-
Name
Dimethyl 2-oxoglutarate
- EINECS
- CAS No. 13192-04-6
- Article Data23
- CAS DataBase
- Density 1.175 g/cm3
- Solubility
- Melting Point
- Formula C7H10O5
- Boiling Point 249.4 °C at 760 mmHg
- Molecular Weight 174.153
- Flash Point 105 °C
- Transport Information
- Appearance Slight Yellow Liquid
- Safety
- Risk Codes
-
Molecular Structure
- Hazard Symbols
- Synonyms Glutaricacid, 2-oxo-, dimethyl ester (6CI,7CI,8CI);Pentanedioic acid, 2-oxo-, dimethylester (9CI);2-Oxoglutaric acid dimethyl ester;Dimethyl 2-oxopentanedioate;Dimethyl a-ketoglutarate;Dimethyl a-oxoglutarate;a-Ketoglutaric acid dimethyl ester;
- PSA 69.67000
- LogP -0.31830
Synthetic route

| Conditions | Yield |
|---|---|
| With boron trifluoride at 65℃; for 0.333333h; | 100% |
| With toluene-4-sulfonic acid In chloroform Heating; | 94% |
| With sulfuric acid for 2h; Heating; | 90% |
-
-
328-50-7
α-ketoglutaric acid

-
-
13192-04-6
dimethyl 2-ketoglutarate

| Conditions | Yield |
|---|---|
| In methanol; benzene for 0.5h; Ambient temperature; | 85% |
-
-
67-56-1
methanol

-
-
328-50-7
α-ketoglutaric acid

-
A
-
13192-04-6
dimethyl 2-ketoglutarate

-
B
-
92684-01-0
2-Oxoglutarsaeure-1-methylester

| Conditions | Yield |
|---|---|
| With 2,2-dimethoxy-propane for 144h; Ambient temperature; | A 81% B 7% |

-
-
186581-53-3, 908094-01-9
diazomethane

-
-
328-50-7
α-ketoglutaric acid

-
-
13192-04-6
dimethyl 2-ketoglutarate

| Conditions | Yield |
|---|---|
| In diethyl ether at 0℃; for 4h; | 76% |

-
-
13192-04-6
dimethyl 2-ketoglutarate

| Conditions | Yield |
|---|---|
| With pyridine In tetrahydrofuran; water at 60℃; | 28% |

-
-
13192-04-6
dimethyl 2-ketoglutarate

| Conditions | Yield |
|---|---|
| With pyridine In tetrahydrofuran; water at 60℃; | 22% |
-
-
62435-72-7
2,5-dimethoxy-2,5-dihydro-furan-2-carboxylic acid methyl ester

-
A
-
13865-19-5
4-oxobutanoic acid methyl ester

-
B
-
13192-04-6
dimethyl 2-ketoglutarate

| Conditions | Yield |
|---|---|
| With hydrogenchloride at 90℃; Erwaermen des Reaktionsprodukts mit Schwefelsaeure enthaltendem Methanol.; |

-
-
13192-04-6
dimethyl 2-ketoglutarate

| Conditions | Yield |
|---|---|
| With hydrogenchloride |

-
-
13192-04-6
dimethyl 2-ketoglutarate

| Conditions | Yield |
|---|---|
| at 360℃; |

-
-
13192-04-6
dimethyl 2-ketoglutarate

| Conditions | Yield |
|---|---|
| at 360℃; |
-
-
41893-54-3
N-(1-Methoxy-5,5-dimethyl-3-oxocyclohex-1-yl)benzamid

-
A
-
13192-04-6
dimethyl 2-ketoglutarate

-
B
-
55-21-0
benzamide

| Conditions | Yield |
|---|---|
| With hydrogenchloride; ozone 1.) methylenechlorid, -60 deg C; 2.) methanol, room temp, 90 min.; Yield given. Multistep reaction. Yields of byproduct given; |
-
-
55590-96-0
1,3-dicarbomethoxy-1-(trimethylsilyloxy)propene

