-
Name
5-Cyanoindole
- EINECS 239-986-4
- CAS No. 15861-24-2
- Article Data76
- CAS DataBase
- Density 1.24 g/cm3
- Solubility Soluble in chloroform, hexane and methanol. Insoluble in water.
- Melting Point 106-108 °C(lit.)
- Formula C9H6N2
- Boiling Point 350 °C at 760 mmHg
- Molecular Weight 142.16
- Flash Point 121.9 °C
- Transport Information
- Appearance off-white crystalline powder
- Safety 26-36-22
- Risk Codes 36/37/38-20/21/22
-
Molecular Structure
-
Hazard Symbols
 Xn;
Xn;  Xi
Xi
- Synonyms 5-Cyanoindol;5-cyano indole;5-Cyano-1H-indole;1H-Indole-5-carbonitrile;
- PSA 39.58000
- LogP 2.03958
Synthetic route

| Conditions | Yield |
|---|---|
| With [(2-di-tert-butylphosphino-2′,4′,6′-triisopropyl-1, 1′-biphenyl)-2-(2′-amino-1,1′-biphenyl)] palladium(II) methanesulfonate In tetrahydrofuran; water at 40℃; for 18h; Inert atmosphere; | 99% |
| With 1,1'-bis-(diphenylphosphino)ferrocene; palladium 10% on activated carbon; zinc(II) formate dihydrate In N,N-dimethyl acetamide at 110℃; for 12h; Inert atmosphere; | 90% |
-
-
174500-89-1
5-cyanoindole-3-carboxylic acid

-
-
15861-24-2
1H-indole-5-carbonitrile

| Conditions | Yield |
|---|---|
| With potassium carbonate In ethanol at 140℃; Schlenk technique; | 99% |
-
-
412947-51-4
4-amino-3-ethynylbenzonitrile

-
-
15861-24-2
1H-indole-5-carbonitrile

| Conditions | Yield |
|---|---|
| With P(p-C6H4F)3; chloro(1,5-cyclooctadiene)rhodium(I) dimer In N,N-dimethyl-formamide at 85℃; for 2h; | 98% |
| With chloro(cyclopentadienyl)bis(triphenylphosphine)ruthenium (II) In pyridine at 90℃; for 0.5h; Inert atmosphere; regioselective reaction; | 98% |
| With pyridine; chloro(cyclopentadienyl)bis(triphenylphosphine)ruthenium (II) at 90℃; for 0.5h; Catalytic behavior; Inert atmosphere; regioselective reaction; | 98% |
-
-
475102-10-4
1-(tert-butoxycarbonyl)-1H-indole-5-carbonitrile

-
-
15861-24-2
1H-indole-5-carbonitrile

| Conditions | Yield |
|---|---|
| In various solvent(s) at 150℃; for 0.0833333h; microwave irradiation; | 98% |
| With 1,1,1,3',3',3'-hexafluoro-propanol at 150℃; for 0.0833333h; Product distribution / selectivity; Microwave irradiation; | 98% |
| With 2,2,2-trifluoroethanol at 150℃; for 1h; Product distribution / selectivity; Microwave irradiation; | 98% |
| With 1,1,1,3',3',3'-hexafluoro-propanol at 150℃; for 0.0833333h; Product distribution / selectivity; Microwave irradiation; | 98% |

| Conditions | Yield |
|---|---|
| palladium diacetate; PS-triphenylphosphine In N,N-dimethyl-formamide at 140℃; for 0.833333h; Irradiation; microwave; | 96% |
-
-
96784-54-2
3-methyl-4-nitrobenzonitrile

