-
Name
Chloroacetamide
- EINECS 201-174-2
- CAS No. 79-07-2
- Article Data108
- CAS DataBase
- Density 1.268 g/cm3
- Solubility Soluble in water(90g/L).
- Melting Point 116-118 °C(lit.)
- Formula C2H4ClNO
- Boiling Point 256 °C at 760 mmHg
- Molecular Weight 93.5129
- Flash Point 108.6 °C
- Transport Information UN 2811 6.1/PG 3
- Appearance White crystal
- Safety 22-36/37-45
- Risk Codes 25-43-62
-
Molecular Structure
-
Hazard Symbols
 T
T
- Synonyms Acetamide, a-chloro- (4CI);2-Chloroacetamide;Chloracetamide;KM 101;KM 101 (fungicide);Mergal AF;Microcide;NSC 54286;NSC 8408;a-Chloroacetamide;
- PSA 43.09000
- LogP 0.41080
Synthetic route
-
-
112-41-4
1-dodecene

-
-
35070-77-0
N-chloro-alpha-chloroacetamide

-
A
-
35077-34-0
2-Chloro-N-(2-chloro-dodecyl)-acetamide

-
B
-
79-07-2
Chloroacetamide

| Conditions | Yield |
|---|---|
| In dichloromethane at 15 - 20℃; Irradiation; | A 95% B 3% |

-
-
120158-03-4
tert-butyl (2-chloroacetyl)carbamate

-
-
79-07-2
Chloroacetamide

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid In dichloromethane for 1h; Ambient temperature; | 94% |
-
-
35070-77-0
N-chloro-alpha-chloroacetamide

-
-
79-07-2
Chloroacetamide

| Conditions | Yield |
|---|---|
| With n-octyne In dichloromethane at 15 - 20℃; Irradiation; | 93% |

| Conditions | Yield |
|---|---|
| With Amberlyst A-26 (OH- form); dihydrogen peroxide In methanol at 20℃; for 2h; Hydrolysis; | 93% |
| With [RuCl2(η6-p-cymene)(P(4-C6H4F)2Cl)]; water at 40℃; for 1h; Sealed tube; Inert atmosphere; | 93% |
| With C19H23Cl2N6PRuS3*3ClH; water at 100℃; for 1h; Inert atmosphere; Schlenk technique; | 88% |
-
-
35077-11-3
α-chloro-N-bromoacetamide

-
-
79-07-2
Chloroacetamide

| Conditions | Yield |
|---|---|
| With 1,2-dimethylcyclohexene In dichloromethane at 15 - 20℃; Irradiation; | 92% |
| With perchloric acid; water; potassium hydrogen phthalate; potassium nitrate; potassium bromide at 25℃; for 1h; Equilibrium constant; |
-
-
591-49-1
1-methylcyclohex-1-ene

-
-
35070-77-0
N-chloro-alpha-chloroacetamide

-
-
35077-36-2, 64226-98-8
2-Chloro-N-((1S,2R)-2-chloro-2-methyl-cyclohexyl)-acetamide

-
B
-
79-07-2
Chloroacetamide

| Conditions | Yield |
|---|---|
| In dichloromethane at 15 - 20℃; Irradiation; | A 88% B 7% |

-
-
67-56-1
methanol

-
-
70737-12-1
methyl 2-chloroacetimidate hydrochloride

-
A
-
74974-54-2
2-chloro-1,1,1-trimethoxyethane

-
B
-
79-07-2
Chloroacetamide

| Conditions | Yield |
|---|---|
| at 20℃; for 24h; | A 65% B n/a |

-
-
112-41-4
1-dodecene

-
-
35077-11-3
α-chloro-N-bromoacetamide

-
A
-
35077-35-1
N-(2-Bromo-dodecyl)-2-chloro-acetamide

-
B
-
79-07-2
Chloroacetamide

| Conditions | Yield |
|---|---|
| In dichloromethane at 15 - 20℃; Irradiation; | A 62% B 30% |

