-
Name
Histamine dihydrochloride
- EINECS 200-298-4
- CAS No. 56-92-8
- Article Data12
- CAS DataBase
- Density 1.14 g/cm3
- Solubility 0.1 g/mL in water
- Melting Point 249-252 °C(lit.)
- Formula C5H11Cl2N3
- Boiling Point 331 °C at 760 mmHg
- Molecular Weight 184.068
- Flash Point 180.3 °C
- Transport Information UN 3335
- Appearance white powder
- Safety 22-26-36/37/39-45-37-36/37
- Risk Codes 36/37/38-42/43-42-22
-
Molecular Structure
-
Hazard Symbols
 Xn,
Xn,  Xi
Xi
- Synonyms 1H-Imidazole-4-ethanamine,dihydrochloride (9CI);Histamine, dihydrochloride (8CI);2-(1H-Imidazol-4-yl)ethanamine dihydrochloride;4-(2-Aminoethyl)imidazole dihydrochloride;Ceplene;Histaminechloride;Histamine dichloride;1H-Imidazole-5-ethanamine,hydrochloride (1:2);
- PSA 54.70000
- LogP 2.21520
Synthetic route
-
-
57090-90-1
1H-imidazole-4-carboxaldehyde oxime

-
-
56-92-8
histamine dichloride

| Conditions | Yield |
|---|---|
| With 5%-palladium/activated carbon; hydrogen; acetyl chloride In methanol at 0℃; for 17.5h; Inert atmosphere; | 98% |

| Conditions | Yield |
|---|---|
| Stage #1: L-histidine With (R)-Carvone In propan-1-ol at 190℃; under 11251.1 Torr; for 0.2h; Sealed tube; Stage #2: With hydrogenchloride In propan-1-ol; water at 190℃; for 0.0833333h; | 92% |
| Multi-step reaction with 2 steps 1: propan-1-ol / 20 - 190 °C / Green chemistry 2: hydrogenchloride / propan-1-ol / 0.2 h / 190 °C / Green chemistry View Scheme | |
| Multi-step reaction with 3 steps 1: copper(I) bromide / 10 h / 100 - 110 °C / Inert atmosphere; Darkness 2: hydrogenchloride / 30 °C 3: hydrogenchloride / methanol / 30 °C View Scheme |

-
-
56-92-8
histamine dichloride

| Conditions | Yield |
|---|---|
| With hydrogenchloride In propan-1-ol at 190℃; for 0.2h; Temperature; Green chemistry; | 92% |
-
-
58995-29-2
Nα-Trityl-L-histidin

-
-
56-92-8
histamine dichloride

| Conditions | Yield |
|---|---|
| Stage #1: Nα-Trityl-L-histidin With (R)-Carvone In propan-1-ol at 0 - 190℃; Green chemistry; Stage #2: With hydrogenchloride In propan-1-ol; water at 190℃; for 0.0833333h; Microwave irradiation; Green chemistry; | 86% |
-
-
74294-62-5
4-(2-Azido-ethyl)-1-(toluene-4-sulfonyl)-1H-imidazole

-
-
56-92-8
histamine dichloride

| Conditions | Yield |
|---|---|
| With hydrogenchloride; hydrogen; palladium on activated charcoal In ethanol for 8h; | 90 mg |

-
-
56-92-8
histamine dichloride

| Conditions | Yield |
|---|---|
| With hydrogenchloride; n-Amyl nitrite Eindampfen des Gemisches und Kochen des Rueckstands mit konz. Salzsaeure; |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 336 mg / pyridine / 2 h / -23 °C 2: 180 mg / lithium azide / dimethylformamide / 18 h / 21 °C 3: 90 mg / aq. hydrochloric acid, hydrogen / palladium-charcoal / ethanol / 8 h View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: 34 mg / ethanol, NAD+, liver alcohol dehydrogenase, catalase, diamine oxidase from pea seedling / 48 h / 37 °C / phosphate buffer (pH 7) 2: 336 mg / pyridine / 2 h / -23 °C 3: 180 mg / lithium azide / dimethylformamide / 18 h / 21 °C 4: 90 mg / aq. hydrochloric acid, hydrogen / palladium-charcoal / ethanol / 8 h View Scheme | |
| With hydrogenchloride In methanol at 30℃; | 4.11 g |
-
-
74294-60-3
NO-bis-p-tolylsulphonylhistaminol

