-
Name
5-Hexynoic acid
- EINECS -0
- CAS No. 53293-00-8
- Article Data34
- CAS DataBase
- Density 1.058 g/cm3
- Solubility Miscible with water.
- Melting Point 27°C (estimate)
- Formula C6H8O2
- Boiling Point 220.6 °C at 760 mmHg
- Molecular Weight 112.128
- Flash Point 99.6 °C
- Transport Information
- Appearance clear yellow liquid
- Safety 26-36
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms 5-Hexyn-1-oic acid;
- PSA 37.30000
- LogP 0.87450
Synthetic route
-
-
928-90-5
5-hexyl-1-ol

-
-
53293-00-8
hex-5-ynoic acid

| Conditions | Yield |
|---|---|
| With jones' reagent In acetone for 1h; Ambient temperature; | 82% |
| With chromium(VI) oxide; hydrogen cation In acetone | 80% |
| With jones reagent at 0℃; | 76% |

| Conditions | Yield |
|---|---|
| In hexane; sulfuric acid; acetone | 72% |
-
-
14918-21-9
5-hexynonitrile

-
-
53293-00-8
hex-5-ynoic acid

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water for 10h; Heating; | 70% |
| With potassium hydroxide | |
| With potassium hydroxide In methanol; water Heating; | |
| With potassium hydroxide In ethanol; water for 4h; Heating; Yield given; |
-
-
279214-91-4
5,6-dibromohexanoic acid

-
-
53293-00-8
hex-5-ynoic acid

| Conditions | Yield |
|---|---|
| With sodium amide In diethyl ether at -33 - 25℃; Dehydrobromination; | 70% |
| With sodium amide In diethyl ether; ammonia at -33℃; for 1.5h; | 65% |
-
-
73448-14-3
hex-5-ynoic acid tert-butyl ester

-
-
53293-00-8
hex-5-ynoic acid

| Conditions | Yield |
|---|---|
| With sodium hydride In N,N-dimethyl-formamide at 70℃; for 20h; | 65% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide; PEG-2000 at 80℃; for 8h; Dehydrobromination; | A 49% B 19.6% |

| Conditions | Yield |
|---|---|
| With chromium(VI) oxide; sulfuric acid; acetone |
-
-
108545-38-6
5-hexynoic acid ethyl ester

-
-
53293-00-8
hex-5-ynoic acid

| Conditions | Yield |
|---|---|
| With potassium hydroxide |
-
-
67-56-1
methanol

-
-
60556-87-8
2-carbonyl-3-butenenitrile

-
-
14657-22-8
allenyltrimethylsilane

-
A
-
53293-00-8
hex-5-ynoic acid

-
B
-
101224-43-5
6-(trimethylsilyl)hex-5-ynoic acid:

| Conditions | Yield |
|---|---|
| Yield given. Multistep reaction. Yields of byproduct given; |

-
-
77758-51-1
methyl hex-5-ynoate

-
-
53293-00-8
hex-5-ynoic acid

| Conditions | Yield |
|---|---|
| With potassium hydroxide for 0.5h; Heating; | |
| With water; potassium hydroxide In methanol at 20℃; for 0.5h; Inert atmosphere; |

| Conditions | Yield |
|---|---|
| With ammonia |
-
-
279214-91-4
5,6-dibromohexanoic acid

-
A
-
53293-00-8
hex-5-ynoic acid

| Conditions | Yield |
|---|---|
| With potassium hydroxide |
-
-
279214-91-4
5,6-dibromohexanoic acid

-
A
-
53293-00-8
hex-5-ynoic acid


| Conditions | Yield |
|---|---|
| With sodium hydroxide; PEG-2000 In water at 82℃; for 8h; |

-
-
2623-87-2
bromobutyric acid

-
-
1216963-74-4
lithium acetylide-ethylenediamine complex

-
-
53293-00-8
hex-5-ynoic acid

| Conditions | Yield |
|---|---|
| In dimethyl sulfoxide |
-
-
1577-22-6
5-hexenoic acid

