-
Name
4-Methoxybenzamide
- EINECS 222-319-6
- CAS No. 3424-93-9
- Article Data403
- CAS DataBase
- Density 1.144 g/cm3
- Solubility
- Melting Point 164-167 °C(lit.)
- Formula C8H9NO2
- Boiling Point 295.803 °C at 760 mmHg
- Molecular Weight 151.165
- Flash Point 161.563 °C
- Transport Information
- Appearance White crystalline powder
- Safety 22-24/25
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xn
Xn
- Synonyms p-Anisamide(6CI,7CI,8CI);Anisamide;NSC 11139;p-Methoxybenzamide;
- PSA 52.32000
- LogP 1.49440
Synthetic route

| Conditions | Yield |
|---|---|
| With C18H57O3P6Ru2(1+)*C6H5O(1-)*C6H6O; water In 1,4-dioxane for 6h; Time; Sealed tube; Inert atmosphere; Schlenk technique; | 100% |
| With [bis(2-methylallyl)cycloocta-1,5-diene]ruthenium(II); water; 2-(diphenylphosphino)-N,N-dimethylpyridine-4-amine In 1,2-dimethoxyethane at 80℃; for 5h; | 99% |
| With C12H24O16Ru3*2H2O In water at 110℃; for 13h; Catalytic behavior; Schlenk technique; Inert atmosphere; | 99% |

| Conditions | Yield |
|---|---|
| With hydroxylamine; C33H28F3N5O3Ru(1+)*Cl(1-) In water at 60℃; for 0.5h; Catalytic behavior; Sonication; | 99% |
| Stage #1: 4-methoxy-benzaldehyde With hydroxylamine hydrochloride; caesium carbonate In water; dimethyl sulfoxide at 100℃; Stage #2: With palladium diacetate In water; dimethyl sulfoxide at 125℃; chemoselective reaction; | 97% |
| With copper(ll) sulfate pentahydrate; hydroxylamine hydrochloride; sodium acetate at 110℃; for 2h; Neat (no solvent); | 97% |

| Conditions | Yield |
|---|---|
| Stage #1: 4-methoxybenzoic acid With 1,3,5-trichloro-2,4,6-triazine; potassium carbonate In tetrahydrofuran for 0.0166667h; Milling; Stage #2: With ammonium thiocyanate In tetrahydrofuran for 0.0833333h; Milling; | 99% |
| With pyridine; urea for 0.00833333h; microwave irradiation; | 90% |
| Stage #1: 4-methoxybenzoic acid With thionyl chloride In tetrahydrofuran at 50℃; for 1h; Stage #2: With ammonium hydroxide In tetrahydrofuran at 0℃; for 0.0833333h; | 84% |
-
-
19486-73-8
N-tert-butyl-4-methoxybenzamide

-
-
3424-93-9
4-methoxyphenylacetamide

| Conditions | Yield |
|---|---|
| scandium tris(trifluoromethanesulfonate) In nitromethane at 170℃; for 1h; Irradiation; | 99% |
| With t-butyldimethylsiyl triflate In toluene at 100℃; for 8h; | 74% |
-
-
619-57-8
p-hydroxybenzamide

-
-
616-38-6
carbonic acid dimethyl ester

-
-
3424-93-9
4-methoxyphenylacetamide

| Conditions | Yield |
|---|---|
| With dmap at 135℃; under 3000.3 Torr; for 7h; Pressure; Temperature; Reagent/catalyst; | 99% |

| Conditions | Yield |
|---|---|
| With ammonia Inert atmosphere; | 98% |
| With ammonia at -10 - 20℃; for 1h; Temperature; | 96.3% |
| With ammonia In water at 20℃; for 1h; | 79% |

