-8
(4-fluorophenyl)(1H-pyrrol-2-yl)methanone

-
C
-
3913-55-1
1-(4-fluorophenyl)pentan-1-ol

| Conditions | Yield |
|---|---|
| Stage #1: lithium pyrrolide With zirconium(IV) chloride In tetrahydrofuran; cyclohexane at -78℃; for 0.333333h; Inert atmosphere; Stage #2: With n-butyllithium In tetrahydrofuran; cyclohexane at -78 - 20℃; Inert atmosphere; Stage #3: 4-fluorobenzaldehyde In tetrahydrofuran; cyclohexane at 20℃; for 48h; Inert atmosphere; | A 13% B 8% C 79% |

-
-
1063716-63-1
Pd(C5H5N)(C13H8N2SO2C6H4NO2)(C6H4CH2OH)

-
-
459-56-3
4-fluorobenzylic alcohol

| Conditions | Yield |
|---|---|
| With Selectfluor In acetonitrile at 50℃; for 0.5h; Product distribution / selectivity; | 78% |
-
-
24167-56-4
N,N-dimethyl-4-fluorobenzamide

-
-
459-56-3
4-fluorobenzylic alcohol

| Conditions | Yield |
|---|---|
| With sodium hydride; sodium iodide; zinc(II) iodide In tetrahydrofuran; mineral oil at 40℃; for 7h; Sealed tube; | 76% |

| Conditions | Yield |
|---|---|
| With zinc In water at 90 - 160℃; for 2h; Microwave irradiation; Green chemistry; | A 13% B 75% |

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate; [4,4′-bis(1,1-dimethylethyl)-2,2′-bipyridine-N1,N1′]bis{3,5-difluoro-2-[5-(trifluoromethyl)-2-pyridinyl-κN]phenyl-κC}iridium(III) hexafluorophosphate; oxygen; caesium carbonate In dichloromethane at 40℃; for 40h; Irradiation; | 75% |

| Conditions | Yield |
|---|---|
| With C24H20ClN2OPRu; potassium tert-butylate; hydrogen In tetrahydrofuran at 110℃; under 10640.7 Torr; for 36h; Inert atmosphere; Schlenk technique; | 73% |
-
-
459-57-4
4-fluorobenzaldehyde

-
-
103-49-1
dibenzylamine

-
A
-
459-56-3
4-fluorobenzylic alcohol

-
B
-
359445-99-1
N,N-dibenzyl-1-(4-fluorophenyl)methanamine

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate; carbon dioxide In tetrahydrofuran at 20℃; Solvent; | A 27% B 72% |
| With sodium tetrahydroborate; carbon dioxide In 1,4-dioxane at 20℃; Solvent; Reagent/catalyst; | A 54% B 35% |

-
-
459-57-4
4-fluorobenzaldehyde

-
A
-
459-56-3
4-fluorobenzylic alcohol

-
B
-
24133-58-2
1,2-bis(4-fluorophenyl)-1,2-ethanediol

| Conditions | Yield |
|---|---|
| With triethylamine; 2-hydroxyethanethiol In acetonitrile for 23h; Irradiation; | A 70% B 18% |
| With lithium sulfate; sulfuric acid; trilithium citrate; lithium chloride In methanol; water Product distribution; Mechanism; Ambient temperature; electrolysis in membrane flow cell with Pb cathode at several pH and cathode potential; | |
| With lithium sulfate; sulfuric acid; trilithium citrate; lithium chloride In methanol; water Ambient temperature; electrolysis in membrane flow cell with a Pb cathode; |

| Conditions | Yield |
|---|---|
| With phosphorus tribromide In benzene at 20℃; | 100% |
| With 1,1,1,2,2,2-hexamethyldisilane In chloroform for 2.5h; Reflux; | 98% |
| With trimethylsilyl bromide In neat (no solvent) at 20℃; for 18h; Green chemistry; chemoselective reaction; | 98% |

| Conditions | Yield |
|---|---|
| With oxygen; perruthenate modified mesoporous silicate MCM-41 In toluene at 80℃; for 1h; Oxidation; | 100% |
| With 2,2,6,6-tetramethyl-piperidine-N-oxyl; oxygen; copper(I) bromide dimethylsulfide complex In chlorobenzene at 90℃; for 6h; | 100% |
| With iron(III) chloride; 4-acetylamino-2,2,6,6-tetramethyl-1-piperidinoxy; oxygen; sodium nitrite In 1,2-dichloro-ethane at 50℃; under 3000.3 Torr; for 5h; Autoclave; | 99% |
-
-
459-56-3
4-fluorobenzylic alcohol

