-
Name
DICYCLOHEXYL KETONE
- EINECS
- CAS No. 119-60-8
- Article Data71
- CAS DataBase
- Density 0.97g/cm3
- Solubility
- Melting Point 57°C
- Formula C13H22 O
- Boiling Point 266.4°Cat760mmHg
- Molecular Weight 194.317
- Flash Point 123.4°C
- Transport Information
- Appearance
- Safety Poison by intravenous route. See also KETONES. A flammable liquid. When heated to decomposition it emits acrid and irritating fumes.
- Risk Codes
-
Molecular Structure
- Hazard Symbols
- Synonyms Cyclohexylketone (7CI,8CI); Bicyclohexyl ketone; Dicyclohexyl ketone;Dicyclohexylmethanone; NSC 49725; NSC 60007
- PSA 17.07000
- LogP 3.71610
Synthetic route

| Conditions | Yield |
|---|---|
| With 6C6H15P*4CF3O3S(1-)*2Ru(2+); acetone In 2,2,2-trifluoroethanol at 20℃; for 3h; Reagent/catalyst; | 99% |
| With tetra-O-acetyl riboflavin; lithium trifluoromethanesulfonate; 1,3-diisopropylthiourea In water; acetonitrile at 22℃; for 24h; Inert atmosphere; Sealed tube; Irradiation; | 90% |
| With isobutyl 4-[bis(acetoxy)iodo]phenylsulfonate; ruthenium trichloride In water; acetonitrile at 20℃; for 8h; | 89% |
-
-
2719-27-9
cyclohexanylcarbonyl chloride

-
-
446065-11-8
cyclohexyltrifluoro-λ4-borane potassium salt

-
-
119-60-8
dicyclohexyl ketone

| Conditions | Yield |
|---|---|
| With potassium fluoride; (1,2-dimethoxyethane)dichloronickel(II); Ir(dF(CF3)ppy)2(bpy)PF6; 4,4'-di-tert-butyl-2,2'-bipyridine In 1,2-dimethoxyethane at 24℃; for 24h; Inert atmosphere; Sealed tube; Irradiation; | 84% |
-
-
1088-01-3
tricyclohexylborane

-
-
14572-78-2
lithium tris(phenylthio)methanide

-
-
119-60-8
dicyclohexyl ketone

| Conditions | Yield |
|---|---|
| With sodium hydroxide; dihydrogen peroxide Ambient temperature; | 80% |

| Conditions | Yield |
|---|---|
| With tetraethyl ammonium permanganate at 20℃; for 0.666667h; | 80% |

| Conditions | Yield |
|---|---|
| With air; sodium nitrite In trifluoroacetic acid at 0 - 20℃; for 5h; | A 79% B n/a |

| Conditions | Yield |
|---|---|
| With [2,2]bipyridinyl; chloro-trimethyl-silane; nickel(II) bromide 2-methoxyethyl ether complex; zinc In tetrahydrofuran; N,N-dimethyl acetamide at 40℃; for 2h; Sealed tube; Inert atmosphere; | 76% |
-
-
95110-49-9
2'-cyclohexyl-2'-phenylsulfinylspiro

-
-
119-60-8
dicyclohexyl ketone

| Conditions | Yield |
|---|---|
| With sodium phenylselenide In ethanol for 16h; Heating; | 75% |
| With sodium phenylselenide In ethanol for 16h; Heating; | 75% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at -80 - 20℃; Inert atmosphere; | 75% |
-
-
91-01-0
1,1-Diphenylmethanol

-
A
-
119-60-8
dicyclohexyl ketone

-
B
-
4453-82-1
Dicyclohexylmethanol

-
C
-
945-49-3
cyclohexylphenylmethanol

| Conditions | Yield |
|---|---|
| With hydrogen In water at 50℃; under 15001.5 Torr; for 6h; chemoselective reaction; | A 7 %Spectr. B 75% C 17% |

