-
Name
CHLOROETHANE
- EINECS 200-830-5
- CAS No. 75-00-3
- Article Data749
- CAS DataBase
- Density 0.884 g/cm3
- Solubility 6710 mg/L at 25℃
- Melting Point -139 °C(lit.)
- Formula C2H5Cl
- Boiling Point 12.7 °C at 760 mmHg
- Molecular Weight 64.5147
- Flash Point <-30 °F
- Transport Information UN 1993 3/PG 2
- Appearance colourless gas
- Safety 9-16-33-36/37-61-45-7-29-36/37/39-26-53
- Risk Codes 45-11-20/21/22-36/37/38-52/53-40-12-39/23/24/25-23/24/25-67-66-22-19
-
Molecular Structure
-
Hazard Symbols
 F+,
F+, Xn,
Xn, T,
T, F
F
- Synonyms Aethylis;Aethylis chloridum;Anodynon;Chlorethyl;Chloridum;Chloroethane;Chloryl;Chloryl Anesthetic;Chloryle anesthetic;Cloretilo;Dublofix;Ether chloratus;Ether hydrochloric;Ether muriatic;Hydrochloric ether;Kelene;Monochloroethane;Muriaticether;Narcotile;R 160;
- PSA 0.00000
- LogP 1.24510
Synthetic route
-
-
87463-00-1
O-ethyl N-methyl-N-phenylthiocarbamate

-
A
-
75-00-3
chloroethane

-
B
-
100244-53-9
(N-methyl-N-phenylcarbamoyl)sulfenyl chloride

| Conditions | Yield |
|---|---|
| With sulfuryl dichloride In chloroform-d1 at 5℃; for 0.166667h; | A 100% B n/a |
| With sulfuryl dichloride In chloroform-d1 at 25℃; |
-
-
945614-32-4
triethyloxonium tris(pentafluoroethyl)trifluorophosphate

-
-
65039-09-0
1-ethyl-3-methyl-1H-imidazol-3-ium chloride

-
A
-
377739-43-0
1-ethyl-3-methyl-imidazolium tris(pentafluoroethyl)trifluorophosphate

-
B
-
60-29-7
diethyl ether

-
C
-
75-00-3
chloroethane

| Conditions | Yield |
|---|---|
| at 80℃; for 3h; Product distribution / selectivity; | A 98.9% B n/a C n/a |

-
-
945614-32-4
triethyloxonium tris(pentafluoroethyl)trifluorophosphate

-
-
30388-20-6
N,N,N',N',N'',N''-hexamethylguanidinium chloride

-
B
-
60-29-7
diethyl ether

-
C
-
75-00-3
chloroethane

| Conditions | Yield |
|---|---|
| at 80℃; for 3h; | A 98.9% B n/a C n/a |


| Conditions | Yield |
|---|---|
| With CoCl2 at 120℃; Product distribution; the thermal stability of the product was investigated by DTA, DTG and TG; | A n/a B 98% |
-
-
945614-34-6
triethyloxonium bis(trifluoromethylsulfonyl)imide

-
-
65039-09-0
1-ethyl-3-methyl-1H-imidazol-3-ium chloride

-
A
-
60-29-7
diethyl ether

-
B
-
75-00-3
chloroethane

-
C
-
174899-82-2
1-ethyl-3-methylimidazolium bis(trifluoromethylsulfonyl)imide

| Conditions | Yield |
|---|---|
| at 80℃; for 3h; | A n/a B n/a C 97.9% |


| Conditions | Yield |
|---|---|
| at 30 - 40℃; for 1h; | A 97% B 87% |


| Conditions | Yield |
|---|---|
| With hydrogenchloride; aluminum (III) chloride; zinc(II) chloride at 70 - 120℃; under 900.09 - 2625.26 Torr; for 2h; Large scale; | 96.3% |
| With hydrogenchloride; zinc(II) chloride at 8 - 130℃; | 65% |
| With hydrogenchloride |
-
-
50966-09-1
diethyl (1-benzamido-2,2-dichlorovinyl)phosphonate