-
-
35070-77-0
N-chloro-alpha-chloroacetamide

-
-
930-66-5
1-chlorocyclohexene

-
A
-
78174-21-7
2-Chloro-N-(2,2-dichloro-cyclohexyl)-acetamide

-
B
-
79-07-2
Chloroacetamide

| Conditions | Yield |
|---|---|
| In dichloromethane at 15 - 20℃; Irradiation; | A 35% B 59% |

-
-
35070-77-0
N-chloro-alpha-chloroacetamide

-
-
932-66-1
1-cyclohexenyl methyl ketone

-
-
35077-39-5, 35077-40-8
N-((1S,2R)-2-Acetyl-2-chloro-cyclohexyl)-2-chloro-acetamide

-
-
35077-39-5, 35077-40-8
N-((1R,2R)-2-Acetyl-2-chloro-cyclohexyl)-2-chloro-acetamide

-
C
-
79-07-2
Chloroacetamide

| Conditions | Yield |
|---|---|
| In dichloromethane at 15 - 20℃; Irradiation; | A 16% B 54% C 30% |

-
-
591-49-1
1-methylcyclohex-1-ene

-
-
35077-11-3
α-chloro-N-bromoacetamide

-
-
35077-37-3, 35077-38-4
N-((1S,2R)-2-Bromo-2-methyl-cyclohexyl)-2-chloro-acetamide

-
-
35077-37-3, 35077-38-4
N-((1R,2R)-2-Bromo-2-methyl-cyclohexyl)-2-chloro-acetamide

-
C
-
79-07-2
Chloroacetamide

| Conditions | Yield |
|---|---|
| In dichloromethane at 15 - 20℃; Irradiation; | A 48% B 14% C 35% |

-
-
35077-11-3
α-chloro-N-bromoacetamide

-
-
110-83-8
cyclohexene

-
-
35077-28-2, 35077-29-3
N-((1S,2R)-2-Bromo-cyclohexyl)-2-chloro-acetamide

-
-
35077-28-2, 35077-29-3
N-(trans-2-bromocyclohexyl)chloroacetamide

-
C
-
79-07-2
Chloroacetamide

| Conditions | Yield |
|---|---|
| In dichloromethane at 15 - 20℃; for 1.5h; Irradiation; Yield given. Yields of byproduct given; | A n/a B n/a C 37% |

-
-
67-56-1
methanol

-
-
110-83-8
cyclohexene

-
-
35077-28-2, 35077-29-3
N-((1S,2R)-2-Bromo-cyclohexyl)-2-chloro-acetamide

-
-
35077-28-2, 35077-29-3
N-(trans-2-bromocyclohexyl)chloroacetamide

-
D
-
79-07-2
Chloroacetamide

| Conditions | Yield |
|---|---|
| With α-chloro-N-bromoacetamide at -70℃; for 17h; Irradiation; Yields of byproduct given; | A 27% B n/a C n/a D 31% |

-
-
498-66-8
norborn-2-ene

-
-
35077-11-3
α-chloro-N-bromoacetamide

-
-
64227-10-7, 64312-52-3
N-((1R,2S,3S,4S)-3-Bromo-bicyclo[2.2.1]hept-2-yl)-2-chloro-acetamide

-
-
64227-10-7, 64312-52-3
N-((1R,2S,3R,4S)-3-Bromo-bicyclo[2.2.1]hept-2-yl)-2-chloro-acetamide

-
C
-
79-07-2
Chloroacetamide

| Conditions | Yield |
|---|---|
| In dichloromethane at 15 - 20℃; Irradiation; Yield given. Yields of byproduct given; | A n/a B n/a C 30% |

-
-
35070-77-0
N-chloro-alpha-chloroacetamide

-
-
110-83-8
cyclohexene

-
-
24281-07-0, 33092-85-2, 35077-14-6, 53297-75-9
N-(trans-2-chlorocyclohexyl)acetamide