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; ammonia; oxygen In water; N,N-dimethyl-formamide at 80℃; under 1520.1 Torr; for 5h; Green chemistry; | 98% |
| Stage #1: 4-Methoxybenzyl alcohol With 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical; ammonia; oxygen; copper(II) nitrate In water; dimethyl sulfoxide at 80℃; under 760.051 Torr; for 5h; Stage #2: With Acetaldehyde oxime In water; dimethyl sulfoxide at 110℃; for 24h; Inert atmosphere; | 97% |
| With ammonia; oxygen In water at 150℃; under 22502.3 Torr; for 3h; | 96.5% |

| Conditions | Yield |
|---|---|
| With manganese(IV) oxide at 130℃; under 4560.31 Torr; for 3h; | 98% |
| With dihydrogen peroxide at 80℃; for 10h; Inert atmosphere; | 96% |
| With (carbonyl)(chloro)(hydrido)tris(triphenylphosphine)ruthenium(II); potassium tert-butylate In tert-butyl alcohol at 70℃; for 12h; | 95% |

| Conditions | Yield |
|---|---|
| With (1,4-diazabicyclo{2.2.2}-octane)zinc(II) tetrahydoborate In tetrahydrofuran for 48h; Ambient temperature; | 97% |
| With 1,3-diadamantane-4,5-dihydroimidazole chloride; potassium tert-butylate; bis(pinacol)diborane; copper(l) chloride In toluene at 25℃; for 4h; Schlenk technique; Inert atmosphere; | 94% |
| With sodium tetrahydroborate; copper(II) sulfate In methanol at 0 - 5℃; for 1h; | 92% |
-
-
874-90-8
4-methoxybenzonitrile

-
A
-
3424-93-9
4-methoxyphenylacetamide

-
B
-
5373-87-5
4-methoxybenzamidoxime

| Conditions | Yield |
|---|---|
| With hydroxylamine In ethanol for 3h; Reagent/catalyst; Reflux; | A 97% B n/a |
| With hydroxylamine hydrochloride; sodium hydroxide In ethanol; water Reflux; |

| Conditions | Yield |
|---|---|
| With 2-(2'-pyridyl)(di(1-adamantyl)phosphino)benzene; ammonia; palladium diacetate In 1,4-dioxane at 120℃; under 1500.15 Torr; for 16h; Autoclave; | 96% |
| With tris-(dibenzylideneacetone)dipalladium(0); (2R)-1-[(1R)-1-[bis(1,1-dimethylethyl)phosphino]ethyl]-2-(diphenylphosphino)ferrocene; ammonium carbamate; sodium hydrogencarbonate In 1,4-dioxane at 100℃; for 20h; Inert atmosphere; | 93% |
| With dmap; bis-triphenylphosphine-palladium(II) chloride; formamide In 1,4-dioxane at 120℃; under 3750.38 Torr; for 18h; | 69% |

-
-
3424-93-9
4-methoxyphenylacetamide

| Conditions | Yield |
|---|---|
| With lithium hydroxide monohydrate In water at 20℃; for 2h; | 96% |
-
-
120158-06-7
tert-butyl (4-methoxybenzoyl)carbamate

-
-
3424-93-9
4-methoxyphenylacetamide

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid In dichloromethane for 1h; Ambient temperature; | 95% |
| With trifluoroacetic acid In dichloromethane at 20℃; for 1h; Reagent/catalyst; |

| Conditions | Yield |
|---|---|
| With ammonium hydroxide; 1,3,5,7-tetramethyl-2,4,8-trioxa-6-phenyl-6-phosphaadamantane; palladium diacetate In toluene at 130℃; under 5171.62 Torr; Temperature; Autoclave; | 95% |
| With 1,4-diaza-bicyclo[2.2.2]octane; N-methoxylamine hydrochloride; sodium iodide; palladium dichloride In acetonitrile at 90℃; under 3800.26 Torr; for 8h; Autoclave; Inert atmosphere; | 93% |
| With 1,4-diaza-bicyclo[2.2.2]octane; palladium 10% on activated carbon; ammonium carbamate; potassium iodide In acetonitrile at 90℃; for 8h; Autoclave; Green chemistry; | 90% |
-
-
3717-21-3, 3717-22-4, 140461-28-5, 140461-29-6, 3235-04-9
p-methoxyl benzaldoxime

-
-
3424-93-9
4-methoxyphenylacetamide

| Conditions | Yield |
|---|---|
| With silver tetrafluoroborate; chloro(1,3-bis(2,6-di-i-propylphenyl)imidazol-2-ylidene)gold(I) at 100℃; for 20h; Beckmann rearrangement; Sealed tube; Neat (no solvent); | 95% |
| With C55H45ClN5P2Ru(1+)*Cl(1-) In water at 110℃; for 12h; Sealed tube; | 94% |
| With C82H80N4O6; mercury dichloride In tetrahydrofuran; water; toluene at 20℃; for 5h; Beckmann Rearrangement; | 93% |

