-
Name
Divinyltetramethyldisiloxane
- EINECS 220-099-6
- CAS No. 2627-95-4
- Article Data18
- CAS DataBase
- Density 0.813 g/cm3
- Solubility insoluble in water
- Melting Point -99 °C(lit.)
- Formula C8H18OSi2
- Boiling Point 138.999 °C at 760 mmHg
- Molecular Weight 186.401
- Flash Point 33.325 °C
- Transport Information UN 1993 3/PG 2
- Appearance Colorless or yellowish transparent liquid
- Safety 16-26-36-37/39
- Risk Codes 11-36/37/38
-
Molecular Structure
-
Hazard Symbols
 F,
F, Xi
Xi
- Synonyms Disiloxane,1,1,3,3-tetramethyl-1,3-divinyl- (6CI,7CI,8CI);1,1,3,3-Tetramethyl-1,3-divinyldisiloxane;1,1'-Divinyltetramethyldisiloxane;1,3-Diethenyl-1,1,3,3-tetramethyldisiloxane;1,3-Divinyl-1,1,3,3-tetramethyldisiloxane;1,3-Divinyltetramethyldisiloxane;Bis(ethenyldimethylsilyl) ether;Tetramethyl-1,3-divinyldisiloxane;sym-Divinyltetramethyldisiloxane;sym-Tetramethyldivinyldisiloxane;Divinyltetramethyldisiloxane;
- PSA 9.23000
- LogP 2.86360
Synthetic route
-
-
18243-27-1
dimethylvinylsilane

-
-
2627-95-4
tetramethyldivinyldisiloxane

| Conditions | Yield |
|---|---|
| With sodium hydroxide at 68℃; for 4h; | 96% |
-
-
5356-83-2
ethoxydimethylvinylsilane

-
-
1351415-92-3
(2-methylallyl)-dimethylvinylsilane

-
-
2627-95-4
tetramethyldivinyldisiloxane

| Conditions | Yield |
|---|---|
| With water; scandium tris(trifluoromethanesulfonate) In acetonitrile at 20℃; for 1h; | 94% |
-
-
5356-83-2
ethoxydimethylvinylsilane

-
A
-
2627-95-4
tetramethyldivinyldisiloxane

-
B
-
79238-55-4
2-methyl-3-butene-2-ol, sodium salt

| Conditions | Yield |
|---|---|
| With water; sodium hydroxide at 20℃; | A 60% B 40% |
-
-
17877-23-5
triisopropylsilanol

-
-
1351415-92-3
(2-methylallyl)-dimethylvinylsilane

-
A
-
2627-95-4
tetramethyldivinyldisiloxane

-
B
-
1351415-84-3
1,1-dimethyl-3,3,3-triisopropyl-1-vinyldisiloxane

| Conditions | Yield |
|---|---|
| With n-heptan1ol; scandium tris(trifluoromethanesulfonate) In water; acetonitrile at 22℃; for 0.5h; | A 19% B 58% |


| Conditions | Yield |
|---|---|
| With hydrogenchloride |

| Conditions | Yield |
|---|---|
| With hydrogenchloride |

| Conditions | Yield |
|---|---|
| With lithium oxide In diethyl ether Yield given; | |
| With water |
-
-
857899-86-6
(1-hydrohexafluoroisobutenyloxy)trimethylsilane

-
-
1719-58-0
Chlorodimethylvinylsilane

-
B
-
1438-79-5
1,1,3,3,3-pentamethyl-1-vinyldisiloxane

-
C
-
2627-95-4
tetramethyldivinyldisiloxane

-
D
-
38755-76-9
dimethyl vinyl fluorosilane

| Conditions | Yield |
|---|---|
| Stage #1: (1-hydrohexafluoroisobutenyloxy)trimethylsilane With cesium fluoride In diethylene glycol dimethyl ether at -10℃; for 2h; Stage #2: Chlorodimethylvinylsilane In diethylene glycol dimethyl ether at -10℃; for 2h; Further byproducts given; |

-
-
206356-44-7
CF3C(O)CH(OSiMe2(C2H3))CF3

-
-
109-81-9
N-methyl-ethane-1,2-diamine

-
A
-
2627-95-4
tetramethyldivinyldisiloxane

| Conditions | Yield |
|---|---|
| for 2h; |


| Conditions | Yield |
|---|---|
| With [D]-sodium hydroxide; water-d2; palladium diacetate; sodium 4-iodo-benzoate at 30℃; for 20h; | 100 %Spectr. |