-
A
-
75-00-3
chloroethane

-
B
-
77113-14-5
azlactone of (1-benzamido-2,2-dichlorovinyl)phosphonochloridic acid

| Conditions | Yield |
|---|---|
| With phosphorus pentachloride at 110 - 120℃; for 5.5h; | A n/a B 96% |

-
A
-
75-00-3
chloroethane

| Conditions | Yield |
|---|---|
| iron(III) chloride In neat (no solvent) heating (120 - 130°C), addn. of further FeCl3, heating (180°C); reprecipitation (toluene); elem. anal.; | A 96% B 94.7% |
-
-
945614-32-4
triethyloxonium tris(pentafluoroethyl)trifluorophosphate

-
A
-
60-29-7
diethyl ether

-
B
-
75-00-3
chloroethane

-
C
-
916807-26-6
1-decyl-3-methylimidazolium tris(pentafluoroethyl)trifluorophosphate

| Conditions | Yield |
|---|---|
| at 80℃; for 3h; | A n/a B n/a C 96% |

-
-
78-38-6
ethylphosphonic acid diethyl ester

-
A
-
75-00-3
chloroethane

-
B
-
1066-50-8
Ethylphosphonic dichloride

| Conditions | Yield |
|---|---|
| With thionyl chloride; N,N-dimethyl-formamide for 18h; Heating; | A n/a B 95.8% |
-
-
589-57-1
diethyl phosphorylchloridite

-
-
17640-25-4
methyl 2,2-dichloro-2-methoxyacetate

-
A
-
5781-53-3
monomethyl oxalyl chloride

-
B
-
553-90-2
Dimethyl oxalate

-
C
-
75-00-3
chloroethane

-
D
-
66298-75-7
phosphonic dichloride

-
E
-
74813-29-9
methyl phosphonochloridate

-
F
-
74813-30-2
ethyl phosphonochloridate

| Conditions | Yield |
|---|---|
| With iron(III) chloride at 105 - 110℃; for 1.16667h; Product distribution; | A n/a B n/a C 95% D n/a E n/a F n/a |

-
-
589-57-1
diethyl phosphorylchloridite

-
-
17640-25-4
methyl 2,2-dichloro-2-methoxyacetate

-
A
-
553-90-2
Dimethyl oxalate

-
B
-
1498-42-6
phosphorodichloridous acid ethyl ester

-
C
-
75-00-3
chloroethane

-
D
-
66298-75-7
phosphonic dichloride

-
E
-
74813-30-2
ethyl phosphonochloridate

| Conditions | Yield |
|---|---|
| With iron(III) chloride at 105 - 110℃; for 0.833333h; Product distribution; | A n/a B n/a C 95% D n/a E n/a F n/a |

-
-
762-04-9
phosphonic acid diethyl ester

-
-
80-10-4
diphenylsilyl dichloride

-
A
-
75-00-3
chloroethane

-
B
-
90285-15-7
C16H22O6P2Si

| Conditions | Yield |
|---|---|
| at 102℃; | A n/a B 94.1% |

-
-
64-17-5
ethanol

-
-
7719-12-2, 52843-90-0
phosphorus trichloride

-
A
-
75-00-3
chloroethane

-
B
-
762-04-9
phosphonic acid diethyl ester

| Conditions | Yield |
|---|---|
| at 85℃; under 30.003 Torr; Addition of ca.3% diethyl phosphite (triethyl phosphite or ethyl chloride are also possible); | A n/a B 94% |

