-
Name
4-tert-Butylbenzaldehyde
- EINECS 213-367-9
- CAS No. 939-97-9
- Article Data274
- CAS DataBase
- Density 0.966 g/cm3
- Solubility Soluble in water.
- Melting Point 245-246 °C
- Formula C11H14O
- Boiling Point 240.8 °C at 760 mmHg
- Molecular Weight 162.232
- Flash Point 96.9 °C
- Transport Information UN 3082 9/PG 3
- Appearance clear to pale yellow liquid
- Safety 36/37-60-61
- Risk Codes 22-43-50/53
-
Molecular Structure
-
Hazard Symbols
 Xn,
Xn, N,
N, Xi
Xi
- Synonyms 4-(1,1-Dimethylethyl)benzaldehyde;Benzaldehyde,p-tert-butyl- (6CI,7CI,8CI);p-tert-Butylbenzaldehyde;para-tert-Butylbenzaldehyde;
- PSA 17.07000
- LogP 2.79660
Synthetic route

| Conditions | Yield |
|---|---|
| With 1H-imidazole; sodium periodate; manganese(III)-porphyrin complex immobilized on polystyrene In water; acetonitrile at 20℃; for 1.16667h; | 100% |
| With oxygen In 1,3,5-trimethyl-benzene at 60℃; under 760.051 Torr; for 7h; Solvent; Reagent/catalyst; | 100% |
| With titanium(IV) oxide; oxygen at 29.84℃; under 760.051 Torr; for 6h; Sealed tube; Irradiation; | 99% |

| Conditions | Yield |
|---|---|
| With polymethylhydrosiloxane; trifuran-2-yl-phosphane; tetrabutyl ammonium fluoride; potassium fluoride; tris-(dibenzylideneacetone)dipalladium(0) In tetrahydrofuran; water at 20℃; for 1h; | 99% |
| With diethylene glycol dimethyl ether; lithium tri-t-butoxyaluminum hydride | |
| With hydrogen; N-ethyl-N,N-diisopropylamine; palladium on activated charcoal In ethyl acetate at 25℃; under 760 Torr; for 1.75h; Yield given; | |
| Multi-step reaction with 2 steps 1: dichloromethane / 0.5 h / 20 °C / Cooling with ice 2: Schwartz's reagent / tetrahydrofuran / 0.5 h / 20 °C View Scheme |

| Conditions | Yield |
|---|---|
| With p-PPh2-C6H4-(OCH2CH2)n-O-C6H4-PPh2-p; ozone Product distribution; various catalysts; | 98% |
| Stage #1: 4-tert-Butylstyrene With oxygen; ozone In dichloromethane at -78℃; Stage #2: With poly(ethylene glycol)-triphenylphosphine In dichloromethane at -78 - 20℃; | 98% |
| With oxygen; benzoic acid; sodium nitrite In toluene at 80℃; for 20h; Reagent/catalyst; Solvent; Temperature; | 82% |
-
-
180261-48-7
4-tert-butyl benzene carbaldehyde oxime

-
-
939-97-9
4-tert-Butylbenzaldehyde

| Conditions | Yield |
|---|---|
| With iron(III) chloride; 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical; oxygen In water; toluene at 60℃; under 760.051 Torr; for 3h; | 98% |
-
-
624286-50-6
(4-tert-butylbenzyloxy)trimethylsilane

-
-
939-97-9
4-tert-Butylbenzaldehyde

| Conditions | Yield |
|---|---|
| With trinitratocerium(IV) bromate for 0.7h; Heating; | 97% |
| With copper(II) nitrate trihydrate; water; silica gel; potassium bromide at 90℃; for 0.5h; Neat (no solvent); | 87% |

| Conditions | Yield |
|---|---|
| With N,N,N,N,-tetramethylethylenediamine; hydrogen; palladium diacetate; propyl di-tert-butylphosphinite In toluene at 100℃; under 3750.38 Torr; for 20h; Inert atmosphere; | 97% |
-
-
19692-45-6
4-tert-butylbenzyl chloride

