-
Name
Boron trifluoride etherate
- EINECS 203-689-8
- CAS No. 109-63-7
- Article Data31
- CAS DataBase
- Density 1.12 g/cm3
- Solubility Reacts with water
- Melting Point -58 °C(lit.)
- Formula C4H10BF3O
- Boiling Point 126 dec
- Molecular Weight 141.929
- Flash Point 118°F
- Transport Information UN 2604 8/PG 1
- Appearance colourless to brown fuming liquid with anacrid odour
- Safety 16-23-26-36/37/39-45-8-28A-43
- Risk Codes 10-14-20/22-35-48/23-34-14/15
-
Molecular Structure
-
Hazard Symbols
 T,
T, C
C
- Synonyms (Diethylether)trifluoroboron;Boron fluoride diethyl ether complex;Boron fluoridediethyl etherate;Boron fluoride etherate;Boron fluoride monoetherate;Boronfluoride-diethyl ether compound;Boron fluoride-ethyl ether complex;Boronfluoride-ethyl etherate;Boron trifluoride etherate(1:1);Boron trifluoride ethyl etherate (1:1);Boron trifluoride monoetherate;Boron trifluoride-diethyl ether (1:1);Boron trifluoride-diethyl ether 1:1complex;Boron trifluoride-diethyl ether complex;Boron trifluoride-diethylether complex (1:1);Trifluoroborane diethyl etherate;Trifluoro[1,1'-oxybis[ethane]]boron;Trifluoro(diethyl ether)boron;Ethylether-boron trifluoride complex;Boron trifluoride-diethyl etherate;Borontrifluoride-ether complex;Boron trifluoride-ethyl ether;Borontrifluoride-ethyl ether (1:1);Boron trifluoride-ethyl ether complex;Borontrifluoride-ethyl etherate;Diethyl ether compound with boron trifluoride;Diethylether trifluoroborane complex;Diethyl ether-trifluoroborane (1:1);Trifluoroboron etherate;Trifluoroborane-1,1'-oxybis[ethane] (1:1);Trifluoroboron-diethyl ether (1:1);(Diethyl ether)trifluoroborane;
- PSA 17.07000
- LogP 1.92260
Synthetic route
-
-
444-40-6
2,4,6-tris(trilfuoromethyl)phenyllithium

-
-
10294-34-5
boron trichloride

-
A
-
99919-68-3
BF2Cl*Et2O

-
B
-
99919-69-4
BFCl2*Et2O

-
C
-
109-63-7
boron trifluoride diethyl etherate

-
D
-
681812-01-1
2,4,6-(CF3)3C6H2BCl2

-
E
-
681812-02-2
bis-(2,4,6-tris-trifluoromethyl-phenyl) fluoroborane

| Conditions | Yield |
|---|---|
| In diethyl ether under an inert atm.; to a soln. of Li-contg. compd. in hexanes and Et2O was added dropwise an excess of B-contg. compd. (1 M in heptane) at -78°C ; the react. mixt. was allowed to warm to room temp. for 6 h; the soln. was filtered and the solvents were removed under vac.; a mixt.of yellow oil and white solid; the mixt. was vac. distd. at 60°C /0.05 Torr; the residue was sublimed at 95°C under vac.; | A n/a B n/a C n/a D 5% E 24% |

-
-
109-63-7
boron trifluoride diethyl etherate

-
-
99179-72-3
3-acetoxy-4-methoxyphenol

-
-
7298-21-7
1-(2,4-dihydroxy-5-methoxyphenyl)ethan-1-one

| Conditions | Yield |
|---|---|
| at 70℃; for 2h; | 100% |
-
-
109-63-7
boron trifluoride diethyl etherate

-
-
675837-54-4
1-[bis(diisopropylamino)boryl]imidazole

-
-
675837-55-5
1-[bis(diisopropylamino)boryl]imidazole(N(3)-B)trifuoroborane

| Conditions | Yield |
|---|---|
| In tetrahydrofuran under Ar; BF3*OEt2 added to soln. of N-compd. in THF at -78°C, mixt. allowed to warm to room temp., stirred for 7 h; evapn. of solvent; | 100% |
-
-
109-63-7
boron trifluoride diethyl etherate

-
-
123-54-6
acetylacetone

-
-
15390-25-7
(acetylacetonato)borondifluoride

| Conditions | Yield |
|---|---|
| With potassium carbonate In dichloromethane at 20℃; for 0.5h; | 100% |
| In dichloromethane for 12h; Inert atmosphere; | 96% |
| With diphenyl acetylene In dichloromethane at 20℃; for 12h; | 83% |
-
-
946613-44-1
C2H5OCOC4N(CH3)2CHC4H2N(OCH3)C4H4N*HCl