-
-
35077-18-0, 35077-19-1
2-Chloro-N-((1S,2R)-2-chloro-cyclohexyl)-acetamide

-
-
35077-18-0, 35077-19-1
2-Chloro-N-((1S,2S)-2-chloro-cyclohexyl)-acetamide

-
D
-
79-07-2
Chloroacetamide

| Conditions | Yield |
|---|---|
| In acetonitrile at 15 - 20℃; Irradiation; Yields of byproduct given; | A 18% B n/a C n/a D 12% |

-
-
498-66-8
norborn-2-ene

-
-
35070-77-0
N-chloro-alpha-chloroacetamide

-
-
64227-07-2, 64281-95-4
2-Chloro-N-((1R,2S,3S,4S)-3-chloro-bicyclo[2.2.1]hept-2-yl)-acetamide

-
-
64227-07-2, 64281-95-4
2-Chloro-N-((1R,2S,3R,4S)-3-chloro-bicyclo[2.2.1]hept-2-yl)-acetamide

-
C
-
79-07-2
Chloroacetamide

| Conditions | Yield |
|---|---|
| In dichloromethane at 15 - 20℃; Irradiation; Yield given. Yields of byproduct given; | A n/a B n/a C 10% |

-
-
35070-77-0
N-chloro-alpha-chloroacetamide

-
-
110-83-8
cyclohexene

-
-
35077-18-0, 35077-19-1
2-Chloro-N-((1S,2R)-2-chloro-cyclohexyl)-acetamide

-
-
35077-18-0, 35077-19-1
2-Chloro-N-((1S,2S)-2-chloro-cyclohexyl)-acetamide

-
C
-
79-07-2
Chloroacetamide

| Conditions | Yield |
|---|---|
| In dichloromethane at 15 - 20℃; for 2.5h; Irradiation; Yield given; | A n/a B n/a C 9% |


| Conditions | Yield |
|---|---|
| With nitrogen trichloride; diethyl ether at -60℃; unter Ausschluss von Feuchtigkeit; |
-
-
64-17-5
ethanol

-
-
4960-82-1
dichlorodiacetamide

-
A
-
105-39-5
chloroacetic acid ethyl ester

-
B
-
79-07-2
Chloroacetamide


| Conditions | Yield |
|---|---|
| With ammonia; water | |
| sodium methylate In methanol | |
| sodium methylate In methanol |

| Conditions | Yield |
|---|---|
| With ammonia | |
| With ammonium hydroxide | |
| With ammonium hydroxide | |
| With ammonium hydroxide at 0℃; | |
| With ammonium hydroxide at 0℃; for 0.25h; Temperature; |

| Conditions | Yield |
|---|---|
| at 160 - 170℃; |

| Conditions | Yield |
|---|---|
| With ammonia | |
| With sulfuric acid; 1,1,1,3,3,3-hexamethyl-disilazane 1.) CH2Cl2, room temp., 2.) CH3OH; Yield given. Multistep reaction; | |
| With ammonia; triethylamine In chloroform at 20℃; for 2h; Substitution; |
-
-
35070-77-0
N-chloro-alpha-chloroacetamide

-
-
78174-28-4
2-Chloro-N-(2,2-dimethoxy-cyclohexyl)-acetamide

-
A
-
35077-41-9
α-chloroacetamidocyclohexanone

-
B
-
822-87-7
2-Chlorocyclohexanone

-
C
-
79-07-2
Chloroacetamide

| Conditions | Yield |
|---|---|
| With water -70 degC, then allowed to warm to r.t.; Yield given. Yields of byproduct given; |


| Conditions | Yield |
|---|---|
| Yield given. Multistep reaction. Yields of byproduct given; |
-
-
26473-57-4
(1-methoxyethylidene)cyclohexane