-
-
939-97-9
4-tert-Butylbenzaldehyde

| Conditions | Yield |
|---|---|
| With 1-dodecyl-3-methylimidazolium iron chloride; periodic acid at 30℃; for 1.5h; | 96% |
| With potassium hydroxide; cetyltrimethylammonim bromide; potassium nitrate In water for 4h; pH=10 - 11; Reflux; | 90% |
| With potassium nitrite; 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical; oxygen; potassium bromide In water for 4h; Reflux; | 90% |
| With sodium nitrate; acetic acid In water for 3.5h; Reflux; | 90% |
| With hexamethylenetetramine; sulfuric acid; sodium carbonate In ethanol; water; toluene | 11.8 g (60%) |

| Conditions | Yield |
|---|---|
| With 1-hydroxy-3H-benz[d][1,2]iodoxole-1,3-dione In fluorobenzene; dimethyl sulfoxide at 80℃; for 8h; | 95% |
| With ammonium cerium(IV) nitrate In water; acetic acid at 85 - 95℃; for 1.5h; | 94% |
| With carbon tetrabromide; oxygen In acetonitrile at 20℃; for 3h; Irradiation; Green chemistry; | 90% |
-
-
19099-48-0
1,1-bis(4-t-butylphenyl)methane

-
-
80077-60-7
1-azidononane

-
A
-
939-97-9
4-tert-Butylbenzaldehyde

-
B
-
1225826-38-9
4-tert-butyl-N-nonylaniline

| Conditions | Yield |
|---|---|
| With water; trifluoroacetic acid; 2,3-dicyano-5,6-dichloro-p-benzoquinone; iron(II) chloride at 60℃; for 5h; | A 85% B 94% |


| Conditions | Yield |
|---|---|
| Stage #1: N-(4-tert-butylbenzyl)-4-tert-butylaniline With oxygen; toluene-4-sulfonic acid; dimethyl sulfoxide In toluene at 100℃; for 119h; Green chemistry; Stage #2: With hydrogenchloride In water at 20℃; for 1h; Reagent/catalyst; Temperature; Green chemistry; | A 93% B 94% |

| Conditions | Yield |
|---|---|
| Stage #1: formic acid; 1-tert-butyl-4-iodobenzene With palladium diacetate; acetic anhydride; tricyclohexylphosphine In N,N-dimethyl-formamide at 30℃; for 1h; Inert atmosphere; Green chemistry; Stage #2: With triethylamine In N,N-dimethyl-formamide at 80℃; for 6h; Inert atmosphere; Green chemistry; | 93% |
| With palladium diacetate; triethylamine; dicyclohexyl-carbodiimide; tricyclohexylphosphine In N,N-dimethyl-formamide at 80℃; for 10h; Inert atmosphere; Sealed tube; | 72% |
-
-
98-51-1
4-tert-butyltoluene

-
A
-
939-97-9
4-tert-Butylbenzaldehyde

-
B
-
98-73-7
4-(1,1-dimethylethyl)benzoic acid

| Conditions | Yield |
|---|---|
| With N-hydroxyphthalimide; oxygen; cobalt(II) acetate In acetic acid at 25℃; under 760 Torr; for 20h; | A 2% B 91% |
| With N-hydroxyphthalimide; oxygen; cobalt(II) acetate In acetonitrile at 25℃; under 760 Torr; for 20h; | A 4% B 77% |
| With oxygen; acetic acid; N-hydroxyphthalimide at 100℃; for 6h; Product distribution / selectivity; | A 3.8% B 66.5% |

| Conditions | Yield |
|---|---|
| With dipotassium hydrogenphosphate; nickel(II) bis(2,2,6,6-tetramethylheptane-3,5-dionate); Ir(dF(CF3)ppy)2(bpy)PF6; zinc dibromide In N,N-dimethyl acetamide at 27℃; for 48h; Inert atmosphere; Irradiation; | 91% |
-
-
18880-00-7
1-(bromomethyl)-4-(1,1-dimethylethyl)benzene

-
-
939-97-9
4-tert-Butylbenzaldehyde

| Conditions | Yield |
|---|---|
| With potassium hydrogencarbonate; dimethyl sulfoxide for 0.0583333h; Microwave irradiation; | 90% |
| With carbon dioxide; copper(II) nitrate | |
| With ethanol; carbon dioxide; hexamethylenetetramine | |
| With 4-methylmorpholine N-oxide at 20℃; for 4h; Reflux; | 246.6 mg |
| Multi-step reaction with 2 steps 1: eosin y; oxygen / dimethyl sulfoxide / 12 h / 25 °C / Irradiation 2: eosin y / dimethyl sulfoxide / 24 h / 80 °C / Irradiation View Scheme |
-
-
39895-55-1
4-(1,1-dimethylethyl)-benzenemethanamine