-
-
18162-48-6
tert-butyldimethylsilyl chloride

-
-
113984-76-2
1-(tert-butyldimethylsilyloxymethyl)-4-fluorobenzene

| Conditions | Yield |
|---|---|
| With 1H-imidazole In tetrahydrofuran at 25℃; | 100% |
| With 1H-imidazole In N,N-dimethyl-formamide at 0 - 20℃; for 18h; | 99% |
| With 1H-imidazole In N,N-dimethyl-formamide at 0 - 20℃; for 18h; | 99% |

| Conditions | Yield |
|---|---|
| With sodium hypochlorite; sodium hydrogencarbonate for 1.33333h; | 100% |
| With diethylene glycol dimethyl ether at 70℃; for 0.5h; Sonication; | 97% |
| With oxygen at 120℃; for 10h; Green chemistry; | 95% |

| Conditions | Yield |
|---|---|
| With thalium(III) chloride tetrahydrate at 20℃; for 0.0666667h; | 99% |
| With polyvinylpolypyrrolidone-bound boron trifluoride In acetonitrile at 20℃; for 2h; | 99% |
| With tribromomelamine In dichloromethane at 20℃; for 0.833333h; Green chemistry; chemoselective reaction; | 99% |
-
-
459-56-3
4-fluorobenzylic alcohol

-
-
18162-48-6
tert-butyldimethylsilyl chloride

| Conditions | Yield |
|---|---|
| With 1H-imidazole In N,N-dimethyl-formamide at 0 - 20℃; for 18h; | 99% |

| Conditions | Yield |
|---|---|
| Stage #1: 4-fluorobenzylic alcohol With 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical; iodine In dichloromethane at 20℃; for 1h; Inert atmosphere; Stage #2: With ammonia; iodine In dichloromethane; water at 20℃; for 2h; Inert atmosphere; | 99% |
| Stage #1: 4-fluorobenzylic alcohol With 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical; iodine In dichloromethane at 20℃; for 1h; Stage #2: With ammonium hydroxide In dichloromethane at 20℃; for 2h; Reagent/catalyst; | 99% |
| With ammonia; oxygen In tert-Amyl alcohol; water at 100℃; under 3750.38 Torr; for 5h; Autoclave; High pressure; | 99% |

| Conditions | Yield |
|---|---|
| at 75℃; | 99% |
-
-
459-56-3
4-fluorobenzylic alcohol

-
-
137-07-5
2-amino-benzenethiol

-
-
1629-26-1
2-(4-fluorophenyl)-benzothiazole

| Conditions | Yield |
|---|---|
| With potassium tert-butylate In toluene at 20 - 110℃; for 48h; | 99% |
| With C15H20MnN2O3S(1+)*Br(1-); potassium hydroxide In neat (no solvent) at 140℃; Inert atmosphere; Green chemistry; | 76% |
| With sodium t-butanolate In toluene at 120℃; for 24h; Schlenk technique; Sealed tube; Green chemistry; | 37% |

| Conditions | Yield |
|---|---|
| With potassium hydroxide In diethyl ether at 20℃; for 12h; | 99% |

| Conditions | Yield |
|---|---|
| With potassium tert-butylate In toluene at 120℃; for 3h; Inert atmosphere; | 99% |
| With [ruthenium(II)(η6-1-methyl-4-isopropyl-benzene)(chloride)(μ-chloride)]2; potassium tert-butylate In toluene at 120℃; for 18h; Inert atmosphere; Sealed tube; | 90% |
-
-
459-56-3
4-fluorobenzylic alcohol

