-
Name
4-Pentyn-1-ol
- EINECS 226-383-6
- CAS No. 5390-04-5
- Article Data37
- CAS DataBase
- Density 0.911 g/cm3
- Solubility Miscible with water.
- Melting Point -24.1°C (estimate)
- Formula C5H8O
- Boiling Point 153.999 °C at 760 mmHg
- Molecular Weight 84.1179
- Flash Point 61.667 °C
- Transport Information UN 1987
- Appearance Clear yellow liquid
- Safety 26-36-37/39
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms (3-Hydroxypropyl)acetylene;1-Hydroxy-4-pentyne;1-Pentyn-5-ol;5-Hydroxy-1-pentyne;NSC 5274;Pent-4-yne-1-ol;
- PSA 20.23000
- LogP 0.39210
Synthetic route

| Conditions | Yield |
|---|---|
| With n-butyllithium In hexane at 0℃; for 2h; Solvent; Temperature; | 99.9% |
| With ammonia; sodium amide | 91% |
| With Iron(III) nitrate nonahydrate; ammonia; sodium Cooling with acetone-dry ice; | 89% |

-
-
5390-04-5
pent-1-yn-5-ol

| Conditions | Yield |
|---|---|
| With indium; water; ammonium chloride In methanol for 0.5h; Heating; | 98% |

-
-
5390-04-5
pent-1-yn-5-ol

| Conditions | Yield |
|---|---|
| With methanol; toluene-4-sulfonic acid In tetrahydrofuran at 70℃; for 24h; deprotection of alcoholic OH; | 94% |
-
-
130018-34-7
5-(2-propenyloxy)-1-pentyne

-
-
5390-04-5
pent-1-yn-5-ol

| Conditions | Yield |
|---|---|
| quinoline-2-carboxylic acid; cyclopentadienylruthenium(II) trisacetonitrile hexafluorophosphate In methanol at 30℃; for 3h; Conversion of starting material; | 94% |
| With quinoline-2-carboxylic acid; cyclopentadienylruthenium(II) trisacetonitrile hexafluorophosphate In methanol at 30℃; for 3h; | 94 % Chromat. |
-
-
197219-01-5
1-methoxy-4-pent-4-ynyloxymethyl-benzene

-
-
5390-04-5
pent-1-yn-5-ol

| Conditions | Yield |
|---|---|
| With trifluorormethanesulfonic acid In dichloromethane at 21℃; for 0.25h; | 91% |

-
-
5390-04-5
pent-1-yn-5-ol

| Conditions | Yield |
|---|---|
| With indium; water; ammonium chloride In methanol for 0.5h; Heating; | 87% |

| Conditions | Yield |
|---|---|
| With lithium aluminium tetrahydride In diethyl ether for 2h; Heating; | 85% |
| With lithium aluminium tetrahydride In tetrahydrofuran at 0 - 20℃; |
-
-
62992-46-5
1-(tetrahydropyranyloxy)-4-pentyn

-
-
5390-04-5
pent-1-yn-5-ol

| Conditions | Yield |
|---|---|
| With indium(III) triflate In methanol; water at 0 - 20℃; for 10h; | 82% |
| With pyridinium p-toluenesulfonate In ethanol at 25℃; for 3h; |

| Conditions | Yield |
|---|---|
| With bis(acetylacetonate)oxovanadium; methyl 3,5-bis((1H-1,2,4-triazol-1-yl)methyl)benzoate; oxygen; sodium acetate at 120℃; for 48h; | A 60% B 62% |
-
-
30428-24-1
nitrite de pentyne-4 ol-1

-
A
-
96-48-0
4-butanolide

-
B
-
18498-59-4
4-pentyn-1-al

-
C
-
5390-04-5
pent-1-yn-5-ol

| Conditions | Yield |
|---|---|
| In benzene at 10 - 20℃; for 18h; Irradiation; | A 2% B n/a C 38% |
| In benzene at 10 - 20℃; for 18h; Product distribution; Irradiation; examination of further acetylenic nitrites, reaction time; | A 2% B n/a C 38% |