-
-
939-97-9
4-tert-Butylbenzaldehyde

| Conditions | Yield |
|---|---|
| With cercosporin; oxygen In methanol at 20℃; Schlenk technique; Irradiation; Green chemistry; chemoselective reaction; | 89% |
| With copper(I) 2-hydroxy-3-methylbenzoate; oxygen; ascorbic acid In N,N-dimethyl acetamide at 45℃; for 2h; chemoselective reaction; | 70% |
| With dipotassium peroxodisulfate; tris(bipyridine)ruthenium(II) dichloride hexahydrate; water In acetonitrile at 20℃; for 12h; Sealed tube; Irradiation; Green chemistry; |
-
-
119072-55-8, 7188-38-7
tert-butylisonitrile

-
-
35779-04-5
1-tert-butyl-4-iodobenzene

-
-
939-97-9
4-tert-Butylbenzaldehyde

| Conditions | Yield |
|---|---|
| Stage #1: tert-butylisonitrile; 1-tert-butyl-4-iodobenzene With triethylsilane; palladium diacetate; sodium carbonate; CyJohnPhos In N,N-dimethyl-formamide at 65℃; for 8h; Inert atmosphere; Sealed tube; Green chemistry; Stage #2: With water Green chemistry; | 88% |

-
-
939-97-9
4-tert-Butylbenzaldehyde

| Conditions | Yield |
|---|---|
| With eosin y In water; acetonitrile at 20℃; for 4h; Irradiation; | 88% |
-
-
32857-63-9
4-t-butylphenylacetic acid

-
-
939-97-9
4-tert-Butylbenzaldehyde

| Conditions | Yield |
|---|---|
| With oxygen; copper diacetate In dimethyl sulfoxide at 120℃; for 18h; Sealed tube; | 87% |
| With (4s,6s)-2,4,5,6-tetra(9H-carbazol-9-yl)isophthalonitrile; 1,8-diazabicyclo[5.4.0]undec-7-ene In acetonitrile at 20 - 25℃; for 6h; Irradiation; | 76% |
| With (4s,6s)-2,4,5,6-tetra(9H-carbazol-9-yl)isophthalonitrile; N,N,N',N'-tetramethylguanidine In acetonitrile at 20℃; for 6h; Irradiation; | 55% |
| With water; copper dichloride at 250℃; under 30003 Torr; for 0.17h; Sealed tube; Green chemistry; | 94 %Chromat. |
-
-
49543-63-7
4-tert-butylbenzyl mercaptan

-
-
939-97-9
4-tert-Butylbenzaldehyde

| Conditions | Yield |
|---|---|
| With dipotassium peroxodisulfate; tetrakis(pyridine)silver(II) peroxodisulfate; oxygen In N,N-dimethyl-formamide at 23℃; under 760.051 Torr; Irradiation; | 87% |

| Conditions | Yield |
|---|---|
| With nitric acid In dichloromethane at 20℃; for 1h; | 86% |
-
-
35779-04-5
1-tert-butyl-4-iodobenzene

-
-
82102-37-2
9-methyl-9H-fluorene-9-carbonyl chloride

-
-
939-97-9
4-tert-Butylbenzaldehyde

| Conditions | Yield |
|---|---|
| With dichloro(1,5-cyclooctadiene)palladium(II); N-Methyldicyclohexylamine; potassium formate; tetra-(n-butyl)ammonium iodide; tricyclohexylphosphine tetrafluoroborate; bis(dibenzylideneacetone)-palladium(0); tri tert-butylphosphoniumtetrafluoroborate In acetonitrile at 80℃; for 18h; Sealed tube; Glovebox; Inert atmosphere; | 86% |

| Conditions | Yield |
|---|---|
| With rhodium(III) iodide; dichloro [1,1'-bis(diphenylphosphino)propane]palladium(II); hydrogen; acetic anhydride; triethylamine; triphenylphosphine In N,N-dimethyl acetamide at 100℃; for 24h; Autoclave; | 85% |
| With dichloro [1,1'-bis(diphenylphosphino)propane]palladium(II); 1,8-diazabicyclo[5.4.0]undec-7-ene In N,N-dimethyl-formamide at 100℃; under 7500.75 Torr; for 20h; Autoclave; Green chemistry; | 81% |