-
-
999-97-3
1,1,1,3,3,3-hexamethyl-disilazane

-
-
14629-55-1
(4-fluorobenzyloxy)trimethylsilane

| Conditions | Yield |
|---|---|
| With Nafion SAC-13 at 20℃; for 0.0666667h; | 98% |
| Stage #1: 1,1,1,3,3,3-hexamethyl-disilazane With C12H24KO6(1+)*Br3H(1-) In acetonitrile at 20℃; for 0.0166667h; Stage #2: 4-fluorobenzylic alcohol In acetonitrile at 20℃; for 0.116667h; | 97% |
| With 1,4-diazabicyclo[2.2.2]octane tribromide supported on magnetic Fe3O4 nanoparticles In neat (no solvent) at 20℃; for 0.666667h; | 97% |

| Conditions | Yield |
|---|---|
| With hydrogen iodide for 1h; Heating; | 98% |
| With chloro-trimethyl-silane; sodium iodide In acetonitrile 0 deg C, 10 min then up to r.t., 1 h; | 97% |
| With natural kaolinitic clay; sulfuric acid; potassium iodide In various solvent(s) for 0.0833333h; Irradiation; microwave; | 82% |
| With bis(1,5-cyclooctadiene)rhodium(I) trifluoromethanesulfonate; hydrogen; iodine; 2,2'-bis-(diphenylphosphino)-1,1'-binaphthyl In 1,2-dichloro-ethane at 20℃; under 22502.3 Torr; Autoclave; Schlenk technique; | 78% |
| With potassium fluoride; tetra-(n-butyl)ammonium iodide; N-ethyl-N,N-diisopropylamine; N,N`-sulfuryldiimidazole In N,N-dimethyl-formamide; trifluoroacetic acid at 20℃; for 0.2h; | 70% |
-
-
459-56-3
4-fluorobenzylic alcohol

-
-
767-00-0
4-cyanophenol

-
-
56442-14-9
4-((4'-fluoro)benzyloxy)benzonitrile

| Conditions | Yield |
|---|---|
| With F-DEAD-2; bis-phenyl-[4-(3,3,4,4,5,5,6,6,7,7,8,8,9,9,10,10,10-heptadecafluorodecyl)phenyl]phosphine In tetrahydrofuran at 20℃; for 3h; Mitsunobu reaction; | 98% |
| With bis-(2-(1-adamantyl)ethyl) azodicarboxylate; triphenylphosphine Mitsunobu reaction; | 95% |
| With bis(3-(perfluoro-t-butyloxy)propyl)diazodicarboxylate; bis-phenyl-[4-(3,3,4,4,5,5,6,6,7,7,8,8,9,9,10,10,10-heptadecafluorodecyl)phenyl]phosphine In tetrahydrofuran at 20℃; for 8h; Mitsunobu reaction; Inert atmosphere; | 90% |
-
-
851166-94-4
4-β-isocyanate-4'-tert-butyldimethylsilyloxy-4'-O-demethyl-4-desoxypodophyllotoxin

-
-
459-56-3
4-fluorobenzylic alcohol

-
-
851167-14-1
4-β-(4-fluorobenzyloxycarbonyl)amino-4'-tert-butyldimethylsilyloxy-4'-O-demethyl-4-desoxypodophyllotoxin

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; | 98% |
-
-
459-56-3
4-fluorobenzylic alcohol

-
-
105-45-3
acetoacetic acid methyl ester

-
-
57-13-6
urea

-
-
283593-12-4
4-(4-fluorophenyl)-5-(methoxycarbonyl)-6-methyl-3,4-dihydropyrimidin-2(1H)-one

| Conditions | Yield |
|---|---|
| at 80℃; for 0.75h; | 98% |
| Stage #1: 4-fluorobenzylic alcohol With bismuth(III) nitrate In various solvent(s) at 50℃; for 0.0833333h; Stage #2: acetoacetic acid methyl ester; urea In various solvent(s) at 90℃; | 88% |


| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; ammonia; oxygen In water; N,N-dimethyl-formamide at 80℃; under 1520.1 Torr; for 5h; Green chemistry; | 98% |
| With tert.-butylhydroperoxide; tetraethylammonium iodide; ammonium bicarbonate In 1,2-dichloro-ethane at 70℃; for 22h; | 77% |
| With tert.-butylhydroperoxide; ammonium hydroxide In water at 100℃; for 16h; | 72% |
| Multi-step reaction with 2 steps 1: cesium carbonate; styrene / iridium pentamethylcyclopentadienyl dichloride dimer / toluene / 24 h / 111 °C 2: 117 mg / hydroxylamine hydrochloride / iridium pentamethylcyclopentadienyl dichloride dimer / toluene / 16 h / 111 °C View Scheme | |
| With ammonium acetate; oxygen In tert-Amyl alcohol at 130℃; under 3800.26 Torr; for 18h; Reagent/catalyst; Autoclave; | 61 %Chromat. |

| Conditions | Yield |
|---|---|
| With 2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine In 1,2,4-Trimethylbenzene at 160℃; for 15h; Inert atmosphere; | 98% |
| With C55H43ClN5P2Ru(1+)*Cl(1-); potassium hydroxide In toluene at 140℃; for 2h; Sealed tube; | 93% |
| With [(η5-pentamethylcyclopentadienyl)IrIIIbis-(κC-1,3-dimethylimidazol-2-ylidene)Cl]BF4; potassium tert-butylate In toluene at 80℃; for 12h; | 92% |
-
-
459-56-3
4-fluorobenzylic alcohol

-
-
109-94-4
formic acid ethyl ester

-
-
1146963-53-2
4-fluorobenzyl formate

| Conditions | Yield |
|---|---|
| With tribromomelamine In neat (no solvent) at 20℃; for 0.733333h; Green chemistry; chemoselective reaction; | 98% |
| With poly(4-vinylpyridinium tribromide) at 20℃; for 0.833333h; neat (no solvent); | 91% |
| Stage #1: formic acid ethyl ester With sulfuric acid; silica gel at 20℃; Stage #2: 4-fluorobenzylic alcohol at 20℃; for 0.0833333h; chemoselective reaction; |

| Conditions | Yield |
|---|---|
| With polystyrene supported triphenylphosphine ruthenium complex In toluene at 120℃; for 20h; Sealed tube; Flow reactor; | 98% |
-
-
766-11-0
5-bromo-2-fluoropyridine

-
-
459-56-3
4-fluorobenzylic alcohol

-
-
936343-08-7
5-bromo-2-((4-fluorobenzyl)oxy)pyridine

| Conditions | Yield |
|---|---|
| Stage #1: 4-fluorobenzylic alcohol With sodium hydride In tetrahydrofuran at 0 - 20℃; for 1.5h; Reflux; Stage #2: 5-bromo-2-fluoropyridine In tetrahydrofuran for 12h; Reflux; | 98% |
| Stage #1: 4-fluorobenzylic alcohol With sodium hydride In tetrahydrofuran; mineral oil at 0℃; for 0.5h; Stage #2: 5-bromo-2-fluoropyridine In tetrahydrofuran; mineral oil at 0 - 80℃; for 3h; |
-
-
459-56-3
4-fluorobenzylic alcohol

-
-
540-88-5
acetic acid tert-butyl ester

-
-
1049037-23-1
tert-butyl 3-(4-fluorophenyl)propanoate

| Conditions | Yield |
|---|---|
| With tri-tert-butyl phosphine; potassium tert-butylate; nickel diacetate In 1,4-dioxane; toluene at 100℃; for 12h; Inert atmosphere; Glovebox; Schlenk technique; | 98% |
| With C22H34Cl2CoF3N5P2; potassium tert-butylate In toluene at 80℃; for 4h; Concentration; Inert atmosphere; Sealed tube; Glovebox; | 82% |
| With Ru(η(2)-2-(2'-pyridyl)phenyl)Cl(CO)(PPh3)2; potassium tert-butylate In toluene at 80℃; for 1.5h; Inert atmosphere; Schlenk technique; Green chemistry; | 82% |
| With C15H17ClIrNOP; potassium tert-butylate In toluene at 60℃; for 12h; Schlenk technique; Inert atmosphere; Glovebox; | 79% |

