-
Name
4-Nitro-2,3-lutidine-N-oxide
- EINECS -0
- CAS No. 37699-43-7
- Article Data18
- CAS DataBase
- Density 1.3 g/cm3
- Solubility
- Melting Point 94-98 °C(lit.)
- Formula C7H8N2O3
- Boiling Point 403.3 °C at 760 mmHg
- Molecular Weight 168.152
- Flash Point 197.7 °C
- Transport Information UN 2811
- Appearance
- Safety 26-36
- Risk Codes 36/37/38-20/21/22
-
Molecular Structure
-
Hazard Symbols
 Xn,
Xn,  Xi
Xi
- Synonyms 2,3-Lutidine,4-nitro-, 1-oxide (7CI);2,3-Dimethyl-4-nitropyridine 1-oxide;2,3-Dimethyl-4-nitropyridine N-oxide;4-Nitro-2,3-dimethylpyridine N-oxide;
- PSA 71.28000
- LogP 2.16330
Synthetic route
-
-
22710-07-2
2,3-dimethylpyridine 1-oxide

-
-
37699-43-7
2,3-dimethyl-4-nitropyridine N-oxide

| Conditions | Yield |
|---|---|
| With potassium nitrite; sulfuric acid at -10 - 85℃; for 2h; Temperature; Green chemistry; | 92.9% |
| With sulfuric acid; nitric acid at 100℃; for 5h; | 78% |
| With sulfuric acid; nitric acid at 95℃; for 20h; | 53% |
-
-
5470-66-6
2-methyl-4-nitropyridine N-oxide

-
-
37699-43-7
2,3-dimethyl-4-nitropyridine N-oxide

| Conditions | Yield |
|---|---|
| Stage #1: 2-methyl-4-nitropyridine N-oxide; methylmagnesium halide In tetrahydrofuran at -60℃; Inert atmosphere; Stage #2: With 2,3-dicyano-5,6-dichloro-p-benzoquinone In tetrahydrofuran at -60 - 20℃; Inert atmosphere; regioselective reaction; | 47% |
-
-
22710-07-2
2,3-dimethylpyridine 1-oxide

-
A
-
35392-67-7
3-methoxy-2-methyl-6-nitropyridine 1-oxide

-
B
-
37699-43-7
2,3-dimethyl-4-nitropyridine N-oxide

| Conditions | Yield |
|---|---|
| With nitric acid In acetic acid at 80℃; for 33h; |
-
-
22710-07-2
2,3-dimethylpyridine 1-oxide

-
-
75-09-2
dichloromethane

-
B
-
37699-43-7
2,3-dimethyl-4-nitropyridine N-oxide

| Conditions | Yield |
|---|---|
| With sodium hydroxide; sulfuric acid; nitric acid In water |

-
-
22710-07-2
2,3-dimethylpyridine 1-oxide

-
-
497-19-8
sodium carbonate

-
A
-
2403-02-3
4-carboxy-2-nitropyridine N-oxide

-
B
-
37699-43-7
2,3-dimethyl-4-nitropyridine N-oxide

| Conditions | Yield |
|---|---|
| With nitric acid |

-
-
583-61-9
2,3-Lutidine

-
-
37699-43-7
2,3-dimethyl-4-nitropyridine N-oxide

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: dihydrogen peroxide; acetic acid / 24 h / Reflux 2: nitric acid; sulfuric acid / 24 h / 90 °C View Scheme | |
| Multi-step reaction with 2 steps 1: 3-chloro-benzenecarboperoxoic acid / chloroform / 18 h / 20 °C 2: nitric acid; sulfuric acid / 95 °C View Scheme | |
| Multi-step reaction with 2 steps 1: 3-chloro-benzenecarboperoxoic acid / dichloromethane / 0 - 20 °C 2: sulfuric acid; nitric acid / water / 5 h / 95 °C View Scheme |
-
-
37699-43-7
2,3-dimethyl-4-nitropyridine N-oxide