-
-
939-97-9
4-tert-Butylbenzaldehyde

| Conditions | Yield |
|---|---|
| With triethylsilane; palladium diacetate; tricyclohexylphosphine In toluene at 100℃; for 20h; Inert atmosphere; Sealed tube; | 85% |
-
-
72390-21-7
1-tert-butoxymethyl-4-tert-butylbenzene

-
-
939-97-9
4-tert-Butylbenzaldehyde

| Conditions | Yield |
|---|---|
| With N-hydroxyphthalimide; nitrogen(II) oxide In acetonitrile at 60℃; for 10h; | 84% |
| 84% |
-
-
67-56-1
methanol

-
-
79135-54-9, 114530-47-1, 123356-27-4, 64392-50-3
(E)-1,1'-(1,2-ethenediyl)bis(4-butyl)benzene

-
A
-
939-97-9
4-tert-Butylbenzaldehyde

-
B
-
26537-19-9
methyl 4-tert-butylbenzoate

| Conditions | Yield |
|---|---|
| With iodine; oxygen; trifluoroacetic acid In ethyl acetate for 20h; Solvent; Reagent/catalyst; Irradiation; | A 84% B 7 %Spectr. |

-
-
84434-23-1
3-(4-(tert-butyl)phenyl)acrylaldehyde

-
-
939-97-9
4-tert-Butylbenzaldehyde

| Conditions | Yield |
|---|---|
| With pyrrolidine; water In acetonitrile at 20℃; for 12h; Inert atmosphere; | 83% |

| Conditions | Yield |
|---|---|
| In 1,4-dioxane for 12h; Heating; | A 82% B 66% |

| Conditions | Yield |
|---|---|
| With rhodium(III) chloride trihydrate; hydrogen; triethylamine; triphenylphosphine In N,N-dimethyl acetamide at 90℃; under 7500.75 Torr; for 12h; Autoclave; | 81% |

| Conditions | Yield |
|---|---|
| With hydrogen bromide; oxygen In acetonitrile at 20℃; for 5h; UV-irradiation; | 100% |
| With tert.-butylhydroperoxide; potassium tert-butylate In water at 60℃; for 5h; | 96% |
| With N-Bromosuccinimide; oxygen In ethyl acetate at 20℃; Irradiation; | 93% |

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate In ethanol at 25℃; for 0.5h; | 100% |
| With sodium tetrahydroborate In ethanol at 20℃; for 0.5h; Inert atmosphere; | 100% |
| With sodium tetrahydroborate In ethanol at 20℃; for 0.5h; Inert atmosphere; | 100% |
-
-
939-97-9
4-tert-Butylbenzaldehyde

-
-
557-20-0
diethylzinc

-
-
127896-25-7
1-(4-tert-butylphenyl)propan-1-ol

| Conditions | Yield |
|---|---|
| With (R,R)-(2,4,6-Me3C6H2-CH=N-CH(Ph))2; (R)-(+)-3,3'-diphenyl-[1,1'-binaphthalene]-2,2'-diol In hexane; dichloromethane 1.) -78 deg C, 4 h, 2.) -20 deg, 1 h; | 100% |
| With (1R,2R)-N,N'-bis[(2,4,6-trimethylphenyl)methylene]-1,2-cyclohexane-diamine; (R)-(+)-3,3'-diphenyl-[1,1'-binaphthalene]-2,2'-diol In hexane; dichloromethane at -78 - -20℃; for 5h; | 100% |
| With titanium(IV) isopropylate at 80℃; for 1h; Time; Inert atmosphere; | 60 %Chromat. |
| With magnesium chloride at 80℃; for 0.5h; Reagent/catalyst; Inert atmosphere; | 24.2 %Chromat. |

| Conditions | Yield |
|---|---|
| With sodium hydroxide for 1.5h; | 100% |
-
-
939-97-9
4-tert-Butylbenzaldehyde

-
-
107-11-9
1-amino-2-propene

-
-
250376-72-8
Allyl-[1-(4-tert-butyl-phenyl)-meth-(E)-ylidene]-amine

| Conditions | Yield |
|---|---|
| With magnesium sulfate In dichloromethane at 20℃; for 20h; | 100% |
-
-
856889-57-1
N-[3-fluoro-4-(4-morpholinyl)phenyl]-N-(4-piperidinylmethyl)-N'-(tetrahydro-2H-pyran-4-yl)urea