-
-
52034-92-1
dicyclohexylacetic acid

-
-
119-60-8
dicyclohexyl ketone

| Conditions | Yield |
|---|---|
| With (2,2'-dipyridyl)bis(5-methyl-2-(4-fluoro)phenylpyridine-N,C)iridium(III) hexafluorophosphate; oxygen; sodium carbonate; 1,1'-diethyl-4,4'-bipyridinium diperchlorate In dimethyl sulfoxide at 20℃; under 760.051 Torr; for 16h; Irradiation; | 65% |

| Conditions | Yield |
|---|---|
| Stage #1: 1-bromocyclohexane With magnesium In tetrahydrofuran at 20℃; for 1h; Stage #2: cyclohexanecarbaldehyde In tetrahydrofuran at 0 - 20℃; for 2h; Stage #3: With 1,3-Diiodo-5,5-dimethyl-2,4-imidazolidinedione; potassium carbonate In tetrahydrofuran; tert-butyl alcohol for 20h; Reflux; Darkness; | 60% |

| Conditions | Yield |
|---|---|
| With (1,2-dimethoxyethane)dichloronickel(II); [4,4’-bis(1,1-dimethylethyl)-2,2’-bipyridine-N1,N1‘]bis [3,5-difluoro-2-[5-(trifluoromethyl)-2-pyridinyl-N]phenyl-C]iridium(III) hexafluorophosphate; potassium carbonate; 4,4'-di-tert-butyl-2,2'-bipyridine In benzene at 20℃; for 16h; Irradiation; Inert atmosphere; | 60% |
| With potassium phosphate; sodium tungstate (VI) dihydrate; [4,4’-bis(1,1-dimethylethyl)-2,2’-bipyridine-N1,N1‘]bis [3,5-difluoro-2-[5-(trifluoromethyl)-2-pyridinyl-N]phenyl-C]iridium(III) hexafluorophosphate; Ni[(4,4′-di-tert-butyl-2,2′-bipyridine)(H2O)4]Cl2; lithium chloride In benzene at 20℃; for 24h; Inert atmosphere; Sealed tube; Schlenk technique; Irradiation; | 52% |
-
-
22651-87-2
cyclohexanecarboxylic acid anhydride

-
-
119-60-8
dicyclohexyl ketone

| Conditions | Yield |
|---|---|
| With [2,2]bipyridinyl; (1,2-dimethoxyethane)dichloronickel(II); Ir[dF(OMe)ppy]2-(dtbbpy)PF6; 1,8-diazabicyclo[5.4.0]undec-7-ene In 1,4-dioxane at 25℃; for 36h; Sealed tube; Inert atmosphere; Irradiation; | 50% |

| Conditions | Yield |
|---|---|
| With potassium iodide In N,N-dimethyl-formamide at 80℃; for 20h; | 47% |
-
-
141694-46-4
trimethyl<<(1Z)-1-cyclohexylhepta-1,6-dienyl>oxy>silane

-
-
119-60-8
dicyclohexyl ketone

| Conditions | Yield |
|---|---|
| With 9,10-Dicyanoanthracene In acetonitrile for 120h; Irradiation; | 37% |
| With 9,10-Dicyanoanthracene In acetonitrile for 65h; Irradiation; | 37 % Chromat. |
-
-
28178-42-9
2,6-diisopropylphenyl isocyanate

-
A
-
119-60-8
dicyclohexyl ketone

| Conditions | Yield |
|---|---|
| With Ru photocatalyst; Ni catalyst In N,N-dimethyl-formamide Mechanism; Irradiation; | A n/a B 16% |


| Conditions | Yield |
|---|---|
| Stage #1: cyclohexylmagnesiumchloride With titanium(IV) isopropylate In diethyl ether at -78 - -30℃; for 0.166667h; Stage #2: carbon dioxide In diethyl ether at -30 - 0℃; for 0.25h; Stage #3: With iodine In diethyl ether at 20℃; for 2h; | A 14% B 0.12 g |

-
-
110-82-7
cyclohexane

-
-
201230-82-2
carbon monoxide

-
A
-
119-60-8
dicyclohexyl ketone

-
B
-
823-76-7
Cyclohexyl methyl ketone

-
C
-
2043-61-0
cyclohexanecarbaldehyde

-
D
-
98-89-5
Cyclohexanecarboxylic acid

| Conditions | Yield |
|---|---|
| With tris(tetrabutylammonium)12-tungstophosphate In acetonitrile at 25℃; under 760 Torr; for 16h; Product distribution; Mechanism; Irradiation; other alkanes and aromatic reactants; var. solvents and times; | A 4.1 % Turnov. B 2.7 % Turnov. C 8% D n/a |