-
-
53293-00-8
hex-5-ynoic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: Br2 / CH2Cl2 / 1.5 h / -40 °C 2: 65 percent / NaNH2 / diethyl ether; liquid ammonia / 1.5 h / -33 °C View Scheme | |
| Multi-step reaction with 2 steps 1: Br2 / CH2Cl2 / 1.5 h / -40 °C 2: NaOH; PEG-2000 / H2O / 8 h / 82 °C View Scheme | |
| Multi-step reaction with 2 steps 1: petroleum ether; bromine 2: ethanolic KOH-solution View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 93.5 percent / Br2 / CH2Cl2 / 1.5 h / -40 °C 2: 70 percent / NaNH2 / diethyl ether / -33 - 25 °C View Scheme | |
| Multi-step reaction with 2 steps 1: 93.5 percent / Br2 / CH2Cl2 / 1.5 h / -40 °C 2: 49 percent / 40 percent aq. NaOH; PEG-2000 / 8 h / 80 °C View Scheme |
-
-
764-60-3
hex-2-yn-1-ol

-
-
53293-00-8
hex-5-ynoic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: NaNH2 / various solvent(s) 2: Jones reagent View Scheme |
-
-
2396-80-7
methyl 5-hexenoate

-
-
53293-00-8
hex-5-ynoic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 2: aq. KOH / 0.5 h / Heating View Scheme |
-
-
108545-33-1
6-(2-Nitro-phenylselanyl)-hexanoic acid methyl ester

-
-
53293-00-8
hex-5-ynoic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 68 percent / aq. H2O2 / tetrahydrofuran / 24 h 3: aq. KOH / 0.5 h / Heating View Scheme |
-
-
18420-41-2, 130233-13-5, 130233-14-6
2-(chloromethyl)tetrahydropyran

-
-
53293-00-8
hex-5-ynoic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: NaNH2, liq. NH3 / tetrahydrofuran / -78 °C 2: Jones' reagent / acetone / 0 °C View Scheme |
-
-
100-72-1
Tetrahydropyran-2-methanol

-
-
53293-00-8
hex-5-ynoic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: SOCl2, pyridine / 10 h / Ambient temperature 2: NaNH2, liq. NH3 / tetrahydrofuran / -78 °C 3: Jones' reagent / acetone / 0 °C View Scheme |
-
-
5390-04-5
pent-1-yn-5-ol

-
-
53293-00-8
hex-5-ynoic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: pyridine / 0.17 h / -10 °C 2: NaCN / dimethylsulfoxide / 0.5 h / 70 °C 3: KOH / ethanol; H2O / 4 h / Heating View Scheme |
-
-
77758-50-0
4-pentynyl-1-tosylate

-
-
53293-00-8
hex-5-ynoic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: NaCN / dimethylsulfoxide / 0.5 h / 70 °C 2: KOH / ethanol; H2O / 4 h / Heating View Scheme |
-
-
14267-92-6
1-chloro-4-pentyne

-
-
53293-00-8
hex-5-ynoic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 53 percent 2: 70 percent / NaOH / H2O / 10 h / Heating View Scheme | |
| Multi-step reaction with 3 steps 1: acetone; sodium iodide 2: aqueous acetone 3: aqueous KOH-solution View Scheme | |
| With hydrogenchloride; potassium hydroxide In water; dimethyl sulfoxide |
-
-
2468-55-5
5-iodopent-1-yne

-
-
53293-00-8
hex-5-ynoic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: aqueous acetone 2: aqueous KOH-solution View Scheme |
-
-
5244-34-8
2,2'-ethane-1,2-diylbissulfanyl-bis-ethanol

-
-
53293-00-8
hex-5-ynoic acid

| Conditions | Yield |
|---|---|
| With n-butyllithium In hexane |

-
-
14918-21-9
5-hexynonitrile

-
-
53293-00-8
hex-5-ynoic acid

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In dichloromethane for 36h; Reflux; | 100% |
| With toluene-4-sulfonic acid In dichloromethane for 24h; Esterification; Heating; | 97% |
| With toluene-4-sulfonic acid In dichloromethane for 24h; Heating; | 96% |
-
-
53293-00-8
hex-5-ynoic acid