-
-
939-97-9
4-tert-Butylbenzaldehyde

| Conditions | Yield |
|---|---|
| With MP-triacetoxyborohydride In dichloromethane at 20℃; for 18h; MP-triacetoxyborohydride; | 100% |
-
-
939-97-9
4-tert-Butylbenzaldehyde

-
-
1826-67-1
vinyl magnesium bromide

-
-
863029-85-0
1-(4-(tert-butyl)phenyl)prop-2-en-1-ol

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 0 - 20℃; for 1h; Inert atmosphere; | 100% |
| Stage #1: 4-tert-Butylbenzaldehyde; vinyl magnesium bromide In tetrahydrofuran at 20℃; for 20h; Heating / reflux; Stage #2: With water; ammonium chloride | |
| In tetrahydrofuran at 0℃; for 1h; |

| Conditions | Yield |
|---|---|
| Stage #1: 4-tert-Butylbenzaldehyde; 2-(3-methoxyphenyl)-1-ethanamine With potassium carbonate In methanol at 20℃; for 2h; Heating / reflux; Stage #2: With sodium tetrahydroborate In methanol at 20℃; for 3h; Heating / reflux; Stage #3: With hydrogenchloride In methanol; water | 100% |

| Conditions | Yield |
|---|---|
| Stage #1: 4-tert-Butylbenzaldehyde; 2-(3-Fluorophenyl)ethylamine In methanol at 20℃; for 2.5h; Heating / reflux; Stage #2: With sodium tetrahydroborate In methanol at 20℃; for 3.08h; Heating / reflux; Stage #3: With hydrogenchloride In methanol; water at 20℃; | 100% |
-
-
939-97-9
4-tert-Butylbenzaldehyde

-
-
13078-79-0
2-(3-chlorophenyl)ethylamine

-
-
875305-16-1
(4-tert-butyl-benzyl)-[2-(3-chloro-phenyl)-ethyl]-amine

| Conditions | Yield |
|---|---|
| Stage #1: 4-tert-Butylbenzaldehyde; 2-(3-chlorophenyl)ethylamine In methanol at 20℃; for 3h; Heating / reflux; Stage #2: With sodium tetrahydroborate In methanol at 20℃; for 4.08h; Heating / reflux; Stage #3: With hydrogenchloride In methanol; water at 20℃; | 100% |
-
-
939-97-9
4-tert-Butylbenzaldehyde

-
-
1583-88-6
4-fluoro-2-phenethylamine

-
-
875305-06-9
(4-tert-butyl-benzyl)-[2-(4-fluoro-phenyl)-ethyl]-amine

| Conditions | Yield |
|---|---|
| Stage #1: 4-tert-Butylbenzaldehyde; 4-fluoro-2-phenethylamine In methanol at 20℃; for 4.5h; Heating / reflux; Stage #2: With sodium tetrahydroborate In methanol at 20℃; for 2.08h; Heating / reflux; Stage #3: With hydrogenchloride In methanol; water at 20℃; | 100% |
-
-
939-97-9
4-tert-Butylbenzaldehyde

-
-
3261-62-9
2-(p-tolyl)ethylamine

-
-
875305-08-1
(4-tert-butyl-benzyl)-(2-p-tolyl-ethyl)-amine

| Conditions | Yield |
|---|---|
| Stage #1: 4-tert-Butylbenzaldehyde; 2-(p-tolyl)ethylamine In methanol at 20℃; for 3h; Heating / reflux; Stage #2: With sodium tetrahydroborate In methanol at 20℃; for 2.38h; Heating / reflux; Stage #3: With hydrogenchloride In methanol; water at 20℃; | 100% |
-
-
875305-55-8
2-(2-fluoro-5-trifluoromethyl-phenyl)-ethylamine hydrochloride

-
-
939-97-9
4-tert-Butylbenzaldehyde

-
-
875305-54-7
(4-tert-butyl-benzyl)-[2-(2-fluoro-5-trifluoromethyl-phenyl)-ethyl]-amine

| Conditions | Yield |
|---|---|
| Stage #1: 2-(2-fluoro-5-trifluoromethyl-phenyl)-ethylamine hydrochloride; 4-tert-Butylbenzaldehyde With potassium carbonate In methanol at 20℃; for 3h; Heating / reflux; Stage #2: With sodium tetrahydroborate In methanol at 20℃; for 2.58h; Heating / reflux; | 100% |
-
-
939-97-9
4-tert-Butylbenzaldehyde