-
-
109-99-9
tetrahydrofuran

-
A
-
1927-59-9
2-tert-butoxytetrahydrofuran

-
B
-
5390-04-5
pent-1-yn-5-ol

-
C
-
190184-80-6
2-(pent-4-yn-1-yloxy)tetrahydrofuran

| Conditions | Yield |
|---|---|
| With tert-butyl peroxyacetate at 110℃; for 12h; Product distribution; Mechanism; | A 5% B 5% C 8% D n/a E n/a F n/a |


| Conditions | Yield |
|---|---|
| With ammonia; sodium amide |

| Conditions | Yield |
|---|---|
| With n-pentylsodium; Petroleum ether | |
| With n-butyllithium; Petroleum ether |
-
-
18137-88-7
2-methyleneoxolane

-
-
5390-04-5
pent-1-yn-5-ol

| Conditions | Yield |
|---|---|
| With n-pentylsodium; Petroleum ether | |
| With n-butyllithium; Petroleum ether |

| Conditions | Yield |
|---|---|
| With tetrahydrofuran; sodium |
-
-
26274-19-1
5-bromo-3,4-dihydro-2H-pyran

-
-
5390-04-5
pent-1-yn-5-ol

| Conditions | Yield |
|---|---|
| With butylsodium; Petroleum ether |
-
-
59287-66-0
4,5-dibromo-1-pentanol

-
-
5390-04-5
pent-1-yn-5-ol

| Conditions | Yield |
|---|---|
| With ammonia; sodium amide |
-
-
14604-44-5
1-methoxy-4-pentyne

-
-
5390-04-5
pent-1-yn-5-ol

| Conditions | Yield |
|---|---|
| durch Entmethylieren; |
-
-
63093-41-4
ethyl 4-pentynoate

-
-
5390-04-5
pent-1-yn-5-ol

| Conditions | Yield |
|---|---|
| With lithium aluminium tetrahydride; diethyl ether | |
| With tetrahydrofuran; potassium m-borate |
-
-
50596-94-6
4-chloro-5-methyl-2,3-dihydro-furan

-
-
5390-04-5
pent-1-yn-5-ol

| Conditions | Yield |
|---|---|
| With lithium diisopropyl amide |
-
-
30428-24-1
nitrite de pentyne-4 ol-1

-
-
5390-04-5
pent-1-yn-5-ol

| Conditions | Yield |
|---|---|
| (photolysis); |


-
-
6581-54-0
3-chlorotetrahydropyran

-
-
7664-41-7
ammonia

-
A
-
110-87-2
3,4-dihydro-2H-pyran

-
B
-
5390-04-5
pent-1-yn-5-ol

-
-
109-99-9
tetrahydrofuran

-
-
6581-49-3
5-chloro-3,4-dihydro-2H-pyran

-
A
-
110-87-2
3,4-dihydro-2H-pyran

-
B
-
5390-04-5
pent-1-yn-5-ol

-
-
110-87-2
3,4-dihydro-2H-pyran

-
-
1822-71-5
n-pentylsodium

-
A
-
31518-14-6
3,4-dihydro-2H-pyran-6-carboxylic acid

-
B
-
5390-04-5
pent-1-yn-5-ol

| Conditions | Yield |
|---|---|
| anschliessend mit festem Kohlendioxid; |


| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: tetrachloromethane / 25 - 35 °C / Einleiten von Chlor in Gegenwart von Jod 2: lithium alanate; diethyl ether 3: sodium amide; liquid ammonia View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: lithium alanate; diethyl ether 2: sodium amide; liquid ammonia View Scheme |

| Conditions | Yield |
|---|---|
| With pyridinium p-toluenesulfonate In dichloromethane for 4.5h; Ambient temperature; | 100% |
| With hydrogenchloride In dichloromethane Ambient temperature; | 100% |
| With toluene-4-sulfonic acid In dichloromethane at 20℃; for 2.5h; | 100% |

| Conditions | Yield |
|---|---|
| With C20H44Cl4N8O4P4Pd2S2; water at 30℃; for 9h; Reagent/catalyst; | 100% |
| With cisplatin In water at 37℃; for 120h; | 67% |
| With water; mercury(II) sulfate |
-
-
5390-04-5
pent-1-yn-5-ol