-
-
55183-45-4
hex-5-in-carboxyl chloride

| Conditions | Yield |
|---|---|
| With oxalyl dichloride Inert atmosphere; | 100% |
| With oxalyl dichloride; N,N-dimethyl-formamide In tetrahydrofuran at 20℃; for 2h; Inert atmosphere; | 83% |
| With oxalyl dichloride; N,N-dimethyl-formamide In benzene at 25 - 35℃; for 2h; | 82% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide | 100% |
| With potassium carbonate In N,N-dimethyl-formamide at 5℃; for 5h; Cooling with ice; | 70% |
| With potassium carbonate In N,N-dimethyl-formamide |
-
-
75-77-4
chloro-trimethyl-silane

-
-
53293-00-8
hex-5-ynoic acid

-
-
101224-43-5
6-(trimethylsilyl)hex-5-ynoic acid:

| Conditions | Yield |
|---|---|
| Stage #1: hex-5-ynoic acid With n-butyllithium In tetrahydrofuran at -78℃; Inert atmosphere; Stage #2: chloro-trimethyl-silane In tetrahydrofuran at -78 - 0℃; Inert atmosphere; Stage #3: With hydrogenchloride In tetrahydrofuran; water at 0 - 20℃; Inert atmosphere; | 100% |
| Stage #1: hex-5-ynoic acid With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 0.75h; Inert atmosphere; Stage #2: chloro-trimethyl-silane In tetrahydrofuran; hexane at -78 - 20℃; Inert atmosphere; Stage #3: With water; acetic acid In tetrahydrofuran; hexane for 0.333333h; | 76% |
| Stage #1: hex-5-ynoic acid With n-butyllithium In tetrahydrofuran at -78℃; for 0.25h; Inert atmosphere; Stage #2: chloro-trimethyl-silane In tetrahydrofuran at -78 - 20℃; for 4h; Inert atmosphere; | 65% |
| Stage #1: hex-5-ynoic acid With n-butyllithium In tetrahydrofuran at -78℃; for 0.0833333h; Inert atmosphere; Stage #2: chloro-trimethyl-silane In tetrahydrofuran at -78 - 20℃; for 1h; Inert atmosphere; Stage #3: With hydrogenchloride In tetrahydrofuran; water | 63% |
| Stage #1: hex-5-ynoic acid With n-butyllithium In tetrahydrofuran; Petroleum ether at -78℃; Stage #2: chloro-trimethyl-silane In tetrahydrofuran; Petroleum ether at -78 - 0℃; |
-
-
53293-00-8
hex-5-ynoic acid

-
-
1219440-76-2
sodium 5-hexynoate

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate In water at 20℃; for 0.166667h; | 100% |
| With sodium hydroxide In water |

| Conditions | Yield |
|---|---|
| With dmap; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In dichloromethane at 20℃; | 100% |
| With 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; dmap In dichloromethane at 20℃; | 100% |
| With N-(3-dimethylaminopropyl)-N-ethylcarbodiimide In dichloromethane at 23℃; Inert atmosphere; | 62% |
-
-
53293-00-8
hex-5-ynoic acid

-
-
6638-79-5
N,O-dimethylhydroxylamine*hydrochloride

-
-
217087-88-2
N-methoxy-N-methylhex-5-ynamide

| Conditions | Yield |
|---|---|
| With dmap; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In dichloromethane at 25℃; Inert atmosphere; | 100% |
| With dmap; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; triethylamine In dichloromethane at 0 - 25℃; for 6h; | 98% |
| With dmap; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In N,N-dimethyl-formamide at 20℃; for 43h; | 94% |
| With 4-methyl-morpholine; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In dichloromethane at 0 - 20℃; for 16h; |
-
-
636-98-6
p-nitrobenzene iodide