-
-
109-63-7
boron trifluoride diethyl etherate

-
-
1318245-52-1
[(C2H5OCOC4N(CH3)2CHC4HN(OCH3)C4H3NH)BF2]

| Conditions | Yield |
|---|---|
| With (C2H5)3N In dichloromethane (C2H5)3N added to a soln. of ligand salt under N2, stirred for 10 min, Bcomplex added, stirred at room temp. for 2 h; poured into an aq. soln. of citric acid, extd. (ether), the org. layer washed with aq. citric acid, dried (Na2SO4), concd. (vac.), filtered (silica, CH2Cl2), concd. (vac.); | 100% |
-
-
109-63-7
boron trifluoride diethyl etherate

-
-
1430883-93-4
(1E,4Z,6E)-5-hydroxy-4-(3-methoxyphenyl)-1,7-bis(4-methoxyphenyl)hepta-1,4,6-trien-3-one

| Conditions | Yield |
|---|---|
| In dichloromethane Reflux; | 100% |
-
-
109-63-7
boron trifluoride diethyl etherate

-
-
1430883-95-6
(1E,4Z,6E)-5-hydroxy-1,4,7-tris(4-methoxyphenyl)hepta-1,4,6-trien-3-one

| Conditions | Yield |
|---|---|
| In dichloromethane Reflux; | 100% |
-
-
109-63-7
boron trifluoride diethyl etherate

-
-
1430883-98-9
methyl 4-((1E,3Z,6E)-3-hydroxy-1,7-bis(4-methoxyphenyl)-5-oxohepta-1,3,6-trien-4-yl)benzoate

| Conditions | Yield |
|---|---|
| In dichloromethane Reflux; | 100% |
-
-
109-63-7
boron trifluoride diethyl etherate

-
-
1430884-00-6
(1E,4Z,6E)-5-hydroxy-1,7-bis(4-methoxyphenyl)-4-phenylhepta-1,4,6-trien-3-one

| Conditions | Yield |
|---|---|
| In dichloromethane Reflux; | 100% |
-
-
109-63-7
boron trifluoride diethyl etherate

-
-
1430884-04-0
(1E,4Z,6E)-5-hydroxy-1,7-bis(4-methoxynaphthalen-1-yl)-4-phenylhepta-1,4,6-trien-3-one

| Conditions | Yield |
|---|---|
| In dichloromethane Reflux; | 100% |
-
-
109-63-7
boron trifluoride diethyl etherate

-
-
1430884-06-2
(1E,4Z,6E)-5-hydroxy-1,7-bis(6-methoxynaphthalen-2-yl)-4-phenylhepta-1,4,6-trien-3-one

| Conditions | Yield |
|---|---|
| In dichloromethane Reflux; | 100% |
-
-
109-63-7
boron trifluoride diethyl etherate

-
-
1430884-08-4
(1E,4Z,6E)-5-hydroxy-1,7-bis(9-methyl-9H-carbazol-3-yl)-4-phenylhepta-1,4,6-trien-3-one

| Conditions | Yield |
|---|---|
| In dichloromethane Reflux; | 100% |

| Conditions | Yield |
|---|---|
| In dichloromethane at -78 - 0℃; for 4h; Inert atmosphere; | 100% |

-
-
109-63-7
boron trifluoride diethyl etherate

| Conditions | Yield |
|---|---|
| Stage #1: 2,2'-[(1E,1'E)-(ethane-1,2-diylbis(azanylylidene)bis(methanylylidene))]bis(4-benzhydrylphenol) With N-ethyl-N,N-diisopropylamine In 1,2-dichloro-ethane for 0.166667h; Heating; Stage #2: boron trifluoride diethyl etherate In 1,2-dichloro-ethane for 0.5h; Inert atmosphere; Heating; | 100% |
-
-
109-63-7
boron trifluoride diethyl etherate

-
-
68-12-2, 33513-42-7
N,N-dimethyl-formamide

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 0℃; | 100% |

| Conditions | Yield |
|---|---|
| In toluene Reflux; | 100% |
-
-
717-27-1
2,5-diacetoxytoluene