-
-
945614-32-4
triethyloxonium tris(pentafluoroethyl)trifluorophosphate

-
-
76-83-5
trityl chloride

-
A
-
60-29-7
diethyl ether

-
B
-
75-00-3
chloroethane

| Conditions | Yield |
|---|---|
| at 80℃; for 10h; | A n/a B n/a C 93.6% |


| Conditions | Yield |
|---|---|
| With calcium chloride at 70℃; for 1h; Product distribution; other time; also in the presence of pyridine; the thermal stability of the product was investigated by DTA, DTG and TG; | A n/a B 93.4% |
| With calcium chloride at 70℃; for 1h; | A n/a B 93.4% |

| Conditions | Yield |
|---|---|
| With manganese(II) acetate at 112℃; for 1h; Product distribution; oter time; the thermal stability of the product was investigated by DTA, DTG and TG; | A n/a B 93% |
-
-
121-44-8
triethylamine

-
-
1005-56-7
phenylcarbonochloridothioate

-
A
-
75-00-3
chloroethane

-
B
-
24486-06-4
O-phenyl-N,N-diethyl thiocarbamate

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; for 1h; Substitution; | A n/a B 93% |


-
-
945614-32-4
triethyloxonium tris(pentafluoroethyl)trifluorophosphate

-
A
-
60-29-7
diethyl ether

-
B
-
75-00-3
chloroethane

-
C
-
945614-40-4
1-hexyl-1-methylpyrrolidinium tris(pentafluoroethyl)trifluorophosphate

| Conditions | Yield |
|---|---|
| at 80℃; for 3h; | A n/a B n/a C 93% |


| Conditions | Yield |
|---|---|
| With thionyl chloride In tetrachloromethane for 5h; Heating; | A 92% B n/a |

-
A
-
75-00-3
chloroethane

| Conditions | Yield |
|---|---|
| With phosphorus pentachloride at 110 - 120℃; for 5.5h; | A n/a B 92% |
-
-
139535-54-9
N-methyl-N-(2,2-dimethyl-3-chloro-3-ethoxypropyl)methanesulfonamide

-
A
-
75-00-3
chloroethane

-
B
-
139535-59-4
N-methyl-N-(2,2-dimethyl-3-oxopropyl)methanesulfonamide

| Conditions | Yield |
|---|---|
| under 9 Torr; Heating; | A 91% B 79% |
-
-
589-57-1
diethyl phosphorylchloridite

-
-
17640-25-4
methyl 2,2-dichloro-2-methoxyacetate

-
A
-
75-00-3
chloroethane

-
B
-
74813-30-2
ethyl phosphonochloridate

| Conditions | Yield |
|---|---|
| With iron(III) chloride at 95 - 110℃; for 1.5h; | A 90% B 38% |

-
-
589-57-1
diethyl phosphorylchloridite

-
-
6957-89-7
Ethyl dichloro-(ethoxy)-acetate

-
A
-
75-00-3
chloroethane

-
B
-
67538-60-7
ethyl phosphonochloridate

-
C
-
814-49-3
diethyl chlorophosphate

-
D
-
5284-10-6
ethyl ethylphosphonochloridate

| Conditions | Yield |
|---|---|
| With iron(III) chloride at 95 - 110℃; for 1.5h; | A 90% B 58% C n/a D n/a |

-
-
371-22-2
fluorophosphoric acid diethyl ester

-
-
17640-25-4
methyl 2,2-dichloro-2-methoxyacetate

-
A
-
75-00-3
chloroethane

-
B
-
67538-58-3
ethyl phosphonofluoridate

| Conditions | Yield |
|---|---|
| With iron(III) chloride at 95 - 110℃; for 1.5h; | A 90% B 45% |

-
-
371-22-2
fluorophosphoric acid diethyl ester

-
-
6957-89-7
Ethyl dichloro-(ethoxy)-acetate

-
A
-
75-00-3
chloroethane

-
B
-
67538-59-4
ethyl phosphonofluoridate

| Conditions | Yield |
|---|---|
| With iron(III) chloride at 95 - 110℃; for 1.5h; | A 90% B 50% |


