-
Name
Cephalothin
- EINECS 200-394-6
- CAS No. 58-71-9
- Article Data3
- CAS DataBase
- Density
- Solubility water: 50 mg/mL
- Melting Point 240 °C
- Formula C16H15N2NaO6S2
- Boiling Point 757.2 °C at 760 mmHg
- Molecular Weight 418.427
- Flash Point 411.8 °C
- Transport Information
- Appearance Crystalline
- Safety 22-36/37
- Risk Codes 42/43
-
Molecular Structure
-
Hazard Symbols
 Xn,
Xn,  Xi
Xi
- Synonyms 5-Thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid,3-[(acetyloxy)methyl]-8-oxo-7-[(2-thienylacetyl)amino]-, monosodium salt,(6R-trans)-;Averon 1;Cefalothine sodium;Cemastin;Cephalothin sodium salt;Cephation;Ceporacin;Cepovenin;Coaxin;Keflin;Lilly 38253;Lospoven;Microtin;Seffin;Sodium(thienylacetamido)cephalosporanate;Sodium 3-acetoxymethyl-7b-(2-thienylacetamido)ceph-3-em-4-carboxylate;Sodium 7-(2-thienylacetamido)-3-acetoxymethyl-3-cephem-4-carboxylate;Sodium7-[2-(2-thienyl)acetamido]cephalosporanate;Sodium cefalotin;Sodiumcephalothin;Sodium cephalotin;Synclotin;Toricelocin;
- PSA 169.38000
- LogP -0.41360
Synthetic route

| Conditions | Yield |
|---|---|
| With sodium 2-ethylhexanoic acid In acetone | 26% |
-
-
39098-97-0
2-thienylacetic acid chloride

-
-
957-68-6
7-Aminocephalosporanic acid

-
-
58-71-9
sodium cephalothin

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate; sodium 2-ethylhexanoic acid In water; acetone at 0 - 5℃; for 0.5h; |

| Conditions | Yield |
|---|---|
| In ethyl acetate; N,N-dimethyl-formamide | 96% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water at 65℃; for 6h; pH=7; | 89% |

-
-
58-71-9
sodium cephalothin

| Conditions | Yield |
|---|---|
| With hydrogenchloride; sodium hydrogencarbonate In ice-water; water | 87.5% |

| Conditions | Yield |
|---|---|
| In water; butan-1-ol at 20℃; for 48h; | 83% |

| Conditions | Yield |
|---|---|
| Stage #1: sodium cephalothin With sodium hydroxide In water at -20 - -10℃; for 0.0375h; Stage #2: With hydrogenchloride In water pH=1.5; cooling; Further stages.; | 80% |
-
-
58-71-9
sodium cephalothin

-
-
1408249-95-5, 1408250-00-9
Sn(2+)*Cl(1-)*C16H15N2O6S2(1-)

| Conditions | Yield |
|---|---|
| With water at 20℃; for 6h; | 72% |

| Conditions | Yield |
|---|---|
| With tetra(n-butyl)ammonium hydrogensulfate; sodium hydrogencarbonate In dichloromethane; water at 20 - 25℃; for 0.5h; Title compound not separated from byproducts; | A n/a B 62% |

-
-
58-71-9
sodium cephalothin

-
-
106-95-6
allyl bromide

-
-
104949-45-3
allyl 7β-(2-(thien-2-yl)acetamido)-3-(acetoxymethyl)-3-cephem-4-carboxylate

| Conditions | Yield |
|---|---|
| In water; N,N-dimethyl-formamide | 51% |
| In water; N,N-dimethyl-formamide for 48h; Ambient temperature; | 48% |
| In water; N,N-dimethyl-formamide at 20℃; for 48h; | 41% |
| In water; N,N-dimethyl-formamide at 20℃; for 48h; | 41% |
| In water; N,N-dimethyl-formamide at 20℃; for 48h; | 41% |
-
-
58-71-9
sodium cephalothin

-
-
61197-32-8
N,N-dimethyl-2-(5-thioxo-4,5-dihydro-tetrazol-1-yl)-acetamide

| Conditions | Yield |
|---|---|
| 44.1% |
-
-
15139-21-6
5-thio-2-nitrobenzoic acid

-
-
58-71-9
sodium cephalothin

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water at 65℃; for 6h; pH=7; | 34% |
| In water at 65℃; for 6h; pH=7.0; |
-
-
98-91-9
thiobenzoic acid

-
-
58-71-9
sodium cephalothin

-
-
862-61-3
(6R)-3-benzoylsulfanylmethyl-8-oxo-7t-(2-thiophen-2-yl-acetylamino)-(6rH)-5-thia-1-aza-bicyclo[4.2.0]oct-2-ene-2-carboxylic acid