-
-
59886-90-7
4-chloro-2,3-dimethylpyridine-N-oxide

| Conditions | Yield |
|---|---|
| With acetyl chloride In ethanol at 65℃; for 5h; | 100% |
| With hydrogenchloride; sodium chloride; benzyltri(n-butyl)ammonium chloride In water; acetonitrile for 12h; Heating / reflux; | 98.5% |
| Stage #1: 2,3-dimethyl-4-nitropyridine N-oxide With hydrogenchloride; benzyltri(n-butyl)ammonium chloride; sodium chloride In water; acetonitrile for 12h; Heating / reflux; Stage #2: In water; acetonitrile pH=9; | 98.5% |
-
-
37699-43-7
2,3-dimethyl-4-nitropyridine N-oxide

-
-
2516-33-8
Cyclopropylmethanol

-
-
206990-47-8
4-Cyclopropylmethoxy-2,3-dimethyl-pyridine 1-oxide

| Conditions | Yield |
|---|---|
| With benzyltri(n-butyl)ammonium chloride; potassium hydrogencarbonate In acetonitrile for 24h; Heating; | 100% |
-
-
67-56-1
methanol

-
-
37699-43-7
2,3-dimethyl-4-nitropyridine N-oxide

-
-
102625-96-7
4-methoxy-2,3-dimethylpyridine 1-oxide

| Conditions | Yield |
|---|---|
| With potassium carbonate for 2h; Heating; | 99% |
| With potassium carbonate at 0 - 65℃; for 4h; | 83% |
| With potassium carbonate for 20h; Heating; | 82% |
| With potassium carbonate In acetonitrile at 0℃; Reflux; | 47% |
| With sodium methylate at 40℃; for 16h; |
-
-
37699-43-7
2,3-dimethyl-4-nitropyridine N-oxide

-
-
75-36-5
acetyl chloride

-
-
59886-90-7
4-chloro-2,3-dimethylpyridine-N-oxide

| Conditions | Yield |
|---|---|
| Stage #1: 2,3-dimethyl-4-nitropyridine N-oxide; acetyl chloride In ethanol at 65℃; Large scale reaction; Stage #2: With sodium hydroxide In ethanol; water; toluene pH=7.5 - 8.5; Large scale reaction; | 96% |
-
-
37699-43-7
2,3-dimethyl-4-nitropyridine N-oxide

-
-
68707-69-7
2,3-dimethyl-4-nitropyridine

| Conditions | Yield |
|---|---|
| Stage #1: 2,3-dimethyl-4-nitropyridine N-oxide With phosphorus trichloride In dichloromethane at -20 - 15℃; for 1h; Stage #2: With sodium hydroxide; water In dichloromethane at -78℃; | 95% |
| With phosphorus trichloride In dichloromethane -20 degC, 15 min, r.t., 15 min; | 92% |
-
-
37699-43-7
2,3-dimethyl-4-nitropyridine N-oxide

-
-
75-89-8
2,2,2-trifluoroethanol

-
-
103577-61-3
2,3-dimethyl-4-(2,2,2-trifluoroethoxy)-pyridine N-oxide

| Conditions | Yield |
|---|---|
| With potassium tert-butylate at 50 - 60℃; for 42h; | 94% |
| With potassium tert-butylate; palladium dichloride In dichloromethane at 83 - 88℃; for 6 - 8h; | 95 - 97 %Chromat. |
| With N-benzyl-N,N,N-triethylammonium chloride; potassium carbonate In water; butanone at 20℃; Concentration; Temperature; Time; Pressure; |
-
-
37699-43-7
2,3-dimethyl-4-nitropyridine N-oxide

-
-
122475-57-4
4-amino-2,3-dimethylpyridine

| Conditions | Yield |
|---|---|
| With hydrogen; acetic acid; palladium on activated charcoal at 60℃; under 2585.7 Torr; for 12h; | 94% |
| With iron; acetic acid In water | 76% |
| With iron In acetic acid at 100℃; for 2h; | 56% |
-
-
37699-43-7
2,3-dimethyl-4-nitropyridine N-oxide