-
A
-
2627-95-4
tetramethyldivinyldisiloxane

-
B
-
1315461-59-6
1,3-bis(2,6-diisopropylphenyl)-2-methylene-2,3-dihydro-1H-imidazole

| Conditions | Yield |
|---|---|
| In benzene-d6 at 80℃; |

| Conditions | Yield |
|---|---|
| With dichlorotricarbonylruthenium(II) dimer; N,N-dimethyl-aniline In 1,4-dioxane at 50℃; under 3750.38 Torr; for 24h; Temperature; Pressure; Solvent; |
-
-
75-77-4
chloro-trimethyl-silane

-
-
1719-58-0
Chlorodimethylvinylsilane

-
A
-
107-46-0
Hexamethyldisiloxane

-
B
-
1438-79-5
1,1,3,3,3-pentamethyl-1-vinyldisiloxane

-
C
-
2627-95-4
tetramethyldivinyldisiloxane

| Conditions | Yield |
|---|---|
| With water at 0 - 10℃; for 2h; | |
| In water at 0 - 20℃; | A 22 %Chromat. B 49 %Chromat. C 28 %Chromat. |

-
-
1066-35-9
dimethylmonochlorosilane

-
-
1719-58-0
Chlorodimethylvinylsilane

-
A
-
3277-26-7
1,1,3,3-Tetramethyldisiloxane

-
B
-
2627-95-4
tetramethyldivinyldisiloxane

-
C
-
55967-52-7
1,1,3,3-tetramethyl-1-vinyldisiloxane

| Conditions | Yield |
|---|---|
| In water at 0 - 20℃; | A 11 %Chromat. B 26 %Chromat. C 62 %Chromat. |

-
-
31989-57-8
bis(triphenylphosphine)palladium(0)

-
-
2627-95-4
tetramethyldivinyldisiloxane

-
-
252008-09-6
[Pd((η(2)-CH2CHSiMe2)2O)P(C6H5)3]

| Conditions | Yield |
|---|---|
| at 25℃; for 0.166667h; Inert atmosphere; | 100% |
-
-
227025-38-9, 242806-77-5
[(C5Me5)Rh(vinyltrimethylsilane)2]

-
-
2627-95-4
tetramethyldivinyldisiloxane

-
-
503091-93-8, 174735-70-7, 174847-75-7
(η(5)-C5Me5)Rh[(η(4)-(CH2=CHSiMe2)2O)]

| Conditions | Yield |
|---|---|
| In not given Rh complex was reacted with ligand; | 99% |
-
-
2627-95-4
tetramethyldivinyldisiloxane

-
-
576-83-0
2,4,6-trimethylphenyl bromide

-
-
769-25-5
2,4,6-trimethylstyrene

| Conditions | Yield |
|---|---|
| With johnphos; bis(η3-allyl-μ-chloropalladium(II)) In N,N-dimethyl-formamide at 40℃; for 24h; | 99% |

| Conditions | Yield |
|---|---|
| In neat (no solvent) CuBr dissolved in divinyltetramethyldisiloxane; crystd. for 1 d, elem. anal.; | 99% |

| Conditions | Yield |
|---|---|
| In neat (no solvent) CuCl dissolved in divinyltetramethyldisiloxane; crystd. for 1 d, elem. anal.; | 99% |

-
-
2627-95-4
tetramethyldivinyldisiloxane

-
-
91608-15-0
tris(2,4,6-trimethoxyphenyl)phosphine

-
-
1222557-48-3
Pd(P(C6H2(OCH3)3)3)(CH2CHSi(CH3)2OSi(CH3)2CHCH2)

| Conditions | Yield |
|---|---|
| In neat (no solvent) (Ar, Schlenk) a suspn. of Rh-complex and phosphine in degassed and driedsiloxane-compound was stirred at room temp. overnight; the solvent was removed under vac., the solid was washed with a small portion of diethyl ether or pentane at -50°C; | 99% |

-
-
189282-65-3
[(cyclopentadienyl)Co(trimethylvinylsilane)2]

-
-
2627-95-4
tetramethyldivinyldisiloxane

-
-
1246272-89-8
[(cyclopentadienyl)Co(1,3-divinyl-1,1,3,3-tetramethyldisiloxane)]

| Conditions | Yield |
|---|---|
| In diethyl ether byproducts: (CH3)3SiCHCH2; (Ar), Schlenk techniques; dropwise addn. of O compd. to soln. of Co complex in Et2O at -30°C over 2 min, stirring for 5 min; evapn. under reduced pressure at -20°C, drying under vacuum; | 99% |

| Conditions | Yield |
|---|---|
| With C38H28Au2F12FeN2O8P2S4 In 1,4-dioxane at 45℃; for 20h; Inert atmosphere; Glovebox; Sealed tube; | 99% |
-
-
1000598-40-2
4-[(dimethyl(vinyl)silyl)]morpholine