-
A
-
75-00-3
chloroethane

| Conditions | Yield |
|---|---|
| With phosphorus pentachloride at 110 - 120℃; for 5.5h; | A n/a B 90% |
-
-
94703-39-6
2,3,3,3-tetrachloro-2-<amino>propionitrile

-
-
122-52-1
triethyl phosphite

-
A
-
75-00-3
chloroethane

-
B
-
814-49-3
diethyl chlorophosphate

-
C
-
94703-45-4
3,3-dichloro-2-<amino>acrylonitrile

| Conditions | Yield |
|---|---|
| In benzene for 1h; Heating; | A n/a B 90% C 25% |

-
-
64317-34-6
N-(pentafluorophenyl)carbonimidoyl dichloride

-
-
121-44-8
triethylamine

-
A
-
75-00-3
chloroethane

-
B
-
120672-75-5
N,N-diethyl-N-pentafluorophenylchloroformamidine

| Conditions | Yield |
|---|---|
| In diethyl ether at 20℃; for 6h; | A n/a B 90% |

-
-
616-47-7
1-methyl-1H-imidazole

-
-
75-00-3
chloroethane

-
-
65039-09-0
1-ethyl-3-methyl-1H-imidazol-3-ium chloride

| Conditions | Yield |
|---|---|
| In ethanol at 35℃; for 10h; Wavelength; UV-irradiation; | 100% |
| 79% | |
| In acetonitrile at 70℃; for 168h; | 71% |

| Conditions | Yield |
|---|---|
| With magnesium In diethyl ether; toluene at 50℃; Inert atmosphere; Flow reactor; | 99.4% |
-
-
75-00-3
chloroethane

-
-
62806-88-6
N,N-dipropylcarbamothioate sodium salt

-
-
759-94-4
S-ethyl N,N-di-n-propylthiocarbamate

| Conditions | Yield |
|---|---|
| In water at 80℃; for 0.25h; in ultrasonic field; | 99.1% |
-
-
75-00-3
chloroethane

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 0℃; for 3h; | 99% |

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate In ethanol at 30 - 40℃; for 4h; | 98.56% |

| Conditions | Yield |
|---|---|
| In water at 70℃; for 0.333333h; in ultrasonic field; | 98.4% |

| Conditions | Yield |
|---|---|
| With ammonium hydroxide In water at 90 - 98℃; under 7500.75 Torr; for 18h; pH=5 - 7; Temperature; Large scale; | 98% |
-
-
75-00-3
chloroethane

-
-
21055-93-6
sodium salt of diethylthiocarbamic acid

-
-
2941-55-1
S-ethyl N,N-diethylthiocarbamate

| Conditions | Yield |
|---|---|
| In water at 80℃; for 0.166667h; in ultrasonic field; | 97.8% |

| Conditions | Yield |
|---|---|
| With tetrabutylammomium bromide In toluene at 40 - 70℃; under 3000.3 - 4500.45 Torr; for 1h; Reagent/catalyst; Autoclave; Inert atmosphere; | 97.63% |
-
-
75-00-3
chloroethane

-
-
56368-46-8
sodium pentamethylenethiocarbamate

-
-
6961-73-5
piperidine-1-carbothioic acid S-ethyl ester

| Conditions | Yield |
|---|---|
| In water at 70℃; for 0.25h; in ultrasonic field; | 97.3% |
-
-
75-00-3
chloroethane

-
-
108-10-1
4-methyl-2-pentanone

-
-
91-68-9
3-diethylaminophenol

-
-
1864-92-2
N,N-diethyl-3-ethoxyaniline

| Conditions | Yield |
|---|---|
| With sodium hydroxide; sodium carbonate | 97.1% |


| Conditions | Yield |
|---|---|
| With magnesium at 50 - 100℃; | 97% |
| With magnesium at 60℃; for 1h; Temperature; | 94% |

| Conditions | Yield |
|---|---|
| With lithium chloride; zinc(II) chloride at 190℃; under 16501.7 Torr; for 10h; Catalytic behavior; Temperature; Pressure; Time; Reagent/catalyst; Autoclave; | 96.7% |
-
-
17771-33-4
7-hydroxy-2,2-dimethyl-chroman-4-one