-
-
259807-92-6
2,3-dimethyl-4-bromopyridine N-oxide

| Conditions | Yield |
|---|---|
| With Acetyl bromide In chloroform for 1h; Heating; | 93% |
| With hydrogen bromide In acetic acid at 100℃; for 48h; Heating; |
-
-
37699-43-7
2,3-dimethyl-4-nitropyridine N-oxide

-
-
147440-89-9
3-methyl-4-nitro-2-pyridinecarboxaldehyde-N-oxide

| Conditions | Yield |
|---|---|
| With selenium(IV) oxide In 1,4-dioxane for 48h; Heating; | 88% |
-
-
37699-43-7
2,3-dimethyl-4-nitropyridine N-oxide

-
-
76-37-9
2,2,3,3-tetrafluoropropanol

-
-
103577-57-7
2,3-dimethyl-4-(2,2,3,3-tetrafluoropropoxy)pyridine-1-oxide

| Conditions | Yield |
|---|---|
| With potassium tert-butylate at 50 - 60℃; | 86% |
-
-
37699-43-7
2,3-dimethyl-4-nitropyridine N-oxide

-
-
107-19-7
propargyl alcohol

-
-
206990-49-0
2,3-Dimethyl-4-prop-2-ynyloxy-pyridine 1-oxide

| Conditions | Yield |
|---|---|
| With benzyltri(n-butyl)ammonium chloride; potassium carbonate In acetonitrile at 90℃; for 3h; | 86% |
-
-
37699-43-7
2,3-dimethyl-4-nitropyridine N-oxide

-
-
1942-46-7
5-decyne

| Conditions | Yield |
|---|---|
| With 1,1,1,3',3',3'-hexafluoro-propanol; 9-(2-mesityl)-10-methylacridinium perchlorate In acetone at 50℃; for 48h; Schlenk technique; Glovebox; Inert atmosphere; Sealed tube; Irradiation; regioselective reaction; | 86% |
-
-
37699-43-7
2,3-dimethyl-4-nitropyridine N-oxide

-
B
-
122475-57-4
4-amino-2,3-dimethylpyridine

| Conditions | Yield |
|---|---|
| With hydrogenchloride; titanium(III) chloride In water; acetonitrile at 20℃; for 1h; | A 17% B 83% |
-
-
37699-43-7
2,3-dimethyl-4-nitropyridine N-oxide

-
-
355-80-6
2,2,3,3,4,4,5,5-octafluoropentan-1-ol

-
-
132332-23-1
2,3-Dimethyl-4-(2,2,3,3,4,4,5,5-octafluoro-pentyloxy)-pyridine 1-oxide

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetonitrile at 90 - 100℃; | 81% |
-
-
37699-43-7
2,3-dimethyl-4-nitropyridine N-oxide

-
-
422-05-9
2,2,3,3,3-pentafluoropropyl alcohol

-
-
103577-60-2
2,3-Dimethyl-4-(2,2,3,3,3-pentafluoropropoxy)pyridine N-oxide

| Conditions | Yield |
|---|---|
| With N,N,N,N,N,N-hexamethylphosphoric triamide; potassium carbonate In butanone at 70 - 80℃; for 108h; | 74% |
-
-
37699-43-7
2,3-dimethyl-4-nitropyridine N-oxide

-
-
375-01-9
2,2,3,3,4,4,4-heptafluorobutanol

-
-
132332-22-0
2,3-Dimethyl-4-(2,2,3,3,4,4,4-heptafluorobutoxy)pyridine N-oxide

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetonitrile at 90 - 100℃; for 41h; | 73% |
-
-
37699-43-7
2,3-dimethyl-4-nitropyridine N-oxide