-
-
109-63-7
boron trifluoride diethyl etherate

-
-
54698-17-8
1-(2,5-dihydroxy-4-methylphenyl)ethanone

| Conditions | Yield |
|---|---|
| at 130℃; for 4h; Inert atmosphere; | 100% |

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In dichloromethane at 20℃; | 100% |

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In dichloromethane at 20℃; for 2h; | 100% |
-
-
109-63-7
boron trifluoride diethyl etherate

| Conditions | Yield |
|---|---|
| Stage #1: 3,3’-dimethoxy-2,2’-diphenyl-6-(pentafluorophenyl)dipyrrin With triethylamine In toluene at 70℃; for 0.5h; Inert atmosphere; Stage #2: boron trifluoride diethyl etherate In toluene at 110℃; for 4h; Inert atmosphere; | 100% |

| Conditions | Yield |
|---|---|
| With triethylamine In toluene | 100% |

| Conditions | Yield |
|---|---|
| Stage #1: diethyl ether; boron trifluoride diethyl etherate With epichlorohydrin In diethyl ether for 1h; Inert atmosphere; Reflux; Stage #2: 1-(4-bromo-2-azidophenyl)-4-phenyl-1H-1,2,3-triazole In dichloromethane | 100% |

-
-
109-63-7
boron trifluoride diethyl etherate


| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; Inert atmosphere; | 100% |

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; for 1h; Inert atmosphere; | 100% |
-
-
109-63-7
boron trifluoride diethyl etherate

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; for 1h; Inert atmosphere; | 100% |
-
-
109-63-7
boron trifluoride diethyl etherate

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; for 1h; Inert atmosphere; | 100% |
-
-
75778-52-8
nitrosyl(5,10,15,20-tetraphenylporphyrinato)(pyridine)cobalt(III)

-
-
109-63-7
boron trifluoride diethyl etherate

-
B
-
100-52-7
benzaldehyde

| Conditions | Yield |
|---|---|
| With benzyl alcohol In 1,2-dichloro-ethane byproducts: BF3-pyridine complex, H2O; under Ar addn. of BF3*Et2O to mixt. of alcohol and complex in (CH2)2Cl2, further addn. of BF3*Et2O, heating in 60°C oil bath for 45 min,addn. 1 drop of pyridine in hexane, removing BF3-pyridine complex pptd.; solvent removing in vac., addn. of petroleum ether to resulting oil, filtration, drying under Ar; | A n/a B 99% |

-
-
375391-20-1
cyclopentadienyl-dicarbonyl-triethoxysilylmethyl-iron

-
-
109-63-7
boron trifluoride diethyl etherate

-
-
375391-21-2
cyclopentadienyl-dicarbonyl-trifluorosilylmethyl-iron

| Conditions | Yield |
|---|---|
| In toluene under N2, excess of BF3(OEt2) was added, stirring for 30 min; volatiles were removed; | 99% |
-
-
109-63-7
boron trifluoride diethyl etherate

-
-
443147-01-1
((CH3)3C)2PCN(CH(CH3)2)2

| Conditions | Yield |
|---|---|
| In tetrahydrofuran treatment THF soln. carbene with 1 equiv. BF3*OEt2; | 99% |
-
-
625-82-1
2,4-dimethyl-1H-pyrrole

-
-
1003-29-8
2-pyrrole aldehyde

-
-
109-63-7
boron trifluoride diethyl etherate

-
-
154793-49-4
5,5-difluoro-1,3-dimethyl-5H-4λ4,5λ,4-dipyrrolo[1,2-c:2',1'-f][1,3,2]diazaborinine

| Conditions | Yield |
|---|---|
| Stage #1: 2,4-dimethyl-1H-pyrrole; 2-pyrrole aldehyde With trichlorophosphate In dichloromethane at -5 - 20℃; for 6h; Inert atmosphere; Stage #2: boron trifluoride diethyl etherate With N-ethyl-N,N-diisopropylamine In dichloromethane Inert atmosphere; | 99% |
| Stage #1: 2,4-dimethyl-1H-pyrrole; 2-pyrrole aldehyde With trichlorophosphate In dichloromethane at 0 - 20℃; for 2h; Stage #2: boron trifluoride diethyl etherate With triethylamine In dichloromethane at 20℃; for 1h; | 36.1% |
| With N-ethyl-N,N-diisopropylamine; trichlorophosphate In dichloromethane (N2) to soln. pyrrole 2-carboxyaldehyde in CH2Cl2 at -5°C, 2,4-dimethylpyrrole was added, stirred for 3 min, POCl3 was dropwise added, stirred at -5°C for 3 h and at room temp. 3 h, i-Pr2NEt and BF3*Et2O were added, stirred for 3 h; react. mixt. was washed with H2O, dried over Na2SO4, evapd., residue waschromd. on silica (hexane/EtOAc, 5:1); | 32.8% |