-
-
53293-00-8
hex-5-ynoic acid

-
-
1084898-19-0
6-(4-nitrophenyl)hex-5-ynoic acid

| Conditions | Yield |
|---|---|
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; triethylamine In tetrahydrofuran at 50℃; for 2h; Inert atmosphere; | 100% |
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; triethylamine In tetrahydrofuran at 50℃; for 2h; Inert atmosphere; | 100% |
| With dichloro bis(acetonitrile) palladium(II); copper(l) iodide; diisopropylamine; triphenylphosphine at 50℃; for 14h; Inert atmosphere; Schlenk technique; | 74% |
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; N-ethyl-N,N-diisopropylamine In acetonitrile at 50℃; for 2h; Sonogashira Cross-Coupling; Inert atmosphere; | 22% |
-
-
53293-00-8
hex-5-ynoic acid

-
-
769-39-1
2,3,5,6-tetraflourophenol

-
-
1462237-48-4
2,3,5,6-tetrafluorophenyl hex-5-ynoate

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane at 20℃; for 24h; Inert atmosphere; Schlenk technique; | 100% |
| With pyridinium p-toluenesulfonate; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In dichloromethane at 20℃; for 16h; Inert atmosphere; |
-
-
62147-49-3
1,3-dihydroxyacetone dimer

-
-
53293-00-8
hex-5-ynoic acid

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane at 0 - 20℃; | 100% |

| Conditions | Yield |
|---|---|
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; triethylamine In tetrahydrofuran at 20℃; for 4.5h; Temperature; Inert atmosphere; | 100% |

| Conditions | Yield |
|---|---|
| With benzotriazol-1-yloxyl-tris-(pyrrolidino)-phosphonium hexafluorophosphate; triethylamine In N,N-dimethyl-formamide at 20℃; for 3h; | 100% |
-
-
53293-00-8
hex-5-ynoic acid

-
-
5636-66-8
6-methylidenetetrahydro-2-pyrone

| Conditions | Yield |
|---|---|
| With [(1,1′-biphenyl-2-yl)di-tert-butylphosphine]gold(I) trifluoromethanesulfonate; silver trifluoromethanesulfonate In chloroform-d1 at 20℃; for 0.0833333h; Reagent/catalyst; | 99% |
| With C42H64P4Pd2S4 at 90℃; for 10h; Inert atmosphere; | 98% |
| With potassium carbonate; gold(I) chloride In acetonitrile at 20℃; for 2h; | 97% |
-
-
863713-59-1
N-(2-hydroxy-1-methylethyl)-3-iodobenzamide

-
-
53293-00-8
hex-5-ynoic acid

-
-
863713-63-7
6-[3-(2-hydroxy-1-methylethylcarbamoyl)phenyl]hex-5-ynoic acid

| Conditions | Yield |
|---|---|
| With pyrrolidine; copper(l) iodide; tetrakis(triphenylphosphine) palladium(0) at 20 - 60℃; for 3.3h; Sonogashira Coupling; | 99% |
| Stage #1: N-(2-hydroxy-1-methylethyl)-3-iodobenzamide; hex-5-ynoic acid With copper(l) iodide; tetrakis(triphenylphosphine) palladium(0) at 60℃; for 3h; Sonogashira coupling; Stage #2: N-(2-hydroxy-1-methylethyl)-3-iodobenzamide; hex-5-ynoic acid In water; acetonitrile for 0.333333h; Further stages.; | 97% |

| Conditions | Yield |
|---|---|
| With dmap; 2,4,6-trichlorobenzoyl chloride; triethylamine In toluene at 20℃; Yamaguchi esterification; Inert atmosphere; | 99% |

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane at 0 - 23℃; for 20h; | 99% |
| With dmap; dicyclohexyl-carbodiimide | 59% |

-
-
53293-00-8
hex-5-ynoic acid

-
-
1391054-29-7
2-[2'-(5-hexynoyloxy)ethylamino]-4,6-di(4'-methoxybenzylamino)-1,3,5-triazine