-
-
501-65-5
diphenyl acetylene

| Conditions | Yield |
|---|---|
| Stage #1: 2,3-dimethyl-4-nitropyridine N-oxide; diphenyl acetylene With tetrafluoroboric acid; 9-(2-mesityl)-10-methylacridinium perchlorate In acetonitrile at 50℃; for 24h; Schlenk technique; Glovebox; Inert atmosphere; Irradiation; Stage #2: In acetonitrile at 50℃; for 12h; Schlenk technique; Glovebox; Irradiation; | 67% |
-
-
37699-43-7
2,3-dimethyl-4-nitropyridine N-oxide

-
-
1942-46-7
5-decyne

| Conditions | Yield |
|---|---|
| With tetrafluoroboric acid; oxygen; 9-(2-mesityl)-10-methylacridinium perchlorate In acetonitrile at 50℃; for 12h; Schlenk technique; Irradiation; | 65% |
-
-
37699-43-7
2,3-dimethyl-4-nitropyridine N-oxide

-
-
100-51-6
benzyl alcohol

-
-
102127-29-7
4-(benzyloxy)-2,3-dimethylpyridin-N-oxide

| Conditions | Yield |
|---|---|
| With benzyltri(n-butyl)ammonium chloride; potassium carbonate In acetonitrile at 90℃; for 20h; | 57% |
-
-
37699-43-7
2,3-dimethyl-4-nitropyridine N-oxide

-
-
7143-01-3
Methanesulfonic anhydride

| Conditions | Yield |
|---|---|
| In dichloromethane for 12h; Heating; | 54% |
-
-
37699-43-7
2,3-dimethyl-4-nitropyridine N-oxide

-
-
109-86-4
2-methoxy-ethanol

-
-
206990-48-9
4-(2-Methoxy-ethoxy)-2,3-dimethyl-pyridine 1-oxide

| Conditions | Yield |
|---|---|
| With potassium carbonate for 20h; Heating; | 53% |
-
-
37699-43-7
2,3-dimethyl-4-nitropyridine N-oxide

-
-
108-24-7
acetic anhydride

-
-
166521-98-8
(3-methyl-4-nitropyridin-2-yl)methyl acetate

| Conditions | Yield |
|---|---|
| at 100℃; for 3h; | 49% |
| at 100℃; for 1.5h; | 11.5 g |
-
-
37699-43-7
2,3-dimethyl-4-nitropyridine N-oxide

-
-
4124-41-8
p-toluenesulfonylanhydride

| Conditions | Yield |
|---|---|
| In dichloromethane for 12h; Heating; | 37% |
-
-
37699-43-7
2,3-dimethyl-4-nitropyridine N-oxide

| Conditions | Yield |
|---|---|
| Stage #1: 2,3-dimethyl-4-nitropyridine N-oxide With trichlorophosphate In dichloromethane at 20℃; for 24h; Stage #2: With triethylamine In dichloromethane at 0℃; for 0.5h; | 20% |
| Multi-step reaction with 4 steps 1: 75 percent / phosphorus oxychloride / CH2Cl2 / 20 °C 2: 63 percent / 3 h / 100 °C 3: 78 percent / aq. hydrochloric acid / 24 h / 20 °C 4: 43 percent / thionyl chloride / CH2Cl2 / 1 h / Heating View Scheme | |
| Multi-step reaction with 2 steps 1: 75 percent / phosphorus oxychloride / CH2Cl2 / 20 °C 2: 37 percent / phosphorus oxychloride; triethylamine / CH2Cl2 / 2 h / 20 °C View Scheme |
-
-
37699-43-7
2,3-dimethyl-4-nitropyridine N-oxide

-
-
7677-24-9
trimethylsilyl cyanide

-
-
1323393-00-5
4-chloro-5,6-dimethyl-2-cyanopyridine

| Conditions | Yield |
|---|---|
| With chloroformic acid ethyl ester In 1,2-dichloro-ethane at -5 - 40℃; | 18% |
-
-
37699-43-7
2,3-dimethyl-4-nitropyridine N-oxide