-
-
13192-04-6
dimethyl 2-ketoglutarate

| Conditions | Yield |
|---|---|
| With 1-methoxy-2-(2-nitrovinyl)benzene; tin(IV) chloride other reagents: boron trifluoride etherate/2-methoxy-β-nitrostyrene, benzaldehyde, benzaldehyde, benzaldehyde dimethyl acetal; |
-
-
7647-01-0
hydrogenchloride

-
-
62435-72-7
2,5-dimethoxy-2,5-dihydro-furan-2-carboxylic acid methyl ester

-
A
-
13865-19-5
4-oxobutanoic acid methyl ester

-
B
-
13192-04-6
dimethyl 2-ketoglutarate

| Conditions | Yield |
|---|---|
| Erwaermen des Reaktionsprodukts mit Methanol und wenig Schwefelsaeure; |

-
-
41893-53-2
N-(1-Methoxy-5,5-dimethyl-3-oxo-1-cyclohexen-1-yl)benzamid

-
-
13192-04-6
dimethyl 2-ketoglutarate

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 30 percent / H2 / Pd/C / methanol / 48 h 2: 1.) Ozon; 2.) HCl / 1.) methylenechlorid, -60 deg C; 2.) methanol, room temp, 90 min. View Scheme |
-
-
611-13-2
2-furoic acid methyl ester

-
-
13192-04-6
dimethyl 2-ketoglutarate

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: sulfuric acid / Electrolysis 2: aqueous hydrochloric acid , 35 percent / 90 °C / Erwaermen des Reaktionsprodukts mit Schwefelsaeure enthaltendem Methanol. View Scheme |
-
-
13192-04-6
dimethyl 2-ketoglutarate

-
-
148728-48-7
3-bromo-2-oxopentane-1,5-dioic acid dimethyl ester

| Conditions | Yield |
|---|---|
| With copper(ll) bromide In chloroform; ethyl acetate for 18h; Heating; | 100% |
| With copper(ll) bromide In chloroform; ethyl acetate at 20℃; Reflux; | 100% |
| With copper(ll) bromide In chloroform; ethyl acetate for 18h; Heating; | 90% |
-
-
13192-04-6
dimethyl 2-ketoglutarate

-
-
89532-94-5
methyl 6-oxo-1,4,5,6-tetrahydropyridazine-3-carboxylate

| Conditions | Yield |
|---|---|
| With acetic acid; hydrazine In methanol at 70℃; for 18h; | 100% |
| With hydrazine hydrate; acetic acid In methanol at 70℃; | 100% |
| With acetic acid; hydrazine In methanol for 5h; Reflux; Inert atmosphere; | 100% |
| With acetic acid; hydrazine In methanol for 12h; Heating; | 92% |
| With acetic acid; hydrazine In methanol Heating; | 92% |
-
-
61-54-1
tryptamine

-
-
13192-04-6
dimethyl 2-ketoglutarate

-
-
79888-13-4
methyl 2,3,5,6,11,11b-hexahydro-3-oxo-1H-indolizino<8,7-b>indole-11b-carboxylate

| Conditions | Yield |
|---|---|
| With acetic acid for 4h; Reflux; Inert atmosphere; | 97% |
| With acetic acid at 120℃; for 3h; Pictet-Spengler reaction; | 86% |
| In benzene for 1.5h; Heating; | 58% |
-
-
608-07-1
2-(5-methoxyindol-3-yl)ethylamine

-
-
13192-04-6
dimethyl 2-ketoglutarate

-
-
129968-05-4
methyl 8-methoxy-2,3,5,6,11,11b-hexahydro-3-oxo-1H-indolizino<8,7-b>indole-11b-carboxylate

| Conditions | Yield |
|---|---|
| With acetic acid for 4h; Reflux; Inert atmosphere; | 97% |
-
-
13192-04-6
dimethyl 2-ketoglutarate

-
-
106-96-7
propargyl bromide

-
-
179815-16-8
dimethyl 2-H-hydroxy-2-<2-(propynyl)>pentanedioate

| Conditions | Yield |
|---|---|
| With aluminium; mercury dichloride In tetrahydrofuran at -78℃; for 4h; | 95% |
| With aluminium; mercury dichloride 1.) 40 deg C, 2.) -78 deg C; Yield given. Multistep reaction; |
-
-
13192-04-6
dimethyl 2-ketoglutarate