-
A
-
1004-55-3
1-(1-chlorocyclohexyl)ethanone

-
B
-
79-07-2
Chloroacetamide

| Conditions | Yield |
|---|---|
| With N-chloro-alpha-chloroacetamide; water 1.) irradiation, CH2Cl2; Yield given. Multistep reaction. Yields of byproduct given; |

| Conditions | Yield |
|---|---|
| With water at 25℃; Rate constant; effect of pH on the rate constants; |

| Conditions | Yield |
|---|---|
| In acetone for 1h; Ambient temperature; | 100% |
-
-
889656-79-5
6-mercapto-3,3-dimethyl-8-dimethylamino-3,4-dihydro-1H-pyrano[3,4-c]-pyridine-5-carbonitrile

-
-
79-07-2
Chloroacetamide

-
-
889656-80-8
1-amino-5-dimethylamino-8,8-dimethyl-8,9-dihydro-6H-pyrano[4,3-d]thieno[2,3-b]pyridine-2-carboxamide

| Conditions | Yield |
|---|---|
| With potassium carbonate In ethanol at 20℃; for 5h; Heating / reflux; | 100% |
| With potassium carbonate In ethanol for 4h; Reflux; Inert atmosphere; |
-
-
890023-58-2
1-(benzylmethylamino)-3-mercapto-6,6-dimethyl-5,6,7,8-tetrahydro-isoquinoline-4-carbonitrile

-
-
79-07-2
Chloroacetamide

-
-
890023-59-3
C22H26N4OS

| Conditions | Yield |
|---|---|
| With potassium carbonate In ethanol Heating / reflux; | 100% |
| With potassium carbonate In ethanol for 4h; Reflux; Inert atmosphere; |
-
-
924-44-7
glyoxylic acid ethyl ester

-
-
79-07-2
Chloroacetamide

-
-
1198115-96-6
ethyl 2-(2-chloroacetamido)-2-hydroxyacetate

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; toluene at 80℃; Inert atmosphere; | 100% |
-
-
2075-46-9
4-nitro-1H-pyrazole

-
-
79-07-2
Chloroacetamide

-
-
32407-64-0
2-(4-nitro-1H-pyrazol-1-yl)acetamide

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetonitrile at 60℃; | 100% |
-
-
552-89-6
2-nitro-benzaldehyde

-
-
79-07-2
Chloroacetamide

-
-
109287-33-4
trans-3-(2-nitrophenyl)oxirane-2-carboxamide

| Conditions | Yield |
|---|---|
| With sodium ethanolate In ethanol at 20℃; for 7h; Darzens Condensation; stereoselective reaction; | 100% |
| With sodium ethanolate In ethanol at 20℃; for 5h; | 100% |
-
-
603-35-0
triphenylphosphine

-
-
79-07-2
Chloroacetamide

-
-
25361-54-0
(2-amino-2-oxo-ethyl)triphenyl-phosphonium chloride

| Conditions | Yield |
|---|---|
| In nitromethane at 105℃; for 19h; Schlenk technique; | 99% |
| In nitromethane at 105℃; for 19h; Inert atmosphere; | 98% |
| In acetonitrile for 11h; Heating; Inert atmosphere; | 96% |
| In various solvent(s) at 100℃; for 16h; | 84% |

| Conditions | Yield |
|---|---|
| With potassium carbonate; potassium iodide In acetone for 48h; Reflux; | 99% |
| With potassium carbonate In N,N-dimethyl-formamide at 110℃; for 2h; | |
| With potassium carbonate In acetonitrile Reflux; | |
| With potassium carbonate; potassium iodide In N,N-dimethyl-formamide at 20℃; for 5h; |
-
-
890023-03-7
3-mercapto-1-dimethylamino-6,6-dimethyl-5,6,7,8-tetrahydroisoquinoline-4-carbonitrile

-
-
79-07-2
Chloroacetamide

-
-
890023-04-8
1-amino-5-dimethylamino-8,8-dimethyl-6,7,8,9-tetrahydrothieno[2,3-c]isoquinoline-2-carboxamide

| Conditions | Yield |
|---|---|
| With potassium carbonate In ethanol Heating / reflux; | 99% |
-
-
890023-33-3
1-dimethylamino-3-mercapto-6,6-dimethyl-6,7-dihydro-5H-[2]pyridin-4-carbonitrile