-
-
75-00-3
chloroethane

-
-
76348-94-2
2,2-dimethyl-7-ethoxy-4-chromanone

| Conditions | Yield |
|---|---|
| With potassium carbonate; potassium iodide In N,N-dimethyl-formamide at 80℃; for 5h; | 96% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 100℃; for 3.5h; | 96% |
-
-
75-00-3
chloroethane

-
-
6375-47-9
3-amino-4-methoxyacetanilide

-
-
19433-93-3
3-acetylamino-6-methoxy-N,N-diethylaniline

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water at 80 - 97℃; under 7500.75 Torr; for 22.5h; pH=6.5 - 7; | 96% |

| Conditions | Yield |
|---|---|
| In acetone at 150℃; under 9000.9 Torr; for 40h; Solvent; Temperature; Pressure; Autoclave; | 95.23% |
| In acetonitrile for 10h; Inert atmosphere; Reflux; | 83.2% |

| Conditions | Yield |
|---|---|
| In acetonitrile at 80℃; for 5h; | 95% |
Ethyl chloride Consensus Reports
Ethyl chloride Standards and Recommendations
ACGIH TLV: TWA 1000 ppm
DFG MAK: Confirmed Animal Carcinogen with Unknown Relevance to Humans
NIOSH REL: (Chloroethane) Handle with caution
DOT Classification: 2.1; Label: Flammable Gas
Ethyl chloride Specification
The Ethane, chloro-, with the CAS registry number 75-00-3, is also known as Octamethylsilanetetramine. It belongs to the product categories of Refrigerants; Organics; Halides (Low Boiling point); Alkyl Chlorides; Gas Cylinders; Monofunctional & alpha,omega-Bifunctional Alkanes; Monofunctional Alkanes; Synthetic Organic Chemistry. Its EINECS number is 200-830-5. This chemical's molecular formula is C2H5Cl and molecular weight is 64.51. What's more, its systematic name is chloroethane. (1)Anesthetic [topical]; (2)Anesthetics; (3)Anesthetics, local; (4)Central Nervous System Agents; (5)Central Nervous System Depressants; (6)Drug / Therapeutic Agent; (7)Mutation data; (8)Peripheral Nervous System Agents; (9)Reproductive Effect; (10)Sensory System Agents; (11)TSCA Flag T [Subject to the Section 4 test rule under TSCA]; (12)Tumor data. It is stable at common pressure and temperature, and it should be sealed and stored in a cool and dry place. Moreover, it should be protected from oxides and water. It is used as a refrigerant, an aerosol spray propellant, an anesthetic, and a blowing agent for foam packaging.
Physical properties of Ethane, chloro- are: (1)ACD/LogP: 1.50; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 1.5; (4)ACD/LogD (pH 7.4): 1.5; (5)ACD/BCF (pH 5.5): 8.14; (6)ACD/BCF (pH 7.4): 8.14; (7)ACD/KOC (pH 5.5): 156.06; (8)ACD/KOC (pH 7.4): 156.06; (9)Index of Refraction: 1.361; (10)Molar Refractivity: 16.16 cm3; (11)Molar Volume: 72.9 cm3; (12)Polarizability: 6.4×10-24cm3; (13)Surface Tension: 17.9 dyne/cm; (14)Density: 0.884 g/cm3; (15)Enthalpy of Vaporization: 24.97 kJ/mol; (16)Boiling Point: 12.7 °C at 760 mmHg; (17)Vapour Pressure: 1170 mmHg at 25°C.
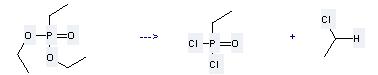