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid; 1,2-bis-(diphenylphosphino)ethane; carbonyl(dihydro)tris(triphenylphosphine)ruthenium(II) In toluene Heating; | 93% |
| With cerium(III) chloride; silica gel; sodium iodide for 0.0666667h; Beckmann rearrangement; microwave irradiation; | 80% |
-
-
104-92-7
1-bromo-4-methoxy-benzene

-
-
15226-74-1, 61091-28-9, 61117-58-6
dicobalt octacarbonyl

-
-
3424-93-9
4-methoxyphenylacetamide

| Conditions | Yield |
|---|---|
| With 1H-imidazole; 1,1'-bis-(diphenylphosphino)ferrocene; palladium diacetate; ammonium chloride; N-ethyl-N,N-diisopropylamine In 1,4-dioxane at 90℃; for 3h; Catalytic behavior; Reagent/catalyst; Solvent; Temperature; Sealed tube; | 93% |
-
-
38388-44-2
2-(4-methoxybenzoylamino)-2-methylpropionic acid

-
-
3424-93-9
4-methoxyphenylacetamide

| Conditions | Yield |
|---|---|
| With copper dichloride In dimethyl sulfoxide at 120℃; for 11h; | 93% |
| With copper dichloride In dimethyl sulfoxide at 120℃; for 11h; | 93% |
-
-
3717-21-3, 3717-22-4, 3235-04-9, 140461-28-5, 140461-29-6
(E)-4-methoxybenzaldoxime

-
-
3424-93-9
4-methoxyphenylacetamide

| Conditions | Yield |
|---|---|
| bis[dichloro(pentamethylcyclopentadienyl)iridium(III)] In toluene at 111℃; for 6h; | 92% |
| With indium(III) chloride; 4-methoxybenzonitrile In toluene for 18h; Reflux; | 91% |
| In ethanol for 20h; Quantum yield; Irradiation; |

| Conditions | Yield |
|---|---|
| Stage #1: p-Methoxybenzyl bromide With ammonia; iodine In water at 60℃; for 4h; Stage #2: With ammonia; dihydrogen peroxide In water at 0 - 20℃; | 92% |

| Conditions | Yield |
|---|---|
| Stage #1: benzylamine With N-chloro-succinimide In acetonitrile at 20℃; for 3h; Green chemistry; Stage #2: p-methylanizole With tert.-butylhydroperoxide; iron(II) acetate In decane; acetonitrile at 82℃; for 22h; Green chemistry; Stage #3: With palladium 10% on activated carbon; hydrogen In methanol at 20℃; for 12h; Green chemistry; | 92% |

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; iron(III) chloride hexahydrate; tetrabutylammonium iodide; ammonium hydroxide In water at 80℃; for 18h; Sealed tube; | 91% |
| With tert.-butylhydroperoxide; ammonium chloride; calcium carbonate In dihydrogen peroxide; acetonitrile at 80℃; for 10h; Catalytic behavior; Inert atmosphere; | 60% |

| Conditions | Yield |
|---|---|
| bis[dichloro(pentamethylcyclopentadienyl)iridium(III)] In toluene at 111℃; for 6h; | 90% |
-
-
123-11-5
4-methoxy-benzaldehyde

-
A
-
3424-93-9
4-methoxyphenylacetamide

-
B
-
874-90-8
4-methoxybenzonitrile

-
C
-
100-09-4
4-methoxybenzoic acid

| Conditions | Yield |
|---|---|
| With H2SO4*2NH2OH; Rh(OH)x/Al2O3 In water for 7h; Heating; | A 90% B 5 % Chromat. C 9 % Chromat. |
| Stage #1: 4-methoxy-benzaldehyde With pyridine; sodium persulfate; tris(2,2-bipyridine)ruthenium(II) hexafluorophosphate; 4-acetylamino-2,2,6,6-tetramethyl-1-piperidinoxy; 1,1,1,3,3,3-hexamethyl-disilazane In acetonitrile at 0.2℃; for 24h; Molecular sieve; Sealed tube; Irradiation; Stage #2: With tetrabutyl ammonium fluoride In tetrahydrofuran; acetonitrile for 1h; Molecular sieve; |