-
-
60-29-7
diethyl ether

-
-
3289-28-9
ethyl cyclohexanecarboxylate

-
-
931-51-1
cyclohexylmagnesiumchloride

-
A
-
92-51-3
cyclohexylcyclohexane

-
B
-
119-60-8
dicyclohexyl ketone

-
C
-
17687-74-0
α,α-dicyclohexyl-cyclohexanemethanol

-
-
542-18-7
cyclohexyl chloride

-
-
3289-28-9
ethyl cyclohexanecarboxylate

-
A
-
119-60-8
dicyclohexyl ketone

-
B
-
17687-74-0
α,α-dicyclohexyl-cyclohexanemethanol

| Conditions | Yield |
|---|---|
| With diethyl ether; magnesium | |
| With sodium; pentane |


| Conditions | Yield |
|---|---|
| With diethyl ether; cyclohexylmagnesiumchloride | |
| With diethyl ether; tert.-butyl lithium | |
| With tert.-butyl lithium In diethyl ether Heating; |
-
-
56547-92-3
exifone

-
-
119-60-8
dicyclohexyl ketone

| Conditions | Yield |
|---|---|
| With ethyl acetate; platinum Hydrogenation; |

| Conditions | Yield |
|---|---|
| With sulfuric acid |

| Conditions | Yield |
|---|---|
| With oxalyl dichloride | |
| With cyclohexanylcarbonyl chloride | |
| Multi-step reaction with 3 steps 1: benzene 2: KOH-solution 3: concentrated sulfuric acid View Scheme |
-
-
66031-37-6
1,1-Dicyclohexyl-2-methoxy-ethanol

-
-
119-60-8
dicyclohexyl ketone

| Conditions | Yield |
|---|---|
| (electrochemical oxidation); |
-
-
52712-18-2
Dicyclohexyl-chlor-methylboronsaeuredimethylester

-
-
119-60-8
dicyclohexyl ketone

| Conditions | Yield |
|---|---|
| With sodium hydroxide; dihydrogen peroxide |
-
-
61259-06-1
1-Cyclohexanecarbonyl-cyclohexanecarboxylic acid; compound with diisopropyl-amine

-
-
119-60-8
dicyclohexyl ketone

| Conditions | Yield |
|---|---|
| at 150 - 200℃; |

| Conditions | Yield |
|---|---|
| (i) THF, (ii) aq. NaOH, H2O2; Multistep reaction; |
-
-
109-06-8
α-picoline

-
-
119-60-8
dicyclohexyl ketone

-
-
102658-00-4
1,1-dicyclohexyl-2-(pyridin-2-yl)ethanol

| Conditions | Yield |
|---|---|
| Stage #1: α-picoline With n-butyllithium In tetrahydrofuran; hexane at -78 - -20℃; Inert atmosphere; Stage #2: dicyclohexyl ketone In tetrahydrofuran; hexane at -20℃; Inert atmosphere; Stage #3: With water; ammonium chloride In tetrahydrofuran; hexane | 100% |
-
-
16520-62-0
4-Phenyl-1-butyne

-
-
119-60-8
dicyclohexyl ketone

-
-
1393658-19-9
1,1-dicyclohexyl-5-phenyl-2-pentyn-1-ol

| Conditions | Yield |
|---|---|
| Stage #1: 4-Phenyl-1-butyne With n-butyllithium In tetrahydrofuran; hexanes at -78℃; for 1h; Stage #2: dicyclohexyl ketone In tetrahydrofuran; hexanes at -78 - 20℃; for 3.5h; | 100% |