-
-
4637-24-5
N,N-dimethyl-formamide dimethyl acetal

-
-
15861-24-2
1H-indole-5-carbonitrile

| Conditions | Yield |
|---|---|
| Stage #1: 3-methyl-4-nitrobenzonitrile; N,N-dimethyl-formamide dimethyl acetal With pyrrolidine In dichloromethane at 50 - 55℃; for 8h; Large scale; Green chemistry; Stage #2: With iron; acetic acid In methanol at 0 - 55℃; for 8h; Reagent/catalyst; Solvent; Temperature; Large scale; Green chemistry; | 96% |
| Stage #1: 3-methyl-4-nitrobenzonitrile; N,N-dimethyl-formamide dimethyl acetal With pyrrolidine In 1,4-dioxane at 102℃; for 2.5h; Leimgruber-Batcho Indole Synthesis; Reflux; Inert atmosphere; Stage #2: With hydrazine hydrate In 1,4-dioxane at 45℃; for 1h; Leimgruber-Batcho Indole Synthesis; | 71% |

| Conditions | Yield |
|---|---|
| With copper(l) iodide; potassium iodide; N,N`-dimethylethylenediamine In toluene at 110℃; for 24h; Rosenmund-von Braun reaction; | 95% |
| Stage #1: 5-bromo-1H-indole; sodium cyanide With potassium iodide; N,N`-dimethylethylenediamine; copper(l) iodide In toluene at 110℃; for 24h; Stage #2: With ammonia In water; ethyl acetate; toluene at 25℃; for 0.166667h; | 95% |
| With copper(l) iodide; tetrakis(triphenylphosphine) palladium(0) In various solvent(s) at 115℃; for 2h; Substitution; | 76% |

| Conditions | Yield |
|---|---|
| With copper(l) iodide; tetrakis(triphenylphosphine) palladium(0) In acetonitrile for 2.5h; Reflux; | 95% |
| With C18H14CuIN4 In acetonitrile at 20℃; for 24h; Inert atmosphere; Sealed tube; UV-irradiation; | 53% |

| Conditions | Yield |
|---|---|
| With 1,4-diaza-bicyclo[2.2.2]octane; copper(I) oxide; phenylsilane; ammonia In 1-methyl-pyrrolidin-2-one at 180℃; under 2280.15 Torr; for 24h; Sealed tube; chemoselective reaction; | 94% |
| With ammonia; hydrogen; nickel dibromide In N,N-dimethyl-formamide at 160℃; for 10h; Sealed tube; | 92 %Chromat. |
-
-
80531-13-1
1-benzyl-1H-indole-5-carbonitrile

-
-
15861-24-2
1H-indole-5-carbonitrile

| Conditions | Yield |
|---|---|
| With potassium tert-butylate; oxygen; dimethyl sulfoxide at 0℃; for 0.333333h; | 93% |

-
-
128810-31-1
1H-indol-5-yl methanesulfonate

-
-
15861-24-2
1H-indole-5-carbonitrile

| Conditions | Yield |
|---|---|
| With 2-[2-(dicyclohexylphosphino)-phenyl]-1-methyl-1H-indole; palladium diacetate; potassium carbonate In water; tert-butyl alcohol at 80℃; for 18h; Inert atmosphere; | 93% |
-
-
32685-23-7
5-Cyano-1-(p-toluenesulfonyl)indole

-
-
15861-24-2
1H-indole-5-carbonitrile

| Conditions | Yield |
|---|---|
| With formic acid; (4,4'-di-tert-butyl-2,2'-dipyridyl)-bis-(2-phenylpyridine(-1H))-iridium(III) hexafluorophosphate; N-ethyl-N,N-diisopropylamine In acetonitrile at 20℃; for 24h; Inert atmosphere; Sealed tube; Irradiation; | 93% |
| With sodium hydride In N,N-dimethyl acetamide at 60℃; for 2h; Inert atmosphere; | 87% |
| With water; sodium hydroxide In methanol at 60℃; for 2h; | 84% |

| Conditions | Yield |
|---|---|
| With potassium fluoride; 4,4'-dimethyl-2,2'-bipyridines; trifluoroacetic acid; sodium iodide; nickel dichloride; zinc In N,N-dimethyl acetamide at 60℃; for 36h; | 93% |
-
-
10075-50-0
5-bromo-1H-indole