| Conditions | Yield |
|---|---|
| With sodium azide; iodine; sodium hydrogencarbonate In water at 100℃; for 2h; Reagent/catalyst; Temperature; Solvent; | 90% |
| With ammonia; iodine In water at 60℃; for 1h; Lieben-Haller-Bauer reaction; | 83% |
| With ammonia; water; iodine In tetrahydrofuran at 20℃; for 24h; Haller-Bauer reaction; | 83% |
| With sodium azide; 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical; oxygen; copper dichloride In water; N,N-dimethyl-formamide at 120℃; under 760.051 Torr; for 15h; Schlenk technique; chemoselective reaction; | 67% |
| With tert.-butylhydroperoxide; ammonia; tetra-(n-butyl)ammonium iodide In water at 100℃; for 16h; Green chemistry; | 53% |

| Conditions | Yield |
|---|---|
| With [{Au(SIPr)}2(μ-OH)][BF4] In tetrahydrofuran at 140℃; for 2h; Reagent/catalyst; Concentration; Microwave irradiation; | 90% |

| Conditions | Yield |
|---|---|
| With water; potassium carbonate In dimethyl sulfoxide at 110℃; for 12h; | 90% |

| Conditions | Yield |
|---|---|
| With ammonia; dihydrogen peroxide; iodine In water; dimethyl sulfoxide at 20℃; for 3h; Irradiation; | 90% |
-
-
2393-23-9
4-methoxy-benzylamine

-
A
-
3424-93-9
4-methoxyphenylacetamide

-
B
-
874-90-8
4-methoxybenzonitrile

| Conditions | Yield |
|---|---|
| With water; oxygen at 140℃; under 3800.26 Torr; for 10h; | A 89% B n/a |
| With ammonium hydroxide; oxygen In tert-Amyl alcohol at 110℃; under 1500.15 Torr; for 16h; | A 13.6% B 79.3% |

| Conditions | Yield |
|---|---|
| With tetraphosphorus decasulfide In 1,4-dioxane at 50 - 68℃; for 3h; Temperature; | 100% |
| With pyridin-1-ium-1-yl[pyridin-1-ium-1-yl(sulfido)phosphinothioyl]sulfanyl-sulfido-thioxo-phosphane In acetonitrile for 0.5h; Reflux; | 88% |
| With Lawessons reagent In tetrahydrofuran at 20℃; Reflux; | 80% |

| Conditions | Yield |
|---|---|
| With barium trifluoromethanesulfonate In toluene for 18h; Reflux; Inert atmosphere; | 100% |
| With C23H24ClIrN3OS(1+)*F6P(1-); caesium carbonate In toluene at 120℃; | 96% |
| With bis[dichloro(pentamethylcyclopentadienyl)iridium(III)]; caesium carbonate In toluene at 140℃; for 12h; | 84% |

| Conditions | Yield |
|---|---|
| With thionyl chloride; carbonic acid dimethyl ester at 80℃; for 7h; Temperature; | 99.2% |
| With trichloromethyl chloroformate In various solvent(s) 0-5 deg C then heated to 60 deg C, 5 min; | 97% |
| With ((1,3,5-triazine-2,4,6-triyl)tris(oxy))tris(triphenylphosphonium) chloride at 140 - 150℃; for 2h; | 97% |

| Conditions | Yield |
|---|---|
| With chloro-trimethyl-silane In acetonitrile at 10 - 20℃; for 24h; Inert atmosphere; | 99% |
| With chloro-trimethyl-silane In acetonitrile at 23℃; | |
| With chloro-trimethyl-silane In acetonitrile at 0 - 20℃; Inert atmosphere; |


| Conditions | Yield |
|---|---|
| With copper(l) iodide; 1,10-Phenanthroline In benzene at 60℃; for 11h; Inert atmosphere; | 99% |
| With copper(l) iodide; 1,10-Phenanthroline In benzene at 60℃; for 11h; Inert atmosphere; | 99% |
-
-
3424-93-9
4-methoxyphenylacetamide