-
-
79-07-2
Chloroacetamide

-
-
890023-34-4
1-amino-5-dimethylamino-7,7-dimethyl-6H-7,8-dihydrocycIopenta[d]thieno[2,3-b]pyridin-2-carboxamide

| Conditions | Yield |
|---|---|
| With potassium carbonate In ethanol Heating / reflux; | 99% |
-
-
942302-89-8
[2-[3-(3-fluoro-benzyloxy)-phenyl]-ethyl]-(furan-2-ylmethyl)amine

-
-
79-07-2
Chloroacetamide

| Conditions | Yield |
|---|---|
| With triethylamine In N,N-dimethyl-formamide at 120℃; for 2h; microwave irradiation; | 99% |

| Conditions | Yield |
|---|---|
| With sodium ethanolate In ethanol at 20℃; for 5h; | 99% |
-
-
6640-27-3
2-chloro-p-cresol

-
-
79-07-2
Chloroacetamide

-
-
1248651-95-7
2-(2-chloro-4-methylphenoxy)acetamide

| Conditions | Yield |
|---|---|
| With potassium carbonate at 82℃; for 0.166667h; Solvent; Microwave irradiation; | 98.7% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone at 56℃; | 98.5% |
| Stage #1: salicylonitrile With potassium carbonate In N,N-dimethyl-formamide at 60℃; for 1h; Stage #2: Chloroacetamide In N,N-dimethyl-formamide at 80℃; for 3h; | |
| With potassium carbonate In N,N-dimethyl-formamide at 80℃; |

-
-
79-07-2
Chloroacetamide

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 70℃; for 10h; | A 98.21% B n/a |

-
-
108-89-4
picoline

-
-
79-07-2
Chloroacetamide

-
-
78572-45-9
1-(2-amino-2-oxoethyl)-4-methylpyridinium chloride

| Conditions | Yield |
|---|---|
| In acetonitrile at 80℃; for 48h; | 98% |
| In acetone at 25℃; for 72h; | 75% |
| In toluene for 5h; Reflux; | 52% |
-
-
147381-62-2
6-(4-chlorophenyl)-3-cyano-4-trifluoromethyl-2(1H)-pyridone

-
-
79-07-2
Chloroacetamide

-
-
155635-60-2
2-[6-(4-Chloro-phenyl)-3-cyano-4-trifluoromethyl-pyridin-2-yloxy]-acetamide

| Conditions | Yield |
|---|---|
| With potassium carbonate; sodium iodide In acetone at 60℃; for 6h; | 98% |
-
-
121221-00-9
N-(O,O'-diisopropylthiophosphoryl)thioacetamide

-
-
79-07-2
Chloroacetamide

| Conditions | Yield |
|---|---|
| With triethylamine In acetonitrile for 3h; Heating; | 98% |

-
-
79-07-2
Chloroacetamide

-
-
136633-82-4
2-(6-amino-3,5-dicyano-4-thiophen-2-yl-pyridin-2-ylsulfanyl)-acetamide

| Conditions | Yield |
|---|---|
| With triethylamine In acetone at 20℃; for 5h; | 98% |

-
-
79-07-2
Chloroacetamide

-
-
1238357-72-6
2-chloro-trans-N-(3-methoxy-propenyl)-acetamide

| Conditions | Yield |
|---|---|
| With oxygen; copper diacetate In dichloromethane; dimethyl sulfoxide at 40℃; for 20h; Molecular sieve; diastereoselective reaction; | 98% |
-
-
1352948-27-6
3-((8-(tert-butoxycarbonyl)-4-oxo-1-phenyl-1,3,8-triazaspiro[4.5]decan-3-yl)methyl)benzoic acid

-
-
79-07-2
Chloroacetamide

-
-
1352948-59-4
tert-butyl 3-(3-((2-amino-2-oxoethoxy)carbonyl)benzyl)-4-oxo-1-phenyl-1,3,8-triazaspiro[4.5]decane-8-carboxylate

