-
Name
Indole-3-carbinol
- EINECS 211-836-2
- CAS No. 700-06-1
- Article Data37
- CAS DataBase
- Density 1.272 g/cm3
- Solubility
- Melting Point 96-99 °C(lit.)
- Formula C9H9NO
- Boiling Point 360.6 °C at 760 mmHg
- Molecular Weight 147.177
- Flash Point 171.9 °C
- Transport Information
- Appearance Off-white powder
- Safety 26-36
- Risk Codes 36/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms Indole-3-methanol(6CI,7CI,8CI);3-(Hydroxymethyl)indole;3-Indolylcarbinol;3-Indolylmethanol;Indinol;NSC 525801;Indole-3-methanol;
- PSA 36.02000
- LogP 1.66020
Synthetic route

| Conditions | Yield |
|---|---|
| With sodium methylate for 8h; Heating; | 100% |
| With methanol; sodium hydrogencarbonate | |
| With sodium methylate |

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate In methanol at 0℃; for 1h; | 100% |
| With formic acid In ethanol at 80℃; for 12h; Catalytic behavior; | 91% |
| With ammonia; calcium In tetrahydrofuran at -33℃; for 2h; other reagents: lithium, sodium, liq. ammonia; | 80% |

| Conditions | Yield |
|---|---|
| Stage #1: 3-methoxycarbonylindole With pentadec-3-en-2-one; trimethylsilyl trifluoromethanesulfonate; triphenylphosphine In dichloromethane at -78 - 0℃; for 1h; Inert atmosphere; Stage #2: With diisobutylaluminium hydride In hexane; dichloromethane at -78℃; Inert atmosphere; Stage #3: With potassium carbonate In methanol; hexane; dichloromethane at 20℃; for 0.5h; Inert atmosphere; | 84% |
-
-
487-89-8
Indole-3-carboxaldehyde

-
-
124-40-3
dimethyl amine

-
A
-
700-06-1
indole-3-carbinol

-
B
-
87-52-5
3-(Dimethylaminomethyl)indole

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate; water In methanol at 55 - 60℃; for 2.5h; | A 25% B 74% |

-
-
487-89-8
Indole-3-carboxaldehyde

-
A
-
700-06-1
indole-3-carbinol

-
B
-
22259-53-6
3-aminomethylindole

-
C
-
93742-14-4
bis(indol-3-ylmethyl)amine

| Conditions | Yield |
|---|---|
| With ammonium hydroxide; sodium tetrahydroborate In methanol at 14 - 18℃; for 20h; | A 72% B 9% C 13% |

-
-
487-89-8
Indole-3-carboxaldehyde

-
-
77287-34-4, 77287-35-5, 60100-09-6
formamide

-
A
-
700-06-1
indole-3-carbinol

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate In methanol at 20℃; for 1.5h; Leukardt-Wallach Amination; | A 30% B 49% |

-
-
87-51-4
indole-3-acetic acid

-
A
-
83-34-1
3-Methylindole

-
B
-
487-89-8
Indole-3-carboxaldehyde

-
C
-
700-06-1
indole-3-carbinol

-
D
-
59027-01-9
3-(3-methylindol-1-yl)methylindole

| Conditions | Yield |
|---|---|
| With 1,3,6,8-tetra-n-butylpyrimido<5,4-g>pteridine-2,4,5,7(1H,3H,6H,8H)-tetrone 10-oxide In acetonitrile for 0.0833333h; Irradiation; | A 14% B 36% C 12% D 8% |
| With 1,3,6,8-tetra-n-butylpyrimido<5,4-g>pteridine-2,4,5,7(1H,3H,6H,8H)-tetrone 10-oxide In acetonitrile for 0.166667h; Product distribution; Ambient temperature; Irradiation; depends on time and wavelength of irradiation and ratio of reacted compounds; |

-
-
110-88-3
1,3,5-Trioxan

-
-
120-72-9
indole

-
-
67-56-1
methanol

-
-
124-41-4
sodium methylate

-
-
700-06-1
indole-3-carbinol

-
-
17206-03-0
1-(1H-indol-3-yl)-N,N-dimethylmethanamine oxide

-
-
700-06-1
indole-3-carbinol

| Conditions | Yield |
|---|---|
| With sodium hydroxide; diethyl ether; water |

| Conditions | Yield |
|---|---|
| With sodium hydroxide; diethyl ether; water |
-
-
87-51-4
indole-3-acetic acid