-
-
123-11-5
4-methoxy-benzaldehyde

-
-
109163-43-1
4-methoxy-N-(4-methoxybenzyl)benzamide

| Conditions | Yield |
|---|---|
| With carbon monoxide In diethyl ether at 150 - 160℃; under 30003 Torr; Sealed tube; | 99% |
| With carbon monoxide; C14H10*Ru(2+)*C11H17O(1-)*F6P(1-) In diethyl ether at 150℃; under 22801.5 Torr; for 22h; Autoclave; | 70% |

| Conditions | Yield |
|---|---|
| With [bis(acetoxy)iodo]benzene; ammonia In methanol at 0 - 20℃; for 2h; Temperature; Hofmann Rearrangement; Inert atmosphere; | 99% |
-
-
3424-93-9
4-methoxyphenylacetamide

-
-
624-31-7
4-tolyl iodide

-
-
39192-94-4
N-(p-methylphenyl)-p-methoxybenzamide

| Conditions | Yield |
|---|---|
| With 2-Phenyl-1,3,2-dioxaborinane In 1,4-dioxane at 115℃; for 30h; Inert atmosphere; | 99% |
-
-
3424-93-9
4-methoxyphenylacetamide

-
-
124-41-4
sodium methylate

-
-
14803-72-6
methyl N-(4-methoxyphenyl)carbamate

| Conditions | Yield |
|---|---|
| Stage #1: 4-methoxyphenylacetamide; sodium methylate In methanol at 20℃; for 0.25h; Stage #2: With bis(1,3-dimethyl-2-imidazolidinone) hydrotribromide In methanol for 4h; Hofmann Rearrangement; Reflux; | 98% |
| With N-Bromosuccinimide In methanol for 0.166667h; Heating; | 87% |

| Conditions | Yield |
|---|---|
| With copper; potassium carbonate; N,N`-dimethylethylenediamine In toluene at 135℃; for 2h; Ullmann type reaction; Inert atmosphere; | 98% |
| With caesium carbonate; palladium diacetate; 4,5-bis(diphenylphosphino)-9,9-dimethylxanthene In 1,4-dioxane at 100℃; for 10h; | 95% |
| With palladium diacetate; caesium carbonate; 4,5-bis(diphenylphosphino)-9,9-dimethylxanthene In 1,4-dioxane at 100℃; | |
| With palladium diacetate; caesium carbonate; 4,5-bis(diphenylphosphino)-9,9-dimethylxanthene In 1,4-dioxane at 100℃; for 10h; Inert atmosphere; Sealed tube; |

| Conditions | Yield |
|---|---|
| With 3-[4-(diacetoxyiodo)phenoxy]-1-propyl-N,N,N-trimethylammonium 4-methylbenzenesulfonate; potassium hydroxide In chloroform at 0 - 20℃; Hofmann Rearrangement; Inert atmosphere; | 98% |

| Conditions | Yield |
|---|---|
| With samarium diiodide; water In tetrahydrofuran at 23℃; for 0.0833333h; Reagent/catalyst; Inert atmosphere; Schlenk technique; chemoselective reaction; | 98% |
| With samarium diiodide; water; triethylamine In tetrahydrofuran at 23℃; for 18h; Inert atmosphere; chemoselective reaction; | 92% |
| With C24H20ClN2OPRu; potassium tert-butylate; hydrogen In tetrahydrofuran at 110℃; under 10640.7 Torr; for 36h; Inert atmosphere; Schlenk technique; | 82% |
-
-
3424-93-9
4-methoxyphenylacetamide

-
-
814-75-5
3-bromo-butanone

-
-
124811-80-9
2-(4-methoxy-phenyl)-4,5-dimethyl-oxazole

| Conditions | Yield |
|---|---|
| With silver(I) trifluoromethoxide In ethyl acetate at 50℃; for 4h; Darkness; | 98% |

| Conditions | Yield |
|---|---|
| With dmap; triethylamine In dichloromethane at 20℃; | 98% |
-
-
3424-93-9
4-methoxyphenylacetamide

-
-
70-11-1
α-bromoacetophenone

-
-
25205-87-2
2-(4-methoxyphenyl)-4-phenyloxazole

| Conditions | Yield |
|---|---|
| With silver(I) trifluoromethoxide In ethyl acetate at 50℃; for 4h; Reagent/catalyst; Temperature; Darkness; | 97% |
| at 140℃; for 1h; | 80% |
| With silver trifluoromethanesulfonate In ethyl acetate at 50℃; Inert atmosphere; | 64% |
| Heating; |
-
-
70-23-5
ethyl Bromopyruvate