| Conditions | Yield |
|---|---|
| at 20℃; | 98% |

| Conditions | Yield |
|---|---|
| With bis-(2-(1-adamantyl)ethyl) azodicarboxylate; triphenylphosphine In tetrahydrofuran at 20℃; Mitsunobu reaction; | 97% |
| With PhP(C6H4-p-(CH2)2C6F13)2; bis(3,3,4,4,5,5,6,6,7,7,8,8,8-tridecafluorooctyl)diazo dicarboxylate In tetrahydrofuran |
4-Fluorobenzyl alcohol Specification
The 4-Fluorobenzyl alcohol with CAS registry number of 459-56-3 is also known as Benzenemethanol,4-fluoro-. The IUPAC name is (4-Flluorophenyl)methanol. It belongs to product categories of Benzhydrols, Benzyl & Special Alcohols; Alcohol; Fluorobenzene; Alcohols; Fluorine Compounds; C7 to C8;Oxygen Compounds. Its EINECS registry number is 207-292-0. In addition, the formula is C7H7FO and the molecular weight is 126.13. This chemical is a colorless or slightly yellow liquid that may cause inflammation to the skin or other mucous membranes and may destroy living tissue on contact. What's more, this chemical is used as intermediate in organic synthesis and should be sealed in ventilated, dry place away from oxidants.
Physical properties about 4-Fluorobenzyl alcohol are: (1)ACD/LogP: 1.09; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 1.09; (4)ACD/LogD (pH 7.4): 1.09; (5)ACD/BCF (pH 5.5): 3.94; (6)ACD/BCF (pH 7.4): 3.94; (7)ACD/KOC (pH 5.5): 92.88; (8)ACD/KOC (pH 7.4): 92.88; (9)#H bond acceptors: 1; (10)#H bond donors: 1; (11)#Freely Rotating Bonds: 2; (12)Index of Refraction: 1.52; (13)Molar Refractivity: 32.69 cm3; (14)Molar Volume: 107.4 cm3; (15)Surface Tension: 38.6 dyne/cm; (16)Density: 1.173 g/cm3; (17)Flash Point: 94.6 °C; (18)Enthalpy of Vaporization: 46.33 kJ/mol; (19)Boiling Point: 202 °C at 760 mmHg; (20)Vapour Pressure: 0.183 mmHg at 25 °C.
Preparation of 4-Fluorobenzyl alcohol: it is prepared by reaction of 4-fluoro-benzaldehyde. The reaction needs reagent sulfurated borohydride exchange resin and solvent methanol at the temperature of 25 °C for 5 minutes. The yield is about 93 %.

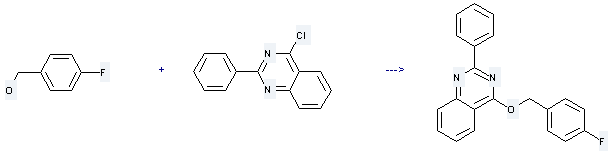
Uses of 4-Fluorobenzyl alcohol: it is used to produce 4-(4-fluorobenzyloxy)-2-phenylquinazoline by reaction with 4-chloro-2-phenyl-quinazoline. The reaction occurs with reagent NaH and other condition of heating. The yield is about 96 %.
When you are using this chemical, please be cautious about it. As a chemical, it is irritating to eyes and respiratory system. Besides, it reacts violently with water and also causes burns. During using it, wear suitable protective clothing, gloves and eye/face protection. Do not breathe gas/fumes/vapour/spray and avoid contact with skin and eyes. In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. If accident happens or you feel unwell seek medical advice immediately. After using it, take off immediately all contaminated clothing.
You can still convert the following datas into molecular structure:
1. Canonical SMILES: C1=CC(=CC=C1CO)F
2. InChI: InChI=1S/C7H7FO/c8-7-3-1-6(5-9)2-4-7/h1-4,9H,5H2
3. InChIKey: GEZMEIHVFSWOCA-UHFFFAOYSA-N
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View