-
A
-
487-89-8
Indole-3-carboxaldehyde

-
B
-
700-06-1
indole-3-carbinol

-
C
-
1861-29-6
3-methylideneoxindole

-
D
-
2005-90-5
2-oxindole-3-methanol

| Conditions | Yield |
|---|---|
| With oxygen In water horseradish peroxidase; |


| Conditions | Yield |
|---|---|
| With myrosinase In water at 37℃; for 4h; Mechanism; | |
| With myrosinase In water for 40h; Mechanism; low water system; |


| Conditions | Yield |
|---|---|
| With myrosinase In water at 37℃; for 4h; Product distribution; other solvent, other substrates; |


| Conditions | Yield |
|---|---|
| With phosphate buffer; potassium bromide; dinitrogen monoxide In water at 20℃; Rate constant; Product distribution; Irradiation; effect of pH on the rate, effect of oxygen on the decay, effect of glutathione on the oxidation; |


| Conditions | Yield |
|---|---|
| With cationic peanut peroxidase; oxygen In acetate buffer at 25℃; pH=5.0; Kinetics; Product distribution; Further Variations:; Reagents; Oxidation; |


| Conditions | Yield |
|---|---|
| With phosphate buffer; goat lung microsomes (cytochrome 450); NADPH at 37℃; for 0.0333333h; pH=7.4; Enzyme kinetics; Oxidation; |

-
A
-
700-06-1
indole-3-carbinol

| Conditions | Yield |
|---|---|
| With sodium hydroxide |

| Conditions | Yield |
|---|---|
| With horseradish peroxidase In ethanol; water at 37℃; for 2h; Kinetics; | |
| With horseradish peroxidase In ethanol; water at 37℃; for 2h; |
-
-
86204-54-8
indolyl-3-methyl thioacetate

-
A
-
700-06-1
indole-3-carbinol

-
B
-
86204-61-7
diindolyl-3,3'-methylsulfide

| Conditions | Yield |
|---|---|
| With potassium hydroxide In tetrahydrofuran at 20℃; |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: ethanol; aqueous H2O2 2: water; NaOH-solution; diethyl ether View Scheme |
-
-
120-72-9
indole

-
-
50-00-0
formaldehyd

-
A
-
700-06-1
indole-3-carbinol

-
B
-
5031-11-8
1-hydroxymethyl-1H-indole

| Conditions | Yield |
|---|---|
| With calcium oxide at 65℃; neat (no solvent); |


-
-
700-06-1
indole-3-carbinol

| Conditions | Yield |
|---|---|
| With water for 72h; |
-
-
1395069-51-8
N-methyl-S-(3-indolylmethyl)carbamodithioate

-
-
700-06-1
indole-3-carbinol

| Conditions | Yield |
|---|---|
| With water |
-
-
86204-60-6
indol-3ylmethanethiol

-
-
700-06-1
indole-3-carbinol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: triethylamine / dichloromethane / 0.33 h / 20 °C 2: water View Scheme | |
| Multi-step reaction with 2 steps 1: triethylamine / dichloromethane / 0.33 h / 20 °C 2: water / 72 h View Scheme |
-
-
86204-54-8
indolyl-3-methyl thioacetate

-
-
700-06-1
indole-3-carbinol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: hydrazine hydrate / tetrahydrofuran / 0.75 h / 80 °C 2: triethylamine / dichloromethane / 0.33 h / 20 °C 3: water View Scheme | |
| Multi-step reaction with 3 steps 1: hydrazine hydrate / tetrahydrofuran / 0.75 h / 80 °C 2: triethylamine / dichloromethane / 0.33 h / 20 °C 3: water / 72 h View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: toluene-4-sulfonic acid / 20 h / Reflux 2.1: sodium hydroxide / ethanol / 40 °C 2.2: 0.42 h / 250 °C / Microwave irradiation View Scheme |
-
-
700-06-1
indole-3-carbinol

-
-
24424-99-5
di-tert-butyl dicarbonate

-
-
96551-22-3
t-butyl 3-hydroxymethylindole-1-carboxylate

| Conditions | Yield |
|---|---|
| With dmap In acetonitrile at 25℃; for 16h; | 100% |
| With dmap; triethylamine In dichloromethane at 20℃; for 1h; | 15% |
-
-
700-06-1
indole-3-carbinol