-
-
18162-48-6
tert-butyldimethylsilyl chloride

-
-
61362-77-4
1-(tert-butyldimethylsilyloxy)-4-pentyne

| Conditions | Yield |
|---|---|
| With 1H-imidazole In N,N-dimethyl-formamide at 25℃; | 100% |
| With 1H-imidazole In N,N-dimethyl-formamide at 25℃; for 4h; Inert atmosphere; | 100% |
| With 1H-imidazole In dichloromethane at 0 - 20℃; for 0.5h; Inert atmosphere; | 100% |
-
-
75-77-4
chloro-trimethyl-silane

-
-
5390-04-5
pent-1-yn-5-ol

-
-
13224-84-5
5-trimethylsilanyl-pent-4-yn-1-ol

| Conditions | Yield |
|---|---|
| With n-butyllithium Inert atmosphere; | 100% |
| Stage #1: chloro-trimethyl-silane; pent-1-yn-5-ol With n-butyllithium In tetrahydrofuran; hexane at -78 - 20℃; for 2h; Inert atmosphere; Stage #2: With hydrogenchloride In water for 3h; Inert atmosphere; | 100% |
| Stage #1: pent-1-yn-5-ol With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 1.08333h; Inert atmosphere; Stage #2: chloro-trimethyl-silane In tetrahydrofuran; hexane at 20℃; | 100% |

| Conditions | Yield |
|---|---|
| With copper(l) iodide; tetrakis(triphenylphosphine) palladium(0); triethylamine In tetrahydrofuran at 20℃; for 12h; Inert atmosphere; | 100% |
| With copper(l) iodide; tetrakis(triphenylphosphine) palladium(0); triethylamine In tetrahydrofuran at 20℃; for 12h; | 100% |
| With copper(l) iodide; tetrakis(triphenylphosphine) palladium(0); triethylamine In tetrahydrofuran at 20℃; for 12h; | 99% |

| Conditions | Yield |
|---|---|
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; triethylamine In acetonitrile at 20℃; for 4.5h; Inert atmosphere; | 100% |
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; triethylamine at 20℃; for 5h; Sonogashira Cross-Coupling; Inert atmosphere; | 99% |
| With copper(l) iodide; palladium diacetate; triphenylphosphine In diethylamine at 20℃; Inert atmosphere; | 89% |
-
-
5390-04-5
pent-1-yn-5-ol

-
-
124-63-0
methanesulfonyl chloride

-
-
68275-03-6
pent-4-yn-1-yl methanesulfonate

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at -20℃; for 0.583333h; | 100% |
| With triethylamine In dichloromethane at 0℃; for 1h; | 100% |
| With triethylamine In dichloromethane at 0℃; for 3h; Inert atmosphere; | 100% |
-
-
5390-04-5
pent-1-yn-5-ol

-
-
58479-61-1
tert-butylchlorodiphenylsilane

-
-
91266-03-4
tert-butyl-pent-4-ynyloxy-diphenyl-silane

| Conditions | Yield |
|---|---|
| With 1H-imidazole In N,N-dimethyl-formamide | 100% |
| With 1H-imidazole In dichloromethane at 25℃; | 100% |
| With 1H-imidazole In dichloromethane at 0℃; for 1h; Inert atmosphere; | 100% |
-
-
5390-04-5
pent-1-yn-5-ol

-
-
57381-43-8
2,5-dibromobenzoic acid methyl ester

-
-
857349-10-1
methyl 2,5-bis(5-hydroxy-1-pentynyl)benzoate

| Conditions | Yield |
|---|---|
| With copper(l) iodide; triethylamine; tetrakis(triphenylphosphine) palladium(0) In dichloromethane at 50℃; Sonogashira reaction; | 100% |
-
-
5390-04-5
pent-1-yn-5-ol

-
-
624-38-4
para-diiodobenzene

-
-
857348-95-9
1,4-bis(5-hydroxy-1-pentynyl)benzene

| Conditions | Yield |
|---|---|
| With copper(l) iodide; triethylamine; tetrakis(triphenylphosphine) palladium(0) In dichloromethane at 50℃; for 24h; Sonogashira reaction; | 100% |
| With copper(l) iodide; tetrakis(triphenylphosphine) palladium(0); triethylamine In dichloromethane at 50℃; Sonogashira Cross-Coupling; |

| Conditions | Yield |
|---|---|
| Stage #1: pent-1-yn-5-ol With pyridinium p-toluenesulfonate In 1,2-dichloro-ethane at 80℃; for 20h; Stage #2: With triethylamine In dichloromethane at 20℃; for 6h; Stage #3: With trifluoroacetic acid In methanol; dichloromethane at 140℃; for 0.0666667h; microwave irradiation; Further stages.; | 100% |

| Conditions | Yield |
|---|---|
| With camphor-10-sulfonic acid In dichloromethane at 0 - 20℃; for 3h; | 100% |
-
-
5390-04-5
pent-1-yn-5-ol