-
-
95-54-5
1,2-diamino-benzene

-
-
21580-64-3
3-(3-oxo-3,4-dihydro-quinoxalin-2-yl)-propionic acid methyl ester

| Conditions | Yield |
|---|---|
| In methanol | 94% |
-
-
7677-24-9
trimethylsilyl cyanide

-
-
13192-04-6
dimethyl 2-ketoglutarate

-
-
96304-82-4
dimethyl 2-cyano-2-(trimethylsilyl)oxyglutarate

| Conditions | Yield |
|---|---|
| for 19h; Ambient temperature; | 93% |
-
-
13192-04-6
dimethyl 2-ketoglutarate

-
-
2446-83-5
diisopropyl (E)-azodicarboxylate

| Conditions | Yield |
|---|---|
| With triphenylphosphine In tetrahydrofuran at 20℃; | 93% |
-
-
13192-04-6
dimethyl 2-ketoglutarate

-
-
343-94-2
tryptamine hydochloride

-
-
79888-13-4
methyl 2,3,5,6,11,11b-hexahydro-3-oxo-1H-indolizino<8,7-b>indole-11b-carboxylate

| Conditions | Yield |
|---|---|
| In methanol for 20h; Heating; | 92% |
| In methanol Heating; | 92% |
-
-
13192-04-6
dimethyl 2-ketoglutarate

-
-
3699-66-9
ethyl 2-diethoxyphosphorylpropionate

-
-
496919-84-7
C12H18O6

| Conditions | Yield |
|---|---|
| With hydrogen; sodium hydride In tetrahydrofuran at 0℃; Inert atmosphere; | 91% |
| With sodium hydride | 71% |
| With sodium hydride Horner-Wadsworth-Emmons Olefination; | 71% |
-
-
17313-66-5
9-aminothioxanthene 10,10-dioxide

-
-
13192-04-6
dimethyl 2-ketoglutarate

| Conditions | Yield |
|---|---|
| With 4 A molecular sieve In toluene for 17h; Heating; | 86% |
-
-
13192-04-6
dimethyl 2-ketoglutarate

-
-
922-67-8
propynoic acid methyl ester

| Conditions | Yield |
|---|---|
| triethylamine In dichloromethane at 0℃; | 85% |
| With triethylamine In dichloromethane at 0℃; | 85% |
-
-
13192-04-6
dimethyl 2-ketoglutarate

-
-
67873-25-0
2-amino-6-(1-benzylhydrazinyl)pyrimidin-4(3H)-one

-
-
1374603-46-9
methyl 3-(7-amino-1-benzyl-4,5-dioxo-1,4,5,6-tetrahydropyrimido[4,5-c]pyridazin-3-yl)propanoate

| Conditions | Yield |
|---|---|
| In water for 3h; Reflux; | 85% |
-
-
13192-04-6
dimethyl 2-ketoglutarate

-
-
95-54-5
1,2-diamino-benzene

-
-
21580-64-3
methyl 3-(3-oxo-3,4-dihydroquinoxalin-2-yl)propanoate

| Conditions | Yield |
|---|---|
| In methanol for 16h; Inert atmosphere; | 84% |
| In ethanol Reflux; |
-
-
13192-04-6
dimethyl 2-ketoglutarate

-
-
38256-25-6
dimethyl (E)-2-oxoglutaconate

| Conditions | Yield |
|---|---|
| Stage #1: dimethyl 2-ketoglutarate With bromine In dichloromethane for 4.25h; Heating / reflux; Stage #2: With triethylamine In diethyl ether at 35℃; for 0.75h; | 83.1% |
| Stage #1: dimethyl 2-ketoglutarate With bromine In dichloromethane for 3h; Reflux; Inert atmosphere; Stage #2: With triethylamine In diethyl ether at 20℃; for 0.5h; | 70% |
| Stage #1: dimethyl 2-ketoglutarate With bromine In dichloromethane for 3h; Heating; Stage #2: With triethylamine In diethyl ether for 0.5h; Further stages.; | 3.6 mmol |

| Conditions | Yield |
|---|---|
| With sodium hydride In tetrahydrofuran for 0.5h; | 82% |

| Conditions | Yield |
|---|---|
| Stage #1: ethyl 2-diethoxyphosphorylpropionate With sodium hydride In tetrahydrofuran for 0.5h; Stage #2: dimethyl 2-ketoglutarate In tetrahydrofuran for 0.5h; | 82% |
-
-
64-17-5
ethanol