-
-
15861-24-2
1H-indole-5-carbonitrile

| Conditions | Yield |
|---|---|
| With tris-(dibenzylideneacetone)dipalladium(0); tris(2-morpholinophenyl)phosphine; potassium carbonate In water; tert-butyl alcohol at 85℃; for 10h; Schlenk technique; Inert atmosphere; | 92% |
| With tetrakis(triphenylphosphine) palladium(0); 1,8-diazabicyclo[5.4.0]undec-7-ene In water; tert-butyl alcohol at 85℃; for 8h; Inert atmosphere; | 85% |
| With sodium carbonate In water; N,N-dimethyl-formamide at 120℃; for 24h; | 76% |

-
-
17422-32-1
5-chloro-1H-indole

-
-
15861-24-2
1H-indole-5-carbonitrile

| Conditions | Yield |
|---|---|
| With 2-[2-(dicyclohexylphosphino)-phenyl]-1-methyl-1H-indole; palladium diacetate; sodium carbonate; triethylamine In water; acetonitrile at 70℃; for 18h; | 91% |

-
-
15861-24-2
1H-indole-5-carbonitrile

| Conditions | Yield |
|---|---|
| With tetraethylammonium perchlorate In N,N-dimethyl-formamide at 20℃; Electrolysis; | 90% |

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide for 20h; Heating; | 88% |
| In 1-methyl-pyrrolidin-2-one for 3h; Inert atmosphere; Reflux; | 82% |
| With 1-methyl-pyrrolidin-2-one at 155℃; for 6h; Inert atmosphere; | 80% |
| In 1-methyl-pyrrolidin-2-one at 100 - 200℃; for 1h; Microwave irradiation; | |
| In 1-methyl-pyrrolidin-2-one at 230℃; for 5h; |

| Conditions | Yield |
|---|---|
| With copper(l) iodide; trans-N,N'-dimethylcyclohexane-1,2-diamine In acetonitrile at 200℃; for 1h; Microwave irradiation; Inert atmosphere; | 88% |

| Conditions | Yield |
|---|---|
| Stage #1: 5-bromo-1H-indole; copper(I) cyanide In 1-methyl-pyrrolidin-2-one for 18h; Heating / reflux; Stage #2: With ammonia In 1-methyl-pyrrolidin-2-one; water | 82% |
| In 1-methyl-pyrrolidin-2-one for 0.5h; Rosenmund-von Braun reaction; microwave irradiation; | 79% |
| With 1-methyl-pyrrolidin-2-one for 4.5h; Heating; | 72.6% |
-
-
10075-50-0
5-bromo-1H-indole

-
-
77287-34-4, 77287-35-5, 60100-09-6
formamide

-
-
15861-24-2
1H-indole-5-carbonitrile

| Conditions | Yield |
|---|---|
| With [2,2]bipyridinyl; aluminum (III) chloride; nickel(II) acetylacetonate; zinc(II) oxide In 1,2-dimethoxyethane at 145℃; for 12h; | 79% |

| Conditions | Yield |
|---|---|
| With copper(l) iodide; triphenylphosphine In N,N-dimethyl-formamide at 140℃; for 24h; Inert atmosphere; | 77% |
-
-
557-21-1
zinc(II) cyanide

-
-
15861-24-2
1H-indole-5-carbonitrile

| Conditions | Yield |
|---|---|
| With bis(1,5-cyclooctadiene)nickel (0); 1,2-bis-(dicyclohexylphosphino)ethane In 1,4-dioxane at 110℃; for 15h; Inert atmosphere; | 76% |

| Conditions | Yield |
|---|---|
| With dmap; 1,1'-bis-(diphenylphosphino)ferrocene; nickel(II) chloride hexahydrate; zinc In acetonitrile at 80℃; for 6.5h; Schlenk technique; Inert atmosphere; Sealed tube; | 72% |
| With dmap; 1,1'-bis-(diphenylphosphino)ferrocene; nickel(II) chloride hexahydrate; zinc In acetonitrile at 80℃; for 6.5h; Inert atmosphere; Sealed tube; | 72% |

| Conditions | Yield |
|---|---|
| With ISOPROPYLAMIDE; sodium carbonate at 110℃; for 15h; | 68% |
-
-
16066-91-4
5-iodo-1H-indole