-
-
2579-20-6
cis, trans-1,3-dimethylaminocyclohexane

-
-
1217526-91-4
C30H42N2

| Conditions | Yield |
|---|---|
| In methanol at 20℃; Molecular sieve; | 100% |
| In methanol at 20℃; Molecular sieve; |

| Conditions | Yield |
|---|---|
| at 20℃; for 12h; | 100% |
-
-
945717-05-5
2-(4-Chloro-3-Ethyl-Phenyl)-Ethyl-Amine-Hydrochloride

-
-
939-97-9
4-tert-Butylbenzaldehyde

-
-
1352659-88-1
[1-(4-tert-butyl-phenyl)-meth-(E)-ylidene]-[2-(4-chloro-3-ethyl-phenyl)-ethyl]-amine

| Conditions | Yield |
|---|---|
| Stage #1: 2-(4-Chloro-3-Ethyl-Phenyl)-Ethyl-Amine-Hydrochloride With sodium hydroxide In water; ethyl acetate Stage #2: 4-tert-Butylbenzaldehyde In ethyl acetate at 20℃; for 2h; | 100% |
| With sodium hydroxide In water; ethyl acetate at 20℃; for 2h; | 100% |

| Conditions | Yield |
|---|---|
| In ethanol at 20℃; | 100% |
-
-
939-97-9
4-tert-Butylbenzaldehyde

-
-
1730-25-2
allylmagnesium bromide

-
-
106027-36-5
1-(4-(tert-butyl)phenyl)but-3-en-1-ol

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; diethyl ether at 0 - 20℃; | 100% |
| With indium In tetrahydrofuran; diethyl ether at 0℃; Inert atmosphere; |

| Conditions | Yield |
|---|---|
| Stage #1: malonic acid; 4-tert-Butylbenzaldehyde With piperidine; pyridine Knoevenagel-Doebner reaction; Reflux; Stage #2: With hydrogenchloride In water Cooling with ice; optical yield given as %de; | 99% |
| With piperidine; pyridine for 2h; Knoevenagel condensation; Reflux; | 88% |
| With piperidine; pyridine | 87% |
-
-
939-97-9
4-tert-Butylbenzaldehyde

| Conditions | Yield |
|---|---|
| With potassium carbonate In ethanol; water for 5.5h; Heating; | 99% |
| With potassium carbonate |

| Conditions | Yield |
|---|---|
| Stage #1: ethyl 2-diethoxyphosphorylpropionate With sodium hydride In tetrahydrofuran; mineral oil at 0℃; for 0.5h; Inert atmosphere; Stage #2: 4-tert-Butylbenzaldehyde In tetrahydrofuran; hexane at 0 - 20℃; Inert atmosphere; | 99% |
| Stage #1: ethyl 2-diethoxyphosphorylpropionate With sodium hydride In tetrahydrofuran at 0 - 25℃; Stage #2: 4-tert-Butylbenzaldehyde In tetrahydrofuran for 1h; Heating; | |
| With sodium hydride In tetrahydrofuran; mineral oil at -78 - 20℃; for 3h; |

| Conditions | Yield |
|---|---|
| With ammonium sulfate; sodium carbonate; sulfur In dimethyl sulfoxide at 120℃; for 10h; Sealed tube; | 99% |
| With pyridine; tris(2,2-bipyridine)ruthenium(II) hexafluorophosphate; ammonium peroxydisulfate; 4-acetylamino-2,2,6,6-tetramethyl-1-piperidinoxy; ammonium carbamate In water; acetonitrile at 20℃; for 24h; Irradiation; Sealed tube; | 98% |
| Stage #1: 4-tert-Butylbenzaldehyde With pyridine; 1,1,1,3,3,3-hexamethyl-disilazane In dichloromethane at 26℃; for 0.0833333h; Stage #2: With 4-acetylamino-2,2,6,6-tetramethylpiperidine-1-oxoammonium tetrafluoroborate In dichloromethane at 26℃; for 18h; | 97% |
-
-
858124-31-9
3-(diethoxyphosphorylmethyl)-4-methoxybenzamide