-
-
56-92-8
histamine dichloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 180 mg / lithium azide / dimethylformamide / 18 h / 21 °C 2: 90 mg / aq. hydrochloric acid, hydrogen / palladium-charcoal / ethanol / 8 h View Scheme |
-
-
605-65-2
5-(dimethylamino)naphth-1-ylsulfonyl chloride

-
-
56-92-8
histamine dichloride

-
-
13285-08-0
N,Nim-Didansylhistamine

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane 1.) 0 deg C, 1 h, 2.) r.t., 20 h; | 100% |

| Conditions | Yield |
|---|---|
| silica gel; palladium In methanol | 100% |

| Conditions | Yield |
|---|---|
| With triethylamine In water Et3N added to aq. soln. of ZnCl2*6H2O, histamine*2HCl and Si-compd. added to suspn., flask closed with rubber septum and shaken vigorously for 20 min; solid collected, recrystd. from MeOH/MeCN (4/1); | 99% |

-
-
56-92-8
histamine dichloride

-
-
88238-58-8
ethyl 2-acetamino-2-ethoxycarbonyl-6-oxohexanoate

-
-
88238-59-9
ethyl 2-acetamino-2-ethoxycarbonyl-9-(4-imidazolyl)-7-azanonanoate

| Conditions | Yield |
|---|---|
| With sodium cyanoborohydride In methanol Ambient temperature; | 98% |
| With sodium cyanoborohydride In ethanol Yield given; |
-
-
22509-74-6
N-ethoxycarbonylphthalimide

-
-
56-92-8
histamine dichloride

-
-
5959-80-8
2-(2-(1H-imidazol-4-yl)ethyl)isoindoline-1,3-dione

| Conditions | Yield |
|---|---|
| With sodium carbonate In water at 20℃; for 2h; | 98% |
| With sodium carbonate In water at 20℃; for 2h; |
-
-
37942-07-7
3,5-di-tert-butyl-2-hydroxybenzaldehyde

-
-
56-92-8
histamine dichloride

-
-
1374255-34-1
2,4-di-tert-butyl-6-{[2-(1H-imidazol-4-yl)-ethylimino]-methyl}-phenol

| Conditions | Yield |
|---|---|
| With formic acid; sodium hydrogencarbonate In methanol for 24h; Inert atmosphere; Reflux; | 98% |

| Conditions | Yield |
|---|---|
| With triethylamine In N,N-dimethyl-formamide at 20℃; for 5h; Cooling with ice; | 97% |

-
-
56-92-8
histamine dichloride

| Conditions | Yield |
|---|---|
| Stage #1: histamine dichloride With sodium methylate In methanol; water Stage #2: C15H17O(1+)*F6P(1-) With acetic acid In methanol; water Reflux; | 96% |

| Conditions | Yield |
|---|---|
| Stage #1: histamine dichloride With sodium hydroxide In ethanol; water at 0 - 70℃; Large scale; Stage #2: isobutyraldehyde In ethanol; water at 0 - 76℃; for 10h; Large scale; Stage #3: With ethanol; acetyl chloride at 0 - 25℃; Large scale; | 96% |
-
-
24424-99-5
di-tert-butyl dicarbonate

-
-
56-92-8
histamine dichloride

-
-
98870-64-5
Nα-(tert-butoxycarbonyl)histamine

| Conditions | Yield |
|---|---|
| With sodium hydroxide In 1,4-dioxane; water at 20℃; for 2h; Inert atmosphere; | 95% |
| With sodium hydroxide In 1,4-dioxane; water at 20℃; for 2h; pH=12; | 85% |
| With triethylamine In methanol; acetonitrile; Petroleum ether |
-
-
383-63-1
ethyl trifluoroacetate,