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate |
-
-
141-90-2
2-thiouracil

-
-
58-71-9
sodium cephalothin

-
-
16082-24-9
(3Ξ,6R)-8,7'-dioxo-7t-(2-thiophen-2-yl-acetylamino)-7'H-spiro[5-thia-1-aza-bicyclo[4.2.0]octane-3,3'-thiazolo[3,2-a]pyrimidine]-2ξ-carboxylic acid

| Conditions | Yield |
|---|---|
| With sodium hydroxide |
-
-
6940-49-4
3-Bromophthalide

-
-
58-71-9
sodium cephalothin

-
-
57427-65-3
(6R)-3-acetoxymethyl-8-oxo-7t-(2-thiophen-2-yl-acetylamino)-(6rH)-5-thia-1-aza-bicyclo[4.2.0]oct-2-ene-2-carboxylic acid (S)-3-oxo-1,3-dihydro-isobenzofuran-1-yl ester
-
-
6940-49-4
3-Bromophthalide

-
-
58-71-9
sodium cephalothin

-
-
57427-64-2
(6R)-3-acetoxymethyl-8-oxo-7t-(2-thiophen-2-yl-acetylamino)-(6rH)-5-thia-1-aza-bicyclo[4.2.0]oct-2-ene-2-carboxylic acid (R)-3-oxo-1,3-dihydro-isobenzofuran-1-yl ester
-
-
68206-45-1
3-nitro-2-pyridinesulfenyl chloride

-
-
58-71-9
sodium cephalothin

-
-
68206-40-6
(6R)-3-acetoxymethyl-8-oxo-7t-(2-thiophen-2-yl-acetylamino)-(6rH)-5-thia-1-aza-bicyclo[4.2.0]oct-2-ene-2-carbothioic acid S-(3-nitro-pyridin-2-yl) ester

| Conditions | Yield |
|---|---|
| With triphenylphosphine In dichloromethane |
-
-
625-56-9
Chloromethyl acetate

-
-
58-71-9
sodium cephalothin

-
-
35390-65-9
(6R)-3-acetoxymethyl-5ξ,8-dioxo-7t-(2-thiophen-2-yl-acetylamino)-(6rH)-5λ4-thia-1-aza-bicyclo[4.2.0]oct-2-ene-2-carboxylic acid acetoxymethyl ester

| Conditions | Yield |
|---|---|
| (i), (ii) MCPBA; Multistep reaction; |
-
-
58-71-9
sodium cephalothin

-
-
128-04-1
sodium dimethyldithiocarbamate

-
-
1055-69-2
(6R)-3-[(dimethyl-thiocarbamoylsulfanyl)-methyl]-8-oxo-7t-(2-thiophen-2-yl-acetylamino)-(6rH)-5-thia-1-aza-bicyclo[4.2.0]oct-2-ene-2-carboxylic acid

| Conditions | Yield |
|---|---|
| In water |
-
-
871-58-9
potassium butylxanthate

-
-
58-71-9
sodium cephalothin

-
-
10002-63-8
(6R)-3-butoxythiocarbonylsulfanylmethyl-8-oxo-7t-(2-thiophen-2-yl-acetylamino)-(6rH)-5-thia-1-aza-bicyclo[4.2.0]oct-2-ene-2-carboxylic acid
-
-
148-18-5
sodium N,N-diethyldithiocarbamate

-
-
58-71-9
sodium cephalothin

-
-
88635-59-0
(6R)-3-[(diethyl-thiocarbamoylsulfanyl)-methyl]-8-oxo-7t-(2-thiophen-2-yl-acetylamino)-(6rH)-5-thia-1-aza-bicyclo[4.2.0]oct-2-ene-2-carboxylic acid
-
-
91840-27-6
Sodium N-methyl-(D-gluco-2,3,4,5,6-pentahydroxyhex-1-yl)dithiocarbamate

-
-
58-71-9
sodium cephalothin

-
-
3352-45-2
(6R)-3-[(D-glucitol-1-yl-methyl-thiocarbamoylsulfanyl)-methyl]-8-oxo-7t-(2-thiophen-2-yl-acetylamino)-(6rH)-5-thia-1-aza-bicyclo[4.2.0]oct-2-ene-2-carboxylic acid
-
-
2801-04-9
sodium bis<2-hydroxyethyl>dithiocarbamate

-
-
58-71-9
sodium cephalothin

-
-
32428-08-3
(6R)-3-{[bis-(2-hydroxy-ethyl)-thiocarbamoylsulfanyl]-methyl}-8-oxo-7t-(2-thiophen-2-yl-acetylamino)-(6rH)-5-thia-1-aza-bicyclo[4.2.0]oct-2-ene-2-carboxylic acid
-
-
58-71-9
sodium cephalothin