-
-
77287-34-4, 77287-35-5, 60100-09-6
formamide

-
-
15861-24-2
1H-indole-5-carbonitrile

| Conditions | Yield |
|---|---|
| With palladium diacetate; 4,5-bis(diphenylphos4,5-bis(diphenylphosphino)-9,9-dimethylxanthenephino)-9,9-dimethylxanthene; trichlorophosphate at 140℃; for 48h; Inert atmosphere; | 67% |
| With copper(l) iodide; triphenylphosphine; trichlorophosphate at 140℃; for 24h; Inert atmosphere; | 66% |
| Stage #1: 5-iodo-1H-indole; formamide With chloro(1,5-cyclooctadiene)rhodium(I) dimer; 4,5-bis(diphenylphos4,5-bis(diphenylphosphino)-9,9-dimethylxanthenephino)-9,9-dimethylxanthene at 20℃; Inert atmosphere; Stage #2: With trichlorophosphate at 135 - 140℃; for 24h; Inert atmosphere; | 50 %Chromat. |
| With zinc diacetate; nixantphos In 1-methyl-pyrrolidin-2-one at 140℃; for 24h; Inert atmosphere; | 86 %Chromat. |
-
-
81881-74-5
5-aminomethylindole

-
A
-
15861-24-2
1H-indole-5-carbonitrile

-
B
-
1196-69-6
1H-indole-5-carboxaldehyde

| Conditions | Yield |
|---|---|
| With dichloro[1,3-bis(2-methylphenyl)-2-imidazolidinylidene](benzylidene) (tricyclohexylphosphine) ruthenium(II); oxygen In N,N-dimethyl-formamide at 120℃; under 760.051 Torr; for 2h; Sealed tube; | A 67% B 10% |
-
-
557-21-1
zinc(II) cyanide

-
-
128810-31-1
1H-indol-5-yl methanesulfonate

-
-
15861-24-2
1H-indole-5-carbonitrile

| Conditions | Yield |
|---|---|
| With dmap; nickel(II) bromide dimethoxyethane; 1,4-di(diphenylphosphino)-butane; zinc In acetonitrile at 80℃; for 12h; Sealed tube; Inert atmosphere; | 65% |
-
-
15861-23-1
indoline-5-carbonitrile

-
-
15861-24-2
1H-indole-5-carbonitrile

| Conditions | Yield |
|---|---|
| With tetrasodium cobalt(II) 4,4',4'',4'''-tetrasulphophthalocyanine In water; ethyl acetate at 20℃; for 36h; Irradiation; Green chemistry; | 65% |
| With potassium tert-butylate In o-xylene at 140℃; for 36h; Inert atmosphere; | 10% |

| Conditions | Yield |
|---|---|
| With 1,10-Phenanthroline; hexakis(acetonitrile)nickel(II) tetrafluoroborate; 2,3,5,6-tetramethyl-1,4-bis(trimethylsilyl)-1,4-diaza-2,5-cyclo-hexadiene at 80℃; for 24h; | 53% |
-
-
15861-24-2
1H-indole-5-carbonitrile

-
-
81881-74-5
5-aminomethylindole

| Conditions | Yield |
|---|---|
| With ammonia; hydrogen; nickel In methanol; water | 100% |
| With [Ru(H)(BH4)(CO)(PPh3)(3-(di-tert-butylphosphino)-N-((1-methyl-1H-imidazol-2 yl)methyl)propylamine)]; hydrogen In isopropyl alcohol at 150℃; for 3h; Inert atmosphere; Autoclave; | 98% |
| With lithium aluminium tetrahydride In tetrahydrofuran for 3h; Inert atmosphere; Reflux; | 94% |
-
-
15861-24-2
1H-indole-5-carbonitrile