-
-
80522-42-5
triisopropylsilyl trifluoromethanesulfonate

-
-
184370-68-1
1-((triisopropylsilyl)oxy)-4-pentyne

| Conditions | Yield |
|---|---|
| With 2,6-dimethylpyridine In dichloromethane at -78℃; | 100% |
-
-
5390-04-5
pent-1-yn-5-ol

-
-
322474-21-5
N,N'-bis-Boc-S-methyl-isothiourea

-
-
1325688-35-4
N,N-bis(tert-butoxycarbonyl)-methyl pent-4-yn-1-ylcarbamimidothioate

| Conditions | Yield |
|---|---|
| With di-isopropyl azodicarboxylate; triphenylphosphine In tetrahydrofuran at -10 - 50℃; for 0.75h; Microwave irradiation; | 100% |
| With di-isopropyl azodicarboxylate; triphenylphosphine In water at -10 - 50℃; for 0.5h; Mitsunobu reaction; Microwave irradiation; | 98% |
| With di-isopropyl azodicarboxylate; triphenylphosphine In tetrahydrofuran at -10 - 50℃; for 0.5h; | 98% |
| With di-isopropyl azodicarboxylate; triphenylphosphine In tetrahydrofuran at -10 - 50℃; Microwave irradiation; |
-
-
5390-04-5
pent-1-yn-5-ol

-
-
622-79-7
benzyl azide

-
-
1198328-29-8
3-(1-benzyl-1H-1,2,3-triazol-4-yl)propan-1-ol

| Conditions | Yield |
|---|---|
| With copper(ll) sulfate pentahydrate; (+)-sodium L-ascorbate In water; tert-butyl alcohol at 20℃; for 2h; | 100% |
| With CuO-600 In water; tert-butyl alcohol at 20℃; for 12h; | 96% |
| With copper-iron nanoparticles In water at 20℃; for 12h; Huisgen cycloaddition; Inert atmosphere; regioselective reaction; | 89% |
-
-
5390-04-5
pent-1-yn-5-ol

-
-
157017-41-9
1-(2-iodo-phenyl)-1H-pyrrole

-
-
1404452-38-5
5-(2-(1H-pyrrol-1-yl)phenyl)pent-4-yn-1-ol

| Conditions | Yield |
|---|---|
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; triethylamine at 25℃; for 16h; Sonogashira Cross-Coupling; Inert atmosphere; | 100% |
-
-
5390-04-5
pent-1-yn-5-ol


| Conditions | Yield |
|---|---|
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; triethylamine In acetonitrile at 20℃; for 0.333333h; Sonogashira Cross-Coupling; | 100% |

-
-
5390-04-5
pent-1-yn-5-ol

-
-
32315-10-9
bis(trichloromethyl) carbonate

| Conditions | Yield |
|---|---|
| Stage #1: pent-1-yn-5-ol; bis(trichloromethyl) carbonate With pyridine In dichloromethane at 0℃; for 0.5h; Stage #2: 1-Boc-2-benzylpiperazine With N-ethyl-N,N-diisopropylamine In tetrahydrofuran at 20℃; for 2h; | 100% |

-
-
5390-04-5
pent-1-yn-5-ol

| Conditions | Yield |
|---|---|
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; triethylamine In N,N-dimethyl-formamide at 60℃; Inert atmosphere; | 100% |
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; triethylamine In N,N-dimethyl-formamide at 80℃; for 8h; Sealed tube; Inert atmosphere; | 80% |
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; triethylamine In N,N-dimethyl-formamide at 80℃; for 20h; Inert atmosphere; Sealed tube; | 58% |
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; triethylamine In N,N-dimethyl-formamide at 80℃; for 8h; Sealed tube; Inert atmosphere; | 2.08 g |

| Conditions | Yield |
|---|---|
| With triethylamine In tetrahydrofuran at 0 - 20℃; for 1.5h; Inert atmosphere; | 100% |

| Conditions | Yield |
|---|---|
| With Jones reagent In acetone at 20℃; for 1h; | 99% |
| With Jones reagent In acetone at 0 - 20℃; for 1h; | 82% |
| With Jones reagent In acetone at 0 - 20℃; for 1h; | 82% |
-
-
5390-04-5
pent-1-yn-5-ol