-
-
56-92-8
histamine dichloride

-
-
76-83-5
trityl chloride

-
-
312937-01-2
2,2,2-trifluoro-N-(1-trityl-1H-imidazol-4-ylethyl)acetamide

| Conditions | Yield |
|---|---|
| Stage #1: histamine dichloride With N-ethyl-N,N-diisopropylamine In methanol Cooling with ice; Stage #2: ethyl trifluoroacetate, In methanol at 20℃; Cooling with ice; Stage #3: trityl chloride With triethylamine In dichloromethane at 0 - 20℃; | 94% |
| Stage #1: histamine dichloride With triethylamine In methanol at 20℃; for 0.166667h; Stage #2: ethyl trifluoroacetate, In methanol; dichloromethane at 0 - 20℃; for 4h; Stage #3: trityl chloride With triethylamine In dichloromethane at 20℃; | 91% |

-
-
56-92-8
histamine dichloride

-
-
1259485-83-0
4-(2-azidoethyl)-1H-imidazole

| Conditions | Yield |
|---|---|
| With copper(ll) sulfate pentahydrate; triflic azide; potassium carbonate In methanol; dichloromethane; water at 20℃; pH=8.3; | 94% |
| With copper(ll) sulfate pentahydrate; triflic azide; potassium carbonate In methanol; dichloromethane; water at 20℃; for 24h; Inert atmosphere; |

-
-
56-92-8
histamine dichloride

| Conditions | Yield |
|---|---|
| Stage #1: histamine dichloride With sodium methylate In methanol; water Stage #2: C13H13O(1+)*F6P(1-) With acetic acid In methanol; water Reflux; | 94% |
-
-
56-92-8
histamine dichloride

-
-
78841-25-5
N-t-butyloxycarbonyl-Nε-(2-chlorobenzyloxycarbonyl)lysine pentafluorophenyl ester

-
-
261784-45-6
Boc-Lys(Z-(2-Cl))-Hia

| Conditions | Yield |
|---|---|
| With triethylamine In chloroform; N,N-dimethyl-formamide for 20h; Substitution; | 93% |
-
-
85-44-9
phthalic anhydride

-
-
56-92-8
histamine dichloride

-
-
5959-80-8
2-(2-(1H-imidazol-4-yl)ethyl)isoindoline-1,3-dione

| Conditions | Yield |
|---|---|
| With acetic acid; triethylamine for 2h; Reflux; | 91.6% |

-
-
56-92-8
histamine dichloride

| Conditions | Yield |
|---|---|
| Stage #1: histamine dichloride With sodium methylate In methanol; water Stage #2: C17H21O(1+)*F6P(1-) With acetic acid In methanol; water Reflux; | 90% |
-
-
56-92-8
histamine dichloride

-
-
791634-10-1
4-(2-bromoethyl)-1H-imidazole hydrochloride

| Conditions | Yield |
|---|---|
| With sulfuric acid; potassium bromide; sodium nitrite at -15℃; | 90% |

| Conditions | Yield |
|---|---|
| With NaOH In methanol; ethanol addn. of solid NaOH to suspn. of histamine in methanol, addn. of aldehyde, stirring 15 min, addn. of V complex and ethanol, stirring overnight; precipitate filtered off and washed with ether, elem. anal.; | 89% |


| Conditions | Yield |
|---|---|
| With sodium hydroxide In methanol for 12h; Heating; | 88% |
-
-
24424-99-5
di-tert-butyl dicarbonate

-
-
56-92-8
histamine dichloride

-
-
127119-12-4
1-t-Butoxycarbonyl-4-{2-(t-butoxycarbonyl-amino)ethyl}imidazole

| Conditions | Yield |
|---|---|
| Stage #1: histamine dichloride With triethylamine In methanol at 20℃; for 0.5h; Inert atmosphere; Stage #2: di-tert-butyl dicarbonate In methanol at 20℃; for 1h; Inert atmosphere; | 88% |
| With triethylamine In acetonitrile at 20℃; for 72h; |
-
-
56-92-8
histamine dichloride