-
-
39234-21-4
sodium N-methyl-N-phenyldithiocarbamate
-
-
877-78-1, 7564-73-0
sodium piperazine-N,N'-bisdithiocarboxylate

-
-
58-71-9
sodium cephalothin
-
-
2720-67-4
potassium propylxanthate

-
-
58-71-9
sodium cephalothin

-
-
10002-60-5
(6R)-8-oxo-3-propoxythiocarbonylsulfanylmethyl-7t-(2-thiophen-2-yl-acetylamino)-(6rH)-5-thia-1-aza-bicyclo[4.2.0]oct-2-ene-2-carboxylic acid
-
-
2720-76-5
potassium-O-hexan-1-yl-dithiocarbonate

-
-
58-71-9
sodium cephalothin

-
-
10002-64-9
(6R)-3-hexyloxythiocarbonylsulfanylmethyl-8-oxo-7t-(2-thiophen-2-yl-acetylamino)-(6rH)-5-thia-1-aza-bicyclo[4.2.0]oct-2-ene-2-carboxylic acid
-
-
2720-77-6
potassium cyclohexyl xanthogenate

-
-
58-71-9
sodium cephalothin

-
-
10111-88-3
(6R)-3-cyclohexyloxythiocarbonylsulfanylmethyl-8-oxo-7t-(2-thiophen-2-yl-acetylamino)-(6rH)-5-thia-1-aza-bicyclo[4.2.0]oct-2-ene-2-carboxylic acid
-
-
32019-63-9
2-methyl-piperidine-1-carbodithioic acid; sodium salt

-
-
58-71-9
sodium cephalothin
Cefalotin sodium Chemical Properties
Name : Cefalotin sodium
Synonyms Cephalothin sodium; Sodium cephalothin; Synclotin; 7-(Thiophene-2-acetamido)cephalosporanic acid sodium salt; Monosodium (6R,7R)-3-acetoxymethyl-8-oxo-7-[2-(thiophen-2-yl)acetylamido]-5-thia-1-azabicyclo[4.2.0.]oct-2-ene-2-carboxylate
Molecular:Structure:
Molecular Formula:C16H15N2NaO6S2
Molecular Weight:418.41
EINECS:200-394-6 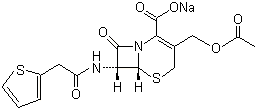
Merck:13,1994
Molecular Formula:240°C
solubility:H2O: 50 mg/mL, clear, faintly yellow
Water Solubility:158 mg/L
storage temp.:2-8°C
Appearance:Crystalline
Cefalotin sodium Toxicity Data With Reference
| 1. | ivn-wmn TDLo:20 mg/kg:BLD | AIMEAS Annals of Internal Medicine. 77 (1972),401. | ||
| 2. | ims-chd TDLo:89 mg/kg/19D-I:BLD AACHAX -,272,63 | |||
| 3. | ims-man TDLo:955 mg/kg/22D-I:BLD AACHAX -,272,63 | |||
| 4. | ipr-rat LD50:7000 mg/kg | JJANAX Japanese Journal of Antibiotics. 28 (1975),188. | ||
| 5. | scu-rat LD50:7500 mg/kg | JIDIAQ Journal of Infectious Diseases. 137 (1978),S51. | ||
| 6. | ipr-mus LD50:5670 mg/kg | JJANAX Japanese Journal of Antibiotics. 27 (1974),746. | ||
| 7. | scu-mus LD50:7500 mg/kg | JACHDX Journal of Antimicrobial Chemotherapy. 6 (Suppl A),(1980),79. | ||
| 8. | ivn-mus LD50:4800 mg/kg | JJANAX Japanese Journal of Antibiotics. 27 (1974),746. |
Cefalotin sodium Consensus Reports
Cefalotin sodium Safety Profile
Human systemic effects by intravenous and intramuscular routes: blood agranulocytosis and thrombocytopenia. When heated to decomposition it emits very toxic fumes of NOx, SOx, and Na2O.
Risk Statements:42/43: May cause sensitization by inhalation and skin contact
Safety Statements:22: Do not breathe dust;36/37: Wear suitable protective clothing and gloves
Hazard Codes:Xn,Xi
WGK Germany:2
Related Products
- Cefalotin sodium
- 58719-23-6
- 58-72-0
- 5872-08-2
- 58722-36-4
- 58725-33-0
- 58725-39-6
- 58725-46-5
- 587-26-8
- 58729-31-0
- 5873-00-7
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View