| Conditions | Yield |
|---|---|
| With (N,N'-dicyclohexyl-2H-imidazol-2-ylidene)CuCl; sodium t-butanolate In toluene at 80℃; for 0.5h; | 99% |
| With sodium t-butanolate; [CuCl(N,N'-bis(cyclohexyl)imidazol-2-ylidene)] In toluene at 80℃; for 0.5h; | 99% |
| With (1,3-bis(9-ethyl-9H-fluoren-9-yl)imidazol-2-ylidene)copper(I) chloride; potassium tert-butylate In tetrahydrofuran at 65℃; for 4h; Catalytic behavior; Solvent; Time; Reagent/catalyst; Temperature; Inert atmosphere; Schlenk technique; | 98% |
-
-
119-60-8
dicyclohexyl ketone

| Conditions | Yield |
|---|---|
| With sodium borodeuteride In methanol at 25℃; for 2h; Cooling with ice; Inert atmosphere; | 99% |

| Conditions | Yield |
|---|---|
| Stage #1: dicyclohexyl ketone With bis-{[N,N′-bis(2,6-(di-isopropyl)phenyl)imidazol-2-ylidene]-(1H-1,2,4-triazol-1-yl)}copper(I) In tetrahydrofuran at 55℃; for 14h; Stage #2: With sodium hydroxide In methanol; water at 25℃; for 1.5h; | 97% |
| With methanol; sodium tetrahydroborate at 0 - 20℃; | 96% |
| With methanol; sodium tetrahydroborate at 0 - 20℃; | 96% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; hexane at -78℃; for 5h; | 96% |
-
-
119-60-8
dicyclohexyl ketone

-
-
1066-54-2
trimethylsilylacetylene

-
-
10562-70-6
1,1-dicyclohexylprop-2-yn-1-ol

| Conditions | Yield |
|---|---|
| Stage #1: trimethylsilylacetylene With n-butyllithium In tetrahydrofuran; hexane at -10℃; for 1h; Inert atmosphere; Stage #2: dicyclohexyl ketone In tetrahydrofuran; hexane at -10 - 0℃; for 3h; Inert atmosphere; Stage #3: With sodium hydroxide In tetrahydrofuran; methanol; hexane at 0 - 20℃; Inert atmosphere; | 94% |

| Conditions | Yield |
|---|---|
| With Montmorillonite clay KSF at 110℃; for 0.25h; Irradiation; | 92% |
-
-
75-77-4
chloro-trimethyl-silane

-
-
119-60-8
dicyclohexyl ketone

-
-
66534-90-5
Dicyclohexyltrimethylsilylenolether

| Conditions | Yield |
|---|---|
| With lithium hexamethyldisilazane | 92% |

| Conditions | Yield |
|---|---|
| With Montmorillonite clay KSF at 75℃; for 0.0333333h; Irradiation; | 91% |
-
-
617-86-7
triethylsilane

-
-
119-60-8
dicyclohexyl ketone

-
A
-
27428-33-7
(cyclohexylidenemethyl)cyclohexane

| Conditions | Yield |
|---|---|
| With [(SIMes)PFMe2][B(C6F5)4]2 In dichloromethane-d2 at 20℃; for 1h; Temperature; Reagent/catalyst; Inert atmosphere; Schlenk technique; Glovebox; | A 11% B 89% |

-
-
119-60-8
dicyclohexyl ketone

-
-
89894-42-8
1,2-dichlorovinyl phenyl ether

| Conditions | Yield |
|---|---|
| Stage #1: 1,2-dichlorovinyl phenyl ether With n-butyllithium In diethyl ether; hexane at -78 - -20℃; for 2h; Stage #2: dicyclohexyl ketone In diethyl ether; hexane at -78 - 20℃; | 88% |
-
-
119-60-8
dicyclohexyl ketone

-
-
3132-64-7
1,2-Epoxy-3-bromopropane

-
-
91540-05-5
4-Bromomethyl-2,2-dicyclohexyl-[1,3]dioxolane

| Conditions | Yield |
|---|---|
| With tin(IV) chloride In tetrachloromethane for 2.5h; Ambient temperature; | 86.8% |

| Conditions | Yield |
|---|---|
| With Fe3O4(at)cellulose-OSO3H nanocomposite In ethanol at 20℃; for 1.5h; Green chemistry; | 85% |


| Conditions | Yield |
|---|---|
| Stage #1: 1-Pentyne With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 0.5h; Inert atmosphere; Stage #2: dicyclohexyl ketone In tetrahydrofuran; hexane at -78 - 20℃; for 3h; Inert atmosphere; | 85% |
-
-
119-60-8
dicyclohexyl ketone