-
-
2969-81-5
4-bromoethylbutanoate

-
-
1229952-77-5
4-(5-cyano-1H-indol-1-yl)butanoic acid

| Conditions | Yield |
|---|---|
| Stage #1: 1H-indole-5-carbonitrile; Ethyl 4-bromobutyrate With tetra(n-butyl)ammonium hydroxide; sodium hydroxide In dichloromethane at 20℃; for 6h; Stage #2: With hydrogenchloride In water | 100% |

| Conditions | Yield |
|---|---|
| With potassium phosphate; copper(l) iodide; N,N`-dimethylethylenediamine In toluene at 110℃; for 24h; | 99% |
| With potassium carbonate In N,N-dimethyl-formamide for 8h; Reflux; | 98% |
| With copper(l) iodide; (1R,2R)-1,2-diaminocyclohexane; potassium carbonate In water at 80℃; for 18h; Buchwald N-arylation; | 91% |
| With copper(l) iodide; 1,10-Phenanthroline; potassium hydroxide In water at 95℃; for 20h; Catalytic behavior; Solvent; Schlenk technique; Inert atmosphere; Sealed tube; | 78% |
| With copper(l) iodide; caesium carbonate In N,N-dimethyl-formamide at 110℃; for 24h; Ullmann-type N-arylation; | 56% |
-
-
15861-24-2
1H-indole-5-carbonitrile

-
-
91897-73-3
(1E,4E)-1-phenyl-1,4-hexadien-3-one

-
-
1128087-20-6
C21H18N2O

| Conditions | Yield |
|---|---|
| Stage #1: 1H-indole-5-carbonitrile; (1E,4E)-1-phenyl-1,4-hexadien-3-one With 2,4-dinitrobenzenesulfonic acid In acetonitrile at 0℃; for 4.5h; Friedel-Crafts alkylation; Inert atmosphere; Stage #2: In methanol at -20 - 20℃; for 48h; Inert atmosphere; | 99% |
-
-
88-12-0
1-ethenyl-2-pyrrolidinone

-
-
15861-24-2
1H-indole-5-carbonitrile

-
-
1262894-55-2
3-(1-(2-oxopyrrolidin-1-yl)ethyl)-1H-indole-5-carbonitrile

| Conditions | Yield |
|---|---|
| With iron(III) chloride In dichloromethane at 40℃; for 6h; regioselective reaction; | 99% |
| With tris(p-bromophenyl)ammoniumyl hexachloroantimonate In chloroform at 20℃; for 0.5h; Friedel-Crafts Alkylation; Green chemistry; regiospecific reaction; | 66% |
| With tris(p-bromophenylammoniumyl) hexachloroantimonate In chloroform at 20℃; for 0.5h; Friedel-Crafts Alkylation; Green chemistry; regioselective reaction; | 66% |
-
-
15861-24-2
1H-indole-5-carbonitrile

-
-
95151-36-3
methyl oct-1-en-3-yl carbonate

| Conditions | Yield |
|---|---|
| With chloro(1,5-cyclooctadiene)rhodium(I) dimer; C39H51N2O3P In acetonitrile at 100℃; for 36h; enantioselective reaction; | 99% |
-
-
15861-24-2
1H-indole-5-carbonitrile

-
-
98-59-9
p-toluenesulfonyl chloride

-
-
32685-23-7
5-Cyano-1-(p-toluenesulfonyl)indole

| Conditions | Yield |
|---|---|
| With tetrabutylammomium bromide; sodium hydroxide In toluene at 20℃; for 1h; | 98% |
| With tetrabutylammomium bromide; sodium hydroxide In water; toluene at 20℃; for 1h; | 98% |
| With tetrabutylammomium bromide; sodium hydroxide In toluene at 20℃; for 1h; | 98.1% |
-
-
15861-24-2
1H-indole-5-carbonitrile

-
-
100-52-7
benzaldehyde

-
-
787631-74-7
3-(hydroxyphenylmethyl)indole-5-carbonitrile

| Conditions | Yield |
|---|---|
| With sodium hydroxide In methanol at 20℃; for 96h; | 98% |
-
-
15861-24-2
1H-indole-5-carbonitrile

-
-
434-45-7
1,1,1-trifluoroacetophenone

-
-
1150561-06-0
3-(2,2,2-trifluoro-1-hydroxy-1-phenylethyl)-1H-indole-5-carbonitrile

| Conditions | Yield |
|---|---|
| With N,N,N′,N′-tetramethyl-N″-tert-butylguanidine; water at 20℃; for 16h; Friedel-Crafts type alkylation; | 98% |
| With TMG In dichloromethane at 20℃; |
-
-
15861-24-2
1H-indole-5-carbonitrile