-
-
939-97-9
4-tert-Butylbenzaldehyde

| Conditions | Yield |
|---|---|
| With sodium tert-pentoxide In N,N-dimethyl-formamide at 20℃; for 1h; Horner-Wadsworth-Emmons olefination; | 99% |
-
-
939-97-9
4-tert-Butylbenzaldehyde

-
-
123-46-6
girard's reagent T

-
-
66932-77-2
diisopropyl (2-oxo-2-(pyrrolidin-1-yl)ethyl)phosphonate

-
B
-
535945-57-4
C17H23NO

| Conditions | Yield |
|---|---|
| Stage #1: 4-tert-Butylbenzaldehyde; diisopropyl (2-oxo-2-(pyrrolidin-1-yl)ethyl)phosphonate With potassium tert-butylate In tetrahydrofuran at 20℃; for 6h; Horner-Wadsworth-Emmons Olefination; Stage #2: girard's reagent T In tetrahydrofuran; methanol stereoselective reaction; | A n/a B 99% C n/a |


| Conditions | Yield |
|---|---|
| With palladium on activated charcoal; hydrogen In methanol at 25℃; under 760.051 Torr; for 0.833333h; | 99% |

| Conditions | Yield |
|---|---|
| With silica gel; toluene-4-sulfonic acid In dichloromethane Reflux; | 99% |
| With boron trifluoride diethyl etherate In dichloromethane Inert atmosphere; | |
| With boron trifluoride diethyl etherate In dichloromethane at 0 - 25℃; |

| Conditions | Yield |
|---|---|
| In neat (no solvent) at 60℃; for 0.0666667h; Microwave irradiation; | 99% |

| Conditions | Yield |
|---|---|
| Stage #1: pentanonitrile; 4-tert-Butylbenzaldehyde With hydrogen In 2-methyltetrahydrofuran at 100℃; under 11251.1 Torr; for 20h; High pressure; Autoclave; Stage #2: With hydrogenchloride In diethyl ether | 99% |
4-TERT-BUTYLBENZALDEHYDE Chemical Properties
MF: C11H14O
MW: 162.23
EINECS: 213-367-9
Product Categories: Aromatic Aldehydes & Derivatives (substituted);Aldehydes;Phenyls & Phenyl-Het;Phenyls & Phenyl-Het;Pharmaceutical Intermediate;C10 to C21;Carbonyl Compounds
bp: 130 °C25 mm Hg(lit.)
density: 0.97 g/mL at 25 °C(lit.)
refractive index: n20/D 1.53(lit.)
Fp: 214 °F
Sensitive: Air & Light Sensitive
BRN: 1906461
4-TERT-BUTYLBENZALDEHYDE's Synonyms: LABOTEST-BB LT00689343;AKOS BBS-00003171;4-TERT-BUTYLBENZALDEHYDE;P-TERT-BUTYL BENZALDEHYDE;TIMTEC-BB SBB008564;4-(1,1-DIMETHYLETHYL)-BENZALDEHYD;4-(1,1-DIMETHYLETHYL)-BENZALDEHYDE;BENZALDEHYDE,-(1,1-DIMETHYLETHYL)- 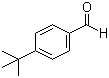
4-TERT-BUTYLBENZALDEHYDE Toxicity Data With Reference
Hazards Identification
Inhalation:Inhalation of material may be harmful.
Skin: may cause burns.
Eyes:may cause burns.
Hazards:Containers may explode when heated.
EC Risk Phrase:R 22 43
EC Safety Phrase:S 23 36/37
UN (DOT):3334
4-TERT-BUTYLBENZALDEHYDE Safety Profile
4-TERT-BUTYLBENZALDEHYDE's afety Information
Hazard Codes Xn,N,Xi
RIDADR UN 3082 9/PG 3
WGK Germany 2
F 8-9-23
Hazard Note Irritant
HazardClass 9
PackingGroup III
4-TERT-BUTYLBENZALDEHYDE Specification
Small spills/leaks:Do not touch or walk through spilled material. Stop leak if you can do it without risk. Prevent dust cloud. Avoid inhalation of ASBESTOS dust.
Stability and Reactivity
Combustion Products:Fire may produce irritating, corrosive and/or toxic gases.
Related Products
- 4-TERT-BUTYLBENZALDEHYDE
- 939-98-0
- 939981-37-0
- 939981-39-2
- 939983-14-9
- 939983-66-1
- 939986-64-8
- 939986-65-9
- 939-99-1
- 939999-69-6
- 940000-26-0
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View