-
-
21487-48-9
3-methyl-1-phenyl-1H-pyrazole-4-carbaldehyde

-
-
1206607-01-3
4-(3-methyl-1-phenylpyrazol-4-yl)spinaceamine

| Conditions | Yield |
|---|---|
| With sodium hydroxide In ethanol; water Heating; | 88% |
-
-
56-92-8
histamine dichloride

-
-
122-03-2
(4-isopropylbenzaldehyde)

-
-
303021-39-8
4-(4-isopropylphenyl)-4,5,6,7-tetrahydro-imidazo[4,5-c]pyridine

| Conditions | Yield |
|---|---|
| With potassium hydroxide In ethanol; water for 1.5h; Heating / reflux; | 87% |

| Conditions | Yield |
|---|---|
| With benzotriazol-1-yloxyl-tris-(pyrrolidino)-phosphonium hexafluorophosphate; N-ethyl-N,N-diisopropylamine In dichloromethane; N,N-dimethyl-formamide at 20℃; for 24h; | 86% |

| Conditions | Yield |
|---|---|
| With benzotriazol-1-yloxyl-tris-(pyrrolidino)-phosphonium hexafluorophosphate; N-ethyl-N,N-diisopropylamine In dichloromethane; N,N-dimethyl-formamide at 20℃; for 24h; | 86% |
-
-
56-92-8
histamine dichloride

-
-
322474-21-5
N,N'-bis-Boc-S-methyl-isothiourea

-
-
255061-12-2
N',N''-bis(tert-butoxycarbonyl)-N-(2-(1H-imidazol-4-yl)ethyl)guanidine

| Conditions | Yield |
|---|---|
| Stage #1: histamine dichloride With sodium carbonate In water at 20℃; for 0.5h; Stage #2: N,N'-bis-Boc-S-methyl-isothiourea In tetrahydrofuran; water at 55℃; for 24h; Reflux; | 86% |
-
-
28320-73-2
((N-(tert-butoxycarbonyl)glycyl)glycyl)glycine