-
-
3424-93-9
4-methoxyphenylacetamide

-
-
78979-61-0
ethyl 2-(4-methoxyphenyl)-1,3-oxazole-4-carboxylate

| Conditions | Yield |
|---|---|
| With silver hexafluoroantimonate In 1,2-dichloro-ethane at 90℃; for 2h; Inert atmosphere; Microwave irradiation; | 97% |
| With silver hexafluoroantimonate In dichloromethane at 90℃; under 3750.38 Torr; for 1h; Sealed tube; Microwave irradiation; | |
| In ethanol at 70℃; for 5h; | |
| Stage #1: ethyl Bromopyruvate; 4-methoxyphenylacetamide With sodium hydrogencarbonate In tetrahydrofuran at 60℃; for 23h; Stage #2: With trifluoroacetic anhydride In tetrahydrofuran at 20℃; for 12h; |
-
-
3424-93-9
4-methoxyphenylacetamide

-
-
24424-99-5
di-tert-butyl dicarbonate

| Conditions | Yield |
|---|---|
| With dmap In neat (no solvent) for 4h; Milling; chemoselective reaction; | 97% |
| With dmap In acetonitrile at 20℃; for 15h; Inert atmosphere; Sealed tube; | 90% |
| With dmap In acetonitrile at 20℃; for 15h; | 86% |
-
-
50-00-0
formaldehyd

-
-
3424-93-9
4-methoxyphenylacetamide

-
-
41125-75-1
5-(4-methoxy)phenyl-1,3,4-oxadiazol-2(3H)-one

| Conditions | Yield |
|---|---|
| With (1,1'-bis(diphenylphosphino)ferrocene)palladium(II) dichloride; ammonium cerium (IV) nitrate; nickel diethyldithiocarbamate; N-fluorobis(benzenesulfon)imide; 1-butyl-methylpyrrolidinium bis(trifluoromethylsulfonyl)amide; 1-hydroxy-3H-benz[d][1,2]iodoxole-1,3-dione In dimethyl sulfoxide at 60℃; for 10h; | 96.8% |
-
-
674-82-8
4-methyleneoxetan-2-one

-
-
3424-93-9
4-methoxyphenylacetamide

-
-
77656-07-6
4-Methoxy-N-(3-oxo-butyryl)-benzamide

| Conditions | Yield |
|---|---|
| With Aliquat 336 In toluene for 10h; Heating; | 96% |
| With chloro-trimethyl-silane; sodium iodide In acetonitrile for 2h; Ambient temperature; | 90% |
| With chloro-trimethyl-silane; sodium iodide In acetonitrile for 2h; Ambient temperature; | 89.5% |
4-methoxybenzamide Chemical Properties
Product Name: 4-Methoxybenzamide
The MF of 4-Methoxybenzamide (3424-93-9) is C8H9NO2.
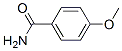
The MW of 4-Methoxybenzamide (3424-93-9) is 151.16.
Synonyms of 4-Methoxybenzamide (3424-93-9): p-Anisamide ; p-Methoxybenzamide
Product Categories: Aromatic Carboxylic Acids,Anilides, Anhydrides & Salts
Form: white crystalline powder
Index of Refraction: 1.546
EINECS: 222-319-6
Density: 1.143 g/ml
Flash Point: 161.5 °C
Boiling Point: 295.8 °C
Melting Point: 164-167 °C
BRN: 1862847
4-methoxybenzamide Toxicity Data With Reference
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mammal (species unspecified) | LD50 | unreported | 1gm/kg (1000mg/kg) | Pharmaceutical Chemistry Journal Vol. 28, Pg. 335, 1994. |
4-methoxybenzamide Safety Profile
Safety information of 4-Methoxybenzamide (3424-93-9):
Risk Statements
36/37/38 Irritating to eyes, respiratory system and skin
Safety Statements
22 Do not breathe dust
24/25 Avoid contact with skin and eyes
WGK Germany 3
RTECS CV5466666
Related Products
- 4-methoxybenzamide
- 3424-98-4
- 34252-44-3
- 34252-54-5
- 34253-01-5
- 34253-03-7
- 3425-61-4
- 34256-78-5
- 34256-82-1
- 342577-38-2
- 3425-89-6
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View