-
-
3282-30-2
pivaloyl chloride

-
-
140872-91-9
pent-4-yn-1-yl 2,2-dimethylpropanoate

| Conditions | Yield |
|---|---|
| With pyridine; dmap In dichloromethane at 0 - 20℃; Inert atmosphere; | 99% |
| With dmap; triethylamine In dichloromethane at 20℃; for 15h; Inert atmosphere; Schlenk technique; | 99% |
| With pyridine In dichloromethane for 1h; 0 to 25 deg C; | 95% |
| With pyridine; dmap In dichloromethane at 0 - 25℃; Inert atmosphere; | 80% |
| With pyridine; dmap at 0 - 20℃; Inert atmosphere; | 76% |

| Conditions | Yield |
|---|---|
| Stage #1: pent-1-yn-5-ol With 1H-imidazole; sodium hydride In tetrahydrofuran; mineral oil at 0℃; for 0.166667h; Inert atmosphere; Stage #2: benzyl bromide In tetrahydrofuran; mineral oil at 0 - 20℃; Inert atmosphere; | 99% |
| With sodium hydride In tetrahydrofuran at 0 - 20℃; for 5h; | 94% |
| Stage #1: pent-1-yn-5-ol With sodium hydride In tetrahydrofuran at 0℃; Inert atmosphere; Stage #2: benzyl bromide In tetrahydrofuran at 0 - 20℃; for 16h; Inert atmosphere; | 94% |

| Conditions | Yield |
|---|---|
| With Amberlyst 15 hydrogen form In hexane at 20℃; for 4h; | 99% |
| Amberlyst H-15 In hexane Ambient temperature; Yield given; |
-
-
5390-04-5
pent-1-yn-5-ol

-
-
610-97-9
o-iodo-methyl-benzoic acid

-
-
152771-70-5
2-(5-hydroxypent-1-ynyl)benzoic acid methyl ester

| Conditions | Yield |
|---|---|
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; triethylamine at 20℃; for 15h; Sonogashira Cross-Coupling; Inert atmosphere; | 99% |
| With copper(l) iodide; triethylamine; trans-bis(triphenylphosphine)palladium(II) chloride at 25℃; for 12h; | 92% |
| With piperidine; copper(l) iodide; tetrakis(triphenylphosphine) palladium(0) Sonogashira coupling reaction; | 86% |
| With triethylamine; bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide at 55℃; Sonogashira coupling; | 85% |
| Stage #1: o-iodo-methyl-benzoic acid With triethylamine for 0.0833333h; Inert atmosphere; Stage #2: pent-1-yn-5-ol With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide at 20℃; for 7h; Sonogashira coupling; Inert atmosphere; | 84% |
-
-
5390-04-5
pent-1-yn-5-ol

-
-
609-73-4
o-nitroiodobenzene

-
-
442155-83-1
2-(5-hydroxy-1-pentynyl)nitrobenzene

| Conditions | Yield |
|---|---|
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; triethylamine at 20℃; Sonogashira coupling; | 99% |
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; triethylamine at 20℃; for 14h; Sonogashira coupling; | 92% |
| With copper(l) iodide; triethylamine; bis-triphenylphosphine-palladium(II) chloride at 25℃; for 16h; Sonogashira cross-coupling; | 91% |
| Stage #1: pent-1-yn-5-ol; o-nitroiodobenzene With bis-triphenylphosphine-palladium(II) chloride; triethylamine; triphenylphosphine In N,N-dimethyl-formamide for 0.5h; Sonogashira coupling; Inert atmosphere; Stage #2: With copper(l) iodide In N,N-dimethyl-formamide at 20℃; for 5.16667h; Sonogashira coupling; Inert atmosphere; | 83% |