-
-
5292-43-3
bromoacetic acid tert-butyl ester

-
-
1229608-56-3
tert-butyl (5-cyano-1H-indol-1-yl)acetate

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetonitrile for 16h; Reflux; | 98% |
-
-
15861-24-2
1H-indole-5-carbonitrile

-
-
610-14-0
2-nitrobenzyl chloride

| Conditions | Yield |
|---|---|
| Stage #1: 1H-indole-5-carbonitrile With tetra(n-butyl)ammonium hydrogensulfate; sodium hydride In N,N-dimethyl-formamide at 0℃; for 0.333333h; Inert atmosphere; Stage #2: 2-nitrobenzyl chloride In N,N-dimethyl-formamide at 0 - 20℃; for 2h; | 98% |
| With dmap; triethylamine In dichloromethane at 20℃; Inert atmosphere; |

| Conditions | Yield |
|---|---|
| Stage #1: 1H-indole-5-carbonitrile With sodium hydride In N,N-dimethyl-formamide; mineral oil at 50℃; for 1h; Inert atmosphere; Stage #2: Isobutyl bromide In N,N-dimethyl-formamide; mineral oil at 20℃; | 98% |
-
-
15861-24-2
1H-indole-5-carbonitrile

-
-
1414941-97-1
2-oxo-3,3′-biindoline-5,5′-dicarbonitrile

| Conditions | Yield |
|---|---|
| With pyridine; bis-[(trifluoroacetoxy)iodo]benzene; bis(pinacol)diborane In toluene at 60℃; for 11h; Reagent/catalyst; | 97.9% |
| With morpholine; copper(I) oxide; tert.-butylhydroperoxide; oxygen In chloroform at 20℃; under 760.051 Torr; for 18h; regioselective reaction; | 56% |
| With pyridine; 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical; water; oxygen; silver nitrate; benzoic acid at 65℃; for 48h; | 37% |
| With 2,2,6,6-tetramethyl-piperidine-N-oxyl; benzoic acid In acetonitrile at 65℃; for 72h; | 15% |
-
-
15861-24-2
1H-indole-5-carbonitrile

-
-
80531-13-1
1-benzyl-1H-indole-5-carbonitrile

| Conditions | Yield |
|---|---|
| With sodium hydride; potassium iodide In mineral oil at 0 - 80℃; | 97.3% |
-
-
15861-24-2
1H-indole-5-carbonitrile

-
-
68-12-2, 33513-42-7
N,N-dimethyl-formamide

-
-
17380-18-6
3-formyl-1H-indole-5-carbonitrile

| Conditions | Yield |
|---|---|
| Stage #1: N,N-dimethyl-formamide With trichlorophosphate for 0.0833333h; Inert atmosphere; Cooling with ice; Stage #2: 1H-indole-5-carbonitrile at 25℃; Inert atmosphere; Cooling with ice; Stage #3: With potassium hydroxide In water at 25℃; Reflux; | 97% |
| Stage #1: N,N-dimethyl-formamide With trichlorophosphate at 0℃; for 0.333333h; Vilsmeier formylation; Stage #2: 1H-indole-5-carbonitrile at 35℃; for 1h; | 94% |
| Stage #1: N,N-dimethyl-formamide With trichlorophosphate at 0 - 5℃; for 0.333333h; Stage #2: 1H-indole-5-carbonitrile at 0 - 30℃; for 3h; Stage #3: With sodium hydroxide In water pH=8 - 9; Reflux; | 92.19% |
-
-
15861-24-2
1H-indole-5-carbonitrile