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 65℃; for 18h; | 98% |

| Conditions | Yield |
|---|---|
| With potassium carbonate; potassium iodide In butanone for 48h; Reflux; Inert atmosphere; | 98% |
-
-
342597-54-0
5-(4-methoxybenzyl)-4-phenyl-4H-1,2,4-triazole-3-thiol

-
-
79-07-2
Chloroacetamide

-
-
874798-09-1
2-[5-(4-methoxybenzyl)-4-phenyl-4H-1,2,4-triazol-3-ylsulfanyl]acetamide

| Conditions | Yield |
|---|---|
| Stage #1: 5-(4-methoxybenzyl)-4-phenyl-4H-1,2,4-triazole-3-thiol With potassium hydroxide In ethanol Reflux; Stage #2: Chloroacetamide In ethanol Reflux; | 98% |

-
-
79-07-2
Chloroacetamide

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 60℃; for 12h; | 98% |

-
-
79-07-2
Chloroacetamide

| Conditions | Yield |
|---|---|
| Heating; | 98% |

| Conditions | Yield |
|---|---|
| Stage #1: diphenylchloromethane With thiourea; potassium iodide In water at 70 - 75℃; for 1.5h; Stage #2: With sodium hydroxide In water at 15 - 24℃; Stage #3: Chloroacetamide With triethylamine more than 3 stages; | 97.9% |
| Stage #1: diphenylchloromethane With thiourea In isopropyl alcohol at 100℃; for 2h; Sealed tube; Stage #2: Chloroacetamide With potassium hydroxide In methanol; isopropyl alcohol at 60℃; for 2h; Sonication; |
-
-
79-07-2
Chloroacetamide

-
-
35077-11-3
α-chloro-N-bromoacetamide

| Conditions | Yield |
|---|---|
| With dibromoisocyanuric acid In dichloromethane for 5h; Bromination; Heating; | 97% |
| With perchloric acid; water; potassium hydrogen phthalate; potassium nitrate; potassium bromide at 25℃; for 1h; Equilibrium constant; | |
| With bromine; trifluoroacetic acid; silver(l) oxide | |
| With dibromoisocyanuric acid In dichloromethane | |
| With sodium hypobromide |
-
-
70182-88-6
7-Amino<1,2,4>triazolo<1,5-c>pyrimidine-5(6H)-thione

-
-
79-07-2
Chloroacetamide

-
-
143212-81-1
(7-amino-s-triazole<1,5-c>pyrimidyl-5)thioacetic acid amide

| Conditions | Yield |
|---|---|
| With sodium ethanolate In ethanol for 3h; Heating; | 97% |

| Conditions | Yield |
|---|---|
| With borohydride exchange resin; nickel diacetate In methanol for 1h; Ambient temperature; | 97% |