-
-
18162-48-6
tert-butyldimethylsilyl chloride

| Conditions | Yield |
|---|---|
| With 1H-imidazole In N,N-dimethyl-formamide at 20℃; for 0.5h; | 100% |

| Conditions | Yield |
|---|---|
| With 1-hydroxy-3H-benz[d][1,2]iodoxole-1,3-dione In dimethyl sulfoxide for 8h; Ambient temperature; | 98% |
| With 1,3,6,8-tetra-n-butylpyrimido<5,4-g>pteridine-2,4,5,7(1H,3H,6H,8H)-tetrone 10-oxide In acetonitrile for 0.0333333h; Irradiation; | 90% |
| With LACTIC ACID; dihydrogen peroxide at 30℃; for 7h; | 87% |
-
-
700-06-1
indole-3-carbinol

-
-
149-30-4
2-Mercaptobenzothiazole

-
-
87549-67-5
2-[(1H-indol-3-yl)methylthio]benzo[d]thiazole

| Conditions | Yield |
|---|---|
| With triphenylphosphine; diethylazodicarboxylate In tetrahydrofuran at 0 - 20℃; for 6.25h; Mitsunobu reaction; | 96% |

| Conditions | Yield |
|---|---|
| Stage #1: indole-3-carbinol With sodium hydride In N,N-dimethyl-formamide at 0℃; for 4h; Stage #2: methyl iodide In N,N-dimethyl-formamide at 0 - 20℃; | 95% |
-
-
700-06-1
indole-3-carbinol

-
-
456-27-9
4-nitrobenzenediazonium tetrafluoroborate

-
-
53330-79-3
3-(4-nitro-phenylazo)-indole

| Conditions | Yield |
|---|---|
| In ethanol; water | 94% |
-
-
700-06-1
indole-3-carbinol

-
-
89337-62-2
tert-butyl((1-methoxy-2-methylprop-1-en-1-yl)oxy)dimethylsilane

| Conditions | Yield |
|---|---|
| With magnesium bis(trifluoromethane solfonyl)imide In dichloromethane for 0.5h; Ambient temperature; | 93% |

| Conditions | Yield |
|---|---|
| With sodium hydride In tetrahydrofuran at 0℃; for 0.25h; | 93% |


| Conditions | Yield |
|---|---|
| With dimethyl sulfoxide; 2,4,6-tripropyl-1,3,5,2,4,6-trioxatriphosphinane-2,4,6-trioxide In ethyl acetate at 20℃; for 5h; | 92% |


| Conditions | Yield |
|---|---|
| With dimethyl sulfoxide; 2,4,6-tripropyl-1,3,5,2,4,6-trioxatriphosphinane-2,4,6-trioxide In ethyl acetate at 20℃; for 5h; | 92% |

-
-
700-06-1
indole-3-carbinol

-
-
137-07-5
2-amino-benzenethiol

-
-
31224-76-7
2-(1H-indol-3-yl)benzo[d]thiazole

| Conditions | Yield |
|---|---|
| With tetrabutyl phosphonium bromide In water at 80℃; for 2h; Catalytic behavior; | 92% |
-
-
700-06-1
indole-3-carbinol

-
-
496-72-0
4-methyl-1,2-diaminobenzene

-
-
940704-02-9
2-(1H-indole-3-yl)-5-methyl-1H-benzimidazole

| Conditions | Yield |
|---|---|
| With tetrabutyl phosphonium bromide In water at 80℃; for 3.5h; Catalytic behavior; | 90% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In N,N-dimethyl-formamide at 25℃; for 3h; Green chemistry; | 87% |
-
-
95-20-5
2-methyl-1H-indole

-
-
700-06-1
indole-3-carbinol

-
-
22546-09-4
(4-methoxyphenyl)(2-methyl-3-indolyl)methane

| Conditions | Yield |
|---|---|
| With ammonium cerium(IV) nitrate In ethanol at 20℃; for 2.5h; ultrasonic irradiation; | 86% |

| Conditions | Yield |
|---|---|
| With dimethyl sulfoxide; 2,4,6-tripropyl-1,3,5,2,4,6-trioxatriphosphinane-2,4,6-trioxide In ethyl acetate at 20℃; for 5.5h; | 86% |


| Conditions | Yield |
|---|---|
| With air | 85.3% |

| Conditions | Yield |
|---|---|
| [ruthenium(II)(η6-1-methyl-4-isopropyl-benzene)(chloride)(μ-chloride)]2 In toluene for 24h; Heating; | 85% |
| With 1,1'-bis(diphenylphosphino)ferrocene; [ruthenium(II)(η6-1-methyl-4-isopropyl-benzene)(chloride)(μ-chloride)]2 In toluene at 20℃; Inert atmosphere; Reflux; Molecular sieve; | 85% |
| With ethanol; sodium ethanolate |
-
-
700-06-1
indole-3-carbinol