-
-
103-63-9
1-phenyl-2-bromoethane

-
-
1262387-08-5
1,1-dicyclohexyl-3-phenyl-propan-1-ol

| Conditions | Yield |
|---|---|
| Stage #1: 1-phenyl-2-bromoethane With magnesium In tetrahydrofuran at -20℃; for 1h; Inert atmosphere; Stage #2: dicyclohexyl ketone In tetrahydrofuran at 20℃; for 3.25h; | 84% |

| Conditions | Yield |
|---|---|
| Stage #1: 3,4-dihydro-2H-pyran With n-butyllithium In tetrahydrofuran; hexane at 0 - 20℃; for 3.16667h; Inert atmosphere; Stage #2: dicyclohexyl ketone In tetrahydrofuran; hexane at -78 - 20℃; for 2h; | 83% |

| Conditions | Yield |
|---|---|
| Stage #1: 2-(tert-butyldimethylsilyloxy)ethyl bromide With magnesium In diethyl ether at 25℃; for 1h; Inert atmosphere; Stage #2: dicyclohexyl ketone In diethyl ether for 3h; Inert atmosphere; | 82.2% |
-
-
95-16-9
1,3-Benzothiazole

-
-
119-60-8
dicyclohexyl ketone

-
-
1401254-53-2
1-(2-benzothiazolyl)-1,1-dicyclohexylmethanol

| Conditions | Yield |
|---|---|
| Stage #1: 1,3-Benzothiazole; dicyclohexyl ketone With tetrakis{[tris(dimethylamino)phosphoranyliden]amino}phosphonium fluoride; tris(trimethylsilyl)amine In toluene at 20℃; for 24h; Inert atmosphere; Stage #2: In tetrahydrofuran | 82% |
-
-
119-60-8
dicyclohexyl ketone

-
-
161607-20-1
3-(benzotriazol-1-yl)-3-ethoxy-1-propene

| Conditions | Yield |
|---|---|
| With n-butyllithium In tetrahydrofuran 1.) -78 deg C, 10 min, 2.) -78 deg C, 3 h; 20 deg C, 12 h; | 81% |
-
-
119-60-8
dicyclohexyl ketone

-
A
-
27428-33-7
(cyclohexylidenemethyl)cyclohexane

-
B
-
3178-23-2
dicyclohexylmethane

| Conditions | Yield |
|---|---|
| With triethylsilane; [(SIMes)PFMe2][B(C6F5)4]2 In dichloromethane-d2 at 50℃; for 24h; Temperature; Reagent/catalyst; Inert atmosphere; Glovebox; Schlenk technique; | A 19% B 81% |
-
-
119-60-8
dicyclohexyl ketone

-
A
-
92107-64-7
1,5-dicyclohexyl-1H-tetrazole

-
B
-
73565-25-0
1-cyclohexyl-5-cyclohexylaminotetrazole

| Conditions | Yield |
|---|---|
| With trifluorormethanesulfonic acid; trimethylsilylazide In dichloromethane at 20℃; for 5h; Inert atmosphere; regioselective reaction; | A 80% B 12% |
-
-
119-60-8
dicyclohexyl ketone

| Conditions | Yield |
|---|---|
| With naphthalene; lithium In tetrahydrofuran for 6.5h; McMurry olefination; Heating; | 78% |
| With titanium(III) chloride In tetrahydrofuran 1.) reflux, 1 h, 2.) reflux, 10 h; | 20% |
-
-
119-60-8
dicyclohexyl ketone