| Conditions | Yield |
|---|---|
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; diethylamine | 99% |
| With copper(l) iodide; tetrakis(triphenylphosphine) palladium(0); triethylamine Inert atmosphere; | 75% |
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; triethylamine In tetrahydrofuran at 20℃; for 16h; Schlenk technique; | |
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; triethylamine at 20℃; for 10h; Inert atmosphere; |

| Conditions | Yield |
|---|---|
| With water; ruthenium hydroxyapatite at 80℃; for 24h; Product distribution; Further Variations:; Catalysts; anti-Markovnikov hydration; | 99% |

| Conditions | Yield |
|---|---|
| johnphos In tetrahydrofuran at 40℃; | 99% |
| With acetylacetonatodicarbonylrhodium(l); johnphos In tetrahydrofuran at 40℃; for 24h; | 91% |
| With johnphos; acetylacetonatodicarbonylrhodium(l) In tetrahydrofuran at 40℃; for 24h; | 91% |
4-Pentyn-1-ol Specification
The 4-Pentyn-1-ol, with the CAS registry number 5390-04-5, is also known as (3-Hydroxypropyl)acetylene. It belongs to the product categories of Miscellaneous; Acetylenes; Acetylenic Alcohols & Their Derivatives; Alkynes; Organic Building Blocks; Terminal. Its EINECS number is 226-383-6. This chemical's molecular formula is C5H8O and molecular weight is 84.12. What's more, its systematic name is pent-4-yn-1-ol. It is stable at common pressure and temperature, and it should be sealed and stored in a cool and dry place. Moreover, it should be protected from light. It is used in the Grignard reagent reaction and aldehyde oxidation reaction.
Physical properties of 4-Pentyn-1-ol are: (1)ACD/LogP: 0.47; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 0.468; (4)ACD/LogD (pH 7.4): 0.468; (5)ACD/BCF (pH 5.5): 1.334; (6)ACD/BCF (pH 7.4): 1.334; (7)ACD/KOC (pH 5.5): 42.788; (8)ACD/KOC (pH 7.4): 42.788; (9)#H bond acceptors: 1; (10)#H bond donors: 1; (11)#Freely Rotating Bonds: 3; (12)Polar Surface Area: 20.23 Å2; (13)Index of Refraction: 1.445; (14)Molar Refractivity: 24.576 cm3; (15)Molar Volume: 92.34 cm3; (16)Polarizability: 9.743×10-24cm3; (17)Surface Tension: 36.394 dyne/cm; (18)Density: 0.911 g/cm3; (19)Flash Point: 61.667 °C; (20)Enthalpy of Vaporization: 45.515 kJ/mol; (21)Boiling Point: 153.999 °C at 760 mmHg; (22)Vapour Pressure: 1.195 mmHg at 25°C.
Preparation: this chemical can be prepared by 2-chloromethyl-tetrahydro-furan at the temperature of -60 °C. This reaction will need reagent NaNH2 and solvent tetrahydrofuran with the reaction time of 1 hour. The yield is about 80%.

Uses of 4-Pentyn-1-ol: it can be used to produce 2-pent-4-ynyloxy-tetrahydro-pyran at the ambient temperature. It will need reagent p-TsOH and solvent diethyl ether. The yield is about 99%.
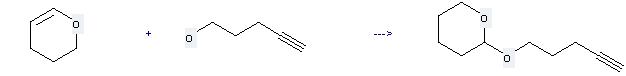
When you are using this chemical, please be cautious about it as the following:
It is irritating to eyes, respiratory system and skin. In case of contact with eyes, you should rinse immediately with plenty of water and seek medical advice. When using it, you need wear suitable protective clothing, gloves and eye/face protection.
You can still convert the following datas into molecular structure:
(1)SMILES: C#CCCCO
(2)Std. InChI: InChI=1S/C5H8O/c1-2-3-4-5-6/h1,6H,3-5H2
(3)Std. InChIKey: CRWVOXFUXPYTRK-UHFFFAOYSA-N
Related Products
- 4-Pentyn-1-ol
- 4-Pentyn-1-ol,5-(triethylsilyl)-
- 53902-12-8
- 53902-17-3
- 53903-49-4
- 53903-51-8
- 53903-58-5
- 539-03-7
- 53905-30-9
- 5390-61-4
- 53906-54-0
- 53906-84-6
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View