| Conditions | Yield |
|---|---|
| In acetonitrile at 20 - 25℃; | 97% |
| With diisopropylamine In acetonitrile at 20℃; for 16h; | 86% |
Chloroacetamide Specification
The CAS registry number of Chloroacetamide is 79-07-2. The IUPAC name is 2-chloroacetamide. Its EINECS registry number is 201-174-2. It is also called Acetamide, 2-chloro-. In addition, the molecular formula is C2H4ClNO and the molecular weight is 93.51. It is a colorless or yellow crystalline substance with characteristic smell, readily soluble in water. What's more, it belongs to the classes of Amides; Carbonyl Compounds; Organic Building Blocks. It should be stored in a airtight, cool and dry place.
Physical properties about this chemical are: (1)ACD/LogP: -0.85; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): -0.85; (4)ACD/LogD (pH 7.4): -0.85; (5)ACD/BCF (pH 5.5): 1; (6)ACD/BCF (pH 7.4): 1; (7)ACD/KOC (pH 5.5): 8.18; (8)ACD/KOC (pH 7.4): 8.18; (9)#H bond acceptors: 2; (10)#H bond donors: 2; (11)#Freely Rotating Bonds: 1; (12)Polar Surface Area: 20.31 Å2; (13)Index of Refraction: 1.447; (14)Molar Refractivity: 19.72 cm3; (15)Molar Volume: 73.7 cm3; (16)Polarizability: 7.82 ×10-24cm3; (17)Surface Tension: 39 dyne/cm; (18)Density: 1.268 g/cm3; (19)Flash Point: 108.6 °C; (20)Enthalpy of Vaporization: 49.35 kJ/mol; (21)Boiling Point: 256 °C at 760 mmHg; (22)Vapour Pressure: 0.0158 mmHg at 25°C.
Preparation of Chloroacetamide: it can be prepared by ethyl chloroacetate and ammonia. At first, cool the ethyl chloroacetate to 10 °C, slowly add ammonia (relative density is 0.9) with stirring. After finish adding ammonia, continue to stir for 1 hour, filter and crystal the product, use ammonia to wash chloride, drained the product as far as possible, use ethanol for recrystallization at last.
![]()
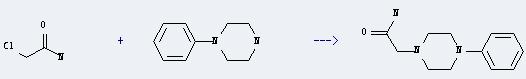
Uses of Chloroacetamide: it can be used in organic synthesis, pharmaceutical intermediates. It can produce organic compounds such as chloroacetonitrile, sulfanilyl-3-methoxypyrazine and sulfanilylmethylpyrazine. In addition, it can be used as preservative of shampoo, cutting oils, plastics, coatings slab and shower gel. What's more, it can react with 1-phenyl-piperazine to get (4-phenyl-piperazino)-acetic acid amide. This reaction will need reagent K2CO3 and solvent toluene. The yield is about 75% by heating.
When you are using this chemical, please be cautious about it as the following:
It is toxic if swallowed and may cause sensitization by skin contact. And it has risk of impaired fertility. During using it, do not breathe dust and wear suitable protective clothing and gloves. In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.).
You can still convert the following datas into molecular structure:
(1)SMILES: ClCC(=O)N
(2)InChI: InChI=1/C2H4ClNO/c3-1-2(4)5/h1H2,(H2,4,5)
(3)InChIKey: VXIVSQZSERGHQP-UHFFFAOYAM
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| dog | LD50 | oral | 31mg/kg (31mg/kg) | Zhonghua Yixue Zazhi. Chinese Medical Journal. Vol. 58, Pg. 462, 1978. | |
| mouse | LD50 | intraperitoneal | 100mg/kg (100mg/kg) | National Technical Information Service. Vol. AD277-689, | |
| mouse | LD50 | intravenous | 180mg/kg (180mg/kg) | U.S. Army Armament Research & Development Command, Chemical Systems Laboratory, NIOSH Exchange Chemicals. Vol. NX#03764, | |
| mouse | LD50 | oral | 155mg/kg (155mg/kg) | Zhonghua Yixue Zazhi. Chinese Medical Journal. Vol. 58, Pg. 462, 1978. | |
| rabbit | LD50 | oral | 122mg/kg (122mg/kg) | Zhonghua Yixue Zazhi. Chinese Medical Journal. Vol. 58, Pg. 462, 1978. | |
| rat | LD50 | oral | 138mg/kg (138mg/kg) | BEHAVIORAL: ATAXIA LUNGS, THORAX, OR RESPIRATION: DYSPNEA | National Technical Information Service. Vol. OTS0536947, |
| rat | LD50 | unreported | 70mg/kg (70mg/kg) | Arzneimittel-Forschung. Drug Research. Vol. 19, Pg. 1073, 1969. |
Related Products
- Chloroacetamide
- Chloroacetamide Oxime
- 79072-90-5
- 790-75-0
- 79079-11-1
- 79079-18-8
- 79080-31-2
- 79080-39-0
- 79082-81-8
- 79-08-3
- 790-83-0
- 79091-24-0
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View