-
-
87-51-4
indole-3-acetic acid

-
A
-
78440-76-3
3-(methoxymethyl)-1H-indole

-
B
-
68232-56-4
2-(1H-indol-3-ylmethyl)-1H-indole-3-acetic acid

| Conditions | Yield |
|---|---|
| In methanol; water | A 6% B 85% |

-
-
700-06-1
indole-3-carbinol

-
-
122-52-1
triethyl phosphite

-
-
17053-23-5
diethyl ((1H-indol-3-yl)methyl)phosphonate

| Conditions | Yield |
|---|---|
| With tetra-(n-butyl)ammonium iodide at 120℃; for 24h; Inert atmosphere; | 83% |
| With tetra-(n-butyl)ammonium iodide In neat (no solvent) at 125℃; for 24h; Michaelis-Arbuzov Synthesis; Inert atmosphere; Schlenk technique; Sealed tube; Green chemistry; | 83% |

| Conditions | Yield |
|---|---|
| With N,N,N,N,-tetramethylethylenediamine; 9-azanoradamantane N-oxyl; oxygen; copper(l) chloride In tetrahydrofuran at 23℃; for 24h; Molecular sieve; | 81% |

| Conditions | Yield |
|---|---|
| With [(bis(2-dicyclohexylphosphinoethyl)amine)Fe(CO)Br2]; sodium triethylborohydride; sodium hydroxide In toluene at 130℃; for 6h; Glovebox; Sealed tube; Inert atmosphere; Flow reactor; High pressure; | 81% |

| Conditions | Yield |
|---|---|
| 80.3% |
-
-
700-06-1
indole-3-carbinol

-
A
-
138250-72-3
2-(indol-3-ylmethyl)-3,3'-methylenediindole

-
B
-
114648-66-7
(1H-indol-3-yl)-(1H-indol-2-yl)-methane

| Conditions | Yield |
|---|---|
| With pH 5 buffer In methanol | A 10% B 80% |
| With pH 5 buffer In methanol | A 10 % Chromat. B 80 % Chromat. |
-
-
83-34-1
3-Methylindole

-
-
700-06-1
indole-3-carbinol

-
-
70423-69-7
2-[(1H-indol-3-yl)methyl]-3-methyl-1H-indole

| Conditions | Yield |
|---|---|
| With ammonium cerium(IV) nitrate In ethanol at 20℃; for 2.5h; ultrasonic irradiation; | 80% |
-
-
700-06-1
indole-3-carbinol

| Conditions | Yield |
|---|---|
| With 1,3-dichloro-5,5-dimethylhydantoin In ethyl acetate at 20℃; | 80% |