Preparation: this chemical can be prepared by ethylphosphonic acid diethyl ester by heating. This reaction will need reagent SOCl2 with the reaction time of 18 hours. This reaction will also need catalyst DMF. The yield is about 95.8%.
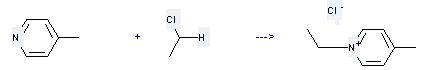
Uses of Ethane, chloro-: it can be used to produce 1-Ethyl-4-methylpyridinium chloride at the temperature of 80 °C. It will need solvent acetonitrile with the reaction time of 5 hours. The yield is about 95%.
When you are using this chemical, please be cautious about it as the following:
This chemical is extremely flammable, so you should keep it away from sources of ignition - No smoking. It has a limited evidence of a carcinogenic effect. It is harmful to aquatic organisms as it may cause long-term adverse effects in the aquatic environment. You should keep the container in a well-ventilated place and take precautionary measures against static discharges. When using it, you need wear suitable protective clothing and gloves. You must avoid releasing it to the environment just refering to special instructions/safety data sheet.
You can still convert the following datas into molecular structure:
(1)SMILES: ClCC
(2)Std. InChI: InChI=1S/C2H5Cl/c1-2-3/h2H2,1H3
(3)Std. InChIKey: HRYZWHHZPQKTII-UHFFFAOYSA-N
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| guinea pig | LCLo | inhalation | 40000ppm/45M (40000ppm) | BEHAVIORAL: SOMNOLENCE (GENERAL DEPRESSED ACTIVITY) BEHAVIORAL: EXCITEMENT LUNGS, THORAX, OR RESPIRATION: ACUTE PULMONARY EDEMA | U.S. Public Health Service, Public Health Bulletin. Vol. 185, Pg. 1, 1929. |
| mouse | LC50 | inhalation | 146gm/m3/2H (146000mg/m3) | "Toxicometric Parameters of Industrial Toxic Chemicals Under Single Exposure," Izmerov, N.F., et al., Moscow, Centre of International Projects, GKNT, 1982Vol. -, Pg. 66, 1982. | |
| rat | LC50 | inhalation | 152gm/m3/2H (152000mg/m3) | BRAIN AND COVERINGS: OTHER DEGENERATIVE CHANGES BEHAVIORAL: GENERAL ANESTHETIC BLOOD: HEMORRHAGE | Toksikologiya Novykh Promyshlennykh Khimicheskikh Veshchestv. Toxicology of New Industrial Chemical Substances. For English translation, see TNICS*. Vol. 6, Pg. 45, 1964. |
Related Products
- Ethyl (13-cis)-9-(4-methoxy-2,3,6-trimethylphenyl)-3,7-dimethyl-2,4,6,8-nonatetraenoate
- ethyl (1R,2R)-1-phenyl-2-(trideuteriomethylamino)cyclohex-3-ene-1-carboxylate,hydrochloride
- Ethyl (1S,2R)-2-(dimethylamino)-1-phenylcyclohex-3-ene-1-carboxylate hydrochloride
- Ethyl (2,4,6-trimethylbenzoyl) phenylphosphinate
- Ethyl (2-amino-4-hydroxy-6-methyl-5-pyrimidinyl)acetate
- Ethyl (2-bromopropionamido)acetate
- Ethyl (2-cyanoimino-5,6-dichloro-1,2,3,4-tetrahydroquinazolin-3-yl)acetate
- ETHYL (2E,4Z)-DECADIENOATE
- Ethyl (2-hydroxyethyl)dimethyl-ammonium benzilate chloride
- Ethyl (4-methylthiophenyl)acetate
- 7500-37-0
- 7500-39-2
- 7500-53-0
- 75006-53-0
- 75006-55-2
- 75007-99-7
- 75010-38-7
- 75011-65-3
- 75-01-4
- 7501-59-9
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View