-
-
24424-99-5
di-tert-butyl dicarbonate

-
-
475102-10-4
1-(tert-butoxycarbonyl)-1H-indole-5-carbonitrile

| Conditions | Yield |
|---|---|
| With pyridine; dmap In dichloromethane at 20℃; for 24h; | 97% |
| With dmap In dichloromethane at 20℃; for 0.75h; | 96.7% |
| Stage #1: 1H-indole-5-carbonitrile With dmap In tetrahydrofuran at 20℃; for 0.5h; Stage #2: di-tert-butyl dicarbonate for 2h; | 96% |
-
-
15861-24-2
1H-indole-5-carbonitrile

| Conditions | Yield |
|---|---|
| With sodium hydride; potassium iodide In mineral oil at 0 - 80℃; | 96.1% |
-
-
15861-24-2
1H-indole-5-carbonitrile

-
-
98-09-9
benzenesulfonyl chloride

-
-
144340-21-6
1-benzenesulfonyl-1H-indole-5-carbonitrile

| Conditions | Yield |
|---|---|
| Stage #1: 1H-indole-5-carbonitrile With tetrabutylammomium bromide; sodium hydroxide In water; toluene for 0.5h; Inert atmosphere; Stage #2: benzenesulfonyl chloride In water; toluene at 20℃; for 25h; Inert atmosphere; | 96% |
| With tetrabutylammomium bromide; sodium hydroxide In water; toluene at 20℃; for 25h; Inert atmosphere; | 96% |
| Stage #1: 1H-indole-5-carbonitrile With sodium hydride In tetrahydrofuran at 0℃; Stage #2: benzenesulfonyl chloride In tetrahydrofuran at 0 - 20℃; | 86% |
-
-
15861-24-2
1H-indole-5-carbonitrile

-
-
1147550-11-5
ammonium thiocyanate

-
-
1044513-31-6
3-thiocyanato-1H-indole-5-carbonitrile

| Conditions | Yield |
|---|---|
| With ammonium cerium (IV) nitrate In methanol at 25℃; for 0.25h; | 96% |
| With Oxone In methanol at 20℃; | 95% |
| With Selectfluor In acetonitrile at 20℃; for 0.166667h; | 92% |
| With Selectfluor In acetonitrile at 20℃; for 2h; Inert atmosphere; | 60% |
-
-
15861-24-2
1H-indole-5-carbonitrile

-
-
13089-11-7
methyl 3,3,3-trifluoropyruvate

-
-
1241940-64-6
(R)-methyl 2-(5-cyano-1H-indol-3-yl)-3,3,3-trifluoro-2-hydroxypropanoate

| Conditions | Yield |
|---|---|
| With (R)-3,3'-bis(1-naphthyl)-5,5',6,6',7,7',8,8'-octahydro-1,1'-binaphthyl posphate alenyl-2,2'-diol In toluene at -78℃; for 94h; Friedel-Crafts alkylation; optical yield given as %ee; enantioselective reaction; | 96% |
| Stage #1: 1H-indole-5-carbonitrile With C78H78N12O6 In tetrahydrofuran at -20℃; for 0.5h; Inert atmosphere; Stage #2: methyl 3,3,3-trifluoropyruvate In tetrahydrofuran at -20℃; for 12h; Inert atmosphere; enantioselective reaction; | 81% |
-
-
15861-24-2
1H-indole-5-carbonitrile

-
-
163797-89-5
N'-hydroxy-1H-indole-5-carboximidamide

| Conditions | Yield |
|---|---|
| With hydroxylamine hydrochloride; triethylamine In ethanol at 80℃; | 96% |
| With hydroxylamine hydrochloride; sodium carbonate In ethanol; water for 3h; Reflux; | 90% |
| With ethanol; hydroxylamine hydrochloride; triethylamine at 0 - 80℃; | 82.3% |
-
-
15861-24-2
1H-indole-5-carbonitrile

-
-
1506-56-5
7-ethoxy-4,7-dioxoheptanoic acid chloride

| Conditions | Yield |
|---|---|
| With dmap; triethylamine In dichloromethane at 20℃; for 72h; Inert atmosphere; | 96% |
-
-
15861-24-2
1H-indole-5-carbonitrile

-
-
1044513-31-6
3-thiocyanato-1H-indole-5-carbonitrile

| Conditions | Yield |
|---|---|
| In acetonitrile for 0.166667h; | 96% |
-
-
15861-24-2
1H-indole-5-carbonitrile