| Conditions | Yield |
|---|---|
| With [Co(PCPMe-iPr)Cl]; potassium tert-butylate In toluene at 80℃; for 16h; Inert atmosphere; Green chemistry; | 79% |
| With trans-[Fe(N,N’-bis(diisopropylphosphino)-N''-dimethyl-2,4,6-triamino-1,3,5-triazine)(CO)Br2]; potassium tert-butylate In toluene at 80℃; for 16h; Inert atmosphere; | 73% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide for 1h; Heating; | 77% |
| With sodium hydroxide In water for 1h; Heating / reflux; | 77% |
| With potato dextrose agar for 6h; | 45% |
Indole-3-carbinol Chemical Properties
Molecular Structure of Indole-3-methanol (CAS NO.700-06-1):
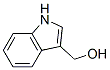
Molecular Formula: C9H9NO
Molecular Weight: 147.1739
IUPAC Name: 1H-Indol-3-ylmethanol
CAS NO: 700-06-1
Appearance:white to off white crystals
Melting point: 96-99 °C
Index of Refraction: 1.705
Molar Refractivity: 44.97 cm3
Molar Volume: 115.6 cm3
Surface Tension: 61.7 dyne/cm
Density: 1.272 g/cm3
Flash Point: 171.9 °C
Enthalpy of Vaporization: 63.98 kJ/mol
Boiling Point: 360.6 °C at 760 mmHg
Vapour Pressure: 7.89E-06 mmHg at 25°C
Classification Code: Anticarcinogenic agents; Antineoplastic Agents; Protective Agents; Reproductive Effect
Synonyms: 3-Hydroxymethylindole ; 3-Indolylcarbinol ; 5-21-03-00045 (Beilstein Handbook Reference) ; AI3-60090 ; BRN 0121323 ; CCRIS 3261 ; EINECS 211-836-2 ; Indinol ; Indole 3 carbinol ; Indole-3-carbinol ; NSC 525801 ; 1H-Indole-3-methanol (9CI)
SMILES: OCc2c1ccccc1nc2
InChI: InChI=1/C9H9NO/c11-6-7-5-10-9-4-2-1-3-8(7)9/h1-5,10-11H,6H2
InChIKey: IVYPNXXAYMYVSP-UHFFFAOYAO
Std. InChI: InChI=1S/C9H9NO/c11-6-7-5-10-9-4-2-1-3-8(7)9/h1-5,10-11H,6H2
Std. InChIKey: IVYPNXXAYMYVSP-UHFFFAOYSA-N
Indole-3-carbinol Uses
Indole-3-methanol (CAS NO.700-06-1) can prevent the development of estrogen-enhanced cancers including breast, endometrial and cervical cancers. It has be shown to inhibit carcinogenesis in several animal species, but it enhances tumor incidence if administered at a post-initiation stage.
Indole-3-carbinol Production
Indole-3-methanol (CAS NO.700-06-1) naturally exists in vegetables such as cabbage, broccoli, brussels sprouts, and kale. It is also widely available in supplement form. In 1985,B Sundberg and his Co-worker did a study about "Identification and Quantification of Indole-3-methanol in Etiolated Seedlings of Scots Pine"(Pinus sylvestris L.). Indole-3-methanol did exist in buffer extract from etiolated seedlings of Pinus sylvestris L.And the stability of indole-3-methanol at different pH levels was investigated.
As a metabolism of indole-3-acetic acid, Indole-3-methanol can be produced by the oxidation of indole-3-acetic acid catalyzed by two cytosolic basic isoperoxidases from Lupinus. This study has been done by A. Barceló in 1990. While indole-3-methanol is only a minor product of the oxidation of indole-3-acetic acid catalyzed by extracellular acidic isoperoxidases, it is the only product of the oxidation of indole-3-acetic acid catalyzed by two cytosolic basic isoperoxidases (EC 1.11.1.7) from lupin (Lupinus albus L.) hypocotyls. It can be produced by the breakdown of the glucosinolate glucobrassicin.
Indole-3-carbinol Toxicity Data With Reference
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| rat | LDLo | subcutaneous | 500mg/kg (500mg/kg) | BEHAVIORAL: COMA | Food and Cosmetics Toxicology. Vol. 18, Pg. 159, 1980. |
Indole-3-carbinol Safety Profile
Safty informations about Indole-3-methanol (CAS NO.700-06-1):
Hazard Codes:Xi
Risk Statements:36/38
36/38 (Irritating to the eyes and skin)
Safety Statements:26-36
26 (In case of contact with eyes,rinse immediately with plenty of water and seek medical advice)
36 (Wear suitable protective clothing)
WGK Germany:3
RTECS:NL9483000
Hazard Note:Irritant
Moderately toxic by subcutaneous route. An experimental teratogen. When heated to decomposition it emits toxic fumes of NOx.
Indole-3-carbinol Specification
Handling and Storage
Handling: Wash thoroughly after handling. Remove contaminated clothing and wash before reuse. Avoid breathing dust, vapor, mist, or gas. Avoid contact with skin and eyes.
Storage: Store in a tightly closed container. Store in a dry area. Keep refrigerated. (Store below 4oC/39oF.)
Related Products
- Indole-3-carbinol
- 70-00-8
- 70008-57-0
- 70010-48-9
- 700-12-9
- 700-13-0
- 700-16-3
- 700-17-4
- 70020-54-1
- 70024-40-7
- 70024-48-5
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View