-
-
2627-95-4
tetramethyldivinyldisiloxane

| Conditions | Yield |
|---|---|
| Stage #1: tetramethyldivinyldisiloxane With phosphoric acid at 105 - 115℃; for 3h; Reflux; Large scale; Stage #2: 4-[(dimethyl(vinyl)silyl)]morpholine at -10℃; Reflux; Large scale; | 97.11% |

| Conditions | Yield |
|---|---|
| With (TFAPDI)Co(2-ethylhexanoate)2 In neat (no solvent) at 23℃; for 1h; | 97% |
-
-
75-54-7
Dichloromethylsilane

-
-
2627-95-4
tetramethyldivinyldisiloxane

-
-
2622-05-1
allylmagnesium bromide

| Conditions | Yield |
|---|---|
| Stage #1: Dichloromethylsilane; tetramethyldivinyldisiloxane With platinum In toluene at 60℃; Inert atmosphere; Stage #2: allylmagnesium bromide In tetrahydrofuran at 60℃; for 1.16667h; Inert atmosphere; Cooling with ice; | 97% |

-
-
107-46-0
Hexamethyldisiloxane

-
-
2627-95-4
tetramethyldivinyldisiloxane

-
-
1438-79-5
1,1,3,3,3-pentamethyl-1-vinyldisiloxane

| Conditions | Yield |
|---|---|
| With 18-crown-6 ether; potassium hydroxide at 80℃; for 1h; | 96.2% |
| With Lewatit SPC 118 for 5h; Ambient temperature; | 17 g |
-
-
1295-35-8
bis(1,5-cyclooctadiene)nickel (0)

-
-
2627-95-4
tetramethyldivinyldisiloxane

-
-
189372-83-6
[Ni2(1,1,3,3-tetramethyl-1,3-divinyldisiloxane)3]

| Conditions | Yield |
|---|---|
| In diethyl ether byproducts: Ni, cyclooctadiene; overnight at ambient temp.; volatiles removed (vac.), taken up into pentane, filtered (Celite), concd.; | 95% |
| In diethyl ether at 20℃; for 24h; Schlenk technique; Inert atmosphere; | 50% |
-
-
2627-95-4
tetramethyldivinyldisiloxane

-
-
35948-25-5
9,10-dihydro-9-oxa-10-phosphaphenanthrene-10-oxide

| Conditions | Yield |
|---|---|
| Stage #1: 9,10-dihydro-9-oxa-10-phosphaphenanthrene-10-oxide With platinum(0)-1,3-divinyl-1,1,3,3-tetramethyldisiloxane complex In 1,4-dioxane at 80℃; for 0.5h; Autoclave; Inert atmosphere; High pressure; Stage #2: tetramethyldivinyldisiloxane In 1,4-dioxane; cyclohexane at 80℃; for 20h; Reagent/catalyst; Solvent; Temperature; High pressure; | 95% |
-
-
2627-95-4
tetramethyldivinyldisiloxane

| Conditions | Yield |
|---|---|
| With platinum(0)-1,3-divinyl-1,1,3,3-tetramethyldisiloxane complex; trichlorosilane In toluene at 20℃; for 18h; Inert atmosphere; Schlenk technique; | 95% |
-
-
2627-95-4
tetramethyldivinyldisiloxane

-
-
947-42-2
diphenylsilanediol

-
-
18586-22-6
1,5-divinyl-3,3-diphenyl-1,1,5,5-tetramethyltrisiloxane

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In hexane at 70 - 80℃; for 2h; Reagent/catalyst; Inert atmosphere; Green chemistry; | 94.6% |
-
-
1112-39-6
dimethyldimethoxysilan

-
-
2627-95-4
tetramethyldivinyldisiloxane

-
-
17980-39-1
1,1,3,3,5,5-hexamethyl-1,5-divinyltrisiloxane

| Conditions | Yield |
|---|---|
| With iron(III) chloride at 70 - 80℃; for 4h; Temperature; Reagent/catalyst; | 94.2% |
| With trifluorormethanesulfonic acid; acetic anhydride; acetic acid at 50℃; for 2.5h; | 51.7% |

| Conditions | Yield |
|---|---|
| Stage #1: Dichloromethylsilane; tetramethyldivinyldisiloxane With dihydrogen hexachloroplatinate In isopropyl alcohol for 0.833333h; Inert atmosphere; Stage #2: acetic anhydride at 100℃; for 12h; Inert atmosphere; | 94% |


| Conditions | Yield |
|---|---|
| Stage #1: tetramethyldivinyldisiloxane With dihydrogen hexachloroplatinate; trichlorosilane In isopropyl alcohol for 0.833333h; Inert atmosphere; Stage #2: acetic anhydride at 100℃; for 12h; Inert atmosphere; | 94% |
-
-
2627-95-4
tetramethyldivinyldisiloxane