-
-
56-92-8
histamine dichloride

-
-
1267456-25-6
N-tert-butoxycarbonyl-glycyl-glycyl-glycyl-histamine

| Conditions | Yield |
|---|---|
| With (benzotriazo-1-yloxy)tris(dimethylamino)phosphonium hexafluorophosphate; triethylamine In acetonitrile at 20℃; for 12h; | 85% |
| With (benzotriazo-1-yloxy)tris(dimethylamino)phosphonium hexafluorophosphate |
Histamine dihydrochloride Chemical Properties
Molecule structure of Histamine dihydrochloride (CAS NO.56-92-8):
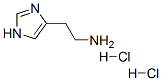
IUPAC: 2-(1H-Imidazol-5-yl)ethanamine
Molecular formula: C5H11Cl2N3
Molecular Weight: 184.07 g/mol
EINECS: 200-298-4
Density: 1.14g/cm3
Melting Point: 249-252 °C
Boiling Point: 331 °C at 760 mmHg
Flash Point: 180.3 °C
Water Solubility: 0.1g/mL, clear, colorless
Stability: Stable. Incompatible with strong oxidizing agents.
Storage Temp.: 2-8 °C
H-Bond Donor: 4
H-Bond Acceptor: 2
Rotatable Bond Count: 2
Tautomer Count: 2
Exact Mass: 183.033003
MonoIsotopic Mass: 183.033003
Topological Polar Surface Area: 54.7
Heavy Atom Count: 10
Canonical SMILES: C1=C(NC=N1)CCN.Cl.Cl
InChI: InChI=1S/C5H9N3.2ClH/c6-2-1-5-3-7-4-8-5;;/h3-4H,1-2,6H2,(H,7,8);2*1H
InChIKey: PPZMYIBUHIPZOS-UHFFFAOYSA-N
Product Categories:Imidazoles & Benzimidazoles;Heterocyclic Compounds;Histidine [His, H];Amino acid salt;Imidazoles & Benzimidazoles
Histamine dihydrochloride Uses
Histamine dihydrochloride (CAS NO.56-92-8) can be used in biochemical research because it can impact on local immune responses, regulating physiological function in the gut and as a neurotransmitter. It has also been used as substrate in determinating histamine enzyme.
Histamine dihydrochloride Production
Histamine dihydrochloride can be extracted from Claviceps purpurea, many plants and animals.
Histamine dihydrochloride Toxicity Data With Reference
| 1. | orl-mus LD50:2534 mg/kg | ARTODN Archives of Toxicology. 2 (1979),371. | ||
| 2. | ipr-mus LD50:184 mg/kg | YKKZAJ Yakugaku Zasshi. Journal of Pharmacy. 97 (1977),1117. | ||
| 3. | scu-mus LDLo:1300 mg/kg | AEPPAE Naunyn-Schmiedeberg’s Archiv fuer Experimentelle Pathologie und Pharmakologie. 166 (1932),437. | ||
| 4. | ivn-mus LD50:370 mg/kg | ATSUDG Archives of Toxicology, Supplement. 2 (1979),371. | ||
| 5. | ipr-rbt LDLo:200 mg/kg | JIDIAQ Journal of Infectious Diseases. 42 (1928),473. | ||
| 6. | ipr-gpg LD50:4602 µg/kg | JPETAB Journal of Pharmacology and Experimental Therapeutics. 77 (1943),54. | ||
| 7. | scu-gpg LD50:1250 µg/kg | TXAPA9 Toxicology and Applied Pharmacology. 8 (1966),339. | ||
| 8. | ivn-gpg LD50:294 µg/kg | JAPMA8 Journal of the American Pharmaceutical Association, Scientific Edition. 33 (1944),80. | ||
| 9. | par-gpg LDLo:570 µg/kg | AEPPAE Naunyn-Schmiedeberg’s Archiv fuer Experimentelle Pathologie und Pharmakologie. 211 (1950),328. |
Histamine dihydrochloride Consensus Reports
Reported in EPA TSCA Inventory.
Histamine dihydrochloride Safety Profile
Poison by subcutaneous, intravenous, intraperitoneal, and parenteral routes. Moderately toxic by ingestion. Experimental reproductive effects. When heated to decomposition it emits very toxic fumes of Cl− and NOx. See also HISTAMINE.
Hazard Codes:  Xn,
Xn,  Xi
Xi
Risk Statements:36/37/38-42/43-42-22
22:(Harmful if swallowed)
36/37/38: (Irritating to the eyes piratory system and skin)
42/43:(May cause sensitization by inhalation and skin contact)
42:(May cause sensitization by inhalation)
Safety Statements:22-26-36/37/39-45-37-36/37
22:(Do not breathe dust)
26: (In case of contact with eyes,rinse immediately with plenty of water and seek medical advice)
36/37/39:(Wear suitable protective clothing, gloves and eye/face protection)
45:(In case of accident or if you feel unwell, seek medical advice immediately (show label where possible))
37:(Wear suitable gloves)
36/37:(Wear suitable protective clothing and gloves)
WGK Germany:2
RTECS:MS1575000
F:1-8-9
Hazard Note:Irritant
Histamine dihydrochloride Specification
Histamine dihydrochloride (CAS NO.56-92-8) is also named as 2-(1H-Imidazol-4-yl)ethanamine dihydrochloride ; 2-(1H-Imidazol-4-yl)ethylamine dihydrochloride ; 4-(2-Aminoethyl)imidazole dihydrochloride ; AI3-24394 ; Bichlorhydrate d'histamine ; Bichlorhydrate d'histamine [French] ; Ceplene ; Histamine HCl ; Histamine chloride ; Histamine dichloride ; Histamine dihydrochloride ; NSC 257873 ; Peremin ; UNII-3POA0Q644U . Histamine dihydrochloride (CAS NO.56-92-8) is white powder.
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View