-
-
1271-55-2
acetylferrocene

-
-
109-63-7
boron trifluoride diethyl etherate

-
-
1162093-13-1
[Fe(C5H5)(C5H4C(OBF3)CH3)]

| Conditions | Yield |
|---|---|
| In diethyl ether under dry N2; mixing of soln. at equimolar ratio; solvent was evapd., crystals were grown by vapor diffusion of pentane into chloroform soln. at -30°C over 2 days; NMR, IR, XRD; | 99% |
BORON TRIFLUORIDE ETHERATE Chemical Properties
Product Name:Boron trifluoride etherate(109-63-7)
CAS No:109-63-7
MF: C4H10BF3O
MW: 141.93
mp : -58 °C(lit.)
bp : 126-129 °C(lit.)
density : 1.15 g/mL(lit.)
vapor density : 4.9 (vs air)
vapor pressure : 4.2 mm Hg ( 20 °C)
refractive index : n20/D 1.344(lit.)
Fp : 118 °F
storage temp. : 2-8°C
form : liquid
color : brown
Water Solubility : Reacts
Sensitive : Moisture Sensitive
Synonyms of Boron trifluoride etherate(109-63-7): 1'-oxybis[ethane]]-trifluoro[(beta-4)-boro;bf3-ethercomplex;Borane, trifluoro-, compd. with 1,1'-oxybis(ethane) (1:1);borane,trifluoro-,cmpdwith1,1'-oxybis(ethane)(1:1);Borane,trifluoro-,compd.with1,1'-oxybis[ethane](1:1);Boron fluoride diethyl etherate;Boron fluoride etherate;Boron fluoride monoetherate
Molecular Structure of Boron trifluoride etherate(109-63-7):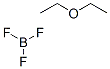
Boron trifluoride etherate(109-63-7) is a fuming liquid. Boron trifluoride etherate may be corrosive to skin, eyes and mucous membranes. Boron trifluoride etherate(109-63-7) may be toxic by inhalation. Upon exposure to water Boron trifluoride etherate may emit flammable and corrosive vapors. Boron trifluoride etherate is used as a catalyst in chemical reactions.
BORON TRIFLUORIDE ETHERATE Toxicity Data With Reference
LD50/LC50: RTECS: Not available. Other: Oral, rat: LD50 = 496 mg/kg
Carcinogenicity: Boron trifluoride etherate - Not listed as a carcinogen by ACGIH, IARC, NTP, or CA Prop 65.
Other: The toxicological properties have not been fully investigated.
BORON TRIFLUORIDE ETHERATE Safety Profile
Hazard Codes : T,C
Risk Statements : 10-14-20/22-35-48/23-34-14/15
Safety Statements : 16-23-26-36/37/39-45-8-28A-43
RIDADR : UN 2604 8/PG 1
WGK Germany : 3
F : 10
HazardClass : 8
PackingGroup : I
BORON TRIFLUORIDE ETHERATE Specification
Handling: Do not allow water to get into the container because of violent reaction. Do not breathe dust, vapor, mist, or gas. Do not get in eyes, on skin, or on clothing. Take precautionary measures against static discharges. Do not ingest or inhale. Use and store under nitrogen. Use only in a chemical fume hood. This product may be under pressure; cool before opening.
Storage: Keep away from heat and flame. Store in a cool, dry place. Store in a tightly closed container. Flammables-area. Store protected from moisture. Store under an inert atmosphere.
Related Products
- Boron carbide
- Boron trifluoride dimethyl etherate
- BORON TRIFLUORIDE ETHERATE
- Boron trifluoride phenol complex
- BORON TRIFLUORIDE-PHENOL COMPLEX (1:2)
- Boronic acid, (10-[1,1'-biphenyl]-4-yl-9-anthracenyl)-
- Boronic acid, [2-[(2-methylphenoxy)methyl]phenyl]- (9CI)
- Boronic acid, 2,5-benzofurandiylbis-
- Boronic acid, 2-dibenzofuranyl-
- Boronic acid, B-2-dibenzothienyl-
- 109637-83-4
- 1096-38-4
- 1096-39-5
- 109640-39-3
- 109-64-8
- 1096-48-6
- 109652-10-0
- 109-65-9
- 109-66-0
- 1096687-52-3
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View