-
-
13192-04-6
dimethyl 2-ketoglutarate

-
-
627076-28-2
(2,4-difluorobenzyl)hydrazine

-
A
-
1297284-15-1
methyl 1-(2,4-difluorobenzyl)-6-oxo-1,4,5,6-tetrahydro-pyridazine-3-carboxylate

-
B
-
1297284-16-2
ethyl 1-(2,4-difluorobenzyl)-6-oxo-1,4,5,6-tetrahydropyridazine-3-carboxylate

| Conditions | Yield |
|---|---|
| hydrogenchloride Reflux; | A 82% B 6.1% |

-
-
13192-04-6
dimethyl 2-ketoglutarate

-
-
100-46-9
benzylamine

-
-
1397970-85-2
methyl 5-(benzylamino)-4,5-dioxopentanoate

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 20℃; for 6h; Inert atmosphere; | 82% |

| Conditions | Yield |
|---|---|
| Stage #1: ethyl 2-diethoxyphosphorylpropionate With sodium hydride In tetrahydrofuran; mineral oil Stage #2: dimethyl 2-ketoglutarate In tetrahydrofuran; mineral oil for 0.5h; | 82% |
-
-
13192-04-6
dimethyl 2-ketoglutarate

-
-
20731-72-0
6-methoxytryptamine-2-carboxylic acid

-
-
129968-06-5
methyl 9-methoxy-2,3,5,6,11,11b-hexahydro-3-oxo-1H-indolizino<8,7-b>indole-11b-carboxylate

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid In 1,4-dioxane; benzene Heating; | 80% |
| With trifluoroacetic acid In 1,4-dioxane; benzene for 24h; Heating; | 80% |
-
-
13192-04-6
dimethyl 2-ketoglutarate

-
-
3699-66-9
ethyl 2-diethoxyphosphorylpropionate

-
-
487-66-1
2-propionic-3-methylmaleic anhydride

| Conditions | Yield |
|---|---|
| Stage #1: dimethyl 2-ketoglutarate; ethyl 2-diethoxyphosphorylpropionate With sodium hydride In tetrahydrofuran for 0.5h; Stage #2: With water; potassium hydroxide In ethanol for 1h; Reflux; | 80% |
-
-
13192-04-6
dimethyl 2-ketoglutarate

-
-
20570-96-1
benzylhydrazine dihydrochloride

-
-
1297284-35-5
methyl 1-benzyl-6-oxo-1,4,5,6-tetrahydropyridazine-3-carboxylate

| Conditions | Yield |
|---|---|
| hydrogenchloride In ethanol for 15h; Inert atmosphere; Reflux; | 79% |
-
-
13192-04-6
dimethyl 2-ketoglutarate

-
-
81077-10-3
methyl 2-hydroxy-4-(metoxycarbonyl)butanoate

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate In methanol at 0 - 20℃; for 24h; Inert atmosphere; | 78% |
| With sodium tetrahydroborate In methanol; dichloromethane for 0.5h; Ambient temperature; | 74% |
| With sodium tetrahydroborate | 65% |
| With sodium tetrahydroborate In methanol |
-
-
13192-04-6
dimethyl 2-ketoglutarate

-
-
2446-83-5
di-isopropyl azodicarboxylate

| Conditions | Yield |
|---|---|
| With 9-phenyl-9-phosphafluorene; Bis(p-nitrophenyl) phosphate; phenylsilane; N-ethyl-N,N-diisopropylamine In toluene at 90℃; for 3.5h; Inert atmosphere; | 76% |
-
-
13192-04-6
dimethyl 2-ketoglutarate