-
-
90-90-4
(4-bromophenyl)(phenyl)methanone

-
-
3139-85-3
4-vinylbenzophenone

| Conditions | Yield |
|---|---|
| With potassium trimethylsilonate; Triphenylphosphine oxide; palladium tris(dibenzylideneacetone) In tetrahydrofuran for 3h; Heating; | 93% |
| With tris-(dibenzylideneacetone)dipalladium(0); potassium trimethylsilonate; Triphenylphosphine oxide In tetrahydrofuran at 66℃; for 5h; Inert atmosphere; | 90% |
-
-
1073-67-2
4-vinylbenzyl chloride

-
-
2627-95-4
tetramethyldivinyldisiloxane

-
-
1123168-01-3
1,3-bis[(E)-4-chlorostyryl]tetramethyldisiloxane

| Conditions | Yield |
|---|---|
| Stage #1: 4-vinylbenzyl chloride; tetramethyldivinyldisiloxane With (carbonyl)(chloro)(hydrido)tris(triphenylphosphine)ruthenium(II) In 1,4-dioxane at 100℃; for 0.0833333h; Inert atmosphere; Stage #2: With copper(l) chloride In 1,4-dioxane at 100℃; for 16h; Inert atmosphere; stereoselective reaction; | 93% |
| With (carbonyl)(chloro)(hydrido)tris(triphenylphosphine)ruthenium(II); copper(l) chloride In 1,4-dioxane at 100℃; for 16h; |

| Conditions | Yield |
|---|---|
| With tetrabutylammomium bromide; potassium acetate; palladium diacetate; tris-(o-tolyl)phosphine In water; N,N-dimethyl-formamide at 100℃; for 48h; Inert atmosphere; | 93% |

| Conditions | Yield |
|---|---|
| With 2,2-dimethoxy-2-phenylacetophenone In tetrahydrofuran at 20℃; for 2h; UV-irradiation; | 93% |
-
-
2627-95-4
tetramethyldivinyldisiloxane

| Conditions | Yield |
|---|---|
| With tetrabutylammomium bromide; potassium acetate; palladium diacetate; tris-(o-tolyl)phosphine In water; N,N-dimethyl-formamide at 100℃; for 48h; Inert atmosphere; | 92% |
-
-
1003-09-4
2-bromothiophene

-
-
2627-95-4
tetramethyldivinyldisiloxane

-
-
13640-78-3, 15332-30-6, 18266-94-9, 34512-02-2, 109958-97-6
1,2-di-[2]thienyl-ethylene

| Conditions | Yield |
|---|---|
| With tetrabutylammomium bromide; potassium acetate; palladium diacetate; tris-(o-tolyl)phosphine In water; N,N-dimethyl-formamide at 100℃; for 48h; Inert atmosphere; | 92% |

| Conditions | Yield |
|---|---|
| With potassium trimethylsilonate; Triphenylphosphine oxide; palladium tris(dibenzylideneacetone) In tetrahydrofuran for 3h; Heating; | 91% |
-
-
2039-82-9
1-bromo-4-ethenyl-benzene

-
-
2627-95-4
tetramethyldivinyldisiloxane

-
-
1123168-00-2
1,3-bis[(E)-4-bromostyryl]tetramethyldisiloxane

| Conditions | Yield |
|---|---|
| Stage #1: 1-bromo-4-ethenyl-benzene; tetramethyldivinyldisiloxane With (carbonyl)(chloro)(hydrido)tris(triphenylphosphine)ruthenium(II) In 1,4-dioxane at 100℃; for 0.0833333h; Inert atmosphere; Stage #2: With copper(l) chloride In 1,4-dioxane at 100℃; for 16h; Inert atmosphere; stereoselective reaction; | 91% |
| With (carbonyl)(chloro)(hydrido)tris(triphenylphosphine)ruthenium(II); copper(l) chloride In 1,4-dioxane at 100℃; for 16h; |
-
-
628-71-7
1-Heptyne

-
-
2627-95-4
tetramethyldivinyldisiloxane

-
A
-
1267749-58-5
1-(1-heptynyl)-1,1,3,3-tetramethyl-3-vinyldisiloxane

-
B
-
1267749-59-6
C18H34OSi2

| Conditions | Yield |
|---|---|
| With carbonylchlorohydridobis(tricyclohexylphosphine)ruthenium(II) In toluene at 120℃; for 24h; Inert atmosphere; | A 91% B 8 %Chromat. |