-
-
64-17-5
ethanol

-
-
1034065-92-3
4-ethoxy-2,3-dimethylpyridine N-oxide

| Conditions | Yield |
|---|---|
| With sodium ethanolate at 70℃; for 24h; Heating; |
4-Nitro-2,3-lutidine-N-oxide Chemical Properties
Molecular structure of 4-Nitro-2,3-lutidine-N-oxide (CAS NO.37699-43-7) is:
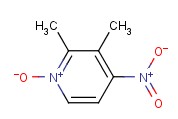
Product Name: 4-Nitro-2,3-lutidine-N-oxide
CAS Registry Number: 37699-43-7
IUPAC Name: 2,3-dimethyl-4-nitro-1-oxidopyridin-1-ium
Molecular Weight: 168.15002 [g/mol]
Molecular Formula: C7H8N2O3
XLogP3-AA: 0.5
H-Bond Donor: 0
H-Bond Acceptor: 3
Melting Point: 94-98 °C(lit.)
Surface Tension: 49.2 dyne/cm
Density: 1.3 g/cm3
Flash Point: 197.7 °C
Enthalpy of Vaporization: 62.9 kJ/mol
Boiling Point: 403.3 °C at 760 mmHg
Vapour Pressure: 2.4E-06 mmHg at 25°C
Product Categories: Pyridines;Pyridines derivates;C7 and C8;Heterocyclic Building Blocks
4-Nitro-2,3-lutidine-N-oxide Uses
4-Nitro-2,3-lutidine-N-oxide (CAS NO.37699-43-7) is used as an intermediate of drug lansoprazole.
4-Nitro-2,3-lutidine-N-oxide Toxicity Data With Reference
| 1. | mmo-sat 100 nmol/plate | GANNA2 Gann. Japanese Journal of Cancer Research. 70 (1979),799. | ||
| 2. | dnr-esc 500 µg/well | CNREA8 Cancer Research. 32 (1972),2369. |
4-Nitro-2,3-lutidine-N-oxide Safety Profile
Safty information about 4-Nitro-2,3-lutidine-N-oxide (CAS NO.37699-43-7) is:
Hazard Codes:  Xi,Xn
Xi,Xn
Risk Statements: 36/37/38-20/21/22
R36/37/38:Irritating to eyes, respiratory system and skin.
R20/21/22:Harmful by inhalation, in contact with skin and if swallowed.
Safety Statements: 26-36
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S36:Wear suitable protective clothing.
RIDADR: 2811
WGK Germany: 3
RTECS: UT2807000
HazardClass: 6.1
PackingGroup: III
4-Nitro-2,3-lutidine-N-oxide Specification
4-Nitro-2,3-lutidine-N-oxide , its cas register number is 37699-43-7. It also can be called 2,3-Dimethyl-4-nitropyridine 1-oxide ; Pyridine, 2,3-dimethyl-4-nitro-, 1-oxide .
Related Products
- 4-Nitro-2-(2,5,5-trimethyl-[1,3]dioxan-2-yl)benzoic acid
- 4-Nitro-2-(2'-oxybenzylidenamino)diphenylamine
- 4-Nitro-2-(3'-nitrosalicylidenamino)diphenylamine
- 4-Nitro-2-(5'-bromosalicylidenamino)diphenylamine
- 4-Nitro-2-(5'-chlorosalicylidenamino)diphenylamine
- 4-Nitro-2-(p-chlorobenzylidenamino)diphenylamine
- 4-Nitro-2-(p-diphenylaminobenzylidenamino)diphenylamine
- 4-Nitro-2-(p-nitrobenzylidenamino)diphenylamine
- 4-Nitro-2-(p-oxybenzylidenamino)diphenylamine
- 4-Nitro-2-(trifluoromethyl)benzoic acid
- 37699-47-1
- 3770-22-7
- 3770-50-1
- 37705-82-1
- 3770-82-9
- 3770-97-6
- 37711-24-3
- 37718-11-9
- 3771-95-7
- 37721-71-4
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View