-
-
57738-66-6
S,S-dimethyl-S-(trifluoroacetoxy)sulfonium trifluoroacetate

-
-
1440957-20-9
(5-cyanoindol-3-yl)dimethylsulfonium perchlorate

| Conditions | Yield |
|---|---|
| Stage #1: 1H-indole-5-carbonitrile; S,S-dimethyl-S-(trifluoroacetoxy)sulfonium trifluoroacetate In dichloromethane at -78 - 20℃; Inert atmosphere; Stage #2: With lithium perchlorate In dichloromethane; water at 20℃; Inert atmosphere; Cooling with ice; | 95.5% |
5-Cyanoindole Specification
The 5-Cyanoindole, with the CAS registry number 15861-24-2 and EINECS registry number 239-986-4, has the systematic name and IUPAC name of 1H-indole-5-carbonitrile. And the molecular formula of this chemical is C9H6N2. It is a off-white crystalline powder which is sensitive to light and air, and it should be stored at 0-6°C. What's more, it is used as medicine intermediate and inhibitor of enzyme.
The physical properties of 5-Cyanoindole are as following:
(1)ACD/LogP: 2.36; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 2.36; (4)ACD/LogD (pH 7.4): 2.36; (5)ACD/BCF (pH 5.5): 36.55; (6)ACD/BCF (pH 7.4): 36.55; (7)ACD/KOC (pH 5.5): 457.47; (8)ACD/KOC (pH 7.4): 457.47; (9)#H bond acceptors: 2; (10)#H bond donors: 1; (11)#Freely Rotating Bonds: 0; (12)Polar Surface Area: 28.72 Å2; (13)Index of Refraction: 1.674; (14)Molar Refractivity: 42.8 cm3; (15)Molar Volume: 113.9 cm3; (16)Polarizability: 16.96×10-24cm3; (17)Surface Tension: 61.5 dyne/cm; (18)Density: 1.24 g/cm3; (19)Flash Point: 121.9 °C; (20)Enthalpy of Vaporization: 59.46 kJ/mol; (21)Boiling Point: 350 °C at 760 mmHg; (22)Vapour Pressure: 4.51E-05 mmHg at 25°C.
Uses of 5-Cyanoindole:
It can react with 2-fluoro-pyridine to produce 5-cyano-N-(2-pyridyl)indole. This reaction will need reagent sodium hydride, and the solvent dimethylformamide. The reaction time is 2 hours with temperature of 95-100°C, and the yield is about 88%.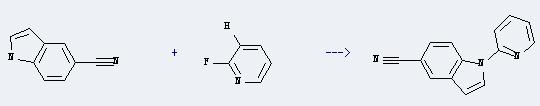
Safety information of 5-Cyanoindole:
You should be cautious while dealing with this chemical. It irritates eyes, respiratory system and skin, and it is also harmful by inhalation, in contact with skin and if swallowed. Therefore, you had better take the following instructions: Do not breathe dust; Wear suitable protective clothing; In case of contacting with eyes, rinse immediately with plenty of water and seek medical advice.
You can still convert the following datas into molecular structure:
(1)SMILES: N#Cc1cc2c(cc1)ncc2
(2)InChI: InChI=1/C9H6N2/c10-6-7-1-2-9-8(5-7)3-4-11-9/h1-5,11H
(3)InChIKey: YHYLDEVWYOFIJK-UHFFFAOYAP
The toxicity data of 5-Cyanoindole is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LD50 | intravenous | 100mg/kg (100mg/kg) | U.S. Army Armament Research & Development Command, Chemical Systems Laboratory, NIOSH Exchange Chemicals. Vol. NX#12275, |
Related Products
- 5-Cyanoindole
- 5-Cyanoindole-3-carboxaldehyde
- 15861-30-0
- 15861-36-6
- 158619-46-6
- 15862-01-8
- 15862-07-4
- 158622-13-0
- 158622-59-4
- 15862-30-3
- 15862-31-4
- 15862-33-6
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View