Divinyltetramethyldisiloxane Specification
The Divinyltetramethyldisiloxane, with the CAS registry number 2627-95-4, is also known as Tetramethyl-1,3-divinyldisiloxane. It belongs to the product categories of Industrial/Fine Chemicals; Monomer; Si (Classes of Silicon Compounds); Siloxanes; Si-O Compounds; Vinylsilanes, Allylsilanes; Organosilicon compound. Its EINECS number is 220-099-6. This chemical's molecular formula is C8H18OSi2 and molecular weight is 186.40. What's more, its systematic name is 1,1,3,3-Tetramethyl-1,3-divinyldisiloxane. This chemical should be sealed and stored in a cool and dry place. This substance is used as raw materials for preparing vinyl polysiloxane, silica gel and vinyl silicone oil.
Physical properties of Divinyltetramethyldisiloxane are: (1)ACD/LogP: 3.053; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 3.05; (4)ACD/LogD (pH 7.4): 3.05; (5)ACD/BCF (pH 5.5): 123.08; (6)ACD/BCF (pH 7.4): 123.08; (7)ACD/KOC (pH 5.5): 1090.87; (8)ACD/KOC (pH 7.4): 1090.87; (9)#H bond acceptors: 1; (10)#H bond donors: 0; (11)#Freely Rotating Bonds: 4; (12)Polar Surface Area: 9.23 Å2; (13)Index of Refraction: 1.419; (14)Molar Refractivity: 57.742 cm3; (15)Molar Volume: 228.612 cm3; (16)Polarizability: 22.891×10-24cm3; (17)Surface Tension: 17.3 dyne/cm; (18)Density: 0.815 g/cm3; (19)Flash Point: 33.325 °C; (20)Enthalpy of Vaporization: 36.075 kJ/mol; (21)Boiling Point: 138.999 °C at 760 mmHg; (22)Vapour Pressure: 8.2 mmHg at 25°C.
Preparation: this chemical can be prepared by dimethyl-vinyl-silane at the temperature of 68 °C. This reaction will need reagent 20% aq. NaOH with the reaction time of 4 hours. The yield is about 96%.

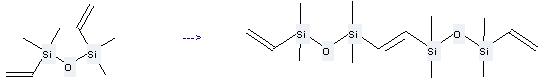
Uses of Divinyltetramethyldisiloxane: it can be used to produce 1,2-bis-(1,1,3,3-tetramethyl-3-vinyl-disiloxanyl)-ethene at the temperature of 80 °C. It will need catalyst RuCl2(CO)3 with the reaction time of 48 hours. The yield is about 49%.
When you are using this chemical, please be cautious about it as the following:
This chemical is highly flammable, so you should keep it away from sources of ignition - No smoking. It is irritating to eyes, respiratory system and skin. In case of contact with eyes, you should rinse immediately with plenty of water and seek medical advice. When using it, you need wear suitable protective clothing, gloves and eye/face protection.
You can still convert the following datas into molecular structure:
(1)SMILES: C[Si](C)(C=C)O[Si](C)(C)C=C
(2)Std. InChI: InChI=1S/C8H18OSi2/c1-7-10(3,4)9-11(5,6)8-2/h7-8H,1-2H2,3-6H3
(3)Std. InChIKey: BITPLIXHRASDQB-UHFFFAOYSA-N
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| rat | LCLo | inhalation | 111gm/m3/1H (111000mg/m3) | BEHAVIORAL: SOMNOLENCE (GENERAL DEPRESSED ACTIVITY) SENSE ORGANS AND SPECIAL SENSES: OTHER CHANGES: OLFACTION BEHAVIORAL: CONVULSIONS OR EFFECT ON SEIZURE THRESHOLD | National Technical Information Service. Vol. OTS0572673, |
| rat | LD50 | oral | > 10gm/kg (10000mg/kg) | BEHAVIORAL: SOMNOLENCE (GENERAL DEPRESSED ACTIVITY) SENSE ORGANS AND SPECIAL SENSES: OTHER CHANGES: OLFACTION SKIN AND APPENDAGES (SKIN): HAIR: OTHER | National Technical Information Service. Vol. OTS0572676, |
Related Products
- Divinyltetramethyldisiloxane
- 26279-64-1
- 2628-16-2
- 2628-17-3
- 262849-64-9
- 262849-65-0
- 262849-66-1
- 262856-01-9
- 26286-54-4
- 26286-55-5
- 26287-62-7
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View