-
-
1966-16-1
5-chloro-2-nitrophenylhydrazine

-
-
143263-49-4
dimethyl (E)-2-(5-chloro-2-nitrophenylhydrazono)glutarate

| Conditions | Yield |
|---|---|
| With hydrogenchloride In methanol at 20℃; for 1h; | 75% |
Dimethyl 2-oxoglutarate Specification
The Pentanedioic acid,2-oxo-,1,5-dimethyl ester with CAS registry number of 13192-04-6 belongs to the classes of Amino Acids 13C, 2H, 15N; Amino Acids & Derivatives; Miscellaneous Reagents; C6 to C7; Carbonyl Compounds; Esters. The IUPAC name is dimethyl 2-oxopentanedioate. In addition, the molecula formula is C7H10O5 and the molecular weight is 174.15. It is a kind of slight yellow liquid.
Physical properties about this chemical are: (1)ACD/LogP: -0.22; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): -0.21; (4)ACD/LogD (pH 7.4): -0.21; (5)ACD/BCF (pH 5.5): 1; (6)ACD/BCF (pH 7.4): 1; (7)ACD/KOC (pH 5.5): 18.21; (8)ACD/KOC (pH 7.4): 18.21; (9)#H bond acceptors: 5; (10)#H bond donors: 0; (11)#Freely Rotating Bonds: 6; (12)Polar Surface Area: 69.67 Å2; (13)Index of Refraction: 1.427; (14)Molar Refractivity: 38.05 cm3; (15)Molar Volume: 148.1 cm3; (16)Polarizability: 15.08 ×10-24cm3; (17)Surface Tension: 36.8 dyne/cm; (18)Density: 1.175 g/cm3; (19)Flash Point: 105 °C; (20)Enthalpy of Vaporization: 48.66 kJ/mol; (21)Boiling Point: 249.4 °C at 760 mmHg; (22)Vapour Pressure: 0.023 mmHg at 25°C.
Preparation of Pentanedioic acid,2-oxo-,1,5-dimethyl ester: this chemical can be prepared by 2-oxo-pentanedioic acid. This reaction will need solvents methanol and benzene. The reaction time is 30 minutes. The yield is about 85%.
.jpg)
Uses of Pentanedioic acid,2-oxo-,1,5-dimethyl ester: it can react with betrimethyl-silanecarbonitrile to get dimethyl 2-cyano-2-(trimethylsilyl)oxyglutarate. The reaction time is 19 hours at ambient temperature. The yield is about 93%.
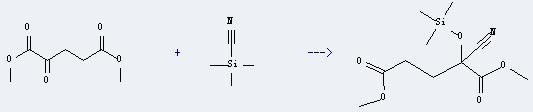
You can still convert the following datas into molecular structure:
(1)SMILES: O=C(C(=O)OC)CCC(=O)OC
(2)InChI: InChI=1/C7H10O5/c1-11-6(9)4-3-5(8)7(10)12-2/h3-4H2,1-2H3
(3)InChIKey: TXIXSLPEABAEHP-UHFFFAOYAN
Related Products
- Dimethyl ( )-2,3-O-Isopropylidene-D-tartrate
- Dimethyl ((1-methyl-5-nitro-1H-imidazol-2-yl)methylene)propanedioate
- Dimethyl (2-oxo-3,3-difluoroheptyl)phosphonate
- Dimethyl (2-oxo-4-phenylbutyl)phosphonate
- Dimethyl (2-oxoheptyl)phosphonate
- Dimethyl (2S, 2'S)-1, 1'-((2S, 2'S)-2, 2'-(4, 4'-(biphenyl-4, 4'-diyl)bis(1H-imidazole-4, 2-diyl))bis(pyrrolidine-2, 1-diyl))bis(3-methyl-1-oxobutane-2, 1-diyl)dicarbamate
- Dimethyl (3-phenoxy-2-oxopropyl)phosphonate
- Dimethyl (R)-(+)-methylsuccinate
- Dimethyl (S)-(-)-methylsuccinate
- Dimethyl (S)-3-hydroxy-L-aspartate
- 13192-12-6
- 131922-15-1
- 131924-52-2
- 131926-98-2
- 131926-99-3
- 131929-60-7
- 131941-25-8
- 13194-48-4
- 13194-60-0
- 1319-46-6
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View