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In tetrahydrofuran at 20℃; for 24h; Inert atmosphere; | 99% |

| Conditions | Yield |
|---|---|
| With chloro-trimethyl-silane In methanol at 20℃; for 16h; | 99% |
-
-
53293-00-8
hex-5-ynoic acid

-
-
28393-04-6
dodeca-5,7-diynedioic acid

| Conditions | Yield |
|---|---|
| With oxygen; ammonium chloride; copper(l) chloride In water at 20℃; | 98% |
| Stage #1: hex-5-ynoic acid With acetyl chloride In methanol at 20 - 80℃; for 12h; Inert atmosphere; Schlenk technique; Stage #2: With copper(II) acetate monohydrate In acetonitrile at 80℃; for 4h; Reflux; Further stages; | 69% |
| With ammonium chloride; water; copper(l) chloride Reagens 4: Luft; |
-
-
53293-00-8
hex-5-ynoic acid

-
-
3740-59-8
3,4-dihydro-6-methyl-2H-pyran-2-one

| Conditions | Yield |
|---|---|
| With triphenylphosphine gold (I) chloride; silver trifluoromethanesulfonate In dichloromethane at 20℃; for 16h; Inert atmosphere; | 98% |
| With zinc(II) carbonate |
-
-
53293-00-8
hex-5-ynoic acid

-
-
910028-80-7
6-bromo-5-hexynoic acid

| Conditions | Yield |
|---|---|
| With N-Bromosuccinimide; silver nitrate In acetone for 2h; | 98% |
| With N-Bromosuccinimide; silver nitrate In acetone at 20℃; for 2h; | 96% |
| With N-Bromosuccinimide; silver nitrate In acetone at 20℃; for 2h; | 72% |
-
-
53293-00-8
hex-5-ynoic acid

-
-
771-61-9
2,3,4,5,6-pentafluorophenol

-
-
1219141-93-1
pentafluorophenyl 5-hexynoate

| Conditions | Yield |
|---|---|
| With dicyclohexyl-carbodiimide In dichloromethane at 0℃; | 98% |
| With dmap; dicyclohexyl-carbodiimide In dichloromethane at 0 - 20℃; for 1h; | 95% |
| With dicyclohexyl-carbodiimide In tetrahydrofuran at 20℃; |

| Conditions | Yield |
|---|---|
| With dmap; 1,2-dichloro-ethane; N-ethyl-N,N-diisopropylamine In dichloromethane Inert atmosphere; | 98% |
-
-
53293-00-8
hex-5-ynoic acid

-
-
3128-06-1
5-ketohexanoic acid

| Conditions | Yield |
|---|---|
| In water at 90℃; for 45h; | 98% |
| With [RhCl2(p-cymene)]2; water at 20℃; for 12h; | 86% |
| With platinum catalyst In water at 37℃; for 30h; | 64% |
-
-
53293-00-8
hex-5-ynoic acid

-
-
157182-49-5
methanandamide

-
-
1313514-82-7
(+)-{(2R)-2-[(5Z,8Z,11Z,14Z)-icosa-5,8,11,14-tetraenoylamino]propyl} 5-hexynoate

| Conditions | Yield |
|---|---|
| Stage #1: hex-5-ynoic acid With dmap; dicyclohexyl-carbodiimide In dichloromethane at 0℃; for 0.5h; Inert atmosphere; Stage #2: methanandamide In dichloromethane at 20℃; for 10h; Inert atmosphere; | 98% |
-
-
53293-00-8
hex-5-ynoic acid

-
-
22323-82-6
(S)-(+)-(2,2-dimethyl-[1,3]dioxolan-4-yl)methanol

-
-
1341126-40-6
(2'R)-2,3-O-isopropylidene-2,3-dihydroxy-1-prop-1-yl pent-4-ynoate