-
-
1779-49-3
Methyltriphenylphosphonium bromide

-
-
57013-04-4
ethene-1,1-diyldicyclohexane

| Conditions | Yield |
|---|---|
| Stage #1: Methyltriphenylphosphonium bromide With n-butyllithium In tetrahydrofuran at 0℃; for 2.75h; Inert atmosphere; Stage #2: dicyclohexyl ketone In tetrahydrofuran at 0 - 20℃; Inert atmosphere; | 78% |
| Stage #1: Methyltriphenylphosphonium bromide With sodium hexamethyldisilazane; sodium t-butanolate In tetrahydrofuran at 0℃; for 0.5h; Stage #2: dicyclohexyl ketone In tetrahydrofuran at 20℃; | 75% |
| Stage #1: Methyltriphenylphosphonium bromide With sodium hexamethyldisilazane In tetrahydrofuran at 0℃; for 1h; Stage #2: dicyclohexyl ketone In tetrahydrofuran at 0 - 20℃; | 41% |
| With n-butyllithium In tetrahydrofuran at 0 - 20℃; for 48h; | |
| Stage #1: Methyltriphenylphosphonium bromide With n-butyllithium In tetrahydrofuran at 0 - 20℃; for 3h; Inert atmosphere; Stage #2: dicyclohexyl ketone at 0 - 20℃; for 48h; Inert atmosphere; |

| Conditions | Yield |
|---|---|
| With lithium tert-butoxide In chloroform at 25℃; for 0.5h; Atherton-Todd Synthesis; | 76% |
Dicyclohexyl ketone Chemical Properties
Empirical Formula: C13H22O
Molecular Weight: 194.3132 g/mol
EINECS: 204-336-0
Index of Refraction: 1.49
Density: 0.97 g/cm3
Flash Point: 123.4 °C
Enthalpy of Vaporization: 50.43 kJ/mol
Boiling Point: 266.4 °C at 760 mmHg
Vapour Pressure: 0.00868 mmHg at 25 °C
Structure of Dicyclohexyl ketone (CAS NO.119-60-8):
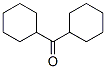
IUPAC Name of Dicyclohexyl ketone (CAS NO.119-60-8): Dicyclohexylmethanone
Canonical SMILES: C1CCC(CC1)C(=O)C2CCCCC2
InChI: InChI=1S/C13H22O/c14-13(11-7-3-1-4-8-11)12-9-5-2-6-10-12/h11-12H,1-10H2
InChIKey: TWXWPPKDQOWNSX-UHFFFAOYSA-N
Product Categories of Dicyclohexyl ketone (CAS NO.119-60-8): Carbonyl Compounds;Ketones
Dicyclohexyl ketone Toxicity Data With Reference
| 1. | ivn-mus LD50:56 mg/kg | CSLNX* U.S. Army Armament Research & Development Command, Chemical Systems Laboratory, NIOSH Exchange Chemicals. (Aberdeen Proving Ground, MD 21010) NX#06751 . |
Dicyclohexyl ketone Consensus Reports
Reported in EPA TSCA Inventory.
Dicyclohexyl ketone Safety Profile
Poison by intravenous route. See also KETONES. A flammable liquid. When heated to decomposition it emits acrid and irritating fumes.
Safety Statements: 23-24/25
S23:Do not breathe vapour.
S24/25:Avoid contact with skin and eyes.
Dicyclohexyl ketone Standards and Recommendations
DOT Classification: 3; Label: Flammable Liquid
Dicyclohexyl ketone Specification
Dicyclohexyl ketone ,its cas register number is 119-60-8. It also can be called Methanone, dicyclohexyl- ; Dicyclohexylmethanone ; Ketone, dicyclohexyl ;and Cyclohexyl ketone .
Related Products
- Dicyclohexyl adipate
- Dicyclohexyl fluorophosphonate
- Dicyclohexyl ketone
- Dicyclohexyl peroxydicarbonate
- Dicyclohexyl phthalate
- Dicyclohexyl(2,4,6-triisopropylphenyl)phosphine
- Dicyclohexyl-3,4,3',4'-tetracarboxylic dianhydride
- Dicyclohexylamine
- Dicyclohexylamine 3-oxo-1-phenyl-2,7,10-trioxa-4-azadodecan-12-oate
- DICYCLOHEXYLAMINE PENTANOATE
- 1196090-89-7
- 1196-09-4
- 1196109-52-0
- 1196145-01-3
- 1196151-13-9
- 1196151-78-6
- 1196152-34-7
- 1196153-86-2
- 1196155-35-7
- 1196155-81-3
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View