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane at 25℃; for 12h; Inert atmosphere; | 98% |
-
-
53293-00-8
hex-5-ynoic acid


| Conditions | Yield |
|---|---|
| Stage #1: hex-5-ynoic acid With N-ethyl-N,N-diisopropylamine; HATU In N,N-dimethyl-formamide at 0 - 20℃; for 0.5h; Stage #2: (2-(4-(tert-butyl)-2-ethoxyphenyl)-4,5-bis(4-chlorophenyl)-4,5-dihydro-1H-imidazol-1-yl)(piperazin-1-yl)methanone In N,N-dimethyl-formamide at 20℃; for 3h; | 98% |
-
-
50-00-0
formaldehyd

-
-
53293-00-8
hex-5-ynoic acid

-
-
1223635-87-7
1-((S)-3-azido-2-isocyanopropyl)benzene

| Conditions | Yield |
|---|---|
| In methanol at 20℃; Passerini Condensation; | 98% |

-
-
53293-00-8
hex-5-ynoic acid

| Conditions | Yield |
|---|---|
| With bromine; magnesium bromide ethyl etherate; sodium bromide In dichloromethane at 20℃; | 98% |
-
-
53293-00-8
hex-5-ynoic acid

| Conditions | Yield |
|---|---|
| With dmap; benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In dichloromethane at 20℃; | 98% |
5-Hexynoic acid Specification
The 5-Hexynoic acid, with the CAS registry number of 53293-00-8, is also known as 5-Hexyn-1-oic acid. It belongs to the product categories of Acetylenes; Acetylenic Carboxylic Acids & Their Derivatives; C6; Carbonyl Compounds; Carboxylic Acids. Its molecular formula is C6H8O2 and molecular weight is 112.13. What's more, its IUPAC name is Hex-5-ynoic acid. In addition, it must be stored in airtight containers and placed in a dry, cool place. Besides, you should ensure the work place is well-ventilated.
Physical properties about the 5-Hexynoic acid are: (1)ACD/LogP: 0.78; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): -0.13; (4)ACD/LogD (pH 7.4): -1.94; (5)ACD/BCF (pH 5.5): 1; (6)ACD/BCF (pH 7.4): 1; (7)ACD/KOC (pH 5.5): 7.7; (8)ACD/KOC (pH 7.4): 1; (9)#H bond acceptors: 2; (10)#H bond donors: 1; (11)#Freely Rotating Bonds: 3; (12)Polar Surface Area: 26.3 Å2; (13)Index of Refraction: 1.463; (14)Molar Refractivity: 29.23 cm3; (15)Molar Volume: 105.9 cm3; (16)Surface Tension: 43.1 dyne/cm; (17)Density: 1.058 g/cm3; (18)Flash Point: 99.6 °C; (19)Enthalpy of Vaporization: 50.36 kJ/mol; (20)Boiling Point: 220.6 °C at 760 mmHg; (21)Vapour Pressure: 0.042 mmHg at 25 °C.
Preparation: this chemical can be obtained by Hex-5-ynenitrile. The reaction needs reagent NaOH and solvent H2O. The reaction time is 10 h. The yield is about 70 %.

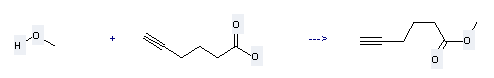
Uses: it is used to produce other chemicals. For example, it is used to produce Hex-5-ynoic acid methyl ester. This reaction needs reagent p-TSA. Meanwhile, it needs solvent CH2Cl2. The reaction time is 24 h. The yield is about 97 %.
When you are using this chemical, please be cautious about it as the following:
As a chemical, it is irritating to eyes, respiratory system and skin. If contact with eyes accidently, rinse immediately with plenty of water and seek medical advice. In addition, during using it, wear suitable protective clothing.
You can still convert the following datas into molecular structure:
(1) SMILES: O=C(O)CCCC#C
(2) InChI: InChI=1/C6H8O2/c1-2-3-4-5-6(7)8/h1H,3-5H2,(H,7,8)
(3) InChIKey: VPFMEXRVUOPYRG-UHFFFAOYAP
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View